Abstract
In an era of environmentally friendly development, methods of the green synthesis of zinc oxide nanoparticles (ZnO NPs) from plant extracts have become a focus of research attention because of the benefits of environmental sustainability, simplicity, and low price. The present review introduces a green mechanism for the synthesis of ZnO NPs using the extracts of plants, exploring factors that influence the morphology of ZnO NPs and their antibacterial properties, and the mechanisms of antibacterial action. The results indicate that the factors that influence morphology include the intrinsic crystallographic morphological properties and conditions of the preparation of ZnO NPs. In terms of preparation conditions, the influence of plant extract concentration, precursor concentration, reaction time, and calcination temperature on NP morphology is related to the species of plants used, with precursor concentration the most significant factor affecting the morphology of ZnO NPs. A pH of 12 appears be the most appropriate alkalinity for the synthesis of ZnO NPs from plant extracts. In addition, the synthesized ZnO NPs display excellent antibacterial properties, the mechanism of which involves photocatalysis, reactive oxygen species, and interactions between ZnO NPs and bacterial surfaces. Factors influencing the antibacterial properties are the type of bacteria and the concentration and morphology of ZnO NPs. Finally, the methods of preparation of antibacterial textiles using synthetic ZnO NPs are discussed in relation to the preparation of antibacterial fibers, fabric, and composite textiles. Here, the future trend of such antibacterial textiles is considered, providing the direction for further research of antibacterial textiles.
Introduction
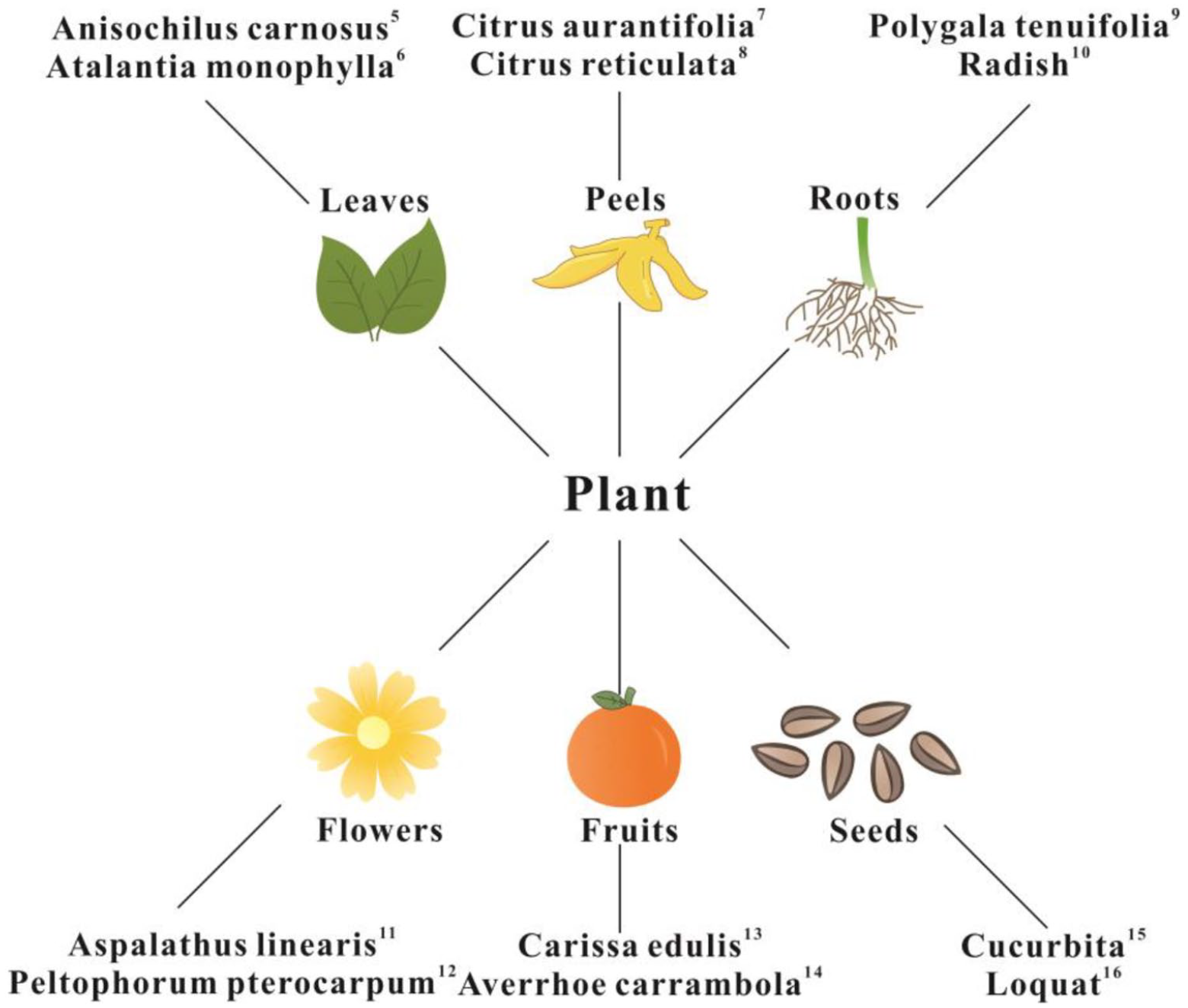
Nanotechnology is an innovative and emerging field, aiming to manufacture novel materials at the nanometer scale. Compared with materials that have an undefined particle size, nanomaterials consist of small particles with a large specific surface area, resulting in materials that display unexpected surface area, volume, quantum size, and macro tunneling effects. Nanomaterials exhibit unique optical, mechanical, catalytic, and biological properties because of these characteristics, resulting in nanomaterials having broad application potential. 1 As a type of nanomaterial, ZnO NPs are widely used in the fields of electrochemistry, medical devices, cosmetics, the textile industry, etc. because of their high specific surface area, biocompatibility, ultraviolet light absorption and scattering, and their antibacterial properties.2,3 The synthesis of ZnO NPs is generally divided into physical and chemical methods, which have the disadvantages of high energy consumption, low purity, uneven particle size distribution, high cost, large quantities of secondary waste, and irreversible pollution of the environment. As applications in which ZnO NPs are utilized increase in number, their synthesis using methods in which the environment is protected is of wide concern, principally because the concept of environmental protection is now deeply rooted in the expectations of the population. Green methods of synthesis refer to those in which microorganisms, enzymes, and plant extracts are used in the fabrication process. No toxic materials should be used, and the process is combined with low energy consumption. It has the advantages of environmental sustainability, eco-friendliness, and low cost, and is therefore an attractive alternative to traditional physical and chemical methods. 4 Plants and their extracts are easy to obtain, and the process requires only zinc salt solution as a metal precursor. ZnO NPs are synthesized by reacting plant extracts mixed with zinc salt solution, representing a method suitable for the green synthesis of ZnO NPs. A number of studies have shown that extracts from leaves, peels, roots, flowers, fruits, and seeds of plants can act as both reducing and stabilization agents for the synthesis of ZnO NPs (Figure 1).5–16 The green synthesis of ZnO NPs from plant extracts results in excellent antibacterial activity against a variety of bacteria,17–19 greater than that observed with chemically synthesized ZnO NPs, without antibiotic resistance. 20 They are non-toxic and compatible with skin, and therefore are suitable for use as additives of products designed to be in contact with the human body. Based on these characteristics, the green synthesis of ZnO NPs from plant extracts can be further used for textiles, adding antibacterial functionality whilst maintaining environmentally-friendly development.
Mechanism of the formation of ZnO NPs from plant extracts
Antioxidants in plants, such as polysaccharides, polyphenols, flavonoids, vitamins, amino acids, alkaloids, tannins, saponins, and terpenoids are reductive. Therefore, plant extracts can be used as reducing agents and capping agents that react with zinc salt solution to form ZnO NPs.21,22
Matinise et al.
23
synthesized ZnO NPs from Moringa oleifera extract and studied the mechanism of their formation. It was found that Zn(NO3)2ž6H2O dissociated into Zn2+ in solution, while
Factors influencing the morphology of synthesized ZnO NPs from plant extracts
Intrinsic crystallographic morphological properties of ZnO
ZnO has a wurtzite crystal structure, with growth readily occurring along the c axis. ZnO NPs grow in the basal direction and have marked shape anisotropy. Due to the effects of surface tension, such nanoparticles are amorphous or crystalline, or can even exhibit a metastable crystallographic phase. 28
Preparation conditions
The most common method of the preparation of ZnO NPs from plant extracts requires the thorough cleaning of the plants with clean or distilled water. Plant extracts are then obtained by drying, grinding into powder, dissolving in solvent, or by direct soaking. The extracts are then mixed with zinc salt solution as the metal precursor from which a precipitate is obtained after being reacted. Finally, ZnO NPs are prepared by calcining the precipitate (Figure 2).

Process of green synthesis of ZnO NPs from plant extracts.
There is a relationship between the morphology and properties of ZnO NPs. 29 It is important to prepare ZnO NPs with a morphology suitable for the purpose intended. Compared with physical and chemical methods, the preparation of ZnO NPs from plant extracts can be controlled so that NPs obtained have a specific size and morphology. 30 In addition, other factors that affect the morphology of NPs include the concentration of plant extract and precursors, duration of reaction, pH value, and calcination temperature (Figure 3).

Factors influencing the green synthesis of ZnO NPs from plant extracts.
As displayed in Table 1, six factors influence the morphology of ZnO NPs.31–42 However, due to differing levels of active reducing compounds in different plant species, 30 the reducing capability is also affected, which fundamentally affects the synthesis of ZnO NPs. Therefore, the effects of plant extract concentration, precursor concentration, reaction time, and calcination temperature are related to the species of plant. Thus, the optimal conditions for the synthesis of ZnO NPs also depend on the species of plant used. However, the overall trend is that the size of ZnO NPs decreases with increasing plant extract and precursor concentration and increases with increasing reaction time and calcination temperature. Rasli et al. 38 found through variance analysis that precursor concentration was the most significant factor affecting the morphology of ZnO NPs synthesized by Aloe vera extract. Abdullah et al. 39 and Shabaani et al. 16 synthesized ZnO NPs with Musa acuminata and Eriobotrya japonica extract at different pH values, respectively, and found that a pH value of 12 was the most appropriate alkalinity for the synthesis of ZnO NPs. Umamaheswari et al. 43 and Jamdagni et al. 44 also confirmed the synthesis by UV-vis spectroscopy. Umamaheswari et al. 43 used Raphanus sativus var. longipinnatus extract to prepare ZnO NPs at pH 8, 10, 12, and 14, which were then characterized by UV-vis spectra. It was found that no absorption peak was observed at pH 14 or pH 8–10, but a characteristic absorption peak was observed at 369 nm when prepared at pH 12. Jamdagni et al. 44 used Nyctanthes arbor-tristis extract to prepare ZnO NPs at pH 9–13. The UV-vis spectra indicated that the absorption lines were almost linear at pH 9, without an absorption peak. Characteristic absorption peaks appeared at pH 12 and 13. However, absorbance and sharpness were both superior at pH 12. Therefore, the authors speculated that the effect of pH on the synthesis of ZnO NPs was not closely related to the species of plant used, with a pH value of 12 possibly the most suitable value for the synthesis of ZnO NPs with any plant extract. The reason may be that the quantity of H+ and OH− will change as pH value is modified, thus affecting the morphology of the ZnO NPs. At a pH value of 12, the relative quantities of OH− and H+ are appropriate. OH− is strongly attracted by the positively charged Zn2+, which promotes the formation of Zn−O bonds in the structure. 39 At lower pH values, the quantity of OH− is reduced, and at a higher pH, the quantity of OH− becomes too great, conditions which are not conducive to the formation of a Zn−O bond.
Factors influencing the green synthesis of ZnO NPs from plant extracts.

The different morphologies of ZnO NPs are displayed in Figure 4.

Antibacterial properties of synthesized ZnO NPs from plant extracts
Bacteria are single-celled organisms with a simple structure that are diverse and have wide distribution, essentially ubiquitous in everyday life. They have strong survival capability, they can rapidly propagate and can easily adapt to environmental conditions, generally harming human health. Increasing antibiotic resistance causes bacteria to pose a serious threat to human life. 45 The green synthesis of ZnO NPs using plant extracts would overcome the problem of antibiotic resistance related to the formation of bacterial biofilms, 46 and because of their good antibacterial properties and biocompatibility, non-toxic, safe, and stable characteristics, they will become a new research direction in the field of antibacterial agents.
Theophil Anand et al. 47 studied the antibacterial properties of ZnO NPs synthesized from Prunus dulcis extract using a disk diffusion method. It was found that the synthesized ZnO NPs inhibited the growth of S. aureus, E. coli, and S. paratyphi. Upadhyaya et al. 48 found that ZnO NPs synthesized from the extract of Lawsonia inermis leaf extract inhibited P. aeruginosa and B. subtilis. Dobrucka and Długaszewska 49 found that the inhibitory effect of ZnO NPs synthesized using the extract of Trifolium pratense flowers on P. aeruginosa was greater than that of gentamicin. Vijayakumar et al. 6 found that ZnO NPs synthesized from the leaf extract of Atalantia monophylla were inhibitory to B. cereus and K. pneumoniae. Stan et al. 31 demonstrated that ZnO NPs synthesized from Allium sativum extract inhibited the growth of S. aureus, E. coli, B. subtilis, P. aeruginosa, L. monocytogenes, and S. typhimurium to a greater extent than chemically-synthesized ZnO NPs. Álvarez-Chimal et al. 50 found that the inhibitory effect of ZnO NPs synthesized from Dysphania ambrosioides extract on S. aureus and S. epidermidis was equivalent to that of chlorhexidine. Ali et al. 51 demonstrated that ZnO NPs synthesized from Aloe vera extract combined with antibiotics were effective at killing methicillin-resistant S. aureus (MRSA) clinical isolates.
These previous studies demonstrate that the green synthesis of ZnO NPs using plant extracts has an inhibitory effect on a variety of bacteria, with antibacterial properties stronger than chemically-synthesized ZnO NPs. Additionally, the effect on a number of bacteria is even greater than that of antibiotics and bactericides. Therefore, they are promising agents which could assist the task of overcoming antibiotic resistance, 52 by using in medical antibacterial textiles, effectively playing a role in sterilization by preventing the growth of bacteria, such as antibacterial bandages, dressings, and gloves.
Antibacterial mechanisms of ZnO NPs synthesized from plant extracts
ZnO NPs created by green synthesis exhibit good antibacterial properties, but the principal antibacterial mechanism remains relatively unclear. For ZnO NPs to be widely used as an antibacterial material, it is first necessary to study the bactericidal mechanisms of ZnO NPs.
Mallikarjunaswamy et al. 53 found that ZnO NPs prepared from Aegle marmelos extract exhibited antibacterial properties, and concluded that the antibacterial mechanism was due to light causing the ZnO NPs to produce reactive oxygen species, causing cells to adopt a state of oxidative stress, leading to protein denaturation in the cells and affecting mitochondrial function and cell metabolic activity, and eventually causing death. Sharmila et al. 54 found that ZnO NPs prepared from Tecoma castanifolia leaf extract displayed antibacterial properties, and found that their effect was on the surface of bacterial cells, leading to the release of ion channels, resulting in an ionic imbalance within the cells that eventually resulted in their death. Ullah et al. 55 demonstrated that the antibacterial properties of ZnO NPs prepared from Thlaspi arvense extract was due to the positive charge of the NPs forming an electrostatic connection with the lipopolysaccharide and phosphoteichoic acid (both negatively charged) components in Gram-positive bacteria, resulting in cell membrane damage, cytoplasmic leakage, and eventually cell death.
In summary, the antibacterial mechanism of ZnO NPs created by green synthesis can be summarized as follows (Figure 5). (a) Photocatalytic mechanism. When ZnO NPs are irradiated by light with energy greater than the bandgap, electrons (e−) in the valence band are excited to transit to the conduction band, leaving a positively charged hole (H+). e− and H+ react with oxygen, hydroxyl groups, and water adsorbed on the surface of the material to produce OH−, O2−, and H2O2, respectively. Of these, H+ and OH− have strong oxidative properties, able to break the chemical bonds of the majority of organic compounds, resulting in the degradation of various components of a microorganism, thus being bactericidal. In addition, O2− has a high reduction capability, which also results in having antibacterial properties. 56 (b) Reactive oxygen mechanism. ZnO NPs produce ROS in cells, leading to damaged bacterial membranes, and thus bacteriolysis and promotion of ZnO NP aggregation within bacteria, eventually leading to their death. 57 (c) Interaction between ZnO NPs and bacterial surface. Because of the large specific surface area of ZnO NPs, they exhibit a strong surface effect. They contact bacterial cell membranes over a large area, interacting with each other, leading to bacterial membrane damage and the release of ion channels, resulting in internal ionic imbalance of the cells, and eventually death. 58

The majority of antibiotics kill bacteria by disrupting the permeability of the cell membrane, inhibiting membrane synthesis, or regulating the enzymatic pathways for the processes of transcription, translation, and replication. Bacteria degrade antibiotics or undergo gene mutation that reduces their binding affinity to target enzymes, preventing or reducing their bactericidal effects. 45 The antibacterial mechanism of ZnO NPs synthesized from plant extracts is difficult to overcome via drug resistance, and therefore they display stable and durable antibacterial properties.
Factors influencing the antibacterial properties of ZnO NPs synthesized from plant extracts
Types of bacteria
Depending on the composition of the cell wall, bacteria can be categorized as either Gram-positive or Gram-negative. It has been found that Gram-positive bacteria are more sensitive to ZnO NPs synthesized from plants than Gram-negative bacteria. Thus, Inamdar et al. 59 found that under the same culture conditions, ZnO NPs prepared with Mimosa pudica leaf extract displayed stronger antibacterial effects on Gram-positive bacteria. The inhibitory concentration of ZnO NPs on S. aureus (Gram-positive bacteria) was 0.00025 μg/mL, but 2.5 μg/mL and 0.0025 μg/mL toward E. coli and B. subtilis (Gram-negative bacteria), respectively. This demonstrates that a lower concentration of ZnO NPs can inhibit Gram-positive bacteria compared with Gram-negative strains. Velsankar et al. 60 found that B. pumilus (Gram-positive bacteria) were more sensitive to ZnO NPs prepared from Echinochloa frumentacea grain extract than S. typhi (Gram-negative bacteria). This is likely due to differences in the composition of the cell wall between the two bacteria. The cell walls of Gram-positive bacteria contain peptidoglycan, teichoic acid, and abundant pores which allow foreign molecules and nanoparticles to enter the cell, resulting in cell membrane damage and cell death. However, the cell walls of Gram-negative bacteria contain lipopolysaccharide, lipoprotein, and phospholipid, representing a barrier that only allows macromolecules to enter, hindering the entry of nanoparticles and weakening the action of ZnO NPs against Gram-negative bacteria. 61
Concentration of ZnO NPs
The antibacterial properties of ZnO NPs vary with the concentration of ZnO NPs. Dobrucka and Długaszewska 49 found that when cultured at concentrations of 125, 256, 516, and 1280 μg/mL, the diameters of zones of inhibition of ZnO NPs prepared from Trifolium pratense flower extract were 22, 26, 30, and 31 mm for S. aureus, and 22, 26, 29, and 31 mm for E. coli, respectively. Thirumoorthy et al. 62 prepared ZnO NPs from Tetraselmis indica extract, resulting in zones of inhibition diameters for S. aureus of 12.3 ± 0.3 mm at a concentration of 50 mg/mL, increasing to 18.4 ± 0.5 mm at a concentration of 100 mg/mL. These studies demonstrate that the zone of inhibition for bacteria increases with increasing ZnO NPs concentration.
Morphology of ZnO NPs
Results have shown that the antibacterial properties of ZnO NPs depend on NP morphology. Saif et al. 2 found that the diameters of zones of inhibition using hexagonal, pyramid, and round ZnO NPs prepared from Cordia myxa extract were 7.2 ± 0.68, 8.0 ± 0.94, and 6.0 ± 0.80 mm for E. coli, respectively, and 8.00 ± 1.00, 9.4 ± 0.20, and 7.2 ± 0.80 mm for S. aureus, respectively, at a concentration of 50 μg/mL. Upadhyaya et al. 48 found that hexagonal ZnO NPs prepared from Lawsonia inermis extract displayed greater inhibition of S. aureus than rod-shaped ZnO NPs at concentrations of 100, 200, and 500 μg/mL, concluding that hexagonal ZnO NPs more easily penetrated the membrane barrier, resulting in cell leakage and death, due to their shape compared with rod-shaped ZnO NPs. Talebian et al. 63 synthesized ZnO NPs with different morphologies via a simple solvothermal method and found that flower-shaped ZnO NPs more strongly killed S. aureus and E. coli than rod-shaped ZnO NPs. These observations suggest that sharp-edged ZnO NPs may have greater antibacterial properties because they more easily penetrate the bacterial cell wall than NPs with smooth edges.
Preparation of ZnO NP antibacterial textiles
The porous structure of textiles causes the accumulation of human sweat and dander which creates a warm and humid environment appropriate for the growth and reproduction of bacteria. For the protection of public health, antibacterial materials are increasingly demanded in a variety of applications, such as sportswear, and medical and protective textiles to prevent bacterial infection. 64 Antibacterial materials would be of particular benefit for the fabrication of medical textiles used to treat wounds. 65 Antibacterial agents are effective at inhibiting the growth and reproduction of bacteria and destroying them. Although the traditional agents used in antibacterial textiles have antibacterial properties, the materials produced by current methods are not ideal. For example, the antibacterial properties are not durable, bacteria develop drug resistance that adversely affects human health, while the production process pollutes the environment. Depending on their source, agents can be divided into three categories: natural antibacterial agents, inorganic antibacterial agents, and organic antibacterial agents. Inorganic antibacterial agents are useful due to their excellent heat resistance, they cause no drug resistance, are not volatile, and have an excellent safety profile with a broad range of antibacterial properties. As an inorganic antibacterial agent, green synthetic ZnO NPs not only exhibit the beneficial properties described above, they also are non-toxic and compatible with the skin, and so are suitable for use in textiles, allowing the fabrication of antibacterial textiles with green environmental credentials, compensating for the shortcomings of traditional antibacterial agents. ZnO NPs can be incorporated as antibacterial agents in green, non-toxic, and harmless antibacterial textiles, and are effective in reducing cross-infection, preventing the spread of disease, thereby ensuring the safety and health of humans. Not only do they fulfill the demands of antibacterial functional textiles, they also satisfy the public’s pursuit of environmental protection. Therefore, the development of environmentally-friendly ZnO NP antibacterial textiles provides the potential to create products with excellent market development prospects in numerous applications.
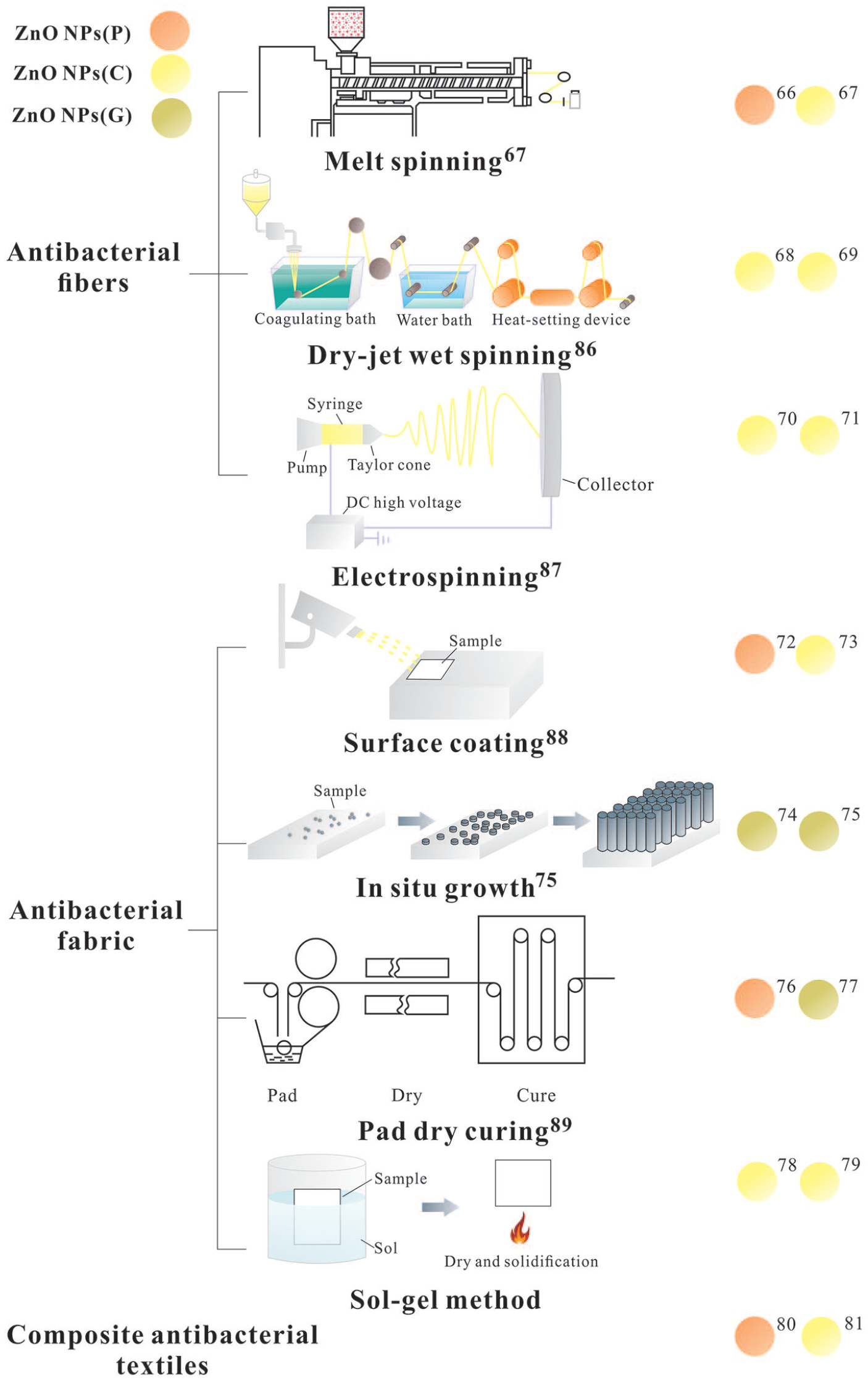
Methods of combining antibacterial agents and textiles usually require the preparation of antibacterial fibers or fabrics. Therefore, the present review will describe the methods by which ZnO NP antibacterial textiles are formulated from ZnO NP antibacterial fibers and ZnO NP antibacterial fabrics. In addition, the techniques by which composite antibacterial textiles are fabricated with ZnO NPs and other antibacterial nanomaterials synthesized from plant extracts are also introduced. Published studies of the green synthesis of ZnO NPs from plant extracts are mostly limited to their synthesis, characterization, and functional evaluation. Few have devoted their investigation to applications in textiles. Therefore, the methods by which antibacterial textiles are prepared using ZnO NPs synthesized from plant extracts are described in the present review, including their methods of preparation using NPs synthesized through chemical methods or commercially sourced. ZnO NPs that have been purchased commercially are termed ZnO NPs(P), while ZnO NPs synthesized by chemical methods are called ZnO NPs(C), and green ZnO NPs synthesized from plant extracts are termed ZnO NPs(G).
Preparation of ZnO NP antibacterial fibers
Antibacterial fibers with ZnO NPs are prepared by mixing the antibacterial agent containing ZnO NPs with the raw fiber material prior to spinning, either melt spinning, dry-jet wet spinning, or electrospinning. Fibers prepared using this latter method exhibit good, stable, and long-lasting antibacterial properties.
Melt spinning
Melt spinning produces antibacterial fibers by mixing an antibacterial agent with a fiber matrix. This method requires good temperature resistance, a small particle size, and good compatibility and dispersion of the polymer suitable for the preparation of ZnO NP antibacterial fibers. Dural Erem et al. 66 prepared ZnO NPs and polyamide composite fibers by melt spinning with commercially-sourced ZnO NPs(P) and tested their antibacterial properties using S. aureus and K. pneumonia. The results indicated that the composite fiber displayed significant antibacterial effects against the two species of bacteria, concluding that the effect was related to the content of ZnO NPs, increasing with the increased ZnO NPs concentration. Zhang et al. 67 synthesized ZnO NPs(C) using zinc acetate and an amino hyperbranched polymer, and then prepared polypropylene (PP)/ZnO fiber by melt spinning method. It was observed that the fibers displayed excellent antibacterial activity against E. coli and S. aureus.
Dry-jet wet spinning
In dry-jet wet spinning, the spinning solution is extruded from a spinneret hole, passing through air, finally entering a wet spinning coagulation bath to form an antibacterial fiber. Li 68 prepared ZnO NPs with Zn(NO3)2·6H2O, C6H8O7·H2O and Al(NO3)2·9H2O. Polyacrylonitrile-based activated carbon fibers loaded with ZnO NPs(C) prepared by dry-jet wet spinning has demonstrated antibacterial activity against E. coli and S. aureus, due to increased ZnO NPs content in the composite fiber that improved it antibacterial properties. Fu et al. 69 synthesized ZnO NPs(C) using sodium dodecyl sulfonate (SDS), Zn(NO3)2·6H2O and NaOH, and then prepared cellulose/ZnO fibers by dry-jet wet spinning. Assessment of its antibacterial properties demonstrated that the fibers exhibited excellent antibacterial activity against S. aureus and E. coli.
Electrospinning
Electrospinning charges a polymer solution or melt within a strong electric field from which a continuous fiber is created by solvent evaporation or solution cooling and curing. Norouzi et al. 70 used zinc acetate dehydrate and NaOH to prepare ZnO NPs(C) and found that fibers created from ZnO NPs and polyvinyl alcohol by electrospinning displayed antibacterial activity against S. aureus and E. coli while being non-toxic to human cells and promoting wound healing. Fibers can be fabricated into a medical functional dressing with anti-inflammatory properties, promoting wound healing. Chen et al. 71 formed ZnO NPs(C) from Zn(CH3COO)2·2H2O and diethylene glycol, and then prepared gelatin/ZnO fibers by electrospinning. An antibacterial assessment indicated that the fibers exhibited excellent antibacterial activity against S. aureus and E. coli with a significant reduction in bacterial numbers of more than 90%.
Preparation of ZnO NP antibacterial fabric
ZnO NP antibacterial fabric is prepared by attaching an antibacterial agent containing ZnO NPs to the surface of the fabric by physical or chemical methods such as surface coating, in situ growth, pad dry curing, and sol-gel. It has the advantage of simplicity and is not time-consuming.
Surface coating
Surface coating is currently a widely-used antibacterial finishing method in which the antibacterial agent and coating agent are mixed to form an antibacterial slurry which is then adhered to the surface of the fabric. This method is easy to perform and suitable for many types of fabric. Jin et al. 72 prepared an antibacterial cotton fabric surface coated with PVDF and purchased ZnO NPs(P). Firstly, PVDF solution and ZnO NP solution were mixed to form an antibacterial slurry into which cotton fabric was immersed. The soaked cotton fabric was then placed in a coagulating non-solvent consisting of a mixture of water and ethanol to solidify the antibacterial coating fixed on the cotton fabric surface. The results indicated that the antibacterial cotton fabric displayed good antibacterial activity against S. aureus and K. pneumoniae. Rani et al. 73 prepared ZnO NPs(C) from Zn(NO3)2·6H2O and NaOH, and subsequently coating them on the Tasar silk fabric. The fabric displayed excellent antibacterial activity against E. coli.
In situ growth
The antibacterial agent is deposited directly onto the surface of the fabric during in situ growth following immersion into a reaction solution. Aladpoosh and Montazer 74 synthesized ZnO NPs(G) in situ on the cellulosic chains of cotton fabric using Keliab extract and Zn(CH3COO)2·2H2O. Firstly, Zn(CH3COO)2·2H2O and cellulosic cotton fabric was impregnated in the synthesis bath stirred for 30 min. Then, Keliab extract was slowly dripped into the synthesis bath, heated to 90°C for 60 min. Finally, the fabric was taken out and dried at 80°C for 30 min, cured at 150°C for 3 min, rinsed with distilled water and dried at room temperature to obtain the fabric sample. It was found that the treated cellulose cotton fabric had excellent antibacterial activity against E. coli and the bacterial reduction rate of S. aureus was 91.66%. Verbič et al. 75 synthesized ZnO NPs(G) on cotton fabric in situ by four methods using Pomegranate peel extract, wood ash extract and Zn(CH3COO)2·2H2O. Compared with the other three methods, the distribution of ZnO wurtzite nanoparticles on the surface of cotton fabric treated by method 4 was more uniform. In this method, the fabric was successively immersed in wood ash extract, Pomegranate peel extract and Zn(CH3COO)2·2H2O, then dried in a continuous dryer. Finally, the fabric sample was firstly dried in a laboratory oven at 100 °C for 30 min. And then temperature will be adjusted to 150 °C, the sample will be kept in the oven for 5 min further, so as to obtain a cotton fabric with evenly distributed ZnO NPs on the surface. Therefore, it is considered that the in situ synthesis of ZnO NP on cotton using method 4 had great potential to advance the development of environmentally friendly functional textiles, reduce the environmental impact of classical chemical textile functionalization and enables the production of protective textiles that could be used in a clothing, medical, or technical textile sector.
Pad dry curing
In pad dry curing, a fabric is coated with a solution of antibacterial agent which then solidifies to make a pad attached to the fabric. Yazhini and Prabu 76 prepared a ZnO NP antibacterial cotton fabric by pad dry curing with purchased ZnO NPs(P). BTCA was used as a cross-linking agent and sodium hypophosphite as the catalyst. Cotton fabric was immersed in the composite colloid solution for 40 min and then extruded, after which the wet fabric was dried in air and cured at 180℃ for 4 min to obtain ZnO NP antibacterial cotton fabric. The fabric displayed good antibacterial activity against both methicillin-resistant S. aureus and P. aeruginosa. Karthik et al. 77 synthesized ZnO NPs(G) from Acalypha indica leaf extract and coated them on a woven cotton fabric using a pad drying curing method. The results demonstrated that the ZnO NP-coated fabric exerted antibacterial activity toward E. coli and S. aureus, the highest rate of antibacterial performance reaching 96% and 99%, respectively.
Sol-gel method
Small molecules are aggregated into a solid phase which then collides and crosslinks within a solvent to form a three-dimensional network structured gel. The sol-gel method solves the problem of insufficiently strong attachment of antibacterial substances to fabric. Farouk et al. 78 prepared ZnO NP antibacterial cotton fabric from ZnO NPs(C) that had been formed using ZnAc·2H2O and LiOH, then dissolved GPTMS in solvent to form a basic sol, into which ZnO was dispersed and mixed to form a ZnO NPs sol, which was then coated on cotton fabric to obtain an antibacterial cotton material with antibacterial activity against E. coli and M. luteum, causing a reduction of 98.8% and 97.3%, respectively. They found that the antibacterial activity of the fabric treated with ZnO NPs increased with increasing ZnO NP content. Kamali and Talebian 79 firstly prepared ZnO sol ZnO NPs(C) with Zn(CH3COO)2·2H2O, 2-propanol, and monoethanolamine, then immersed the cotton fabric in the sol prior to sonicating for 50 min, drying at 100℃ for 5 min, then finally curing at 150℃ for 3 min. The treated cotton fabric displayed high antibacterial activity against E. coli.
Preparation of ZnO NP composite antibacterial textiles
In addition to using ZnO NPs as a single antibacterial agent, they can also be combined with other nanomaterials that have antibacterial properties to prepare materials with superior antibacterial performance. Abbasipour et al. 80 prepared an Ag/ZnO/chitosan composite-coated cotton gauze with ZnO NPs provided by the enterprise, and evaluated its antibacterial properties in comparison to chitosan, chitosan/Ag, chitosan/ZnO, and chitosan/Ag/ZnO-treated and untreated cotton gauze. The results demonstrated that Ag/ZnO/chitosan eliminated 99% of S. aureus and 96% of E. coli. The other cotton gauze treatments were less effective against S. aureus. Babu et al. 81 prepared ZnO NPs(C) using Zn(NO3)2·6H2O, PVP, and NaOH. Then they prepared Ag/ZnO (AZ) and Ag/ZnO/Fe3O4 (AZF) nanocomposites and compared their antibacterial properties with ZnO against E. coli and S. aureus. The results indicate that the antibacterial properties of AZ against these bacteria were greater than that of ZnO, while the performance of AZF against E. coli was superior to that of ZnO. These studies demonstrate that ZnO composites have antibacterial properties superior to that of ZnO alone.
In addition to ZnO NPs, Ag, and Fe3O4 NPs utilized in composites can also be synthesized environmentally with plant extracts, not only improving the antibacterial properties of materials but also ensuring the green sustainability of the preparation process. Thomas et al. 82 synthesized Ag NPs with Passiflora edulis leaf extract, finding that they displayed antibacterial activity against E. coli and S. aureus. Rahmawati et al. 83 synthesized Fe3O4 NPs with Saccharum officinarum extract. In addition to Fe3O4, Aisida et al. and Mohamed et al. also synthesized FeO and Fe2O3 using plant extracts, observing that they had inhibited bacteria. Aisida et al. 84 synthesized FeO nanorods (FeO-NRs) with Moringa oleifera leaf extract, and found that the FeO-NRs inhibited the growth of S. aureus, P. aeruginosa, E. coli, Shigella, Salmonella typhi, and P. multocida. Mohamed et al. 85 synthesized Fe2O3 NPs with Hyphaene thebaica fruit extract, and studied the antibacterial properties against different strains of bacteria. The results indicated that Fe2O3 NPs also exhibited antibacterial activity with the following ranking: B. subtilis > P. aeruginosa > K. pneumonia > E. coli > S. epidermidis.
In summary, green synthetic ZnO NP antibacterial fibers can be prepared by melt spinning, dry-jet wet spinning, or electrospinning, with good antibacterial properties. Compared with fibers prepared by melt spinning and dry-wet spinning, those prepared by electrospinning have a smaller diameter, greater uniform stability, higher specific surface area, with stable and durable antibacterial performance, and so can be used in antibacterial textiles in applications where there was a high demand for bactericidal properties. However, the preparation procedure is complex, using sophisticated equipment. Green synthetic ZnO NP antibacterial fabric can be prepared by surface coating, in situ growth, pad drying, or sol-gel methods, and in each case, the antibacterial fabric exhibits antibacterial activity. Surface coating and in situ growth methods the most simple to execute, although the duration of the antibacterial effect is rather poor, performance gradually weakening due to friction and washing as the fabric is used. Dry curing and sol-gel methods improve the attachment of antibacterial agents to the fabric. Using these methods, an antibacterial coating can be produced that has uniform composition, high purity, few restrictions for the fabric substrate, combined with low production costs. Therefore, ZnO NP antibacterial textiles can be prepared that have durable and stable antibacterial properties. The green synthesis of antibacterial nanomaterials with plant extracts described above can be used to prepare composite antibacterial materials with ZnO NPs that are effective in improving the antibacterial properties of textiles with green credentials.
However, as shown in Figure 6, at present, ZnO NPs synthesized by chemical methods or purchased commercially are used mostly in researching ZnO NP antibacterial textiles. ZnO NPs synthesized from plant extracts are rarely used, creating a void in the available research results, despite their tremendous research potential.
As science and technology have progressed and individuals’ living standards have grown, there is a generally higher expectation of comfort and cleanliness. ZnO NP antibacterial textiles can be washed multiple times, demonstrating antibacterial persistence following direct contact with human skin. Although there are limited numbers of research publications on this subject, there is considerable potential for different research directions for ZnO NP antibacterial textiles in the future.
Conclusions
Over recent years, green methods of synthesizing ZnO NPs using plant extracts have attracted considerable attention due to the benefits to environmental protection, lack of toxicity, and low cost. The present review has summarized recent progress of their synthesis over recent years, and their prospects for applications in antibacterial textiles explored. As can be observed from a survey of the literature presented here, the morphology of ZnO NPs is dependent on plant species, plant extract concentration, precursor concentration, reaction time, pH value, and calcination temperature. In terms of the conditions for preparation, the plant species is the most fundamental factor affecting NP morphology, while other factors are associated with it, including precursor concentration, which is the most significant factor affecting their morphology. However, pH value is less dependent on plant species, and a pH value of 12 appears to be the most suitable for the synthesis of ZnO NPs from plant extracts. In addition, ZnO NPs synthesized in this way demonstrate antibacterial activity against a variety of bacteria that is greater than that provided by ZnO NPs created by chemical synthesis. Furthermore, they do not suffer the problem of antibiotic resistance. The mechanism of antibacterial action is photocatalysis, reactive oxygen species formation, and direct interaction of the nanoparticles with bacterial surfaces. The type of bacteria, and concentration and morphology of nanoparticles are factors that affect antibacterial properties. Compared with Gram-negative bacteria, Gram-positive strains are more sensitive to ZnO NPs synthesized from plant extracts. The antibacterial effect increases with increased ZnO NP concentration. ZnO NPs with sharp edges have better antibacterial performance than those with smooth edges. Compared with traditional physically and chemically synthesized ZnO NPs, NPs synthesized using this method are non-toxic and compatible with skin and so are suitable for the preparation of antibacterial textiles. Such textiles with environmental credentials satisfy the market demand for sustainable products and so have excellent market development potential and prospects for various applications. However, little research has as yet been performed on the comfort and antibacterial durability of ZnO NPs antibacterial textiles, and this should be the research direction in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
