Abstract
Fibers are the unit component for product development. They can be divided into two types: synthetic and natural fibers. Recently, emerging nanotechnology has played a vital role in advancing next-generation fabrics. The nanomaterials provide several unique properties such as higher conductivity, self-cleaning, water-resistant, and others. Owing to their advanced properties, the fabrics are being developed by coating and integrating with nanomaterials. Therefore, in the presented work two cotton samples were modified with titanium dioxide (TiO2) and zinc oxide (ZnO). These samples were further examined under various techniques including scanning electron microscopy (SEM), UV-visible spectroscopy, X-ray fluorescence (XRF), and Fourier-transform infrared spectroscopy (FTIR). Furthermore, these samples were evaluated at varying wavelengths with UV light and the obtained results demonstrated that the nano-coated fiber samples can be differentiated at 365 nm.
Introduction
Nanotechnology is a widely progressing discipline employing distinct nanostructures including nanofibers, nanotubes, nanocrystals, and nanofoams in various fields. Nanofiber technology is introduced in applications of composites for aircraft, fabrics, batteries, energy transfer systems, and drug delivery.1,2 Fabrics and clothing are the rapidly growing sector worldwide. Fabrics have various applications in sectors such as defense, healthcare, clothing, environmental protection, packaging, industry, and machinery. Nanomaterials are incorporated in fabrics to provide exceptional properties including stain proof, self-cleaning, antimicrobial, anti-static, waterproof, oil repellent, ultraviolet light protection, and temperature controlling. 3 The nano fabrics can be used for multiple applications such as fabric development, food processing for sterile food wrapping, filter substances, and gauzes used for treatments. The studies have shown that the cotton fabric once coated with nanomaterials improves the quality of fabric. 4
Fibers are classified into two categories; synthetic and natural fiber. To modify these fibers, several nanomaterials have been synthesized. The nanomaterials coated fibers overcome the drawbacks of synthetic and natural fibers including wrinkle-resistant, 5 fire-resistant, 6 and others. The graphene oxide nanosheets modified with titanium dioxide (TiO2) nanoparticles and further coated on the cotton fabrics provide self-cleaning properties, ultraviolet (UV) protection, and enhances electrical conductivity. 7 Carbon nanotubes (CNTs) are useful for improving the mechanical strength of the composite. 8 Few nanomaterials such as copper oxide (CuO), 9 and silver (Ag) 10 help in the development of antimicrobial fibers. In addition, ZnO and TiO2 nanomaterials provide anti-bacterial and self-cleaning properties11–15 in a much better way. Therefore, these nanomaterials were incorporated in this present study.
The fibers can be modified with nanomaterials using two methods: (a) Before spinning, the nanomaterials are blended with raw fiber material; (b) Coating nanomaterials on the developed fiber. 16 The most widely used nanomaterials for fiber modification are ZnO and TiO2. These nanomaterials provide significant benefits over other nanomaterials. There are various studies that have been reported for the modification of fibers to achieve exceptional properties (water and fire resistant, self-cleaning, microbial repellent). A study describes the modification of the polyester surface with TiO2, Iron oxide (Fe3O4), Ag nanocomposite, to enhance antibacterial, self-cleaning, and impart magnetic activities. The presence of TiO2 nanoparticles increases the photocatalytic activity while Fe3O4 and Ag nanoparticles are nucleated on the TiO2 nanoparticles modified surface. This enhances the electron-hole pair separation and thus the rate of recombination is extended. 17 Another study shows a surface modification of cotton fabric with cerium oxide (CeO2), ZnO, and TiO2 using the cross-linking method. To investigate the properties of these modified cotton samples, various techniques were incorporated to determine the number of nanoparticles used in the sample such as X-ray fluorescence (XRF), and energy dispersive X-ray spectroscopy (EDX). The Field emission scanning electron microscopy (FESEM) was used to observe the distribution of nanoparticles on the surface. The samples were evaluated for Gram-positive and Gram-negative bacteria to check the antibacterial properties. 18 Further, the modification of cotton fibers with manganese (Mn) doped TiO2 nanoparticles was reported. The modified sample shows a 100% reduction of Gram-positive and Gram-negative bacteria. The fabric also shows other properties including mechanical strength, breathability, and resistance. 19 TiO2 nanoparticles have several applications which can be incorporated in polyesters and provide indoor decontamination.20,21 Moreover, the modification of polyester and cotton fabrics with TiO2 nanoparticles leads to provide wrinkle-resistant, anti-UV, and air permeability of the fabric.22,23
The research conducted discusses the characteristics of cotton fibers once coated with TiO2 and ZnO nanoparticles. The cotton samples were characterized with various techniques including scanning electron microscopy (SEM), UV-visible spectroscopy, XRF, and Fourier-transform infrared spectroscopy (FTIR).
Materials and methods
Chemicals and reagents
Titanium tetra isopropoxide (TTIP) was purchased from Sigma-Aldrich, U.S.A., nitric acid, zinc nitrate (Zn(NO3)2.6H2O), and potassium hydroxide (KOH) were acquired from Sisco Research Laboratories Pvt Ltd (SRL), Delhi, India. Distilled water (DW) was used throughout the experiments. Prior to experiments all the apparatus were autoclaved. TiO2 and ZnO nanoparticles were synthesized in the laboratory.
Cotton gauze cloth of plain weave was purchased from the local vendor each piece weighing 80 mg. For the comparative study, the cloth was coated with nanoparticles (TiO2 and ZnO) separately. The control experiments were performed with the non-coated cotton samples.
Synthesis of TiO2 nanoparticles
The synthesis of TiO2 nanoparticles was performed in the laboratory, using TTIP as a precursor and nitric acid as a reducing agent. To synthesize TiO2 nanoparticles a solution of TTIP, absolute ethanol, and DW was prepared in the ratio of 1:1:4. The pH of this solution was maintained at 7. The solution was continuously stirred for 30 min and kept undisturbed for up to 24 h. The obtained gel was dried at 120°C for 2 h. The product was again heated at 450°C for 2 h, and the sample obtained was grind thoroughly into fine powder.
Synthesis of ZnO nanoparticles
The ZnO nanoparticles were prepared using Zn(NO3)2.6H2O as precursor and KOH as reducing agent. A solution of 4.87 g of Zn (NO3)2 in 80 mL of DW was prepared, the solution was kept for continuous stirring while adding KOH solution drop-wise till the white gel is formed. The formed gel was centrifuged and washed with DW repeatedly three times, and the final wash was carried out with alcohol. The gel was dried at 100°C, and a white color powder was obtained. The white powder was heated up to 500°C for 3 h.
Coating procedure
The samples were washed with DW and dried at 100°C for 5 min to remove the moisture. Suspensions of concentration (0.4 g/L) for each TiO2 and ZnO nanopowder were made separately. These suspensions were sonicated for 30 min. The samples were immersed in the suspension for 10 min and then padded. Thereafter, the samples were annealed at 75°C for 1 h. The samples loaded with nanopowder were washed with DW in an ultrasonic bath for 5 min to remove the unbounded nanoparticles. The control samples were also washed with DW and dried at 100°C for 5 min and annealed at 75°C. The control samples were marked as “S,” TiO2 nanoparticles coated samples were marked as “S1,” and ZnO nanoparticles coated samples were marked as “S2.”
Results and discussion
Characterization of TiO2 and ZnO nanoparticles
The size of the TiO2 nanoparticles was obtained, with dynamic light scattering (DLS) at AINT, Amity University Uttar Pradesh (AUUP), Noida, India (Zetasizer Nano-S90 by Malvern Panalytical). Figure 1 shows the DLS of the nanoparticles, Z-average for TiO2 nanoparticles was found to be 115.7 nm. The ZnO nanoparticles were characterized with ultraviolet-visible spectroscopy at AINT, Amity University Uttar Pradesh (AUUP), Noida, India (UV- 2600, Shimadzu). The UV peak for ZnO nanoparticles was observed at 371 nm (Figure 2).

Dynamic light scattering (DLS) of TiO2 nanoparticles.

Ultraviolet spectroscopy (UV) of ZnO nanoparticles.
The surface morphology of the samples was studied with SEM as shown in Figure 3. The study was carried out at AIMS, Amity University Uttar Pradesh (AUUP), Noida, India (EVO 18 Special Edition, Zeiss). The samples were coated with gold-palladium, by sputtering to make them conductive before SEM analysis. In Figure 3(a) there are spherical shape structures on the surface, showing the attachment of TiO2 nanoparticles on the sample. In Figure 3(b) elongated shapes are attached to the surface shows the coating of ZnO nanoparticles on the cotton sample. Figure 3(c) shows no structure, this describes that there is no nanomaterial coating on the surface, so this sample was used for control experiments.

Scanning electron microscopy (SEM) for samples (S, S1, and S2): (a) S1 (TiO2 nanoparticle coated) at 10 µm, (b) S1 (TiO2 nanoparticle coated) at 2 µm, (c) S2 (ZnO nanoparticle coated) at 10 µm, (d) S2 (ZnO nanoparticle coated) at 2 µm, (e) S (non-coated) at 10 µm, and (f) S (non-coated) at 2 µm.
The composition elements of the samples were studied with energy dispersive X-ray spectroscopy as illustrated in Figure 4. Figure 4(a) shows titanium (Ti) and oxygen (O) composition, confirming the presence of TiO2 nanoparticles on the surface of sample S1. Figure 4(b) illustrates the composition of Zn and O, hence, the presence of ZnO nanoparticles is confirmed on the surface of sample “S2.” Figure 4(c) describes the composition of sample S, where no titanium or zinc is present therefore, it was used as a control. The presence of carbon and oxygen in the control sample may come from substrate cotton which is nothing but cellulose. In all three samples, the gold and palladium were present, because of prior coating to make the sample conducting for SEM analysis.

Energy dispersive X-ray analysis (EDX): (a) EDX for S1 (TiO2 nanoparticles coated), (b) EDX for S2 (ZnO nanoparticles coated), and (c) EDX for S (non-coated).
Comparative study of coated and non-coated cotton samples using non-destructive traditional techniques
Visualization under distinct magnifications
The fibers were examined by using Nirvis Docubox Dragan in white light at 3× (Figure 5(a)) and 75× (Figure 5(b)) magnifications. However, these fibers could not be differentiated under both the magnifications. Since the sizes of coated particles are in nanoscale range these particles do not interact with visible region light.

Examination under magnification for samples using Nirvis Docubox Dragan: (a) magnification at 3× (i) TiO2 nanoparticle coated cotton sample, (ii) ZnO nanoparticle coated cotton sample, and (iii) control sample; and (b) magnification at 75× (i) TiO2 nanoparticle coated cotton sample, (ii) ZnO nanoparticle coated cotton sample, and (iii) control sample.
Optical differentiation through UV Light
The UV light analysis of the samples was carried out with the VISPEC instrument (Foster Freeman), at different wavelengths 254, 313, 365 nm. It was observed that the control sample “S” shows no transition at 254 and 313 nm as illustrated in Figure 6(a) and (b), respectively. Whereas it shows fluorescence effect at 365 nm as represented in Figure 6(c). Similarly, no transition was obtained with nano-coated samples at wavelength 254 and 313 nm. However, at 365 nm an enhancement of fluorescence in ZnO nanoparticles coated sample was observed as presented in Figure 6(c). The enhancement in the intensity of fluorescence for ZnO nanoparticles coated cotton fiber is because the ZnO nanoparticles show fluorescence property under UV Light. 24 Also, at 365 nm quenching of fluorescence in TiO2 nanoparticles coated sample was obtained as described in Figure 6(c). In this case, TiO2 nanoparticles coated sample, the coating works as a barrier between the cotton surface and UV light. It can be described as interaction area of the surface decreases, which causes the suppression in the intensity of fluorescence and as a result, a good difference among these three samples at 365 nm.
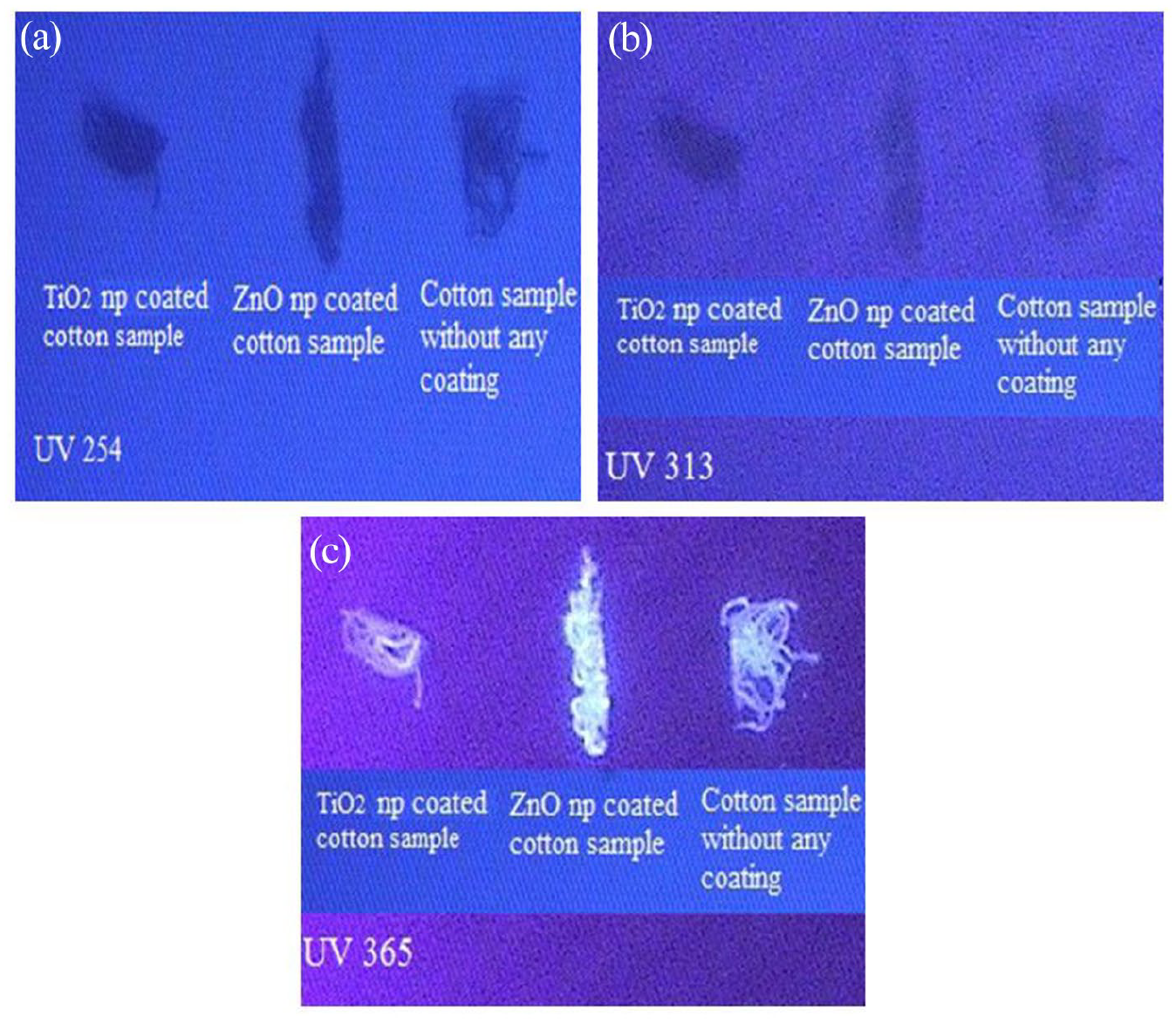
Comparison under ultraviolet light: (a) UV 254 nm, (b) UV 313 nm, and (c) UV 365 nm.
FTIR measurements
FTIR was performed using the Perkin Elmer Spectrum GX FTIR system in ATR mode with a horizontal attenuated reflection accessory. The three samples (S, S1, and S2) were scanned within a range from 4000 to 400 cm−1 as shown in Figure 7. The spectra of “S1” (Figure 7(a)) and “S2” (Figure 7(b)) were compared. No appreciable difference was observed from 4000 to 600 cm−1. However, expanding the spectra between 600 and 400 cm−1 as illustrated in Figure 7(a) and (b) it was found that, an extra peak exists at 417.18 cm−1 in the spectra of sample S1. Further, it was also observed that the two peaks at 469.91 and 463.57 cm−1 in spectra of sample S (Figure 7(c)) are merged into one peak at 465.06 cm−1. Similarly, the two peaks in the spectra of “S” sample at 454.54 and 450.00 cm−1 are replaced by one peak at 453.71 cm−1 in the spectra of “S1.” No such merging of peaks was observed in the spectra of sample “S2,” but the peak at 411.74 cm−1 was split into 413.76 and 409.74 cm−1. Extra peaks at 419.24, 498, and 508 cm−1 were also observed in the spectra of “S2” (Figure 7(b)). The splitting of peaks at wavelength 557.75 and 492.05 cm−1 was observed in both the nano-coated samples “S1” and “S2.” Merging of the peaks in TiO2 coated cotton sample “S1,” the appearance of extra peaks in both the nano-coated samples, and the splitting peaks in these samples can be used for differentiating the TiO2 coated, ZnO coated, and control samples. The reason for splitting, merging, and appearance of extra peaks is beyond this paper and it is a matter of further research.

Fourier-transform infrared spectroscopy analysis: (a) FTIR for S1 (TiO2 nanoparticles coated), (b) FTIR for S2 (ZnO nanoparticles coated), and (c) FTIR for S (non-coated).
X-ray fluorescence analysis
The samples were examined with XRF (Reny EDS- 720 model, SHIMADZU). Samples were kept in a Mylar cup with a 6 µm thickness of Mylar film. The following instrument parameters were used for the analysis: target material (Rhodium (Rh)), operating voltage (50 kV), and acquisition range (0–40 keV). The XRF results are presented in Figure 8. XRF spectrum of the control sample is showing that most of the X-ray energy has been transmitted through the sample, and continuous background spectra with strong and weak characteristics peaks were obtained for target material at 20 and 3 keV, respectively. Some peaks of calcium (Ca), sulfur (S), potassium (K), iron (Fe), titanium (Ti), copper (Cu), thulium (Tm), cobalt (Co), zinc (Zn) were also observed in the control sample. The presence of these elements may be due to the nutrient part of the cotton or from the environment. The result obtained for both nano-coated samples (S1 and S2) shows that the intensity of background, as well as peaks of target material, and the element present in the control be suppressed significantly. Whereas, an intense peak of the element of nanomaterial is recorded. This is due to the interaction of X-ray with coated nanomaterial.

X-ray fluorescence analysis (XRF): (a) XRF for S1 (TiO2 nanoparticles coated), (b) XRF for S2 (ZnO nanoparticles coated), and (c) XRF for S (non-coated).
The data collected for element percentage composition is displayed in Table 1. The data represented in the table clearly shows the high percentage of “Ti” in sample “S1,” and a high percentage of “Zn” in sample “S2.” This is due to the coating of TiO2 and ZnO in samples “S1,” and “S2” respectively. Table 2 describes different nanomaterials used in fibers and their purpose. The present work was carried out for nano-based analysis of cotton fabric which differentiates them from other reported purposes of the nanomaterials incorporated for fibers.
Percentage of elements in different samples (S, S1, and S2).

S: control cotton sample; S1: TiO2 nanoparticle coated cotton sample; S2: ZnO nanoparticle coated cotton sample.
Comparison table for reported literature of nanomaterials in textile modification and presented work.
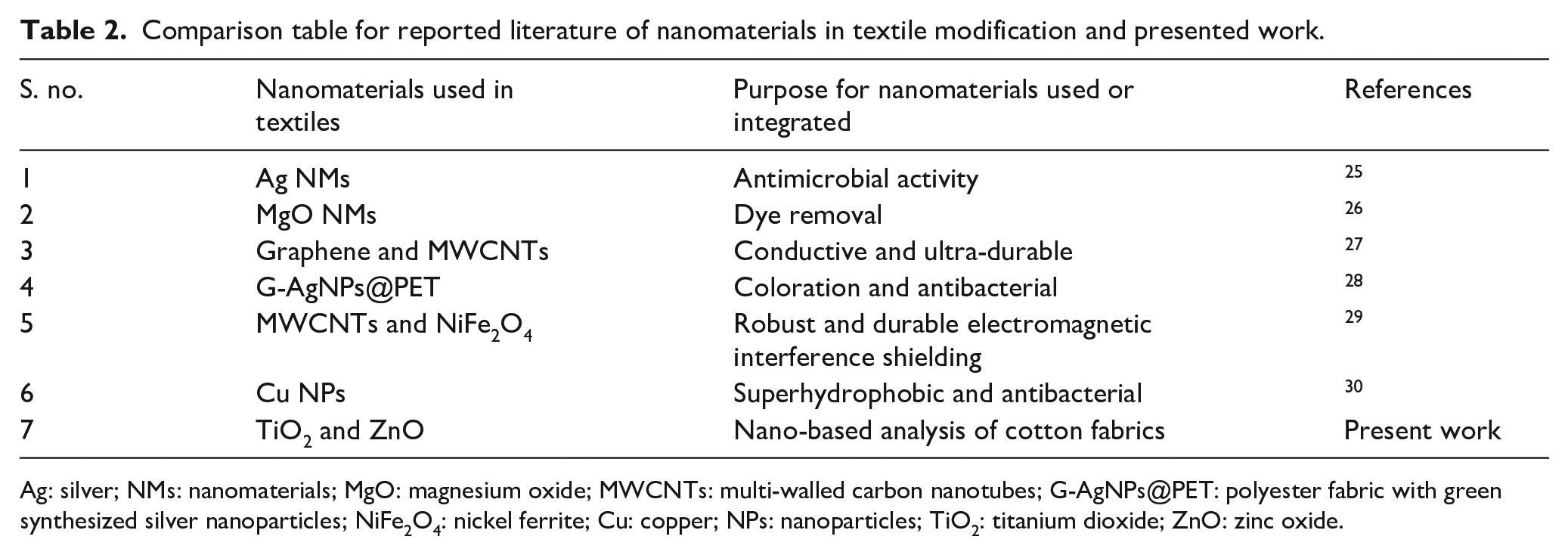
Ag: silver; NMs: nanomaterials; MgO: magnesium oxide; MWCNTs: multi-walled carbon nanotubes; G-AgNPs@PET: polyester fabric with green synthesized silver nanoparticles; NiFe2O4: nickel ferrite; Cu: copper; NPs: nanoparticles; TiO2: titanium dioxide; ZnO: zinc oxide.
Conclusion
In order to differentiate surface modification of cotton fiber for wider analysis, a comparative study for two nanomaterials; TiO2 and ZnO coated on the surface of cotton fibers were performed. Samples were characterized by using techniques like SEM, EDX, UV, FTIR, XRF, and magnification. It was observed that the samples can be easily differentiated using UV light at wavelength 365 nm. This technique has various advantages over other methods such as low cost, user-friendly, and non-destructiveness. Since the fibers were previously made up of organic materials only, thus, it was difficult to obtain XRF data. The present study indicates that the UV and XRF analysis together can provide quality information about the nano-coated sample. Besides, the results obtained from FTIR samples also indicate that the peaks can be significantly determined by modified fabric samples coating with TiO2 and ZnO nanoparticles exhibiting functional groups present on the surface, helping in differentiating them clearly. This study may immensely facilitate in retrieving the information of fibers or fabrics samples for wider analytical applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
