Abstract
Nanotechnology modifications play a major role in textile industry due to extraordinary properties exhibit in fabrics due to nanomaterials. It offers different functionalities namely self-cleaning, wrinkle resistance, flame retardancy, protection from UV radiations or antibacterial property. Further, it is important to maintain cleanliness in aircraft upholstery always but the airliners have to bear a considerable amount of money to clean up the aircraft upholstery. Interestingly, nanotechnology can provide stain repellence property for fabrics in aircraft upholstery. This study covers a method of developing a stain repellent fabric which was stable even after 25 standard laundering conditions. In this study nanotechnology was used to modify a cotton fabric with stain repellant property. After nanotechnology modification, the surface wettability of the treated fabrics was characterized by static water contact angle measurements before and after 25 washes carried out under standard laundering conditions. Similar testings were carried out for the untreated fabric samples. The static water contact angle for the treated fabric was 161° with the recovery of 97.5% after 25 washing cycles. SEM and AFM micrographs were used to analyze the coatings. Further, the stability of hydrophobicity in the modified cotton fabric after 25 washes was also tested for tea, coffee, and water solutions which are vulnerable stain types in aircraft upholstery. It clearly proved that the modified cotton fabric even after 25 washes showed hydrophobicity for tea, coffee, and water. Therefore, it could be concluded that the developed modified cotton fabric can consider to be used as an aircraft upholstery.
Introduction
Researchers have been working to mimic the biological surfaces such as lotus leaves, butterfly wings, mosquito eyes, moth eyes, red rose petals, gecko feet, desert beetle, and spider silks which exhibit excellent hydrophobicity.1–5
The surfaces with hydrophobic and/or super hydrophobic properties have received a great scientific and industrial attention because of their wide range of applications.4,6 Hydrophobic or super hydrophobic surfaces can be made by chemically treated methods to manufacture products in various domains, namely in automotive, aerospace, marine applications, and textiles. Super hydrophobic surfaces bring in tremendous enhancement in the functional performances compare with their conventional counterparts. 6 The existence of contamination often affects the efficiency and safety of transportation. Therefore, self-cleaning is an important and interesting property in transportation sector. 7 Hydrophobic fabrics have also self-cleaning properties which can be used in upholstery, usual cloths, mattresses, and also in sport cloths. 8
As per the statistics released in 2018 by the International Air Transport Association (IATA) the estimate air passenger numbers could hit 8.2 billion per year by 2037. 9 Increased demand for capacity means, there should be more aircraft and necessary improvements to the ecosystems to keep those aircraft flying safely. However, corrosion is a constant threat to the structural integrity of aircraft occurred mainly due to saltwater air. Meanwhile, inside the cabin, spills of coffee and soft drinks can damage to the interior of the aircraft. In order to maintain the proper standards of the aircraft it must undergo regular inspections which airlines have to spend a huge sum for maintenance. There are several research articles published related to super hydrophobic nanomaterials on aircraft surfaces, air conditioner, and refrigerator tubes which lead to have a freeze less surface when in touch with moisture. 8 In addition, the literature,10–13 highlighted the super hydrophobicity in aircraft paints and varnishes, pipe coating, and fabric. However, there wasn’t any published literature related to the hydrophobic properties of aircraft upholstery fabric materials which is found in the carpeting, wall fabric, and seat covers on an aircraft.
The two main requirements to create an excellent super hydrophobic surface are surface roughness and low surface energy. 7 Several technologies such as E-beam lithographic, etching, stretching, spin coating, plasma deposition can be used to develop hydrophobic surfaces. 7 In addition, electrospinning can also be used to manufacture nanofibers with coatings from a charged solution.14,15 Furthermore, 16 nanotechnologies also can be used to produce super hydrophobic or hydrophobic surfaces.
Sol-gel technique has been applied to the fabrication of superhydrophobic surfaces on fabrics, 17 fabrics are vulnerable to have stains of tea, coffee, and wine.18,19
In sol-gel method, hydrolysis reaction of the precursor material is carried out to form a nano-colloidal solution in order to provide preferable durability of stain repellency for the fabric which was treated by TiO2 sol. The precursors are often based on metal organic compounds such as titanium (IV) isopropoxide Ti(OC3H7)4. 20 Introducing of crosslinking groups are advantageous in order to improve the binding sites. 21 This will reduce washing cleaning cycles as durability of such highly hydrophobic fabrics. This will lead to reduce cost for cleaning and as well as the cost for labor. The binding group can be reacted with cellulose which is available on fabric surface. The application of fluorocarbon finishes on fabrics to impart stain repellency has shown considerable growth during last decade. However, the use of fluorocarbon on fabric is also a major environmental concern as there are many adverse effects of it on environment. 22 There are plenty of evidences concerning possible persistence, bioaccumulation, and/or toxicity of these types of fluorocarbon chemicals in the environment. 23
In contrast to the extensive studies on fabricating superhydrophobic cellulose fabrics, only very few research mentioned about the performance of repellent property even after cleaning. Polycarboxylic acids (PA) are one of the multi-functional organic molecules with chemical and thermal stability. It could make ester linkage with hydroxyl groups of cellulose at an elevated temperature on fabric. 24 It is known that carboxylic acids could be esterified with cellulose –OH, and also could co-condense with Ti-OH groups in the nano sol solution which leads to the formation of interfacial ester bonds. 25
In this method non-toxic 1,2,3,4, butane tetracarboxylic acid (BTCA) was used to bind both Ti-OH and cellulose –OH to offer an alternative approach in improving durability of hydrophobicity in fabric finished by sol-gel method (Scheme 1). To lower the surface energy further stearic acid was also used as it has 18-carbon chain. Stain repellency of the modified aircraft fabric was evaluated by static water contact angle measurement. The objective of this study was to modify cotton fabric with superhydrophobic properties which provide protection for the fabric upon spillage of variety of beverages. In addition, introduction of superhydrophobic fabric in cotton fabric will lead to provide self-cleaning property which save huge amount of cost for regular cleaning and also the cost to purchase new fabrics.

Chemical binding mechanism of cotton fabric.
Methodology
A piece of fabric was considered as a control sample and other three fabrics were treated with the binder as explained in section 2.2. After the treatment with the binder only two fabric pieces were treated with TiO2 sol as discussed in section 2.3. The left behind fabric piece was characterized to verify the formation of esteric bonds. After treating with TiO2 sol, only one fabric piece was further modified with stearic acid in order to further improve the hydrophobicity as explained in section 2.4. All the experiments were repeated twice.
Materials
Four fabric pieces which are being used as cotton fabrics having the dimensions of 5 cm × 5 cm were used for the experiments. These fabrics were obtained after scouring and bleaching process. These fabrics had 1 × 1 rib structure with a weight of 196 g/m2.Titanium isopropoxide (AR grade purity > 99.9%, from Sigma-Aldrich Co, USA), ethanol (AR grade purity > 99.9% from Heyman UK), acetic acid (AR grade purity > 99.7% from Sigma-Aldrich Co, USA), 1,2,3,4-butane tetra carboxylic acid (BTCA) (1% w/w) (assay 99%, Sigma Aldrich Co, USA), sodium hypophosphite monohydrate (NaH2PO2) (0.6% w/w) (Sigma Aldrich Co, USA), stearic acid (Sigma-Aldrich Co, Germany), and acetone (AR grade purity > 99.5%, Sigma-Aldrich Co, USA) were used without further purification. Deionized water was used for all the experiments.
Esterification of cellulose fabric with BTCA as the binder
Two cotton fabric pieces were immersed in aqueous solution of 1,2,3,4-butane tetra carboxylic acid (BTCA) (1%, w/w) with NaH2PO2 as catalyst (0.6%, w/w) 16 for 10 min at room temperature and then padded with a wet pickup of 70%–80%. The sample was dried at 80 °C for 5 min in an oven followed by curing at 170°C for 2 min in an oven.
Preparation and grafting of TiO2 sol
Titanium-IV tetraisopropoxide Ti(O-iPr)4 (TIP), ethanol, and acetic acid were maintained in a molar ratio of 1:100:0.05 respectively. Ti(O-iPr)4 was hydrolyzed using acetic acid at room temperature. To the prepared solution, the required amount of ethanol was added drop by drop under vigorous stirring for 2 h and the stirring was continued until a clear solution of TiO2 nano-crystals were formed.26,27 BTCA treated two fabric pieces were washed once using water to remove excess BTCA followed by immersed in the TiO2 sol solution for 10 min and then padded with a wet pickup of 70%–80%. The samples were dried at 90°C for 3 min and cured at 120°C for 1 h. 24 TiO2 sol was coated on the remaining fabric piece to compare the hydrophobic property with the fabric which were treated by both BTCA and TiO2 sol.
Surface energy lowering of the treated fabric with stearic acid
To lower the surface energy of the fabric, stearic acid was used individually. One of the fabric pieces which was treated by BTCA and TiO2 sol was immersed in a stearic acid (0.5%, w/w) solution of acetone for 10 min, then padded, and cured at 110°C for 1 h. 28
Characterization
Characterization for the synthesized TiO2 sol
The nano particles were characterized using the following technique:
Particle Size Analyzer (PSA) – Particle sizes of the titania sols prepared were measured using three samples with the particle size analyzer (Mastersizer 2000, Malvern Instruments Ltd, UK).
Characterization for untreated and treated cotton fabrics
Fourier Transform Spectroscopy (FTIR) – An untreated and treated cotton fabrics were analyzed using a FTIR (Vertex model 80, Bruker). The diffuse reflectance mode was used and the transmittance spectra were recorded over the range 4000–600 cm−1. The resolution was 4 cm−1 and the number of scans was 64 for each spectrum.
Scanning Electron Microscopy (SEM) – A small piece of untreated and treated cotton fabrics before and after 25 washes were viewed at high magnification using SEM (Hitachi SU 6600, Japan). Applied voltage was 5.0 kV and used secondary electron mode for the analysis.
Atomic Force Microscopy (AFM) – An untreated and treated cotton fabrics before and after 25 washes were characterized using Atomic Force Microscope (AFM), Park system. Images were obtained using an XE-100 microscope. The measurements were taken in air at room temperature using non-contact mode, with Si tips of the 1650-00 type scanning at frequencies 0.5 Hz.
Evaluation of hydrophobicity for the modified cotton fabric
The wettability of fabric samples is mostly characterized by static water contact angle (SWC). Static contact angles were evaluated for untreated and treated cotton fabric pieces before and after 25 washes
At room temperature, a 10 µl droplet of deionized water was placed in to five different positions on the modified cotton fabric surface using a micro pipette. The polynomial line fitted on the water bubble and tangent line plotted at the intersection point to calculate the static water contact angle (θ) as shown in Figure 1. The average static contact angle value was calculated based on the average of five measurements.

Side view of a drop of water laid on modified cotton fabric.
Evaluation of retainability of hydrophobicity during washing in the modified cotton fabric
The modified cotton fabric pieces were washed separately under standard laundering conditions. 29 The samples were cut in to a specific size (4 inch × 4 inch) and put into a beaker. Four hundred milliliter of distilled water was added with 2 g of detergent into the beaker. The samples were subjected to stirring at 40°C for 30 min. Each test specimen was rinsed three times in beaker using water for 1 min by hand squeezing. The specimens were dried in an oven at 70°C and then fabrics were conditioned at standard humidity and temperature for at least 1 h before the evaluation of properties. Washing was done 25 times for each fabric piece as per the standards for cotton fabric materials. 30
Results and discussion
Characterization of TiO2 sol
The TiO2 nano sol particle size distributions have been determined using three samples from the particle size analyzer. The Z-average values of the samples are found to be 0.3444 and 33.43 nm respectively as shown in Figure 2.

Average particle size distribution of TiO2 sol.
Characterization of untreated and treated cotton fabrics
The structure of untreated cotton fabric (control) and treated cotton fabrics were confirmed by FTIR analysis. The FTIR analysis was done for the fabric after treating with BTCA. BTCA treated fabric was washed once in order to remove excess BTCA, which were deposited on fabric. The carboxylic groups in BTCA and the hydroxyl groups of the fabric surface results in ester bonds. The free carboxylic acid groups available in BTCA, attached with TiO2 sol after treating with TiO2 sol. The untreated cotton fabric was compared with modified cotton fabric.
The transmittance spectrum (Figure 3(a)) of untreated fabric exhibit O-H stretching at around 3434 cm−1. After modification with BTCA according to Figure 3(b), the transmittance spectrum exhibit O-H stretching absorption at around 3412 cm−1 and C-H stretching vibration at 2903 cm−1.

FTIR spectra for untreated and treated cotton fabric: (a) an untreated cotton fabric, (b) BTCA grafted cotton fabric, (c) BTCA grafted cotton fabric treated with 0.1 M NaOH, (d) BTCA and 0.1 M NaOH grafted cotton fabric after treated with 1 M HCl, and (e) BTCA, TiO2 sol, and stearic acid treated cotton fabric.
The method of determination of ester binded fabric, employing infrared spectroscopy FTIR, was developed by Yang. 31 Carbonyl groups on cotton fabric treated with polycarboxylic acids are retained in three forms: ester, carboxyl acid, and carboxylate anion. If the polycarboxylic acid contains three or more carboxyl groups all the three forms could be appear in a single molecule. 32 The ester carbonyl and carboxyl carbonyl bands overlap in the spectra of the BTCA treated fabric and the both peaks occurs in the range between 1690 and 1760 cm−1. In Figure 3(b), band at 1719 cm−1 represent both ester carbonyl and carboxyl carbonyl bands. The standard procedure is to treat the cotton fabric with 0.1 M NaOH solution, to convert all the free carboxyl groups in the fabric to carboxylate. After treating with 0.1 M NaOH as shown in Figure 3(c), the band at 1718 cm−1 represents the carbonyl of ester, while the band at 1576 cm−1 represents the carbonyl of carboxylate, the basic form of the free carboxylic acid. According to the literature,33–37 after treating with 0.1 M NaOH two carbonyl bands at 1725 and 1552 cm−1 appear in the spectrum.
According to the Figure 3(c), 0.1 M NaOH treated cotton fabric shows a band at 1576 cm−1 which represents the carbonyl of carboxylate which is basic form of the free carboxylic acid available in the coated BTCA of the fabric. The fabric which was treated by 0.1 M NaOH again re-treated with 1 M HCl, the carbonyl of carboxylate groups converted to carboxylic acid groups and the peak at 1576 cm−1 disappear and only the peak at 1733 cm−1 appears as shown in Figure 3(d).
Scheme 1 illustrates the estimated chemical binding mechanism of modified cotton fabric which was drawn by ChemDraw software version 15. Even though TiO2 sol bind to BTCA as shown in Scheme 1, there is no band can be clearly associated to the Ti-OH group in FTIR spectra. 38
As illustrates in Scheme 1, untreated aircraft upholstery cotton fabric contains O-H bonds which has bonding capability with BTCA, 39 in the presence of NaH2PO2 as shown in step 1. In step 2, nano sol solution of TiO2 was introduced and it was reacted with OH groups in the carboxyl acid, 40 which substitute by BTCA to the cotton fabric. In step 3, stearic acid was dissolved in acetone and it was allowed to react with the product from step 2, which consist with unreacted OH groups available in TiO2 sol solution. 41 As stearic acid consists with many hydrocarbon groups it assists to improve the hydrophobicity of the cotton fabric, which indicates as the end product of step 3 in the Scheme 1.
Surface wettability of treated and untreated cotton fabric
Surface wettability was examined by measuring the static contact angle. The untreated fabric absorbs water droplets quickly (within approximately 2 s) due to hydrophilicity and the capillary effect of the fabric. Therefore, the untreated fabric completely wetted with water, which is common and well known for cotton fabric. 28 The fabric treated only with TiO2 sol showed poor water repellency, while the cotton fabric treated with BTCA, TiO2 sol, and stearic acid showed super hydrophobicity even for 25 washes. Five measurements were taken for each fabric and average value was determined for static contact angle.
According to the data shown in Table 1, BTCA, TiO2 sol, and stearic acid coated fabric showed highest static contact angle without washing and even after 25 washes this modified fabric showed highest static contact angle. Untreated fabric and only BTCA coated fabric showed hydrophilicity all the time, while only stearic acid and only TiO2 sol treated fabrics showed hydrophobicity without washing and completely hydrophilicity after 25 washes. This may be due to wash off of coated TiO2 sol and stearic acid. In the system having both BTCA and TiO2 sol showed hydrophobicity without washing and even after 25 washes hydrophobicity was observed. In the presence of BTCA binder TiO2 sol cotton fabric showed super hydrophobicity even after 25 washes.
Static water contact angles on untreated and treated cotton fabric.

Effect of BTCA and stearic acid on washing durability of cotton fabric
It is clearly seen from the results that the static water contact angles were significantly discrepancies of hydrophobicity between the ester-bridged fabric and TiO2 sol treated fabric. Free carboxylic acid groups on BTCA and stearic acid were proposed to have strengthened the stability of TiO2 sol coating, and ultimately the durability of the hydrophobicity provided by esterification. 1,2,3,4-Butane tetracarboxylic acid (BTCA) attracted most attention for washing durability. BTCA and stearic acid treated fabric particularly exhibited a high static contact angle of 161° with the recovery of 97.5% even after washing, presenting quite promising durability. Further, it is clear that when there are more interaction sites on the surface in TiO2 sol treated cotton fabric, the more carboxylic acid groups in polycarboxylic acid molecules can be blinded. Therefore, when there are more interaction sites it is more difficult to break the ester-bridge on the surface of the modified fabric which helps to withstand for better laundering ability.
Morphology of untreated and treated cotton fabrics
Typical longitudinal fibril structure with natural veins, which was clearly observed on untreated fabric (Figure 4(a)), disappeared after coated with TiO2 sol as shown in Figure 4(b). The vanished characteristic parallel ridges and grooves could be due to the weakly cross-linked condensation layer formed by TiO2 sol hydrolyzed under acidic conditions. 42 Also, there is no depositions could observe in Figure 4(b), which showed the absence of formation of TiO2 nano particles. After 25 washes a cluster of broken fragments could be clearly observed on fibers (Figure 4 (c)), which implied that the TiO2 sol coating was cracked after washing as there was no BTCA binder. As seen from Figure 4(c), the thickness of the TiO2 sol coating is approximately 1 µm.

SEM morphology of cotton fabrics: (a) an untreated cotton fabric, (b) treated cotton fabric with TiO2 sol only, (c) TiO2 sol grafted cotton fabric after 25 washes, (d) BTCA, TiO2 sol, and stearic acid treated cotton fabric without washing, and (e) BTCA, TiO2 sol, and stearic acid treated cotton fabric after 25 washes.
In the BTCA, TiO2 sol and stearic acid treated fabric showed no depositions on the cotton fabric surface after 25 washes as shown in Figure 4(e), while there were several depositions observed on the fabric surface before washing as shown in Figure 4(d).
Nevertheless, the SEM cannot provide clear explanation about why the BTCA, TiO2 sol, and stearic acid treated fabric is stable upon water interactions. 43
AFM analysis were conducted to the fabric samples to correlate the super hydrophobic behavior of the fabric samples with the surface roughness. Similar study was found in the literature, 43 as well.
According to the 3D image shown in Figure 5(a), riggers were clearly visible in untreated cotton fabric, where as in Figure 5(b) shown depositions of TiO2 sol and stearic acid which were responsible for hydrophobicity. The Figure 5(c) shows after 25 washes the degree of depositions were reduced but still deposition could be observed on the surface of the fabric. This leads to give further, hydrophobicity on the cotton fabric by reducing surface energy.
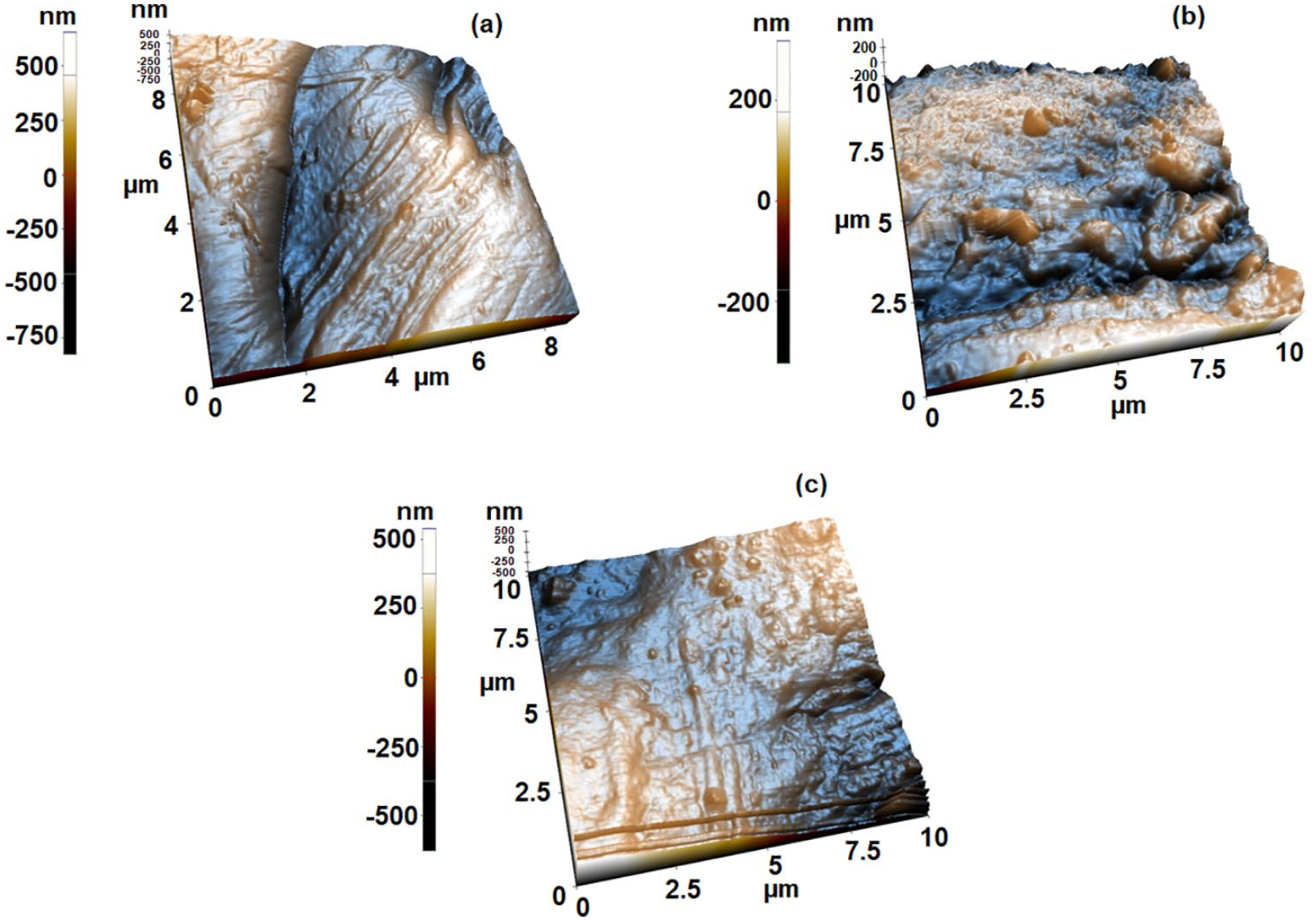
3D AFM topography images of untreated and treated cotton fabrics: (a) an untreated cotton fabric, (b) BTCA, TiO2 sol, and stearic acid treated cotton fabric without washing, and (c) BTCA, TiO2 sol, and stearic acid treated cotton fabric after 25 washes.
Based on the AFM results shown in Figure 5, it can be observed that the surface porosity is also improved in the wetting properties of the surface.
Evaluate the stain repellency of the modified cotton fabric
Generally, tea, coffee, and water can be identified as main water based drinks found in aircraft during a flight. Hence, aircraft upholstery is highly vulnerable for tea, coffee, and water stains. Figure 6, illustrates tea, coffee, and water liquid drops setting on the modified cotton fabric after 25 washes. Further as per literature,44,45 there are evidences that cotton has been used in aircraft applications.

Side views of the droplets of (a) tea, (b) coffee, and (c) water laid on modified cotton fabric.
A tea bag contained 5.00 g of powdered tea was added to 5 ml of boiled hot water in a beaker. Five gram of coffee powder was also added to a separate beaker consist of 5 ml of boiled hot water. In addition, 5 ml of boiled hot water was added to another beaker as a control solution. All the solutions were allowed to cool and when the temperature reached to the room temperature, all the solutions were filtered. Ten microliter of droplet from each solution was placed on three modified cotton fabric having the dimension of 10 cm × 10 cm using a micro pipette. Five droplets were placed at a time from the same solution on a modified cotton fabric piece. The rest two fabrics were also added with the five droplets from each solution respectively. Static water contact angles were measured and calculate the average static water angle for each solution. The results are given in Table 2.
Average static water contact angles of treated cotton fabric pieces after 25 washes for tea, coffee, and water solutions.

As shown in Figure 6 and based on the average static water contact angle values given in Table 2, it is evident that the modified cotton fabric after 25 washes showed hydrophobicity for tea, coffee, and water which are vulnerable stain types in an aircraft upholstery. The stability of hydrophobicity can be confirmed as this experiment was carried out 25 washes.
Conclusions
In this work, BTCA was used as the binder to improve the washing durability of cotton fabric which was achieved hydrophobicity by sol-gel technology combining TiO2 sol and stearic acid. The results showed that the BTCA binder and stearic acid treated cotton fabric could effectively improve the washing durability of cotton fabric, while TiO2 sol only and stearic acid only coated cotton fabrics showed weak hydrophobicity without washing and complete hydrophilicity after 25 washes. Untreated cotton and only BTCA coated cotton fabrics showed no hydrophobicity at all. The BTCA and TiO2 sol coated cotton fabric showed hydrophobicity without washing and hydrophilicity after 25 washes. The BTCA, TiO2 sol and stearic acid treated cotton fabric was durable for laundering at a recovery percentage of static contact angle of 97.5%. This study demonstrated that the surface treatment using BTCA and stearic acid to TiO2 sol is a promising alternative for achieving durable hydrophobic in cotton fabrics. The stability of hydrophobicity in the modified cotton fabric after 25 washes was also tested with tea, coffee, and water solutions which are vulnerable stain types in aircraft upholstery. It clearly proved that the modified cotton fabric even after 25 washes showed hydrophobicity for tea, coffee, and water. Therefore, it can be concluded the developed modified cotton fabric can consider to be used as an aircraft upholstery.
Footnotes
Acknowledgements
The author would like to express her gratitude to Sri Lanka Institute of Nanotechnology, Pitipana, Homagama, Sri Lanka for supporting to characterize all the samples.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
