Abstract
This article introduce a novel, convenient and efficient finishing approach for the creation of quaternary ammonium groups (QAGs) into polyamide-6 (PA-6) macromolecules via polymerization of vinyl monomer is named methacryloyloxyethyl trimethyl ammonium chloride (MAETAC). The graft copolymerization reaction proceeds in the presence of nano-metal oxides TiO2, ZnO and its hybrid mixture. The finishing method of PA-6 fabrics was carried out by using pad-dry-cure technique in presence K2S2O8 as initiator. The reaction mechanism was suggested. The surface topography was studied using Scan Electron Microscope (SEM) and Energy dispersive X-Ray (EDX). Evidence the chemical bonding between poly (MAETAC) and amide groups as well as the ionic interaction between nanoparticles (NPs) and carboxylic end groups in the chemical structure of the PA-6 fabrics were confirmed by FT-IR. The antimicrobial activity of the finished fabrics were evaluated against Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans. The achieved antimicrobial functions on the PA-6 fabrics are durable in repeated laundering processes. Even after laundering five times the PA-6 fabrics treated with QAs/Ti NPs could still provide 32%, 80.7%, 22.6%, and 60.5%, while PA fabrics treated with QAs/Zn or QAs/(Ti+Zn) showed excellent antimicrobial activity against Staphylococcus aureus, E. coli, P. aeruginosa, and C. albicans before laundering, respectively. Also, it has been found that, PA-6 fabrics finished with the suggested method even after five washing cycles, displayed excellent ultraviolet protection effectiveness. The results of mechanical properties showed that this finishing approach had not destructive effect on the physical properties of the fabrics.
Introduction
Improving current characteristics and preparing new materials with high add values are the main factors for textile functionalization.1–4 The modification of fibers with quaternary ammonium compounds (QACs)5–10 is one of the efficient ways to achieve antimicrobial fibers and textiles. Cationic surface active agents (cationic surfactants), including especially quaternary ammonium salts (QASs), are significant biocides known to be efficient antiseptic and disinfectant substances for many years.11–13
Despite many beneficial features, there is an fundamental weak point in applying QASs as a finishing agent: leaching from the fibers. The QAS structure does not contain any active functional groups to enable its chemical bonding to the fibers. Leaching of the QAS happens due to the weakness of physical bonding, leading in a rapid decline in concentration below the minimum inhibitory concentration (MIC), the smallest chemical concentration, generally an antimicrobial agent that prevents noticeable microbial growth. Furthermore, the durability of QASs against washing is poor. Many studies proposed new techniques of incorporating QAS into PA structure.5–10
Many ways have been reported to finish textiles using QAS to endow them antimicrobial activity.14,15 Ionic interactions between reactive groups on the polymers and QAS can be utilized in the functional finishing of fabrics. Immobilization of QACs on PA-6 knitted fabrics based on reaction with carboxylic end groups in polymer macromolecules has been studied. Son and Sun, 16 were employed these ionic interactions to accomplish required long-lasting antimicrobial features in the chemical finishing of PA fabrics. Shalaby et al., 17 used partial acid hydrolysis for pretreatment of PA-6 fibers, then, QAC definitely DMABAC was immobilized on the surface of the hydrolyzed fibers. The salt structure has been formed (—NH3+. . . .¯OOC—) between the PA-6 hydrolyzed surfaces and the QAC.
Some scientists used polymerizable vinyl monomers comprising quaternary ammonium with groups of acrylate or methacrylate to impart antimicrobial activity to PA fabrics.18–20 Such monomers of vinyl were called surfactants or surfmers. These compounds have the capacity to polymerize into a polymer network under particular response circumstances with a polycationic framework, along with side QA groups chemically linked to the primary polyacrylate chain. The advantage of fixed binding to the surface of the PA is that the QAGs can behave as a bio-barrier and by contact kill microorganisms. In addition, the formation of a polymer network on the surface of the fibers significantly improves the antimicrobial agent’s durability and washes strength.
Nano-metal oxides containing polyQAS offer the opportunity to develop a unique class of textile nano-finishing products. However, owing to bad fixation of these NPs on the textile surface, the nano-metal oxides finish does not resist subsequent washing. Enhancing the bonding of NPs with textile surfaces not only improves the durability property, but also offers an environmentally friendly consideration to prevent the release of unbound NPs into the environment. 20
This paper revealed a novel, convenient and effective finishing strategy, involving covalently grafting chains of a polyvinyl quaternary ammonium salt (PVQAS) monomer is named methacryloyloxyethyl trimethyl ammonium chloride (MAETAC) onto polyamide-6 fabrics in presence nano-metal oxides (TiO2 or ZnO and TiO2/ZnO mixture). The grafting reaction assisted by K2S2O8 as initiator. The PA-6 fabrics were finished with Poly VQAS in order to improve their antimicrobial activity against different micro-organism classes such as bacteria and fungi. To provide more functional finishing characteristics for PA-6 fabrics such as UV protection, and strengthening their antimicrobial activity the PA-6 fabrics were finished with aqueous solutions of organic-inorganic hybrid mixture from nano-metal oxides containing VQAS. This research defines the preparation, implementation of the above method as a novel approach to finishing and characterization of appropriate TiO2 or ZnO NPs and their combination with PVQAS for PA fabrics.
Experimental work
Materials
Polyamide-6 fabric used throughout this study was in the form of knitted fabric cloth made from filament yarns (warp 42 ends/cm, weft 36 picks/cm) which was kindly supplied by El-Nasr (Shourbagy) Co., Cairo, Egypt.
Chemicals
Vinyl quaternary ammonium salt named methacryloyloxyethyl trimethyl ammonium chloride (MAETAC), its concentration is 65% in aqueous solution b, brouchesed from Sigma-Aldrich. MAETAC was of pure grade and used without further treatment.

TiO2 ~ 21 nano-powders (Sigma-Aldrich)
ZnO ~ 50 nano-powders (Sigma-Aldrich)
The initiator employed in this study was Potas-sium persulphate (K2S2O8), used without further purification.
All chemical used in this study were analytical grade chemicals.
Microorganisms and growth conditions
The antimicrobial potency of finished fabrics was tested using:
One Gram-positive bacteria (Staphylococcus aureus NRRL B-767);
Two Gram-negative bacteria (Escherichia coli ATCC 25955, and Pseudomonas aeruginosa ATCC 10145); and
One yeast, Candida albicans ATCC 10231.
Bacterial and yeast strains were cultured overnight at 28°C to 30°C in nutrient broth (NB) and potato dextrose broth (PDB), respectively, These cultures were used as inoculums in antimicrobial test.
The finishing approach
Finishing of PA-6 with MAETAC
The PA-6 fabrics (20 × 20 cm2) were padded at room temperature with completely soluble padding solution of MAETAC (10 %) containing 2.0 g/L of K2S2O8. The fabrics were then removed from the pad bath, squeezed to 80 % (wt/wt) wet pick-up using a laboratory padder (ROACHES, UK), and dried at 80°C for 3.0 min in an oven (ROACHES, UK). Then, the fabrics were cured at 120°C for 5.0 min. After the elapsed reaction time, the treated fabrics were cooled to room temperature in a dessicator until attaining a constant weight. The dried samples were washed five times with hot water to remove the homopolymer of poly methacryloyloxyethyltrimethyl ammonium chloride (PMAETAC). The finished PA-6 fabrics dried in an oven at 30°C for 24 h, cooled again as previously indicated and weighed.
Finishing of polyamide-6 with the hybrid mixture
The treatment of PA-6 fabrics with TiO2, ZnO NPs and its mixture was carried out according to the same above mentioned method and by adding 1.0 g/L NPs to the finishing medium.
Analysis
SEM and EDX
A JEOL-Model JSM T20 scanning electron microscope (SEM) operating at 19 kV was used to obtain photomicrographs of fibers surfaces.
FT-IR
The chemical structure was determined using the Fourier transformation infrared (FT-IR) spectrometer, model NEXUS 670, NICOLET USA. The measurements were carried in spectral range from 4000 to 500 cm−1. Reflection percentage measurement technique was applied (R%) to all investigated samples.
Antimicrobial assay
Turbidity method
The antimicrobial activity of finished fabrics samples was determined by turbidity method according to the method described in Rama.21,22 Briefly, 30 µL of the tested microorganism (106 colony-forming units (CFU)/mL) were inoculated into 3.0 mL of NB or PDB medium in standard test tubes containing tested textile (2 × 2 cm2). Test tubes were incubated at 30°C for 24 h under shaking conditions. Antimicrobial activity was evaluated by measuring the Optical density (OD) at 600 nm and comparing to the blank sample.
AATCC100 method
The antimicrobial activity of the finished fabrics samples was determined according to the standard test method AATCC100 23 with some modifications. Samples (1 × 1 cm2) were placed in test sterile glass tubes and moistened with 100 µL sterile phosphate buffer. Overnight grown tested cultures were used for inoculation of the fiber then incubated at 30°C for 24 h. Tenfold serial dilutions of each sample were prepared and were spread onto nutrient agar or potato dextrose agar plates (three replicates per dilution) and incubated at 28°C to 30°C for 24 h. Microbial colonies that grown on these plates were counted and compared to the control sample (blank). The reduction percentage was calculated from the following equation.
Where:
CFUU is OD at 600 or the number of colony forming units of blank and CFUT is OD at 600 or the number of colony forming units of treated samples. The data was the mean of three replica.
Washing durability of antibacterial activities
Treated PA-6 fabrics were subjected to a Launder-Ometer washing test using AATCC standard method 61-1994 to determine the washing durability of antibacterial activities. In general, one cycle of a Launder-Ometer washing using this AATCC test method is considered equivalent to five machine washes in a home laundry.
The PA samples were introduced in a glass bottle and immersed in aqueous solution containing 2.0 g/L nonionic detergent, at 71°C. The material-to-liquor ratio (M:L) was 1:50. After 45 min, the samples were rinsed repeatedly with distilled water and allowed to dry in the open air.
UPF factor
UPF factor was measured using UV-Shimadzu 3101 PC-Spectrophotometer. It is a double beam direct ratio measuring system. It consists of the photometer unit and a pc computer. UPF factor was determined according to the method described in Australian/New Zealand standard AS/NZS 4399: 1996. UPF values were calculated automatically and classified according to Table 1.
Protection and classification according to AS/NZS 4399:1966.

The mechanical properties
The finished and parent samples of PA-6 were tested for their tensile strength and elongation behaviors using a Textile Tensile Strength Tester No. 6202 – ASANO Machine MFG, Japan. The results listed in this paper are the mean of five times measurements, each sample area is 5 × 10 cm2. The measurement was proceed according to Standard Test methods for tensile properties- ASTM -D5034.
Results and discussion
Fixation of VQAs on PA-6 fabrics
The process of grafting of polyamide-6 fibers is done through the free radical mechanism which is carried out in three steps. The first step is initiation, cracking of potassium persulphate under the effect of reaction temperature and the formation of potassium sulphate radicals (equation (1)). The second step is propagation; the attack of formed radicals is either the amide groups in polyamide macromolecule and/or the double bond in VQAs (equation (2)). The third step is termination, by decay the radicals formed in the reaction medium by interacting with each other or with the growing chain of polyamide-6 grafted with polyvinyl quaternary ammonium salt or by the interaction of two growing chains from polyvinyl quaternary ammonium with one another, which is leading to the formation of a homopolymer.
The abovementioned reaction mechanism was confirmed by FT-IR analysis and through the new imparted functional properties to PA-6 fabrics washed one and five cycles according to the standard test method.
O O
║ ║ Heat
K+OSO–OSO¯ +K 2KSO4• (2R•) (1)
║ ║
O O
R• = Radical
~(CH2)5 — CON — (CH2)5 ~ + R • ~ (CH2)5 —CON• — (CH2)5 ~
(PA) PA macroradical (2)
H
R • + VQAs R— VQas•
PA macroradical + VQAs ~ (CH2)5 —CON— (CH2)5 ~
VQAs = Vinyl quaternary ammonium II
[VQAs+]2 (3)
R– VQAs• + VQAs R—[VQAs•]2
II + R • or [Poly VQAs•]m ~ (CH2)5 —CON— (CH2)5 ~
PVQAs = Polyvinyl quaternary ammonium
[VQAs+]n+m (4)
Grafted Polymer
[PVQAs•]n + [Poly VQAs•]m [PVQAs•]n+m
Homopolymer
Loading PA with nanoparticles
It is well known that, PA-6 have carboxylic end groups. These groups have the ability to react with NPs (Ti or Zn or their hybrid mixture via ionic interaction). Also, may be some of NPs encapsulated between the thin layers of PVQAs chains chemically bonded with PA-6 fabric on its surfaces.
Characterization of PA-6 fabrics loaded with ZnO NPs
In order to evaluate the NPs adhesion to the PA-6 fabrics, the treated fabrics were washed 1 and five times according to AATCC standard test method (61-1989). EDX analysis verified the existence of TiO2 and ZnO NPs on the surface of PA fabrics. Figure 1 shows EDX spectra of PA-6 fabrics loaded with TiO2 and ZnO NPs following one washing cycle. On the basis of these spectra, it is noteworthy to conclude that the deposited material consisted of Ti or Zn and O2. This demonstrates that TiO2 and ZnO are still present on the ground panel of PA fabrics even after one washing cycle (five home washings). EDX measurements also show greater levels of Ti and Zn on the functionalized PA fabrics (Ti and Zn atomic weight percentage was Ti = 11.32 and Zn = 9.97).

EDX micrographs of finished PA-6 fabrics* (3000×). (a) PA fabric blank (b) PA → PVQAs (c) PA → PVQAs → Ti NPs (d) PA → PVQAs → Zn NPs (e) PA → PVQAs → (Ti + Zn) NPs.
Figure 2 shows EDX spectra of PA-6 fabrics loaded with TiO2 and ZnO NPs following five washing cycles. This shows that even after five washing cycles (25 home washings), TiO2 and ZnO are still present on the PA fabrics surfaces (Table 2). EDX measurements also reveal higher Ti content on PA fabrics (Ti and Zn atomic weight percentage was Ti = 6.77 and Zn = 6.18). This means that TiO2 nanoparticles have sufficient adhesion toward the PA-6 fabric surface.
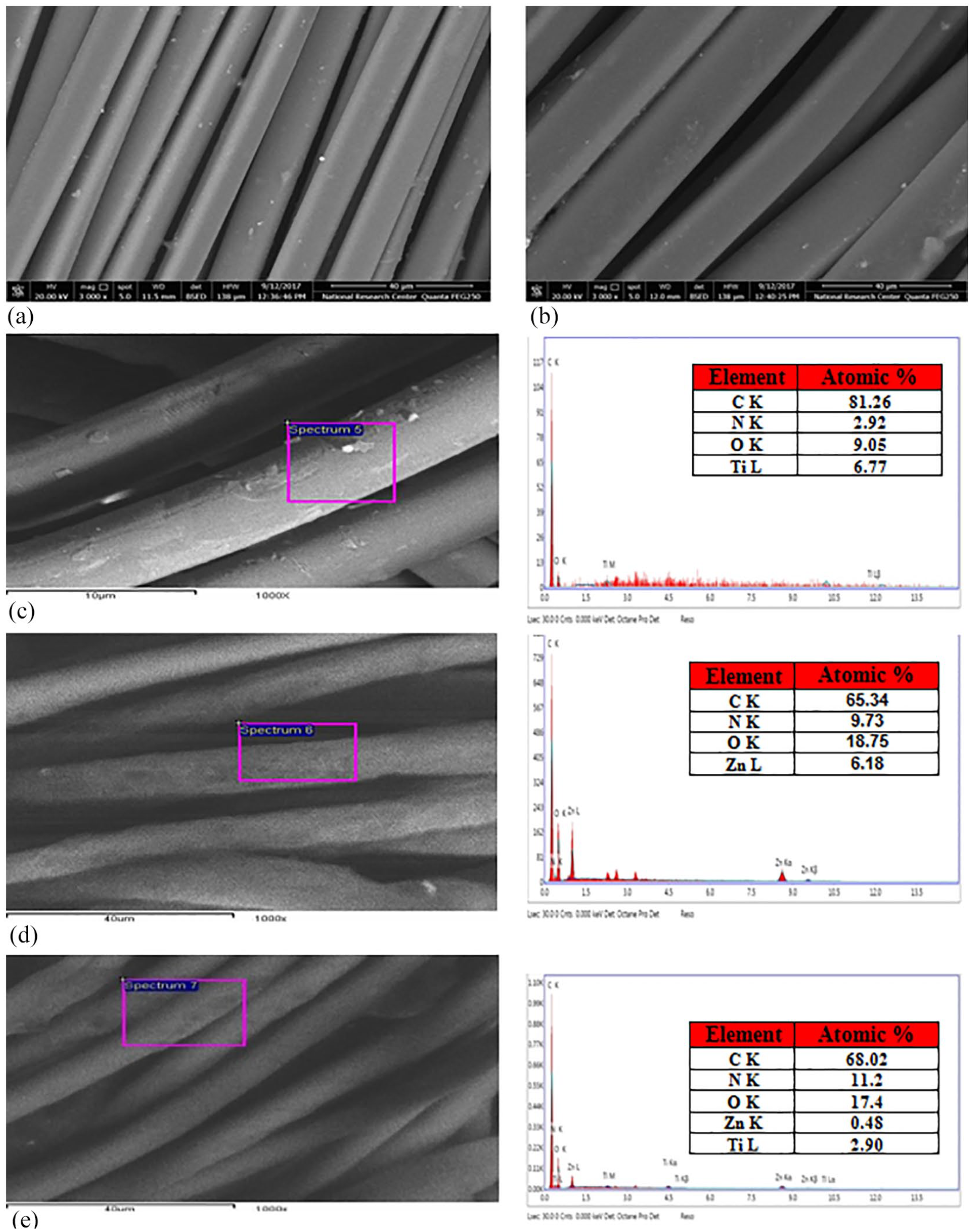
EDX micrographs of finished PA-6 fabrics* (3000×). (a) PA fabric blank (b) PA → PVQAs (c) PA → PVQAs → Ti NPs (d) PA → PVQAs → Zn NPs (e) PA → PVQAs → (Ti + Zn) NPs.
Effect of the finishing method on the amount of NPs loaded on PA-6 fabrics.
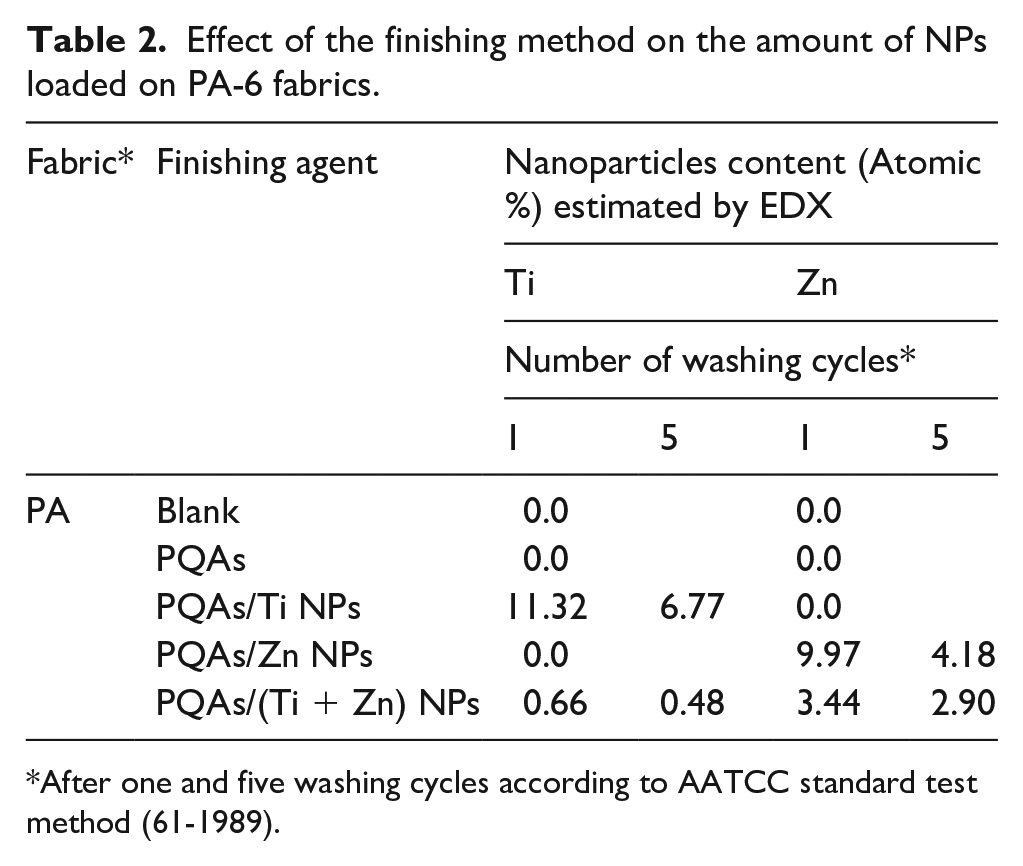
After one and five washing cycles according to AATCC standard test method (61-1989).
Similar findings were revealed when polyamide fabrics finished with hybrid blend under inquiry. This implies that NPs are sufficiently adherent to finished PA fabrics either by Ti or Zn alone or by mixing them both.
Surface topography
Scan electron microscope (SEM)
In order to investigate the morphology of the finished PA-6 fabrics by PVQAS and loaded with TiO2 or ZnO NPs and its mixture, SEM images of samples were recorded. Figure 1 shows the images of the finished fabrics followed by one washing cycle washing cycle (five home washings).
Figure 1(a) shows that the surfaces of parent PA clean and smooth. After treatment by PVQAS (Figure 1(b)) a thin layer was appeared on the surfaces of PA fabrics. The finished PA fabrics by PVQAS in presence NPs (Figure 1(c), (d) and (e)) are covered by continuous deposited materials. The size and the shape of such deposits material (like a white powder), vary according to the PA-6 fabrics used during the finishing process. We think, it is a granule of Ti and Zn as previously proven by EDX. The noted modifications that occurred on the surface topography of polyamide-6 fabrics completed with PVQAS in the presence of NPs are a direct sign that NPs are directly connected to the surfaces of the PA-6 fabric.
FT-IR
Functional groups of parent PA-6 fabric were investigated by using FT-IR spectra analysis as shown in Figure 3(a). The IR spectrum of parent PA fabric (Figure 3(a)) has a various distinctive absorption peaks.

FT-IR spectra of finished PA-6 fabrics*. (a) PA fabric blank (b) PA → PVQAs (c) PA → PVQAs → Ti NPs (d) PA → PVQAs → Zn NPs (e) PA → PVQAs → (Ti + Zn) NPs
The absorption bands in the region 3000 to 3800 cm−1 were attributed to stretching vibration modes of O—H groups. The band at 2906 cm−1 is determined by υ(C—H2) asymmetric stretching vibration. Moreover, there is an absorption band at 2267 cm−1 of O—H stretching, which associated to carboxylic end group. The peak located at 1735 cm−1 is assigned to the stretching vibration of >C=O group. The band at 1035 cm−1 is related to C—O and C—O—C stretching modes, whereas the peak appeared at 1577 is determined by amide group.
Figure 3 shows that, the bands of amide (>C=O stretching) is shifted from 1730 and to be 1660, 1666, 1664 cm−1, respectively.
The band of the amide group in PA finished with PVQAS (N—H bending) at 1565 cm−1 increases to be 1573, 1569, and 1566 cm−1, respectively.
Fabrics loaded by NPs, the bands of amide group (C—N stretching) are shifted from 1234 cm−1 to be 1241, 1272 in ZnO, TiO2 individually and to be at 1267 cm−1 when TiO2 and ZnO mixed together. Also, the band of C—O stretching at 961 cm−1 was shift to be 968, 964, and 975 cm−1 in TiO2, ZnO, and their mixture respectively.
At the same time, the intensity ratio between the amide of parent PA band to amide of PA-6 finished with PQAS and loaded with NPs band was decreased from 10.5 to 1.6, 2.5, and 1.7 for TiO2, ZnO and their mixture, respectively. Figure 2 shows, the band at 2969 cm−1 is determined by (CH3) υ(C—H3) asymmetric stretching vibration. Moreover, the bands of υ(–C-H3), υ(–C-H2), υ(>C=O) are shifted slightly.
In fact, the decrease in the intensity of the amide bands and appearance υ (C—H3) band might take place by bonding the amide group to the poly vinyl quaternary ammonium salt.
In nylon fabrics coated by TiO2 or ZnO have bands at around 701, 588, and 534 cm−1 could be attributed to Zn—O vibrational modes. In addition, the bands at around 497 to 582 cm−1 could be attributed to Ti—O band of TiO2. In another way, nylon coated by TiO2 and ZnO NPs mixture is contained the bands of both TiO2 and ZnO. We can conclude that, TiO2, ZnO and its mixture were coated nylon surface successfully similar finding was reported in Al-Balakocy et al. and Shalaby et al.24,25
Antimicrobial activity
The antimicrobial activity of PA-6 fabrics finished with PVQAS on one hand, and loaded with NPs, on the other hand, and its hybrid mixture, was investigated against Gram-positive Staphylococcus aureus, and Gram-negative, E. coli and Pseudomonas aeruginosa as well as non-filamentous fungus C. albicans. The activity by shake flask method is quantified by the measurement the optical density (OD). Table 3 indicates the antimicrobial activity of PA-6 fabrics finished with PVQAS and loaded with NPs. It is seen from the data listed is this (Table 3) that, all finished fabrics showed, after one washing cycle, a high antimicrobial activity against the previously mentioned three microorganisms. In fact, the % CFU reduction for all tested PA-6 fabrics samples is significant, whereas no dedication is found for parent PA-6 fabric.
Effect of vinyl quaternary ammonium finishing on the antimicrobial activity of PA-6 fabrics*.

After 1 and 5 washing cycles according to AATCC standard test method (61-1989).
The antibacterial and antifungal activities of the treated PA-6 textile samples may be due presence QAGs which not only attack the cell wall and cell membrane destroying its permeability and releasing the intracellular constituents (ribose, sodium, glutamate, and etc) but also interfere with membrane function, for example, electron transport chain, nutrient uptake, protein and nucleic acid synthesis, and also affect enzyme activity. The bioactive compounds might have several invasive targets that could lead to inhibition of the bacteria (Mechanisms of metal microbial toxicity). 26
Several mechanisms were proposed for the antimicrobial activity of metal ions. It was reported that the toxic effects of metals on microorganisms can be due to antioxidant depletion, DNA damage, impaired membrane function and/or interference with nutrient assimilation. Some metal ions such as Cu2+, Co2+, Zn2+, and Mn2+ increase intracellular reactive oxygen species, leads to DNA damage, membrane activity disturbance, and inhibition of particular enzyme activities that are vital for cell growth. Metal ions bind to the proteins, enzymes and other electronegative chemical groups of the cell wall and cell membrane of the microorganism causing structural changes that disrupt the permeability leading to lyses of the cells. Metal ions can access inside the bacterial cell, complexing with apoenzyme or displacing any metal ion that essential for the enzymatic action preventing protein synthesis and/or inhibiting nucleic acid synthesis. Metal catalyzed oxidation of several amino acids leading to loss of catalytic activity of the enzymes. Also, some metals activate cell autolytic enzymes leading to bacterial death (Zn2+).27,28
The function of activating PA-6 fabrics with PVQAs on antimicrobial activity in the presence of NPs appears to be more important as the samples were constantly laundered in laundry-Ometer (five washing cycles). This demonstrates the feasibility of activating PA-6 fabrics with PQAS and NPs hybrid polymer on its antimicrobial finish.
Ultraviolet protection properties
The impact of activating PA fabrics on UV protection effectiveness was explored either with PQAS or in the presence of TiO2, ZnO NPs and their mixtures. The rate of UV protection was quantified and expressed via UPF values that are given in Table 4. It was found that the UPF factors for untreated PA-6 and PA-6 finished with PQAS fabrics are equal to 12.9 and 44.5, respectively. Activation with NPs deposition on the above described PA fabrics resulted in a substantial increase in the UPF factor to the level correlating to the 50 + UPF rating, which assigns excellent UV protection after one washing cycle. These findings indicate outstanding durability of laundering of PA-6 fabrics activated with PQAS and loaded by NPs. It was discovered that PA-6 fabrics activated with PQAS and loaded with NPs showed greater effectiveness of UV protection relative to PQAS (50+) treatment, which assigns great protection against UV. These UV protection effectiveness is even greater after a washing process (five washing cycles), showing the outstanding durability of laundering.
Effect of finishing by vinyl quaternary ammonium on the UPF of PA-6 fabrics*.

After 5 washing cycles according to AATCC test method (61-1989).
The mechanical properties
The results of tensile strength and elongation for the parent and finished PA-6 fabrics were listed in Table 5. Generally, for finished fabrics the tensile strength and elongation slightly decreased it may be due to the effect of finishing with nanoparticles on PA-6 fabrics which can cause the reduction of the elongation. However, the maximum loss in tensile strength and elongation were 1.66% and 2.66%, respectively. This maximum reduction in strength and elongation is still in the acceptable range. The results of mechanical properties measurements confirm that this approach can be used on PA-6 without significant adverse effect on the physical properties of the fabrics.
Effect of finishing by vinyl quaternary ammonium on the mechanical properties of PA-6 fabrics*.

After 5 washing cycles according to AATCC test method (61-1989).
Conclusion
A novel, convenient and efficient finishing approach was reported in this article, involving covalently grafting Poly VQAS chains onto PA-6 fabrics in presence nano-metal oxides (TiO2 or ZnO and its mixture). The grafting reaction assisted by K2S2O8 as initiator. The finished fabrics with QAS/Zn and QAS/Ti+Zn exerted, after five washing cycles, a high antimicrobial activity against the pathogenic microorganisms (Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans). PA-6 fabrics triggered with PVQAs and loaded by NPs also showed a high effectiveness of UV protection relative to PVQAS (50+) treatment, which assigns great UV protection. The data revealed that, PA-6 finished by the suggested approach imparted antimicrobial activity and UV protection, even after five washing cycles. The results of mechanical properties measurements confirm that this approach can be used on PA-6 without significant adverse effect on the physical properties of the fabrics. Also, NPs containing PVQAS as a mixture are suitable for the development of textiles with excellent activities for antibacterial and ultraviolet protection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

