Abstract
Circulating tumor cells (CTCs) are cancerous cells that detach from the tumor and circulate in the bloodstream, resulting in metastasis by invading and colonizing new organs. These cells, however, pose a great significance from the clinical perspective as they can be used for the detection and prognosis of cancer. Traditionally used cancer diagnosis treatments are invasive and cannot detect tumor heterogeneity. CTCs, on the other hand, can be recovered through various non-invasive techniques such as liquid biopsies. Further, they can potentially aid in accurate diagnosis and prognosis, real-time monitoring of the body’s therapeutic response, and in providing personalized therapy. Hence, CTCs can conceivably help in reducing breast cancer-related mortality. Numerous techniques have been developed for the accurate detection and capture of CTCs. However, there is still much scope for improvement before CTCs can be used in clinical settings. In this review, we discuss about CTC characteristics and their role in breast cancer. The clinical significance of CTCs in breast cancer is elaborated. Further, various technologies developed and the ongoing trials for CTC detection are reviewed. Potential for improvement in currently employed methods and future research expectations are also discussed.
Introduction
Breast cancer is the most prevalent form of cancer in women, worldwide and the leading cause of cancer deaths among women. 1 Although the mortality rate due to breast cancer has reduced due to advancements in treatment strategies, these strategies are effective only in early-diagnosed breast cancer. Most breast cancer deaths occur due to metastasis and tumor relapse, as well as the lack of effective treatment and drug resistance.2–6 Detection of the metastasis and disease progression offer the best chance for effective treatment and a better prognosis.7–9
Presently, radiological examinations and tissue biopsies are the primary methods used to diagnose breast cancer.10–13 Advancements in radiography and tissue biopsy have immensely increased the early detection and diagnosis of breast cancer. 6 However, each of these methods has its challenges. Tissue biopsies, due to their invasive nature, often have the risk of bleeding and/or infections. The localized sampling done in biopsies fails to represent the full extent of heterogeneity such as the genetic and molecular profiles of diverse locations and cell types of the tumor. They further do not allow longitudinal follow-up to identify modifications occurring in response to the therapy. Radiological and imaging techniques pose a risk of radiation exposure, among other safety issues. Serum-based tumor markers such as CA15-3 and carcinoembryonic antigen (CEA) have been used to monitor the effect of therapy on metastatic breast cancer (MBC), but they lack sufficient sensitivity and specificity. Hence, liquid biopsy which includes circulating tumor cells (CTCs), tumor-derived extracellular vesicles (exosomes), circulating free DNA (cfDNA), and circulating tumor DNA (ctDNA) has emerged as an alternative tool for diagnosis, monitoring, and prognosis of breast cancer. Blood sampling is comparatively much easier, making this approach non-invasive and repeatable.14–17
Among all the components of liquid biopsy, CTCs are the most valuable as provide dynamic information on genetics, epigenetics, and proteomic information of the cancer cell. The study of these pioneers of metastasis allows us to get deep insight into the development and treatment of blood-borne metastasis.8,9 Besides, these cells can even be detected in smaller quantities, perhaps enabling diagnosis even before any tumor is diagnosed by usual imaging techniques. 6 Further, CTC analysis also helps in detecting residual tumors and follow-up on the therapy response of each patient, enabling treatment design and modification according to the patient’s requirement. This indicates the importance of CTCs in the field of personalized and customized medicine.
Circulating tumor cells
CTCs were first noticed by Ashworth in 1869, resembling the primary tumor cells in the blood of a patient with metastatic cancer. 18 Although derived from primary tumors, the characteristic features of CTCs are distinct from the origin cells in terms of stemness characteristics and epithelial-to-mesenchymal transition (EMT) properties that enable them to escape the primary tumor, leading to intravasation into the bloodstream and enhancement of their ability to initiate metastasis.7,19
The molecular mechanisms that the CTCs undergo to disseminate from the primary site, travel through the bloodstream, and colonize to form a secondary tumor can be broadly classified into three steps: a) Intravasation and Angiogenesis, b) Survival Maintenance, and c) Extravasation and Colonization. 20
Intravasation and angiogenesis
The CTCs escape from the tissue through easily accessible blood vessels and enter the bloodstream, both passively and actively. 21 In the case of active intravasation, CTCs exhibit dynamic motility and migration. The cells undergo morphological changes through EMT, a process through which epithelial tumor cells lose their intercellular adhesion and gain mesenchymal and invasive features. 22 EMT is mediated through Transforming Growth Factor Beta (TGFβ), Wnt, and Notch signaling. Transcription factors such as FOXC2, SNAI1, SNAI2, and TWIST1 regulate the process. 23 During dissemination, as CTCs migrate to distant areas, tumor cells use EMT activation to separate themselves from the basement membrane and enter the circulation directly.20,24–26 However, in passive intravasation, the external forces (including tumor vasculature and the microenvironment) aid in CTC movement by dragging or pushing the cells. 20
EMT is crucial for metastatic spread of CTCs and determines the cancer progression. 27 However, the cell composition during EMT displays dynamic changes with respect to the type of cancer. The highest lung metastatic capacity was found to be correlated with epithelial-type CTCs having a restricted mesenchymal transition, whereas mesenchymal-type CTCs demonstrated limited lung metastasis. 8 On the other hand, in breast cancer specimens, mesenchymal CTCs have been reported to be positively correlated with cancer progression. Remarkably, rather than single CTCs, clusters of CTCs were found to be more strongly associated with mesenchymal markers. This suggested mesenchymal transitions of pre-existing CTC clusters or proliferation of single CTCs that had previously undergone EMT.7–9
Angiogenesis is further a notable hallmark of cancer. Vascular endothelial growth factor (VEGF) secretion is instigated by carcinomas to promote the production of new blood vessels that in turn aid in providing oxygen and necessary nutrients to continuously growing cells.20,28
Survival maintenance
Upon entering the blood vessels, CTCs are faced with various environmental stresses (such as oxidative stress, shearing force, collisions with other blood components, and many others). Transition to a mesenchymal-like feature does help in CTC survival, but some undergo a specific apoptosis, called anoikis.20,29,30 Most epithelial tumor cells detach themselves from the extracellular matrix to survive within the circulation and prevent anoikis. CTCs also express hypoxia-inducible factor (HIF) 1α which discourages anoikis by metabolic reprogramming.31,32 Platelet aggregation around CTCs protects them from shear stress and immune responses, the latter also prevented via CTC interaction with neutrophils and cancer-associated fibroblasts (CAFs).32,33
An interesting adaptive mechanism displayed by CTCs for survival is clustering. CTC clusters are groups of more than two or three tumor cells that are connected with strong cell-to-cell junctions. They are extremely rare; representing 2–5% of all the CTCs. CTC clusters, however, are extremely metastatic. Studies have associated metastasis of CTC clusters to be 20–100 times more efficient with lower prognosis and OS compared to single CTCs. In CTC clusters, the strong cell-to-cell connections are speculated to prevent anoikis. Heterotypic CTC clusters, formed by the aggregation of tumor cells along with other cell types including neutrophils, platelets, and CAFs, display even better survival mechanisms than homotypic CTC clusters which occur just between the tumor cells. This is because the microenvironment provided by the other cell types protects the CTCs from attacks, minimizes shear stress, and even aids in colonization.9,34–37
Homotypic clustering is formed by cell adhesion molecules (such as plakoglobin, claudins, and CD44). Hypoxic conditions trigger the expression of these molecules and result in the initiation of cluster formation. Epigenetic changes are promoted by CTC clustering (e.g., hypomethylation of binding sites for OCT4, NANOG, and SOX2), which in turn facilitates metastasis seeding.37,38 Further, overexpression of the Heparanase (HPSE) enzyme has been associated with mediating homotypic clustering by suppressing anoikis and promoting survival. 39 On the other hand, in heterotypic clusters, cytoskeleton genes such as GLU, VIM, and TUB were found to be upregulated which encouraged the formation of cytoskeleton bridges between CTCs and other cells.37,40
Extravasation and colonization
As CTCs extravasate, they subsequently go through an opposite process known as MET (Mesenchymal Epithelial Transition) and multiply to create macro-metastases.24–26 This is required because mesenchymal-like CTCs, though highly motile, do not exhibit the cell growth required by cancer cells. MET aids the CTCs in regaining their epithelial-like properties and suppressing the mesenchymal characteristics. The CTCs then penetrate vessel walls, leaving the bloodstream and crossing the basement membrane to reside in the stroma where they will develop into a secondary tumor.20,41–43 This extravasation is facilitated by the secretion of Endothelial focal adhesion kinase by the primary cancer which results in the upregulation of the E-selectin that in turn promotes the homing of cancer cells. 44 Apart from this, target organs may also release certain chemokines, such as the C-X-C motif 12 ligand (CXCL12), attracting the CTCs and further facilitating their migration. 45 Some CTCs, however, lie in a dormant state in the bone marrow. They are known as disseminated tumor cells (DTCs). They may remain in the dormant state for decades before metastasizing. This has been observed in breast cancer, colorectal, and lung cancer which eventually led to bone metastases.20,46
In comparison to single cells, CTC clusters also exhibit better colonization which is facilitated by the cytokines and growth factors secreted by the associated non-tumor cells that result in remodeling of the target site. CTC clusters also have higher adhesion to endothelial walls leading to a higher extravasation rate. 35 A study also reported the reorganization of CTC clusters in reversible chain-like structures for migrating through capillary-sized vessels to reduce hydrodynamic resistance. Upon exiting the vessel, the clusters then regain their original morphology. This transition is possible via a series of steps including rotation of cells inside the clusters, selective cleavage, and preserving specific cellular adhesions. 37 All these mechanisms further promote metastasis to distant locations.
CTCs at distinct metastatic sites display molecular and cellular heterogeneity. Current evidence suggests that ctDNA and CTCs represent similar molecular characteristics of the overall disease and can be used to provide an extensive view of the genomic landscape of the entire cancer. However, it is still not proven if the CTCs separated from different organs are shed in a homogenous manner. This is due to the fact that even though CTCs regain certain characteristics of the primary tumor, they also acquire new traits of the metastatic organ.47,48 CTCs exhibit heterogeneity with respect to their gene expression as well. The gene expressions are specific to the type of cancer and the destination for metastasis. For instance, in the case of breast cancer, the CTCs that tend to metastasize to the brain often exhibit gene expressions of Notch1, Her2, and EGFR but an absence of EpCAM is seen.20,49
CTCs of the epithelial, epithelial-mesenchymal transition (EMT), hybrid (epithelial/EMT +), irreversible EMT +, and circulating tumor stem cell types can all be seen in cancer patients’ CTC pools. 50 It can be difficult to separate these extremely uncommon CTCs from the vast pool of circulating blood cells. 51 Nevertheless, the new CTC separation methods have led us to separate different types of CTCs for cancer screening, clinical outcome evaluation, and prognosis assessment. 7
Detection strategies of circulating tumor cells in breast cancer
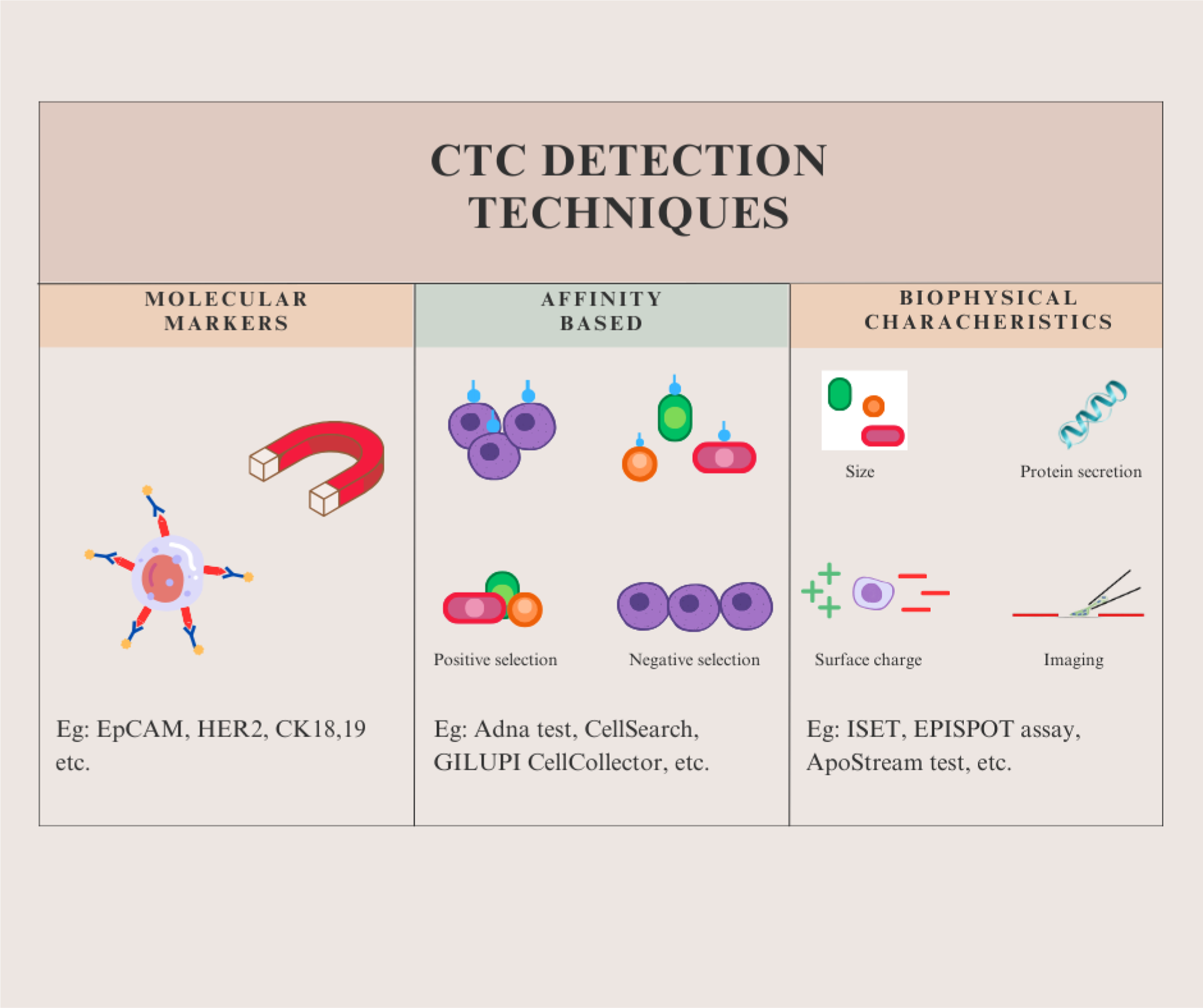
The analysis of CTCs is usually a three-step procedure, consisting of enrichment, detection, and release. Both enrichment and detection are critical for the specificity and sensitivity of the overall approach.52,53 The first strategy involves a specific contact between CTCs and materials via physical interactions or antigen-antibody interactions.53,54 The second strategy uses various optical detection methods, such as flow cytometry, fluorescence microscopy or spectrophotometry, surface-enhanced Raman scattering, electrical impedance, etc.53,55 In the final step, analysis of released CTCs occurs through CTC cultures for genomic, transcriptomic, and proteomic studies.53,56,57 Several commercialized CTC detection technologies have been developed which can be categorized based on the method of CTC identification as affinity-based or biophysical-based methods.
58
Each of these categories is further divided based on detection strategies. All the categories are summarized in Figure 1. Types of CTC detection techniques. *CTC detection techniques can be broadly categorized into three types—detection on the basis of 1) molecular markers, 2) affinity, and 3) biophysical characteristics such as size, density, surface charge, protein secretion, etc.
59
Affinity-based capture and enrichment
Molecular markers used to identify CTCs.a

Affinity-based CTC detection.a

aNumerous technologies have been developed commercially that capture and recover CTCs with the help of specific markers present on them, utilizing antibodies or magnetic forces for precise detection. 70
Biophysical methods for detection of circulating tumor cells in breast cancer
Biophysical characteristic–based CTC detection.a

aVarious technologies have also been developed for identification and capture of CTCs on the basis of their biophysical attributes such as size, inertial focusing, surface charge, etc. 70
Cell release for molecular profiling and in vitro proliferation
With the growth of various technologies to detect and capture CTC, research in this field is no longer limited to the detection and quantification of CTC in different cancers. Single-cell multi-omics methods have provided genomic, epigenomic, transcriptomic, and proteomic information on CTCs.38,72–75 Analysis of a single CTC can yield valuable insight into the molecular mechanisms and acquired genomic variations of CTCs, which can be correlated with the clinical characteristics of the patients at different stages of cancer. Therefore, CTC analysis can facilitate the personalization of therapeutic options.75,76
CTCs are also good candidates for generating preclinical models, especially 3D organoid cultures and CTC-derived xenograft models which could be applied in screening therapeutic drugs, and understanding mechanisms of development of cancer and loss of cancer immunity.77–80 The numerous clinical applications of CTCs are showcased in Figure 2. Applications of CTCs analyzed from liquid biopsies. *Liquid biopsy enables in isolating single CTCs as well as CTC clusters. These CTCs can provide various information about the tumor metastasizing including the tumor heterogeneity and type, its gene sequence and mutations, and also its response to therapies and drugs. This information can in turn aid in cancer diagnosis, prognosis, and treatment.
59

However, ex vivo CTC cultures have been observed to show a low success rate, limiting their feasibility in clinical trials. 81 Further, it is necessary to address other issues as well such as low CTC purity, huge blood quantities required, clogging of the device, high time duration, variations in size and shape leading to cell loss, etc.82,83 Also, epitope expression and plasticity need to be better understood, and molecular and functional assays need to be made more efficient before the cultures can be used in clinical settings.78,83
Clinical utility of determining CTC in breast cancer patients
CTCs enter the circulation from tumor lesions in the form of heterogeneous groups. These CTCs exhibit molecular and phenotypic variations according to the microenvironment and the treatment. CTC heterogeneity can be divided into two groups—intrinsic and extrinsic heterogeneity. Extrinsic heterogeneity reflects the overall tumor heterogeneity, which can be used in understanding the primary tumor heterogeneity at the genomic level, whereas intrinsic heterogeneity is related to the metastatic characteristics of single CTCs. Both of these variations can be used to guide treatment according to their molecular and functional features. Hence, CTC detection and analysis can aid in understanding tumor heterogeneity and providing precision medicine.84–86
In 2016, the ESMO/ASCO Recommendations for a Global Curriculum in Medical Oncology Edition highlighted the relevance of liquid biopsy in cancer diagnosis, prognosis, and personalized cancer therapy. Since then, the presence of CTCs has been reported in the peripheral blood of all major carcinomas, and studies on breast, prostate, colorectal, small cell, and non-small cell lung cancers have shown the efficacy of CTCs in predicting prognosis.87,88 As of 20th January 2024, there are 362 studies associating CTCs with breast cancer at ClinicalTrials.gov, which reflects the ongoing curiosity about CTCs as biomarkers for the diagnosis and prognosis of cancers. 89 It has been shown by previous studies that cancer recurrence as well as metastasis can be predicted and a patient’s prognosis can be evaluated through CTC load.
The DETECT studies, which were reported at the San Antonio Breast Cancer Symposium in 2020, illustrated the examination of blood samples from MBC patients for the DETECT III and IV trials. The DETECT tests began to define the significance of characterizing the CTC and its use in targeted therapy as the largest study program on CTC-based therapy from around the world. The purpose of the studies was to assess the safety of chemotherapy or endocrine therapy combined with dual HER2-targeted therapy. This study was the first to register patients based on the HER2 phenotype of their CTCs. Further, the patients were categorized based on the tumor’s HER2 status. This trial was divided into several DETECT studies, and clinical success was predicted by CTCs and progression-free survival (PFS). The examination of the patients chosen for these investigations demonstrated the significance of CTC enumeration in the prognosis of MBC. There was a striking correlation between overall survival (OS) and CTC status. CTC count was found to be indirectly proportional to OS. Further, shorter OS was seen in patients displaying CTCs with stronger HER2 with respect to those with negative or moderate HER2-stained CTCs.90,91
CTCs can also be used to determine the endocrine levels in a patient and thus alter the treatment accordingly. For instance, a new study found CTCs in 50% of ER-positive MBC patients. The majority of these individuals had ER-negative CTCs and one-third of these patients had both ER-positive and ER-negative CTCs. This method of ER assessment via CTCs is less invasive and can help in modifying therapy according to the patient’s response. Further in this study, 10 individuals experienced disease stabilization or even regression, compared to 14 patients who displayed advancement. Patients under progression showed a change in their CTC status from negative to positive. The study used chemotherapy in place of endocrine treatment to reduce the number of CTCs. In the majority of samples, both the conversion to a CTC-negative status and the decline in CTC number from the baseline to the completion of therapy were significant. Given that these findings suggest that a decrease or an increase in the number of CTCs is connected to stable illness or advancement, respectively, it was necessary to address the differences in the two samples according to the clinical response. It was discovered that blood samples from patients whose diseases were progressing showed a substantially larger rise in the CTC number than did blood samples from patients whose diseases were stable. These findings demonstrated a substantial link between CTC counts and progression-free survival as well as a solid association between the rise in CTC numbers and lower OS. Monitoring ER-CTC status can therefore be added to the CTC count as a predictive factor and may aid in the prediction of medication resistance.90,92
Another study examined the prognostic value of cytokeratin (CK-19) positive CTC in BC patients receiving adjuvant chemotherapy right after primary surgery in order to identify the existence of any residual tumor and customize adjuvant therapy to eliminate the tumor. This study showed that patients with detectable CK-19 mRNA-positive CTCs post-chemotherapy had substantially worse OS. It also indicated that CTCs can be used as an indicator of chemotherapy resistant residual disease.93,94 Another notable study is the TREAT-CTC Trial in 2010. Although, this study didn’t achieve the aim with which it was initiated but it did demonstrate the importance of CTC in the prognosis of breast cancer. In the results published, 7.2% of the 1317 HER2-negative patients were found to be CTC-positive post-adjuvant therapy. The trial thus concluded that CTC screening can aid in early breast cancer prognosis and further CTC-positive patients exhibit a higher risk of relapse.94,95 An additional study demonstrated that detection of CTCs in triple-negative breast cancer patients undergoing neoadjuvant chemotherapy indicated tumor micro-metastatic spread at distant sites and correlated with an increased chance of relapse. 96
Although until now research work related to CTCs has focused on its prognostic significance, recently research has also been initiated to determine its utility in detecting and differentiating breast cancer cases from benign cases. This approach is based on the concept that in some circumstances, metastatic spread may occur from an unknown primary (an unidentified lesion). A recent study by Jin et al evaluated the value of CTCs in early diagnosis and staging of BC. Their study indicated that CTCs could be used to distinguish BC patients from healthy volunteers and patients with benign breast cancer (p < .0001). 97
Clinical studies currently in phase III/IV determining the clinical utility of CTCs.a

aVarious ongoing clinical trials are determining the significance of CTCs in MBC patients. Such trials analyze the correlation of CTCs with cancer progression, progression-free, disease-free and OS, treatment response, therapeutic resistance, etc. 89
Identification of resistance mechanisms and drug selection
Breast cancer therapy focuses on ER/PR and HER-2 status. Clinical data indicate that the HER-2 status of distant metastatic tumor cells and CTCs differs considerably from that of original tumor cells in around 30% of instances, which raises the possibility that therapies that target the initial tumor may not be suitable for treating metastatic tumors. A non-invasive technique like CTCs is the appropriate replacement for traditional tissue biopsies, which may cause harm to patients when discovering new features and characteristics of malignancies. Apart from ER/PR and HER-2 status, the identification of immune checkpoint regulators like PD-L1 in recent years has made it feasible to successfully treat MBC, which has emerged as an intriguing new therapeutic target. 6 Mazel and his colleagues were the first to discover the expression of PD-L1 on CTC surface in breast cancer patients with HER2(−) and ER or PR(+). Going forward, clinical trials involving breast cancer patients who are undergoing immune checkpoint therapy can use the established assays of CTC/PD-L1. 99 Other studies have also tried to metabolically classify CTCs which can then aid in identifying CTC subpopulations and administering targeted therapies. This has been done by correlating the expression of markers in CTCs with disease progression. 100 For instance, PD-L1 (+) and CD 47 (+) CTCs have been associated with reduced progression-free and OS. 101 Similarly, TLR4 (+) CTCs were present at higher rates in metastatic patients, while higher rates of pSTAT3 (+) CTCs were seen in early-stage breast cancer patients. 102
Drug resistance remains a major challenge in oncology. It can render traditional cancer therapies useless often resulting in fatal outcomes. 103 To prevent this, it is important to identify chemoresistance in the early stages. This can be achieved via CTCs. 100 Molecular and genomic characteristics of CTCs can be analyzed to determine the tumor’s clonal dynamics by comparing it with the clinical course and identification of the mutation that is leading to the resistance so as to administer a targeted therapy. 104 For instance, a study identified AR gene expression and AR-V7 splice variant in ER(+) breast cancer with bone metastasis which resulted in resistance to endocrine therapy. Thus, it concluded that an anti-tumor therapy targeting the AR expression may benefit the patients. 105 Another study reported that a short course of trastuzumab (a HER2-targeted cancer drug) was able to prevent chemotherapy resistance in breast cancer patients with HER2 mRNA and CK-19 mRNA-positive CTCs and bone marrow disseminated tumor cells. Secondary adjuvant therapies can potentially then yield better results. 106 Other chemoresistance-associated genes have also been identified in CTCs such as MRP1, MRP2, MRP4, MRP5, MRP7, ERCC1, and MDR1. 102
Real-time therapy monitoring
Real-time detection of CTCs in patients is required to monitor and evaluate their status after treatment, such as surgery or chemotherapy, as well as to forecast and estimate survival time. By examining each patient’s real-time CTC data throughout treatment, the clinical validity of the CELLSEARCH approach for monitoring CTC quantification in patients with MBC was evaluated. 107 Bidard et al. requested 51 European centers to provide anonymized data on individual MBC patients who participated in trials between January 2003 and July 2012. After analysis of this data, they concluded that CTC counts instead of tumor markers enhance the prediction of prognosis for MBC. 108
CTCs can also aid in the detection of minimal residual disease (MRD). Residual or disseminated tumor cells can often go undetected in clinical or radiological tests and later lead to recurrence in remission patients. Many studies have linked CTCs to MRD in breast cancer patients.100,109 For instance, Radovich et al. conducted a randomized trial, analyzing 196 early-stage triple-negative breast cancer patients and reported the presence of CTCs and ctDNA is associated with reduced disease-free survival and OS. 110 Another study performed a comprehensive liquid biopsy analysis on early-stage breast cancer patients. The analysis included numerous liquid biopsy biomarkers such as CTC enumeration and their phenotypic analysis, gene expression (for ALDH1, CK-19, CD24, CD44, and TWISTI) and analysis of ESR1 and PIK3CA mutations in EpCAM (+) CTCs and DNA methylation of ESR1 in CTCs. The study reported that patients who showed relapse had tested positive for at least one of these markers demonstrating the significance of said biomarkers in detecting RD. 111
A high CTC count has been associated with metastasis, poor prognosis, disease relapse, shorter progression-free, disease-free, or OS and therapeutic resistance. Thus, real-time monitoring of CTCs can not only detect and diagnose disease progression but also be utilized in the assessment and monitoring of treatments and early detection of relapse.59,112–114
Limitations and future prospects
The correlation between CTC analysis in breast cancer patients for detection, prognosis, and therapeutic response has been displayed by innumerable studies. These studies can aid in predicting tumor progression, monitoring patient’s responses to therapies, and providing personalized therapy. As discussed earlier, various technologies have been developed for CTC detection and capturing. 115 However, there are certain limitations which need to be addressed.
For instance, temporal and spatial heterogeneity in CTCs prevents accurate CTC quantification. Dynamic changes occur in the phenotype of CTCs due to which they can go undetected. The fundamental issue is the lack of a thorough study of CTC heterogeneity at the genetic, phenotypic, and morphological levels to determine the attributes linked to the development of metastatic BC. 116 CTC rarity further can lead to difficulty in CTC identification. 54 This can be avoided by using technologies that account for more elaborate functional and molecular attributes of CTCs. 70 It is also necessary to standardize methods for clinical CTC detection and quantification for uniform results. Further easier, faster, and more efficient systems have to be developed for the establishment of CTC analysis in clinical settings. 117
Further, other liquid biopsy biomarkers like ctDNA can be supplemented with CTC analysis for better results.90,118 Besides this, 2D as well as 3D cultures derived from CTCs and xenograft models can aid in better monitoring of cancer progression and drug screening through organoids. 119 This was demonstrated by Sachs et al in 2018, when they generated organoid lines from samples of patients to create a living biobank of various breast cancer subtypes. 120
Various companies such as the European Liquid Biopsy Society (ELBS) and the European Liquid Biopsy Academy (ELBA) are working towards the development of novel approaches for CTC capture and analysis. 121 Overcoming the above-mentioned limitations and developing robust and systematized methods for CTC identification of all cancer types can bring CTCs into the spotlight of personalized medicine in the future as minimally intrusive yet very insightful sources of biomarkers. 122
Conclusion
Circulating tumor cells show great potential as the biomarker for cancer detection, progression, and treatment. Through liquid biopsy, they provide a non-invasive method for screening and management of breast cancer. Analyzing the molecular characteristics of CTCs can aid in understanding the primary tumor and determining the right course of treatment. Thus, CTCs give way to precise and personalized medicine. However, to achieve the same, certain limitations have to be overcome. Even though CTCs were first reported more than 150 years ago, the literature available is still limited. Primarily, a gold standard has to be developed for CTC identification. Moreover, concepts like tumor heterogeneity and CTC clustering need to be explored more for better understanding. Lastly, use of computational tools for multi-omics study and model development can aid in better monitoring and management of the disease. Hence, more research is required to address the challenges of this procedure and develop standardized guidelines for its use in clinical settings.123,124
Statements and declarations
Footnotes
Acknowledgements
We would like to acknowledge Amity Institute of Biotechnology and Amity University, Noida campus for providing us with the facilities and infrastructure to work on this review.
Author contributions
All the authors equally contributed to the preparation and revision of the manuscript.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
