Abstract
Male fertility has declined markedly in recent decades, with reductions in sperm count, motility, and quality posing a growing global concern. Among the multifactorial causes, oxidative stress plays a central role by disrupting redox balance, damaging sperm DNA, impairing membrane integrity, and reducing motility. Natural antioxidants, particularly plant-derived bioactives, have gained significant attention as potential modulators of male reproductive health. Gum Acacia (GA), a dried exudate obtained mainly from Acacia senegal and Acacia seyal, is a highly branched arabinogalactan-rich polysaccharide with excellent solubility, biocompatibility, and bioactivity. Preclinical studies suggest that GA exerts protective effects on testicular function by reducing oxidative stress, modulating inflammation, inhibiting apoptosis, and improving metabolic and hormonal profiles, including testosterone and luteinizing hormone. GA has also shown promise in mitigating reproductive damage caused by diabetes, chemotherapy, and environmental toxins, as well as enhancing semen cryopreservation as a plant-based alternative to conventional extenders. Despite these encouraging findings, evidence remains limited, fragmented, and largely restricted to animal studies, with human data virtually absent. This review critically appraises current experimental and biomedical evidence on GA as a modulator of male fertility, highlights potential mechanisms of action, and identifies key knowledge gaps. Findings indicate that GA could emerge as a safe, accessible, and cost-effective adjunct for improving male reproductive outcomes, warranting further investigation in standardized and clinically oriented studies.
Introduction
In recent decades, male fertility has become an increasing global concern, with a marked decline observed in sperm count, motility, and quality. Multiple factors contribute to this trend, including lifestyle changes, environmental pollutants, oxidative stress, and systemic diseases such as diabetes and cancer (World Health Organization [WHO], 2021). Recent global evidence confirms a significant decline in male fertility, with a 51.6% reduction in sperm concentration and a 62.3% drop in total sperm count between 1973 and 2018, according to a large meta-analysis of 223 studies (Levine et al., 2023). Among the numerous contributors to male infertility, oxidative stress plays a particularly critical role. It arises from an imbalance between reactive oxygen species (ROS) and antioxidant defense systems, leading to DNA damage in sperm, reduced motility, compromised membrane integrity, and impaired acrosome reaction—factors essential for successful fertilization (Bilodeau et al., 2000; Engdawork et al., 2024). The testicular microenvironment is especially vulnerable to oxidative injury due to its high cellular turnover and the abundance of polyunsaturated fatty acids in sperm membranes. Elevated oxidative stress has been strongly linked to reduced sperm concentration, increased DNA fragmentation index (DFI), and impaired function of Sertoli and Leydig cells, thereby disrupting spermatogenesis and testosterone synthesis (Dong et al., 2022; Jalili et al., 2024). Antioxidants, whether enzymatic (e.g., superoxide dismutase, catalase, glutathione peroxidase) or non-enzymatic (e.g., vitamins C and E, polyphenols), are essential for restoring redox balance, and enhancing these defenses through dietary or pharmacological means has become a central focus in addressing oxidative-stress-induced infertility (Eidan et al., 2015; Jara et al., 2019).
In this context, natural antioxidants have attracted growing interest in reproductive medicine for their potential to protect testicular tissue, improve spermatogenesis, and restore hormonal balance (A. M. Firouzabadi, 2024; A. R. Firouzabadi et al., 2025). Plant-derived bioactives such as resveratrol, curcumin, ginseng, and ginger exhibit not only free radical scavenging activity but also the ability to modulate inflammation, apoptosis, and hormone regulation—making them particularly valuable in the multifactorial pathology of infertility (Ralli & Alhalmi, 2025; Sueishi et al., 2019). One promising yet underexplored natural compound is Gum Acacia (GA), a dried exudate obtained mainly from the hardened sap of Acacia senegal and Acacia seyal. Its principal component, arabinogalactan—a highly branched polysaccharide composed of arabinose and galactose—confers functional properties such as high solubility, emulsification, and viscosity (Mohamed et al., 2025). These characteristics, along with trace amounts of proteins and minerals like calcium and potassium, enhance its applicability in both industrial and pharmaceutical formulations. GA is recognized as safe (GRAS) and has demonstrated minimal toxicity in animal and human studies, making it a promising candidate for nutritional and medicinal interventions, including fertility enhancement (Al-Baadani et al., 2021). Beyond its established applications, GA has demonstrated prebiotic, anti-inflammatory, and antioxidant properties, and has been traditionally used to treat inflammation, gastrointestinal disorders, and renal conditions (Al-Jubori et al., 2023). GA also exhibits prebiotic activity, modulating gut microbiota composition and promoting the growth of beneficial bacteria. This gut-mediated systemic effect may contribute to improved metabolic profiles, hormonal regulation, and reproductive outcomes. Studies in animal models, including zebrafish and rodents, have demonstrated improvements in glucose and lipid metabolism, particularly in diabetic states—conditions often linked to impaired fertility. Notably, certain findings suggest sex-specific effects, highlighting the importance of tailored evaluation (Barkeer et al., 2024; Saha & Dey, 2024).
Although emerging animal studies suggest that GA may confer reproductive benefits—such as preserving testicular architecture, improving sperm quality, and modulating reproductive hormones under conditions of oxidative stress, toxic exposure, or metabolic dysfunction—the current body of evidence remains limited, fragmented, and predominantly preclinical (Nofal et al., 2023; Table 1). Few studies have explored its mechanisms of action in detail, and clinical data in human populations are virtually absent. Moreover, the potential translational value of GA in male infertility management has yet to be systematically assessed. Addressing this knowledge gap is essential for determining whether GA could serve as a safe, accessible, and cost-effective adjunct in reproductive health care. Accordingly, the present review critically appraises the existing experimental and biomedical evidence on GA’s role as a modulator of male fertility, with the aim of identifying mechanistic insights, evaluating the strength of current findings, and outlining priorities for future research (Table 2).
Summary of Gum Acacia Effects on Male Reproduction.

Characteristics of Original Studies Investigating the Effects of Gum Arabic (GA) on Male Reproductive Function.

Method
A comprehensive literature search on the effects of GA as a modulator of male fertility was conducted using three major scientific databases: PubMed, Scopus, and Web of Science. The search included all studies published up to August 2025 and employed a combination of keywords and MeSH terms such as “Gum Arabic,” “Gum Acacia,” “Acacia senegal,” “Vachellia senegal,” “male infertility,” “sperm quality,” “sperm motility,” “sperm viability,” “testicular function,” “testosterone,” “oxidative stress,” “antioxidants,” and “reproductive outcomes.” Specific combinations of keywords were also used, including “Gum Acacia and male infertility,” “Gum Arabic and sperm motility,” and “Acacia gum and reproductive hormones.” The inclusion criteria for this review were restricted to original experimental studies (in vivo or in vitro) that specifically investigated the effects of GA on male reproductive parameters, articles published in the English language from the inception of the databases until August 2025, and studies with available full-text providing comprehensive data on outcomes such as sperm characteristics, reproductive hormones, and testicular histology. Conversely, studies were excluded if they were unrelated to the topic, lacked full-text access, were not published in English, or were case reports, conference abstracts, or review articles. From an initial pool of 34 articles identified through title and abstract screening, duplicates and studies not meeting the predefined inclusion and exclusion criteria were removed, leaving 20 articles for full-text evaluation. After a thorough assessment, 16 studies fully complied with all eligibility requirements and were selected for the final review (Figure 1).

Flowchart of Study Selection According to PRISMA Checklist.
Role of GA in Male Reproductive Health
Evidence from experimental models indicates that GA exerts multifaceted protective effects on male reproductive health under pathological conditions (A. A. Ahmed et al., 2020; Nofal et al., 2023). Its beneficial actions are particularly evident in three major contexts: (a) disease-induced testicular dysfunction, where GA counteracts oxidative stress, inflammation, and apoptosis caused by chemotherapy, diabetes, or environmental toxins; (b) semen cryopreservation, where GA serves as a plant-based alternative to conventional extenders and enhances sperm survival during freeze–thaw cycles; and (c) functional fertility outcomes in animal models, where GA supplementation improves sperm quality, hormonal balance, and reproductive performance, although some contradictory results have also been reported (Abo et al., 2020; A. A. Ahmed et al., 2020; M. Ali et al., 2017; Nofal et al., 2023).
Through these diverse applications, GA has been consistently associated with improvements in spermatogenesis, testicular integrity, and endocrine regulation. The following subsections critically examine the available evidence, highlighting both the therapeutic promise and the mechanistic basis of GA’s role in male reproductive health (A. A. Ahmed et al., 2020; Al-Doaiss & Al-Shehri, 2020; Nofal et al., 2023).
Protection Against Disease-Induced Testicular Dysfunction
Chemotherapeutic agents such as cisplatin, while highly effective in cancer treatment, often result in severe testicular toxicity and long-term infertility (Ghafouri-Fard et al., 2021; Nofal et al., 2023). Cisplatin generates excessive ROS, disrupts steroidogenesis, and induces apoptosis in germinal cells, ultimately impairing spermatogenesis. Evidence indicates that co-treatment with GA alleviates these adverse outcomes by restoring redox balance and suppressing pro-apoptotic signaling (Nofal et al., 2023; Park et al., 2022). GA enhances the activity of antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px), while reducing lipid peroxidation markers such as malondialdehyde (MDA; Musa et al., 2016). In addition, it downregulates inflammatory cytokines such as Tumor Necrosis Factor-alpha (TNF-α) and Interleukin 6 (IL-6), suppresses apoptotic markers like caspase-3, and promotes proliferating cell nuclear antigen (PCNA) expression, which supports germinal cell proliferation (N. Ahmed et al., 2022; Fedail et al., 2016). Histological examinations confirm the preservation of testicular architecture in GA-treated animals, with seminiferous tubules maintaining organized germinal epithelium (Nofal et al., 2023; Singhal et al., 2018). Functionally, significant improvements in sperm motility, sperm count, and hormonal recovery—including testosterone and luteinizing hormone (LH)—have been observed in individuals with impaired sperm parameters following GA administration, with motility increasing from ~45% to ~75%, sperm count rising from ~100 ×106 to ~180 ×106, testosterone from 2.18 ng/ml to 3.03 ng/ml, and LH from 0.20 mIU/ml to 0.25 mIU/ml (Nofal et al., 2023). These findings suggest that GA not only mitigates structural injury but also restores functional fertility potential, positioning it as a candidate adjunct therapy for fertility preservation in patients receiving chemotherapy.
Similar protective effects have been demonstrated in the context of diabetes mellitus, a systemic metabolic disorder with profound negative consequences for male fertility (A. A. Ahmed et al., 2020; Azouz & Hassanen, 2020). Hyperglycemia and insulin resistance increase oxidative stress within the testes, disrupt Sertoli and Leydig cell functions, and suppress testosterone production, leading to impaired spermatogenesis and reduced sperm quality (Harrer et al., 2025). GA supplementation in diabetic models has been shown to counteract these pathological processes through its antioxidant and anti-inflammatory activities (A. A. Ahmed et al., 2020; Azouz & Hassanen, 2020). Treated animals exhibit preserved seminiferous tubule morphology with reduced vacuolization and germ cell loss, indicating structural protection (Azouz & Hassanen, 2020). At the molecular level, GA modulates the expression of metabolic genes such as glucose transporter 1 (GLUT1), glucose transporter 3 (GLUT3), and lactate dehydrogenase, thereby enhancing testicular energy metabolism and supporting germ cell survival (A. A. Ahmed et al., 2020). These changes are accompanied by increases in testosterone and LH levels, reflecting partial restoration of hypothalamic–pituitary–gonadal (HPG) axis function (A. A. Ahmed et al., 2020; Azouz & Hassanen, 2020). Moreover, GA downregulates caspase-3 and upregulates Visfatin, providing additional protection against metabolic stress-induced apoptosis while promoting steroidogenesis (Abo et al., 2020). Collectively, these findings point to GA as a promising therapeutic agent for managing diabetic infertility through mechanisms that integrate antioxidant defense, metabolic regulation, and hormonal balance.
Environmental toxins and lifestyle-related exposures also play a substantial role in male reproductive decline (Rotimi & Singh, 2024). Agents such as aluminum chloride (AlCl3) and hookah smoke induce oxidative and nitrosative stress, activate inflammatory pathways—including Nuclear Factor kappa B (NF-Κb) and Interleukin 1β—and disrupt endocrine function, culminating in defective spermatogenesis (Z. Li et al., 2023). GA has been shown to counteract these deleterious effects by activating the Nuclear factor erythroid 2-related factor 2 (NRF2) antioxidant pathway, thereby upregulating the transcription of protective enzymes (SOD, CAT, GSH-Px) and reducing oxidative injury (B. H. Ali et al., 2020; Imbabi et al., 2023). At the same time, it suppresses pro-inflammatory mediators including TNF-α, IL-6, and NF-κB, mitigating chronic testicular inflammation (B. H. Ali et al., 2020). Functionally, animals exposed to toxins and treated with GA demonstrate improved sperm motility, morphology, and membrane integrity, alongside preserved testicular architecture with active spermatogenesis (Abo et al., 2020; A. A. Ahmed, 2021). Importantly, hormonal recovery is evident, as reflected by significant increases in serum testosterone and LH levels. Specifically, the co-administration of GA significantly mitigated the suppressive effects of toxic exposure (Cisplatin or Hookah Smoke), elevating testosterone levels from 2.18 ± 0.07 ng/ml to 3.03 ± 0.51 ng/ml in the cisplatin model, and from 2.0 ± 0.2 ng/ml to 4.1 ± 0.2 ng/ml in the hookah smoke model. Similarly, LH levels showed recovery, rising to 0.2534 ± 0.031 mIU/ml in the co-treated group compared to the lower values in the exposed groups (B. H. Ali et al., 2020; Nofal et al., 2023). These results reinforce the role of GA as a natural protective agent capable of mitigating reproductive toxicity caused by environmental and lifestyle hazards, with strong mechanistic support from its antioxidant, anti-inflammatory, and anti-apoptotic properties.
GA in Semen Cryopreservation
Semen cryopreservation plays a pivotal role in assisted reproductive technologies, serving as a cornerstone in both livestock breeding programs and fertility preservation strategies for valuable genetic resources (Castro et al., 2025). Traditional protocols rely heavily on egg yolk-based extenders, which are effective in protecting sperm during freeze–thaw cycles but raise several concerns. These include risks of microbial contamination, variability in biochemical composition between egg yolk batches, and biosecurity limitations that can restrict their use in large-scale or international breeding programs (Bustani & Baiee, 2019). Consequently, there is growing interest in safer, plant-based alternatives that can combine cryoprotective efficacy with standardization and biosecurity. In this context, GA has attracted significant attention due to its polysaccharide-rich structure, antioxidant potential, and high biocompatibility.
Experimental studies in animal models, including rams, stallions, and bulls, demonstrate that preheated GA at concentrations of 3–6% maintains post-thaw sperm motility and viability at levels comparable to or exceeding those of conventional extenders (M. Ali et al., 2017; Khanzadeh et al., 2021; Ramezani et al., 2018; Shehab-El-Deen et al., 2023). Mechanistically, GA contributes to cryoprotection by stabilizing sperm plasma membranes, reducing osmotic shock, and preserving acrosome integrity, which is essential for fertilization (M. Ali et al., 2017; Khanzadeh et al., 2021; Ramezani et al., 2018; Shehab-El-Deen et al., 2023). Furthermore, GA attenuates cryo-induced oxidative stress by enhancing the activity of antioxidant enzymes such as SOD, catalase, GSH-Px, and glutathione reductase and by suppressing lipid peroxidation, thereby reducing the accumulation of ROS that typically compromises sperm viability after thawing (Khanzadeh et al., 2021; Šterbenc et al., 2014). These findings are consistent with GA’s protective mechanisms observed in disease-induced models of testicular dysfunction, reinforcing the notion that its antioxidant and anti-inflammatory capacities are transferable across different reproductive contexts.
The addition of exogenous antioxidants such as cysteine or ascorbic acid to GA-based extenders has been shown to amplify these benefits, providing synergistic protection against ROS accumulation during freeze–thaw cycles (Imbabi et al., 2023; Kao et al., 2008; Nofal et al., 2023). Under such conditions, parameters, including sperm motility, velocity, linearity, and acrosomal integrity, are significantly improved compared to egg yolk extenders alone [42]. Moreover, the incidence of morphological abnormalities is reduced, indicating not only structural preservation but also an enhancement of the functional competence of sperm cells (Nofal et al., 2023). This dual effect—structural stabilization and functional improvement—suggests that GA has potential beyond merely serving as a replacement for egg yolk, positioning it as an additive with unique biological properties that actively promote sperm resilience during cryopreservation.
The efficacy of GA-based extenders, however, is not uniform across all experimental conditions and is strongly influenced by semen collection methods. Studies reveal that semen obtained via artificial vagina (AV) responds more favorably to GA supplementation than semen collected through electro-ejaculation (EE; Khanzadeh et al., 2021; Ramezani et al., 2018). This discrepancy may be attributed to the higher baseline quality and reduced stress factors associated with AV-derived samples, which allow GA’s protective actions to manifest more fully (Shehab-El-Deen et al., 2023). Optimal cryoprotective effects are generally observed at concentrations between 3% and 5%, with higher doses exerting potentially adverse effects on sperm viability, possibly due to increased viscosity or osmotic imbalance introduced by excessive GA [37, 38].
Taken together, the current body of evidence underscores the potential of GA as a biosecure, plant-based cryoprotective agent capable of addressing the limitations of traditional extenders. Its combined antioxidant activity, membrane-stabilizing properties, and compatibility with additional antioxidants provide a strong mechanistic foundation for its application in semen preservation. Nonetheless, further studies are warranted to optimize dosing strategies, standardize protocols across species, and evaluate long-term fertilization outcomes. Such advancements would help establish GA not merely as an experimental alternative but as a reliable and scalable solution in modern reproductive biotechnology.
Reproductive Outcomes and Functional Fertility in Animal Models
Rodent studies consistently provide robust evidence that GA supplementation exerts beneficial effects on male reproductive outcomes (Azouz & Hassanen, 2020; Nasir et al., 2020). Experimental data repeatedly demonstrate increases in testicular weight, sperm count, sperm motility, and circulating testosterone levels following GA treatment (Nasir et al., 2020). Fertility trials extend these findings by reporting larger litter sizes and enhanced offspring viability, underscoring that GA not only improves spermatogenic parameters but also translates these changes into functional reproductive success (Abi Assaf et al., 2024; Nasir et al., 2020). Histopathological examinations complement these observations, revealing preserved seminiferous tubule organization, active germinal epithelium, and reduced vacuolization in GA-treated animals (Nasir et al., 2020). Mechanistically, these improvements are associated with GA-induced upregulation of PCNA, which supports germ cell proliferation, and suppression of caspase-3, which limits apoptosis within testicular tissues (Azouz & Hassanen, 2020; Hamza et al., 2016). Importantly, several studies have documented that GA’s positive effects persist even after treatment cessation, suggesting a capacity to induce long-lasting or even semi-permanent improvements in reproductive health rather than temporary symptomatic relief (Beytur et al., 2012; Sawhney et al., 2005; Spermon et al., 2006).
Despite this encouraging body of evidence, the literature also contains contradictory or adverse reports that introduce necessary caution. For instance, administration of very high doses of GA was linked to reduced fertility in female zebrafish, while extracts derived from Acacia nilotica—a different but related species—were shown to induce irreversible male infertility in certain models (Abi Assaf et al., 2024). Such discrepancies may arise from species-specific responses, variations in the phytochemical composition of different Acacia species, or the dose-dependent duality of bioactive compounds, whereby moderate concentrations confer protection but excessive exposure triggers toxicity (Jeon et al., 2024; Pedro et al., 2022). Moreover, inconsistencies in treatment duration, extraction methods, and experimental design contribute to the heterogeneity of findings (J. Li et al., 2024). These considerations highlight the importance of cautious interpretation of preclinical outcomes and the pressing need for standardized experimental protocols. Dose–response studies, comparative evaluations across animal models, and clear distinctions between Acacia senegal (true GA) and other Acacia species are essential prerequisites before any serious clinical translation can be considered.
The mechanistic basis of GA’s fertility-modulating actions appears to be multifaceted, reflecting the compound’s diverse biochemical properties. In addition to upregulating PCNA and downregulating caspase-3, GA enhances the expression of Visfatin, a key regulator of energy metabolism, and steroidogenic acute regulatory protein (StAR), which is critical for cholesterol transport into mitochondria during steroid hormone synthesis. These molecular adaptations align with observed improvements in sperm quality, testicular architecture, and androgen production (A. A. Ahmed, 2021; B. H. Ali et al., 2020). Furthermore, GA’s antioxidant and anti-inflammatory capacities help neutralize ROS, reduce lipid peroxidation, and suppress pro-inflammatory cytokine signaling (e.g., TNF-α, IL-6, and NF-κB), thereby creating a biochemical environment conducive to sustained spermatogenesis and endocrine balance (A. A. Ahmed, 2021; B. H. Ali et al., 2020; Imbabi et al., 2023; Khanzadeh et al., 2021; Figure 2). This integrated protective profile provides a mechanistic rationale for the broad spectrum of reproductive benefits associated with GA supplementation.
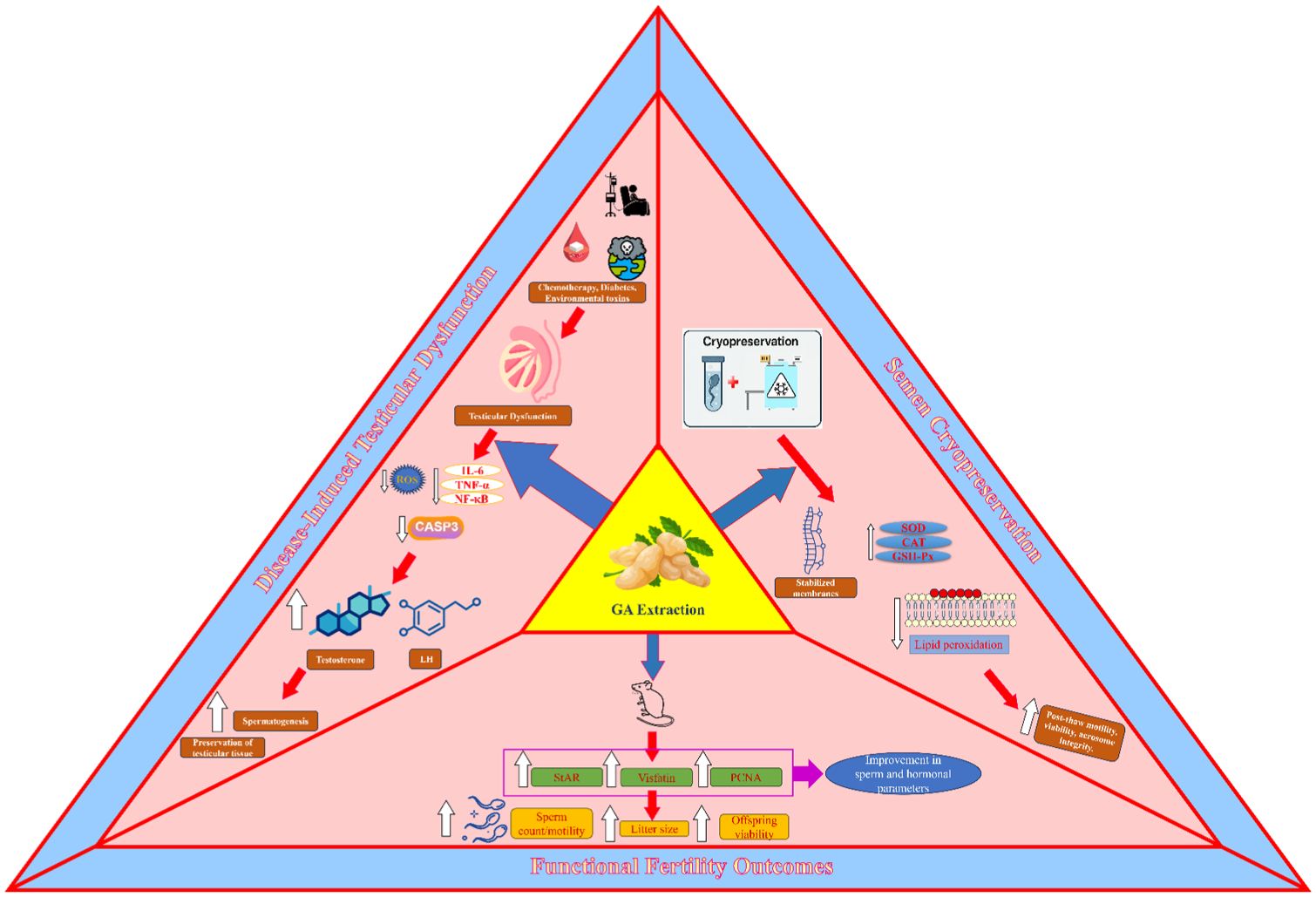
Protective Roles of GA in Male Reproductive Health. This Schematic Summarizes the Multifaceted Protective Actions of Gum Acacia (GA) on Male Reproductive Function. In Models of Disease-Induced Testicular Dysfunction, GA Alleviates Oxidative Stress, Inflammation, and Apoptosis Triggered by Chemotherapy, Diabetes, or Environmental Toxins, Thereby Preserving Testicular Architecture, Supporting Spermatogenesis, and Restoring Testosterone and Luteinizing Hormone (LH) Levels. In Semen Cryopreservation, GA Functions as a Plant-Based Cryoprotective Agent by Stabilizing Sperm Plasma Membranes, Reducing Lipid Peroxidation, and Enhancing Post-Thaw Motility, Viability, and Acrosome Integrity. In Animal Models, GA Supplementation Improves Sperm Quality, Testicular Weight, Androgen Production, and Reproductive Outcomes, Including Larger Litter Sizes and Greater Offspring Viability. Despite These Promising Effects, Some Contradictory Findings Highlight the Need for Dose Optimization, Species-Specific Evaluation, and Standardized Experimental Protocols Before Translation Into Clinical Practice.
From a translational perspective, these findings carry potential implications for managing male infertility associated with oxidative stress, metabolic disorders, or environmental exposures. If validated in higher animal models and eventually in clinical studies, GA could be developed as a dietary supplement, adjunct therapeutic, or even as a bioactive component of semen preservation strategies. However, several critical gaps remain unresolved: the optimal therapeutic dose, the duration of administration required to achieve long-term benefits, the potential for cumulative toxicity at high intake levels, and the variability in response between different species or genetic backgrounds. Equally important is the need to investigate whether GA interacts with conventional fertility drugs, hormonal therapies, or assisted reproduction protocols, as such interactions could either potentiate or interfere with treatment outcomes.
Future Directions and Research Opportunities
Despite the promising evidence regarding the protective and fertility-enhancing effects of GA, several critical gaps remain that warrant systematic exploration. Future research should address the following key directions:
Mechanistic Insights
Future studies should clarify the molecular pathways underlying GA’s antioxidant, anti-inflammatory, and steroidogenic effects. Multi-omics approaches such as transcriptomics, proteomics, and metabolomics can provide a comprehensive understanding of its impact on spermatogenesis, endocrine regulation, and energy metabolism, while the potential gut–testis axis mediated by GA’s prebiotic activity should also be investigated.
Standardization and Dose Optimization
Dose–response studies are required to define the optimal therapeutic range of GA that maximizes benefits while minimizing potential toxicity. Clear differentiation between Acacia senegal and other Acacia species, along with standardized extraction and formulation protocols, will be necessary to ensure consistency and reproducibility across studies.
Advanced Preclinical Models
Future investigations should first expand and deepen preclinical research, extending beyond rodent models to higher mammals with reproductive systems more comparable to humans. In particular, well-characterized models of idiopathic infertility should be incorporated, and long-term safety, fertility outcomes, and potential transgenerational effects should be systematically evaluated. In parallel, the reproductive effects of different types of smoke exposure (e.g., cigarette, waterpipe, and environmental smoke) warrant detailed investigation in controlled preclinical models to address existing knowledge gaps and establish a robust evidence base before translation to human studies.
Synergistic and Comparative Studies
The potential interactions of GA with established antioxidants, hormonal therapies, and assisted reproductive technologies (ART) require careful study. Comparative evaluations with conventional fertility supplements, as well as its role in semen cryopreservation focusing on fertilization and embryonic outcomes, will further clarify its translational utility.
Clinical Translation
Well-designed randomized controlled trials are urgently needed to validate GA’s safety, tolerability, and efficacy in men with oxidative stress–induced, diabetic, or idiopathic infertility. Its potential applications as a dietary supplement, functional food additive, or adjunct in ART should be systematically explored, while accounting for sex-specific and population-specific responses influenced by genetics, diet, and lifestyle.
Conclusion
GA emerges as a promising natural modulator of male reproductive health, with preclinical evidence supporting its multifaceted protective effects. GA exerts antioxidant, anti-inflammatory, and anti-apoptotic actions, preserves testicular architecture, enhances spermatogenesis, and improves hormonal balance. Its beneficial effects are evident across diverse pathological contexts, including diabetes, chemotherapy-induced toxicity, and environmental or lifestyle-related reproductive stress. Moreover, GA shows potential as a plant-based cryoprotectant for semen preservation, demonstrating both structural and functional support for sperm cells. Despite these encouraging findings, the current evidence is predominantly preclinical, limited in scope, and occasionally inconsistent, highlighting the need for caution.
Footnotes
Acknowledgements
During the preparation of this work, the authors used Gemini to improve the quality of the text of the article, as well as to write the “Future Directions and Research Opportunities” section, and also to check. After using Gemini, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Author Contributions
AMF was responsible for overall supervision. AMF drafted the manuscript, which was revised by AMF.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable, as no new data were generated or analyzed during this study.
