Abstract
Since routine sperm analysis cannot detect molecular-level defects, efforts continue to find additional markers for selecting the “highest quality” sperm. This study analyzes ejaculates from normozoospermic and non-normozoospermic men, including teratozoospermics (T), asthenoteratozoospermics (AT), and oligoasthenoteratozoospermics (OAT), by two approaches. The first is distribution of sialic acid residues, molecules critical for sperm migration and survival in female reproductive tract, using lectins with distinct binding specificities (SNA I, MAL I, WGA). The second is the localization of membrane proteins, tetraspanins (CD9, CD63, CD81, CD151), which are involved in regulation of numerous cellular processes. Flow cytometry showed no significant differences in overall lectin positivity between diagnostic groups. However, in relation to sperm viability based on propidium iodide (PI) staining, SNA I binding was significantly higher in PI-low sperm across all groups. MAL I labeling increased in PI-intermediate and PI-high sperm, particularly in T and OAT ejaculates. WGA positivity decreased in the viable sperm of non-normozoospermic groups. Immunofluorescence analysis revealed tetraspanins in a subset of spermatozoa in both groups, most frequently in the acrosome and/or equatorial region, the primary site of sperm-egg fusion. Our results suggest that lectin-staining combined with PI could provide valuable insights into sperm status. SNA I and MAL I-based analysis could support early detection of potentially poor-quality sperm, that is, negative selection.
Introduction
Male infertility is a rising issue mainly due to sperm abnormalities from environmental, genetic, and lifestyle factors (Craig et al., 2017; Y. Ma et al., 2019; Sciorio et al., 2024; Skakkebaek et al., 2016). Despite significant progress in reproductive technologies, the effectiveness of human reproductive treatments in terms of optimal method for sperm selection has not improved dramatically (Pedrosa et al., 2020). While routine analysis of male ejaculate cannot detect sperm defects at the molecular level, there is an effort to find suitable markers to help detect hidden problems that may be behind infertility. Reflecting the current state of knowledge and our previous analyses in the fields of glycobiology (Sečová et al., 2024) and the physiology of tetraspanins in mammalian spermatozoa (Jankovičová, Neuerová, et al., 2020; Jankovičová, Sečová, et al., 2020), we tested two approaches to the evaluation of human spermatozoa in ejaculates from normozoospermic (N) and non-normozoospermic (non-N) patients. The first approach involved mapping the sialic acids (Sia), the terminal part of the sperm surface structure, called the glycocalyx, which contains glycoproteins, glycolipids, and glycosylphosphatidylinositol-anchored glycoproteins. Glycans are synthesized during spermatogenesis, further acquired and modified during epididymal maturation, via seminal plasma during ejaculation, and also in the process of sperm capacitation in the female reproductive tract (Pini et al., 2018; Tecle & Gagneux, 2015). Glycocalyx, especially Sia, is important for sperm motility, progression, and penetration through cervical mucus (Tollner et al., 2012), suppression of the female reproductive tract immune system (Tecle et al., 2019) and masking receptors for zona pellucida binding (Lassalle & Testart, 1994).
The second approach was based on detecting the distribution of proteins, tetraspanins that are integral components of the sperm membranes. Tetraspanins are crucial regulators of cell physiology and are involved in many cellular processes, including fertilization. They can associate with a range of other proteins and with each other, forming a multimolecular network called the tetraspanin web, thereby coordinating protein interactions, adhesion, signaling, and membrane dynamics (Hemler, 2001). CD9, CD81, and CD151 have been previously described in bull and also in human spermatozoa (Antalíková et al., 2015; Jankovicova et al., 2016, 2020), and our recent study documented the presence of CD63 on bull sperm (Jankovičová et al., 2024). Several studies on CD9 and CD63 have reported the possible involvement of tetraspanins in human sperm fertility (Al-Dossary et al., 2015; García-Herrero et al., 2010; Martins et al., 2020; Salvolini et al., 2013). Structural modeling of the CD9–CD81 interaction in human spermatozoa suggested that CD81 may regulate dynamic changes within the membrane, whereas CD9 appears to stabilize the web (Frolikova et al., 2018).
Tetraspanins are important markers of extracellular vesicles (EVs). These nanostructures can transfer proteins, lipids, and nucleic acids between cells, thus mediating intercellular communication in many physiological and pathological processes in mammals. In males, they are involved in the development, maturation, and maintenance of sperm function. With growing experimental data, extracellular vesicles are increasingly being considered for use in reproductive technologies to improve infertility treatment.
This study aimed to investigate the distribution of: (i) sialic acid moieties on the surface of spermatozoa using lectins with different binding specificities (SNA I, MAL I, and WGA); (ii) tetraspanins within the sperm membranes and/or extracellular vesicles using antibodies (anti-CD9, CD63, CD81, CD151) on spermatozoa in ejaculates from normozoospermic and non-normozoospermic patients.
Materials and Methods
Sperm Preparation
Semen specimens were obtained after a period of abstinence of 2–5 days, collected in sterile containers via masturbation. The samples were liquefied at room temperature (RT) within 30 minutes. Ejaculates were classified according to World Health Organization manual (World Health Organization [WHO], 2010). All methods were carried out in accordance with relevant guidelines and regulations. The informed consent was obtained from all subjects.
Lectin Staining
Sialic acids were analyzed using lectins conjugated with fluorescein isothiocyanate (FITC) (Vector Laboratories): Maackia amurensis lectin I (MAL I), Sambucus nigra lectin I (SNA I), and Wheat germ agglutinin (WGA) at final concentration 1 μg/mL. The specificity of these lectins was determined in the study by Bojar et al. (2022) using a combination of machine learning algorithms and expert annotation. Flow cytometry was performed on 7 normozoospermic (N), 11 teratozoospermic (T), 6 asthenoteratozoospermic (AT), and 9 oligoasthenoteratozoospermic (OAT) ejaculates. The SpermFlowEx Kit (Exbio) was used to evaluate sperm viability and acrosome integrity. Three aliquots of sperm ejaculates diluted to 5×106 of sperm/mL with PBS were incubated with one of three lectins for 30 min at RT in the dark, then with propidium iodide (PI) for 20 min at RT and analyzed using BD FACS Canto II (BD Biosciences). Data was processed using Floreada.io (free web-based software). The fluorochromes have no emission overlap (no compensation was applied). A minimum of 10,000 events was recorded for each sample. Two-dimensional plots of FITC versus PI fluorescence events were drawn, and individual subpopulations were divided by gates: PI-negative (viable), PI-low (pre-moribund), PI-intermediate (moribund), and PI-high (dead) and quadrants (lectin-positive and negative events) (Figure S1, S2), quantified and statistically evaluated. In addition, reaction patterns of lectins were evaluated using fluorescence microscopy. Both intact (non-permeabilized) sperm and sperm with membranes permeabilized by acetone-methanol (1:1) were incubated with lectins as described above, then washed and mounted with Fluoroshield-DAPI. Slides were examined by epifluorescence microscopy at 400× magnification. Images were recorded via a Leica DFC340 FX digital camera and processed via Leica Advanced Fluorescence software (Leica Microsystems). Representative images are shown.
Tetraspanin Staining
Tetraspanins were analyzed by indirect fluorescent assay using the primary antibodies: anti-CD9 mouse monoclonal antibody (C-4, sc13118, Santa Cruz Biotechnology, 0.2 mg/mL, dilution 1:50), anti-CD81 rabbit polyclonal antibody (NBP2-20564, Novus Biologicals, 1.01 mg/mL, dilution 1:100), anti-CD151 rabbit polyclonal antibody (ABIN6748303, antibodies-online.com, 0.5 mg/mL, dilution 1:50), anti-CD63 goat polyclonal antibody (ABIN1440014, antibodies-online.com, 3 mg/mL, dilution 1:300), rabbit IgG isotype control (Novus Biological, 5 mg/mL dilution 1:500), mouse IgG1 isotype control (EXBIO, 1 mg/mL dilution 1:100); and secondary antibodies: goat anti-mouse IgG (H+L) Alexa Fluor 488 (1:500), donkey anti-rabbit IgG (H+L) Alexa Fluor 488 (Abcam) (1:500), donkey anti-goat IgG (H+L) Alexa Fluor 488 (Invitrogen) (1:500). We analyzed samples from 8 normozoospermic (N) and 6 non-normozoospermic (non-N) ejaculates, which were frozen using CryoSperm™ Sperm Freezing Medium (CooperSurgical) at a 1:1 ratio, a medium commonly used for insemination or IVF samples. We also analyzed sperm from 6 normozoospermic ejaculates processed by density gradient centrifugation (DGC) using PureCeption™ 40% upper phase and 80% lower phase (SAGE Media™, CooperSurgical). Spermatozoa from all ejaculates were prepared as smears, permeabilized with acetone-methanol (1:1), and then blocked with Super Block® Blocking Buffer (Thermo Scientific) for 1 hour at 37 °C. Primary antibodies were applied overnight at 4 °C and secondary antibodies for 1 h in the dark at RT. The presence of acrosomes was monitored by Peanut agglutinin (PNA)-TRITC (Vector Laboratories, 5 mg/mL, dilution 1:300) staining for 30 min in the dark at RT. Slides were mounted with Fluoroshield-DAPI, evaluated and processed as previously stated. The staining patterns were quantified by scoring a minimum of 200 spermatozoa for each sample. Representative results are shown.
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 8.0.1 (GraphPad Software, Inc., La Jolla, CA, USA). Normality of data distribution was assessed with the Shapiro-Wilk test. Depending on the distribution, repeated measures (RM) one-way ANOVA with Holm-Sidak’s multiple comparisons test or the Friedman test was applied for lectin analyses. Comparisons of sperm proportions between diagnostic groups were analyzed using one-way ANOVA or Kruskal–Wallis test, as appropriate. Data in the table are presented as mean ± SEM (%), while median and interquartile range (25th–75th percentile) is shown in box plots. For tetraspanin analyses, normality was confirmed by the Shapiro–Wilk test, and either a t-test or Mann–Whitney test was used. Post hoc pairwise comparisons were adjusted using Bonferroni correction. A p-value <0.05 was considered statistically significant.
Results
Analysis of Sperm Sialic Acid Moieties by Lectins
Flow cytometric analysis revealed no significant differences in overall lectin positivity between sperm from normozoospermic and non-normozoospermic (T, AT, OAT) ejaculates (Figure 1), likely due to substantial individual variability.
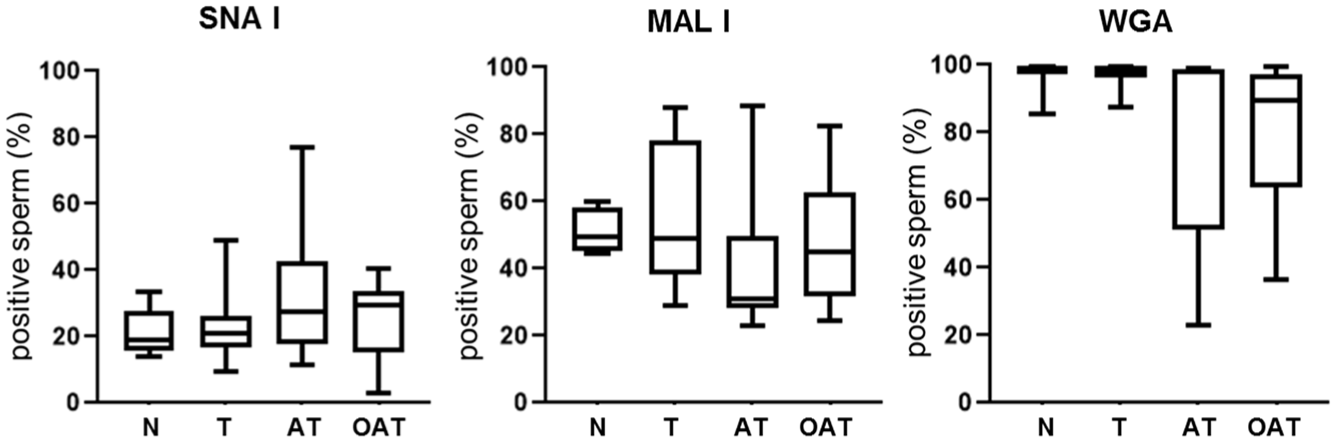
Comparison of Lectin Positivity in Normozoospermic and Non-Normozoospermic Ejaculates.
A significant difference in the labeling of individual lectins (Figure 1) was observed when evaluated simultaneously with PI labeling. Based on PI staining, spermatozoa were categorized as follows: PI-negative (viable), PI-low (pre-moribund), PI-intermediate (moribund), and PI-high (dead). The proportions of these subpopulations within each diagnostic group are shown in Table 1.
Distribution of Sperm Subpopulations Based on PI Staining in Ejaculates from Different Diagnostic Groups.

Note. N, normozoospermia; T, teratozoospermia; AT, asthenoteratozoospermia; OAT, oligoasthenoteratozoospermia. Significant differences were found compared to the corresponding sperm subpopulation in normozoospermic ejaculates.
p = 0.0428; *** p = 0.0002; **** p < 0.0001.
SNA I positivity was significantly higher in pre-moribund sperm compared to viable sperm across all diagnostic groups (N: p = 0.0225; T: p < 0.0001; AT: p = 0.0021; OAT: p = 0.0092) and was also elevated in the moribund and dead subpopulations in T ejaculates (p = 0.0102; p = 0.0494). MAL I labeling was significantly increased in moribund (p = 0.0225) and especially in dead sperm (p = 0.0005) in normozoospermic samples. A marked increase in MAL I positivity was observed across all sperm subpopulations in T (p < 0.0001) and OAT (pre-moribund: p = 0.0025; moribund: p = 0.0026; dead: p = 0.0022) ejaculates, while in AT samples significance was restricted to pre-moribund (p = 0.0021) and moribund (p = 0.0437) sperm. WGA positivity remained uniformly high in normozoospermic ejaculates (93%–97%), but was significantly reduced in viable sperm compared to pre-moribund and moribund subpopulations in T (p = 0.0001; p = 0.0002), AT (p = 0.0105; p = 0.0048), and OAT (p = 0.0004; p = 0.0115) ejaculates (Figure 2).
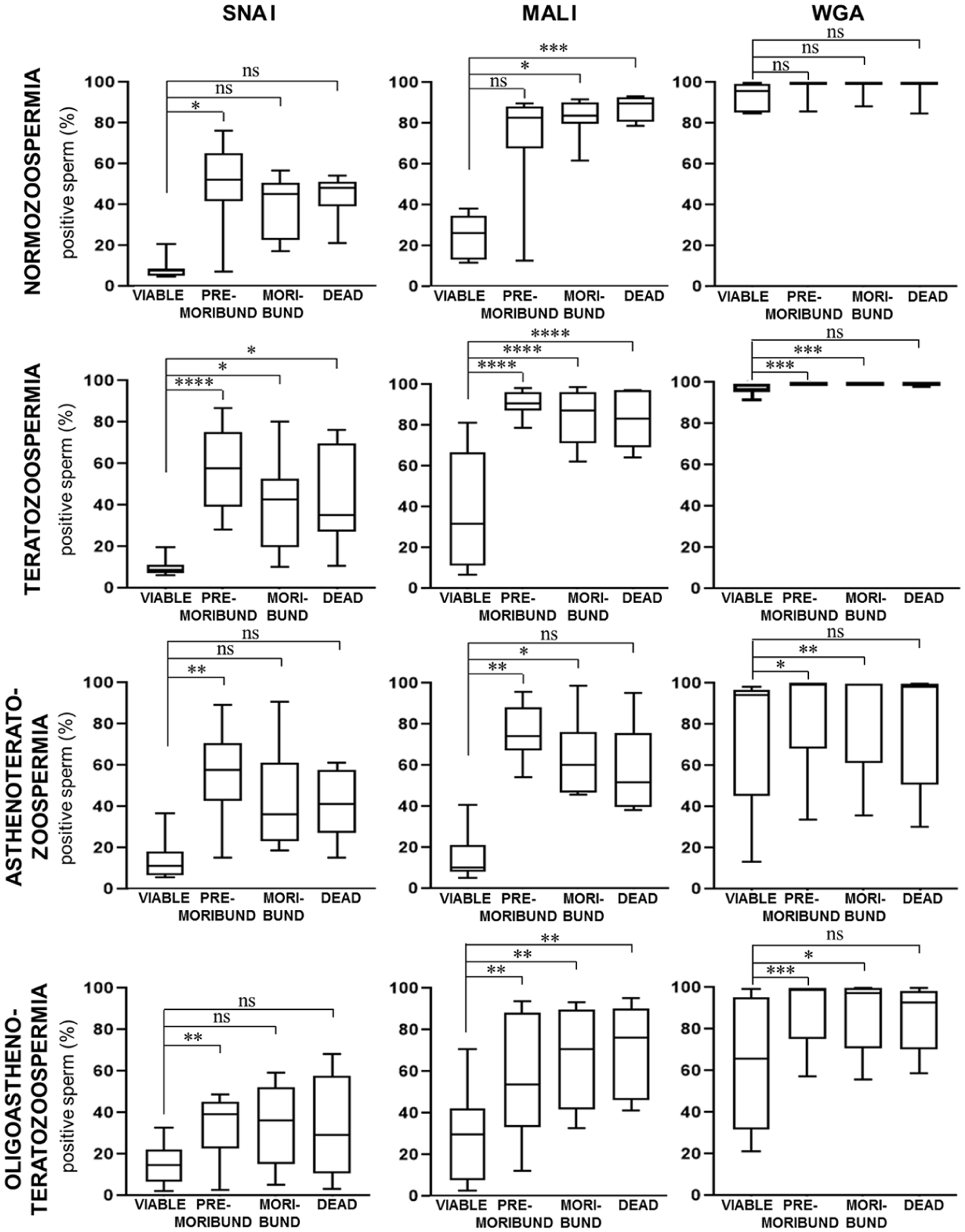
The Proportion of Sperm labeled with SNA I, MAL I, and WGA Lectins and Propidium Iodide Based on Cytometric Analysis of Normozoospermic and Non-Normozoospermic Ejaculates.
The flow cytometric analysis also included the detection of acrosomal integrity using a commercial kit containing an antibody against an intra-acrosomal protein, detectable on sperm with damaged membrane. The obtained data showed the proportion of sperm with intact acrosomes as follows: N 49.71 ± 3.94%; T 53.73 ± 3.75%; AT 42.30 ± 7.12% and OAT 42.86 ± 3.08%, which corresponds with the proportion of viable sperm reported in Table 1.
Considering that lectin binding sites are located on glycans both on the sperm surface and under the plasma membrane, we performed lectin cytochemistry to analyze labeling patterns on sperm with non-permeabilized membranes (to assess surface-accessible glycans, analogous to flow cytometry) as well as on sperm with permeabilized membranes. In the case of SNA I, almost the entire sperm population was stained in the acrosomal region and throughout the head and tail after permeabilization. Similarly for MAL I, most of the permeabilized spermatozoa showed a signal in the acrosomal region with an intensely stained equatorial region. The signal in the sperm tail was observed independently of permeabilization. In the case of WGA, the same reaction patterns were observed in permeabilized and non-permeabilized spermatozoa, namely, the acrosomal region, the entire head, and the tail (Figure 3).
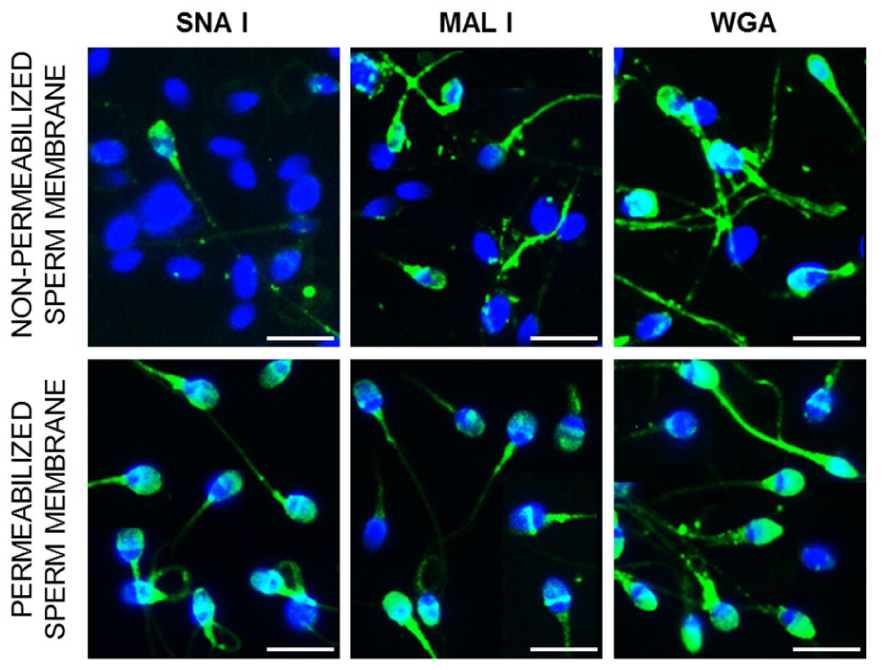
Lectin Labeling of Sperm with Non-Permeabilized and Permeabilized Membranes.
Analysis of the Tetraspanins Distribution
Given the presumed role of CD9 as a tetraspanin web stabilizer capable of interacting with other molecules, we identified its potential partners from the tetraspanin family in the STRING database using CD9_HUMAN (UniProt: P21926) as a query protein (Szklarczyk et al., 2015). Provided protein-protein network suggested that the tetraspanins CD9, CD81, CD151 and CD63 may function as a complex in sperm (Figure 4). We assumed that diagnosis could influence the distribution of individual tetraspanins or the complex, and therefore we investigated their localization in normozoospermic and non-normozoospermic ejaculates. Immunofluorescence analysis showed the occurrence of several sperm subpopulations based on tetraspanin labeling on the sperm head and tail. In all analyzed tetraspanins, we observed two predominant reaction patterns on the sperm head, where the signal was detectable in the equatorial region of the acrosome or spread over the acrosome in both normozoospermic and non-normozoospermic ejaculates (Figure 5). We detected CD9 labeling in the equatorial region (N 29.83 ± 8.32%, non-N 20.80 ± 6.86%) and throughout the acrosome (N 10.72 ± 7.93%, non-N 5.32 ± 3.39%). In the case of CD81 the signal was detectable predominantly in the equatorial region (N 50.99 ± 4.85%, non-N 36.32 ± 2.32%) and, to a lesser extent, in the tail (N 13.43 ± 1.77%, non-N 14.97 ± 3.03%) or exclusively in the tail (N 8.97 ± 1.68%, non-N 8.89 ± 0.81%). The presence of CD151 was detected in the equatorial region (N 37.03 ± 5.34%, non-N 27.82 ± 5.14%) or the whole acrosomal area (N 38.75 ± 4.51%, non-N 35.59 ± 4.68%). For CD63 tetraspanin, in addition to the labeled tails (100%), an extra signal was observed in the equatorial and post-equatorial region (N 39.66 ± 6.39%, non-N 23.18 ± 2.69%), over the entire acrosome (N 9.55 ± 1.53%, non-N 10.71 ± 3.25%), and the whole sperm head (N 31.75 ± 7.16%, non-N 35.08 ± 3.26%). There was no significant difference in the total positivity and individual reaction patterns of tetraspanins between N and non-N ejaculates, and similarly, between sperm selected by DGC separation and non-separated sperm. It is worth noting that a high degree of individual variability was observed, especially for CD9-positivity, which showed a range of 13% to 75% in normozoospermics and 0% to 51% in non-normozoospermics, regardless of sperm separation (Figure 5).

Protein–Protein Interaction Network of Tetraspanins CD9, CD63, CD81, and CD151 Generated by STRING Analysis (CD9_HUMAN, Uniprot ID: P21926).
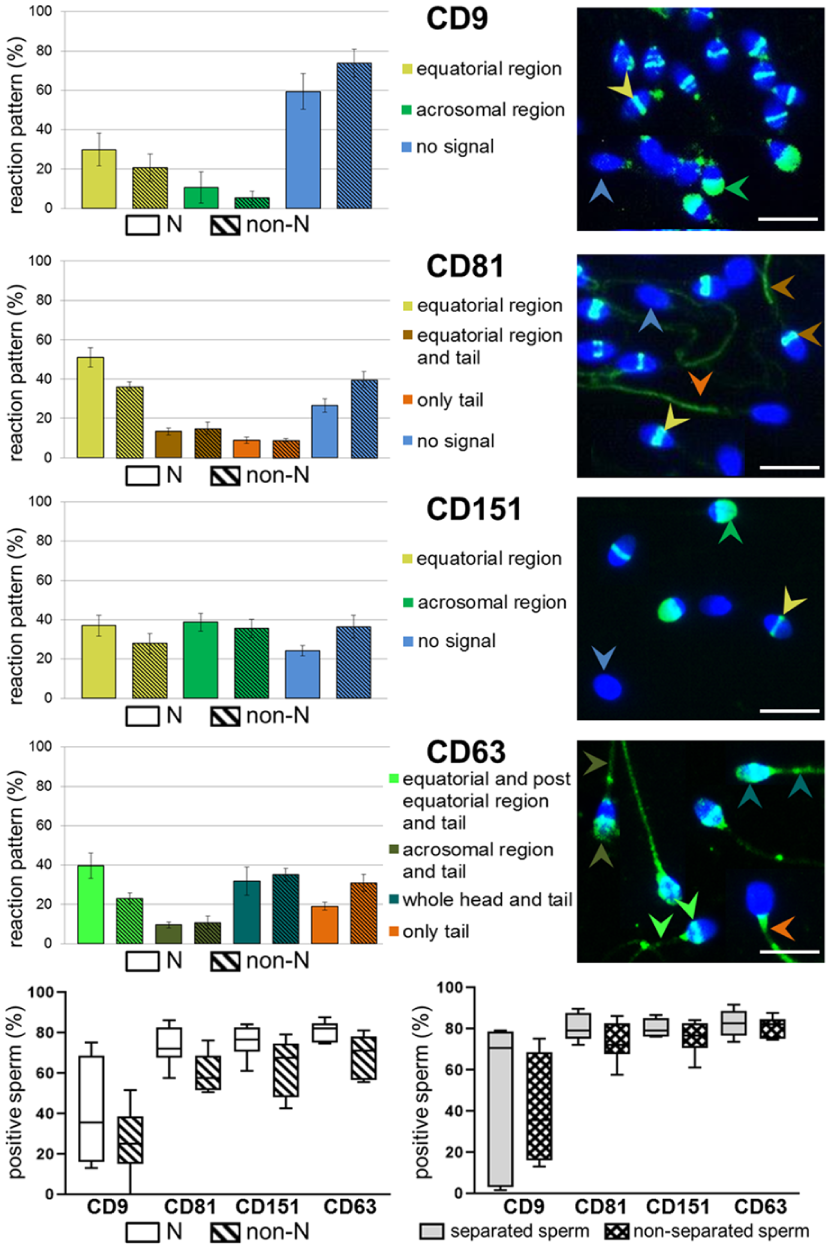
Tetraspanin-Positive Spermatozoa: Proportion and Distribution of Reaction Patterns.
Discussion
Human ejaculate is made up of a heterogeneous pool of spermatozoa. According to WHO (2010), normal semen should contain 39×106 spermatozoa per ejaculate, 40% of motile sperm, 50% of viable sperm, and 4% of sperm with normal morphology. Neglecting sperm count, motility, and viability, the “reference sample” itself may contain 4% of morphologically “normal” sperm, there is a fine line between normozoospermic and non-normozoospermic ejaculates, which greatly complicates any comparative analysis with problematic ejaculates aimed at finding a marker defining “sperm quality.” Although sperm morphology remains a valuable indicator of sperm function, sperm with abnormal morphology may still be able to fertilize an egg (Check et al., 1992). Consequently it is essential to understand the molecular mechanisms and focus on detecting subcellular changes in sperm to improve the diagnosis of idiopathic male infertility and assisted reproductive techniques (Bracke et al., 2018; Galimov et al., 2022; Nikolettos et al., 1999).
The terminal components of the sperm glycocalyx is critical for sperm maturation, capacitation, and fertilization; its alteration or disruption has been associated with male infertility (F. Ma et al., 2012; Yi et al., 2023). In addition, residues of sialic acid (Sia) significantly contribute to the negative surface charge of sperm, which has already been utilized in sperm separation techniques (Ainsworth et al., 2005). Devices such as the Cell Sorter-10 (CS-10) and the Felix device have been developed for this purpose (Fleming et al., 2008; Villeneuve et al., 2023; Zhang et al., 2024).
Several studies have emphasized the necessity of thorough validation of these methods using different sperm samples (Ainsworth et al., 2005; Simon et al., 2016) and highlighted the need to consider the complex relationship between sperm membrane charge and functional properties (Zhang et al., 2024). The sperm surface charge (Ainsworth et al., 2005, 2007; Fleming et al., 2008) arises from a complex glycan structure characterized by variable sugar isomers, branching patterns, linkage types, and terminal modifications (Sharon & Lis, 2004), as well as by the presence of different sialic acid variants (Sato & Kitajima, 2021; Varki, 2017). To investigate these structures in more detail, we employed lectins, glycan-binding proteins that are capable, to a certain extent, of discriminating among these structural configurations. We applied three lectins with different binding preferences: WGA with the broadest specificity, binding Sia and N-acetylglucosamine-containing glycans, MAL I binding α2-3-sialylated N-acetyllactosamine and terminal 3-O sulfated galactose on N-acetyllactosamine, and SNA I, exclusively recognizing α2-6-sialylated N-acetyllactosamine (Bojar et al., 2022). Flow cytometric analysis comparing normozoospermic and non-normozoospermic ejaculates revealed no significant differences in overall lectin positivity, likely due to substantial individual variability of ejaculates. However; a considerable difference in the labeling of individual lectins has been observed when evaluated simultaneously with PI labeling. All non-normozoospermic (T, AT, OAT) showed a significantly higher proportion of sperm with bound lectins (SNA I, MAL I, WGA) in the PI-low (pre-moribund) compared to the PI-negative (viable) subpopulation. In N ejaculates, a significant difference between these categories was observed only for SNA I binding. It appears that SNA I and MAL I are more sensitive indicators of sperm status (viable vs. pre-moribund/moribund/dead). Based on observations of sperm following permeabilization, which exposes glycans under the plasma membrane to lectin binding, it should be considered that SNA I and MAL I may also recognize glycan structures located in these regions. We propose that lectin-based analysis could assist in the early detection or preliminary exclusion of potentially poor-quality or unsuitable sperm, that is, in negative sperm selection as was previously described for PNA lectin in cattle (Odhiambo et al., 2011). Although WGA displays consistently high values across all normozoospermic subpopulations, reduced positivity of viable sperm in all non-normozoospermic ejaculates suggests that additional factor/s probably affect WGA binding to sperm. The interquartile range in AT and OAT compared to T and N suggests that these factors could be associated with sperm motility.
Mammalian sperm must exhibit a specific molecular configuration necessary for the efficient execution of reproductive functions. These molecules are primarily involved in DNA protection (Champroux et al., 2016; Ward, 2010), ensuring sperm motility, signaling pathways that initiate sperm capacitation, membrane reorganization, and the acrosome reaction (Puga Molina et al., 2018). Equally important are molecules functioning as surface receptors and molecules maintaining the integrity of sperm cells and their membranes. While tetraspanins are molecules with essential roles in the formation, stabilization, and organization of cell membrane complexes, the second aim of our study was to compare the tetraspanin profile of spermatozoa from normozoospermic and non-normozoospermic ejaculates. We analyzed the distribution of CD9, CD63, CD81, and CD151. According to a STRING analysis performed using CD9_HUMAN (Uniprot identifier: P21926) as the query protein (Szklarczyk et al., 2015), these tetraspanins are directly linked and likely interact functionally. Such associations support their proposed roles in gamete maturation, fertilization, and embryo development (Jankovičová, Neuerová, et al., 2020; Jankovičová, Sečová, et al., 2020).
We observed several reaction patterns without significant differences in abundance and proportion of sperm positivity when comparing normozoospermic and non-normozoospermic ejaculates. As mentioned above, even normozoospermic ejaculate is highly heterogeneous; we attempted to select “good quality sperm” using the DGC method and characterize them in terms of tetraspanin profile. We did not observe any significant differences in localization of CD9, CD81, CD151, and CD63 between spermatozoa separated by DGC from the non-separated sperm. It is probably due to the high individuality of each ejaculate, and the fact that this method relies on sperm motility may not accurately reflect the condition of the sperm membrane, as even sperm with considerable morphological defects can still display progressive motility (Sutovsky et al., 2024). However, it is worth noting that all analyzed tetraspanins were found in the equatorial region of the sperm head. This region is considered the primary site for sperm-egg membrane fusion (Yanagimachi & Noda, 1970), playing a critical role in maintaining stability and facilitating protein interactions necessary for successful fertilization. The equatorial region remains intact until the acrosome reaction, at which point proteins from other regions are relocalised as it was described for Izumo1 (Satouh et al., 2012; Sebkova et al., 2014), CD46 and integrins (Frolikova et al., 2016) or tetraspanins CD81 and CD9 (Glander & Schaller, 1993; Jankovicova et al., 2016). We previously suggested the possible role for CD81 in the regulation of dynamic changes and CD9 in the stabilization of sperm plasma membrane during gamete interaction (Frolikova et al., 2018). We have shown the localization of CD151 at this primary fusion site in human and bull sperm (Jankovicova et al., 2020). Based on this knowledge, we assume that the shared presence of tetraspanins in the equatorial region seems to be a positive sign in terms of stabilizing this critical area.
In the part of the sperm population, we observed CD9, CD151, and CD63 additionally in the acrosomal region of the head. This is interesting in the context of our findings regarding CD9 in bull sperm (Antalíková et al., 2015, 2022; Jankovičová et al., 2024). In fresh ejaculate, CD9 was detected in more than 75 % of sperm in this region (Antalíková et al., 2015) and changing localization of CD9 in the epididymal spermatozoa toward the cauda was found. In the caput, the signal was predominantly observed in the equatorial region; however, in most of the caudal sperm, the signal was spread over the acrosome (6.5-fold higher proportion than in the caput) (Jankovičová et al., 2024). We hypothesized that CD9 in the equatorial region of spermatozoa entering the epididymis is derived from spermatogenesis in the testis (Antalíková et al., 2022), and during passage through the epididymis, further transport of CD9 to the acrosomal region probably via EVs occurs. Transfer of specific proteins through CD9-EVs to bull sperm has been already documented (Belleannée, 2015; Caballero et al., 2013; Frenette et al., 2010; Sullivan & Saez, 2013). Our results did not show the significantly different distribution of CD9 in the normozoospermic and non-normozoospermic human ejaculates. On the other hand, considerable individual variability in CD9 positivity was observed independent of diagnosis. We hypothesized that these differences might reflect a distinct association of CD9-EVs with sperm during maturation processes. Given the existence of sperm compartmentalization, where specific areas are responsible for specific functions, it is also important to consider not only the presence of tetraspanins on sperm but also their specific reaction pattern and its representation. The role of CD63 has been previously suggested in humans (García-Herrero et al., 2011). Fresh sperm from infertile men undergoing ICSI showed overexpression of CD63 transcripts in the non-pregnant compared to the pregnant group. A different distribution of CD9 and CD63 in prostasomes was found in normozoospermic and oligozoospermic men (Janković et al., 2021). In our recent study, we detected CD63 in bull spermatozoa from different parts of the epididymis, epididymal EVs, and ejaculated spermatozoa along the entire length of the sperm tail and partially in the post-equatorial region of the sperm head (Jankovičová et al., 2024). We could therefore speculate that the patterns found in this study could indicate the presence of CD63-positive EVs also on human spermatozoa. Several studies have suggested EVs as a promising diagnostic marker of male fertility (Gervasi et al., 2020; Giacomini et al., 2020; Lange-Consiglio et al., 2022; Roca et al., 2006, 2022; Rodriguez-Martinez & Roca, 2022). EVs originating from different parts of the male reproductive tract can be internalized by sperm (Barrachina et al., 2022), affecting their maturation and regulating their functions (Murdica, Giacomini, et al., 2019; Rimmer et al., 2021), sperm motility acquisition, oxidative stress reduction, and the ability to undergo capacitation and acrosome reaction (Machtinger et al., 2016; Murdica, Giacomini, et al., 2019). CD63-EVs from human semen improved sperm function (Luo et al., 2024). Interestingly, EVs from seminal plasma of normozoospermic, but not asthenozoospermic men, have been found to increase sperm motility and induce capacitation (Barceló et al., 2018; Murdica, Cermisoni, et al., 2019).
Considering our results in a broader context, spermiogram abnormalities are closely linked to oxidative stress, which contributes to membrane lipid peroxidation, protein modifications, and increased DNA fragmentation (Kaltsas, 2023; Wang et al., 2025). DNA fragmentation is influenced by multiple intrinsic and extrinsic factors, including defective spermatogenesis, apoptosis, varicocele, infections, diabetes, obesity, environmental toxins, smoking, and radiation (Andrabi et al., 2024; Panner Selvam et al., 2021). As mentioned above, oxidative stress not only damages DNA (Mannucci et al., 2021; Muratori et al., 2015) but also induces significant changes in the sperm proteome, affecting proteins essential for sperm function. Proteomic analyses have demonstrated differential expression of proteins under oxidative stress, including proteins involved in motility, membrane function, and other critical processes (Intasqui et al., 2013; Sharma et al., 2013). Although these studies do not directly map topographical redistribution of proteins on the sperm membrane, the results indicate that oxidative stress alters the expression of membrane-localized and membrane-associated proteins, as well as components of structural and signaling complexes, which can lead to functional changes in protein distribution within the cell. Regarding our future research directions, the simultaneous analysis of oxidative stress and DNA fragmentation may enable a more comprehensive assessment of membrane sperm quality markers.
In conclusion, our results suggest an initial framework for the use of SNA I and MAL I lectins in negative sperm selection. The shared presence of tetraspanins in the equatorial region of the sperm head could be considered a positive factor in stabilizing the area crucial for successful sperm-egg fusion. It is becoming increasingly apparent that further consideration should be given to the evaluation of human semen. We believe that our findings could contribute to the development of procedures for more accurate assessment of human sperm quality at the subcellular level. Furthermore, given the declining fertility of other animal species, including livestock, these findings may also be of interest in studying the sperm physiology of other mammalian species.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883261420405 – Supplemental material for Sialic Acids and Tetraspanins: An Insight into their Potential as Indicators of Human Sperm Quality
Supplemental material, sj-docx-1-jmh-10.1177_15579883261420405 for Sialic Acids and Tetraspanins: An Insight into their Potential as Indicators of Human Sperm Quality by Katarína Michalková, Petra Sečová, Jana Jankovičová, Ľubica Horovská, Žaneta Dzurillová, Michal Ješeta and Jana Antalíková in American Journal of Men's Health
Footnotes
Acknowledgements
The authors would like to thank their colleagues Pavla Postlerová, Kateřina Komrsková, Veronika Kraus, and Ondřej Šimoník from the Laboratory of Reproductive Biology at the Institute of Biotechnology of the Czech Academy of Sciences (BIOCEV) for their valuable discussion on some methodological aspects of the part of the study, and Mrs. Beáta Holíková from Nitra for her skillful technical assistance.
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by Ethics Commission of the Nitra Self-Governing Region, Slovak Republic (reference number 1-2/2025) and by the Ethics Committee of the University Hospital Brno, Czech Republic (reference number 11-110123/EK).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Author Contributions
K.M.: Investigation, Data curation, Formal analysis, Methodology, Writing – review & editing; P.S.: Investigation, Data curation, Formal analysis, Methodology, Writing – review & editing; J.J.: Funding acquisition, Investigation, Writing – review & editing; Ľ.H.: Methodology; Ž.D.: Data curation, Formal analysis; M.J.: Data curation, Project administration, Writing – review & editing; J.A.: Conceptualization, Supervision, Project administration, Writing – original draft, review & editing. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Grant Agency of the Ministry of Education, Science, Research and Sport of the Slovak Republic and the Slovak Academy of Sciences (VEGA-2/0074/24); the Ministry of Education, Youth and Sport of the Czech Republic (MSMT INTER-COST LUC 23009); the Andronet COST Action (CA20119); and the bilateral project of Czech Academy of Sciences and Slovak Academy of Sciences (CAS-SAS-2024-02).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the first author/corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
