Abstract
This study aimed to investigate the effects of different timing of branched-chain amino acid (BCAA) supplementation (pre-/post-exercise) on the recovery of delayed-onset muscle soreness (DOMS) and associated inflammatory factors after resistance training. A double-blind randomized controlled experimental design was used in this study. Twenty-four untrained male college students volunteered to receive BCAA supplementation and completed resistance training. Participants were randomly assigned to the BCAA and placebo groups and sequentially performed two experiments of pre-exercise supplementation and post-exercise supplementation. Thus, four groups were formed: the BCAA-PRE group, the BCAA-POST group, the PLCB-PRE group, and the PLCB-POST group. Muscle soreness, countermovement jump (CMJ), and related blood parameters [interleukin-6 (IL-6), C-reactive protein (CRP), creatine kinase (CK), blood lactate (B[La])] were measured 30 min, 24 and 48 hr after resistance training. BCAA post-exercise supplementation significantly reduced muscle soreness scores compared to the placebo group at 48 hr after resistance training (p < .05). At 24 hr after resistance training, the BCAA group significantly reduced serum IL-6 and CRP (p < .05), in addition, the BCAA-POST group had lower serum IL-6 and CRP than the BCAA-PRE group (p < .05). No significant difference between groups was detected for CMJ or B[La] (p > .05). Resistance training induced the development of DOMS accompanied by elevated inflammatory factors (CRP and IL-6) and muscle proteins (CK). Compared to pre-exercise BCAA supplementation, post-exercise supplementation was observed to be more effective in alleviating the symptoms of DOMS and reducing inflammatory factors. However, it does not change the state of neuromuscular recovery.
Keywords
Introduction
The delayed-onset muscle soreness (DOMS) induced by resistance training has been one of the research hotspots in the field of exercise science (Cheung et al., 2003). Branched-chain amino acid (BCAA), an essential amino acid, has been demonstrated in previous studies to enhance the benefits of exercise by participating in energy metabolism (Nie et al., 2018), improving muscle strength (Spillane et al., 2012), and reducing central fatigue (Blomstrand, 2006). In recent years, with the deepening of related research, the efficacy of BCAA supplementation to enhance exercise benefits has been affected by various factors, including exercise intensity (Fedewa et al., 2019), supplementation regimen (Estoche et al., 2019; Watson et al., 2004), and exercise environment (Watson et al., 2004). In particular, there is still a great controversy regarding the optimal timing of BCAA supplementation: pre-exercise or post-exercise (Ra et al., 2018).
Resistance training is the process by which the body overcomes or triumphs over resistance to achieve muscle gain and strength enhancement (Westcott et al., 2012). Numerous studies have demonstrated the positive effects of resistance training on bone health, muscle mass, cardiovascular function, and metabolic function, in addition to the most basic function of developing strength qualities (Alizadeh et al., 2023). Nevertheless, resistance training inevitably results in damage to muscle fibers and the production of muscle inflammation (Calle & Fernandez, 2010; Peake et al., 2017), which manifests as symptoms of DOMS. It has been demonstrated that acute resistance training results in elevated plasma C-reactive protein (CRP), interleukin-6 (IL-6), and other inflammatory factors approximately 24 hr after recovery (Forti et al., 2017). In addition, serum creatine kinase (CK), an indirect indicator of muscle damage, increases with the onset of muscle pain (Baird et al., 2012). Therefore, the search for scientific and rational nutrition supplementation that attenuates DOMS, eliminates muscle inflammation, accelerates muscle repair and remodeling, and optimizes the effects of resistance training has received increased attention from researchers in recent years.
BCAA constitute approximately one-third of human skeletal muscle tissue and are composed of leucine, isoleucine, and valine (Neinast et al., 2019). Although the positive effects of BCAA in promoting protein synthesis, accelerating fatigue recovery, and reducing muscle soreness have been previously demonstrated in the literature (Osmond et al., 2019; Salem et al., 2024; Weber et al., 2021), the effects of BCAA remain highly controversial with regard to the optimal timing of supplement intake (Fedewa et al., 2019; Ra et al., 2018). Shimomura et al. demonstrated that 100 mg/kg BCAA supplementation before exercise was an effective means of inhibiting muscle damage (Shimomura et al., 2010). However, Andreo et al. investigated 24 untrained young adults and showed that BCAA supplementation before resistance training did not improve recovery of muscle inflammatory factors from immediate to 48 hr after exercise (Estoche et al., 2019). In addition, a review study by Rahimi et al. found that taking BCAA was more effective than passive recovery after all forms of exhaustive and destructive exercise, with positive results in reducing muscle soreness, improving muscle function, and preventing recovery from exercise injury (Rahimi et al., 2017). It is worth noting that the supplementation protocols in most of the previous studies varied in dosage, frequency, and timing (Kephart et al., 2016; Ra et al., 2018), and it was not possible to accurately compare the effects of pre- and post-exercise BCAA supplementation on muscle recovery under the same conditions. Therefore, the effects of the timing of BCAA supplementation (pre-/post-exercise) need to be further investigated.
In summary, this study aimed to compare the effects of BCAA intake at different timings (pre-/post-exercise) on DOMS and related inflammatory factors after resistance training.
Methods
Participants
Twenty-four male general college students with no athletic training experience were selected as participants. The characteristics of the participants are shown in Table 1. An a priori power analysis was conducted (G*Power version 3.0.1) for an F test. Based on a statistical power (1−β) of 0.8, a moderate effect size (0.5), and an overall level of significance of 0.05, at least 12 participants were required in each group. To participate in this study, the researchers verified through a questionnaire that all volunteers met the inclusion/exclusion criteria: (a) Exercise less than 2 times per week; (b) No supplements affecting athletic performance and muscle recovery were ingested during the 3 months before the official experiment; (c) No heavy exercise 1 week before the experiment; (d) Good health, no major illnesses, not injured or operated on in the last 6 months. Participants volunteered to take part in the experiment. Prior to the start of the experiment, the researcher informed participants of the possible risks and benefits of the experiment, as well as the schedule and details of the experimental procedures.
Participant Characteristics.

All values are mean ± SD. BCAA = branched-chain amino acid, PLCB = placebo.
Research Design
The study adopted a randomized, double-blind, placebo-controlled research design. The participants were scheduled to visit the laboratory on three occasions at the same time of day. Participants entered the laboratory 1 week before the start of experiments to familiarize themselves with the procedures. The next two visits were formal experiments. Twenty-four subjects were randomized into two groups, one group of participants supplemented with BCAA supplementation (BCAA group, n = 12) and the other group of participants supplemented with placebo (PLCB group, n = 12). Each group of 12 subjects participated in two separate experiments (pre-exercise supplementation and post-exercise supplementation), thus forming four groups: the BCAA-PRE group, the BCAA-POST group, the PLCB-PRE group, and the PLCB-POST group. The wash-out period between the two experiments was 1 week. Participants signed an informed consent form. All experimental procedures were reviewed by the Ethics Committee of Hebei Sport University, which was conducted by the 1964 Helsinki Declaration.
Preliminary testing
On the initial visit to the laboratory, participants underwent a one-repetition maximum (1RM) test to determine their base level. Specific test procedure: the participants were first predicted to be 60% 1RM weight, and completed 10 repetitions of weight-bearing knee flexion according to the prescribed apparatus, with a 2-min rest. The weight was increased to 65% to 70% of the estimated weight, completing 5 repetitions and then resting for 2 min. The weight was increased to 80% to 85% of the predicted weight and 3 repetitions were completed with 3 to 5 min of rest. Subsequently, the weight was increased or decreased based on the completion of the weighted knee flexion maneuver, resulting in a final projected 1RM weight for the participant’s instrumented weighted knee extension.
Procedure
The day of the experiment started with a 5-min 100W warm-up on a power bike. The pre-exercise supplementation group ingested the appropriate supplement 30 min before resistance training and baseline parameters were collected. Subsequently, the participants entered into resistance training, the specific resistance training program: (a) 90% 10RM weighted knee extension equipment exercises, each group of 10, a total of 5 groups, 3 min rest between groups; (b) Vertical jumping exercise: 15 repetitions per group, 3 groups in total, with 3 min rest between groups. Participants jumped as hard as they could each time; (c) 30 m continuous frog jump, a total of 2 groups, 3 min rest between groups, requiring subjects to jump forward as far as possible each time. The entire duration of the resistance training is 1 hr. Stretching and relaxation activities were performed after training. VAS, CMJ, and blood indices were collected at 30 min, 24 hr, and 48 hr of post-training recovery. After the 7-day wash-out period, the test was repeated by exchanging supplementary protocols to minimize the effects of familiarizing with the motion model. The post-exercise supplementation group was supplemented with appropriate supplements immediately after the completion of resistance training (Figure 1).

Flowchart of the experiment.
Supplementation and Dietary Control
The supplements were BCAA capsules (40 mg/kg) with a composition ratio of leucine:isoleucine:valine = 2:1:1. The placebo utilized maltodextrin capsules with the same shape and consistent total calories. Plasma BCAA concentrations increased within 15 min and peaked after 30 min after ingestion of BCAA supplements, as reported in previous studies. The participants were allowed to ingest the supplement 30 min prior to the experiment. The dietary intake of the participants was controlled during the test period. participants were instructed to follow their normal total calorie intake throughout the duration of the study. Also, the daily dietary status and protein intake ratio of the participants were monitored 3 days before the experiment to provide a reference for the diet on the experimental day and to avoid the effect of a high-protein diet on the BCAA intake effect.
Measurements
Muscle Soreness
A visual analog scale (VAS; Langley & Sheppeard, 1985) was used to determine the degree of muscle soreness in the participants. Participants marked the level of pain in the quadriceps muscle on a scale based on self-perception after completing three self-weighted standard deep squats. The VAS consisted of a 10-cm line whose endpoints were labeled with “no pain” (left) and “unbearable pain” (right). The same investigator did this for all the participants to avoid inter-investigator measurement errors. Qualified personnel were responsible for the supervision of each participant during the test. The test–retest reliability determined using an intra-class correlation coefficient (ICC) was ≥ .97.
Countermovement Jump (CMJ)
Participants performed a CMJ on a jump mat (Omegewave, Finland) to assess maximal jumping height. When the participants heard the command to start, they quickly squatted in place until the knee joints were at 90°, and then promptly jumped upward three times, recording the highest value. The reliability of this procedure was determined by conducting a test–retest analysis, which yielded a coefficient of variation of 1.3%.
Blood parameters (B[La], IL-6, CRP, CK)
Venous blood was collected at 4-time points (baseline, 30 min, 24 hr, and 48 hr after exercise), injected into test tubes for coagulation, and then serum was separated by centrifugation at 3,000 rpm for 10 min and stored in a refrigerator at −20° C for measurement. Enzyme immunoassay for human serum interleukin-6 (IL-6) and C-reactive protein (CRP) was tested by DR-200BS enzyme labeling instrument. Colorimetric assay for human serum Creatine Kinase (CK) was performed by Myriad BS-420 Automatic Biochemistry Instrument. Blood lactate (B[La]) was obtained by testing ear blood with a blood lactate meter (Lactate Scout, Germany).
Statistical analysis
Data are presented as the mean ± standard deviation (SD). The normality and homogeneity of the outcome measures were evaluated using Shapiro–Wilk’s and Levene’s tests, respectively. The base indicators (age, body mass, and height et al.) were analyzed using independent samples t-tests. A two-way repeated measures analysis of variance (RM-ANOVA) was conducted, with supplementation (BCAA-PRE, BCAA-POST, PLCB-PRE, and PLCB-POST) and time (baseline, 30 min, 24 hr, and 48 hr after exercise) as the independent variables. Dependent variables included VAS, B[La], IL-6, CRP, CK, and CMJ. Violation of sphericity was corrected using the Greenhouse–Geisser method. When significant differences were confirmed with ANOVA, multiple comparison testing was performed using Bonferroni post hoc correction to identify these differences. The level of statistical significance was set at p ≤ .05. The statistical analyses were conducted using the SPSS Statistics for Windows version 27.0 software (IBM Corp., Armonk, NY, USA).
Results
Muscle soreness
This study analysis unveiled an effect of time [F(3,42) = 361.239, p < .001, η2 partial = .963], intervention [F(3,44) = 2.553, p = .048, η2 partial = .148], and an intervention × time interaction [F(3,44) = 21.067, p < .001, η2 partial = .590]. At 48 hr of recovery, VAS scores were significantly lower in group BCAA-POST compared to the PLCB (pre, post) group (3.72 ± 0.43 vs. 2.73 ± 0.41; p = .003) (Figure 2).
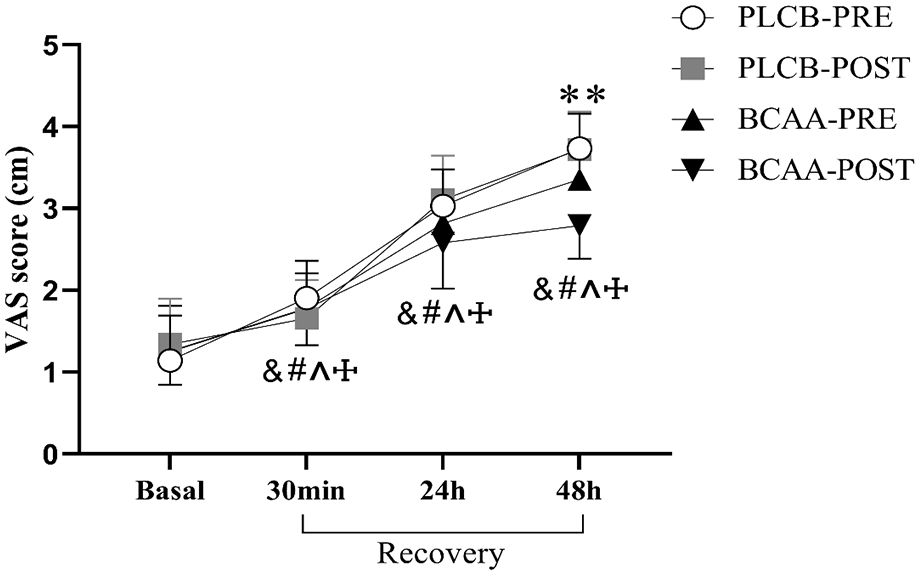
The changes in VAS score after resistance training in different BCAA supplement timing. &: significant differences from baseline in Group PLCB-PRE. #: significant differences from baseline in Group PLCB-POST. ^: significant differences from baseline in Group BCAA-PRE. †: significant differences from baseline in Group BCAA-POST. **: significant differences between PLAC and BCAA-POST.
Countermovement Jump (CMJ)
This study analysis unveiled an effect of time [F(3,42) = 93.913, p < .001, η2 partial = .870] and an intervention × time interaction [F(3,44) = 8.848, p < .001, η2 partial = .376]. The intervention effect was found to be non-significant [F(3,44) = 0.129, p = .942, η2 partial = .009]. CMJ in group BCAA (pre, post) was significantly higher in the recovery period at 24 hr (37.30 ± 4.45; p < .001), and 48 hr (37.57 ± 4.51; p < .001), compared to 30 min (34.72 ± 4.48). CMJ in group PLCB (pre, post) was significantly higher in the recovery period at 48h (37.10 ± 3.64; p = .044), compared to 30 min (35.92 ± 4.19) (Figure 3).

The changes in CMJ after resistance training in different BCAA supplement timing. &: significant differences from baseline in Group PLCB-PRE. #: significant differences from baseline in Group PLCB-POST. ^: significant differences from baseline in Group BCAA-PRE. †: significant differences from baseline in Group BCAA-POST. *: Significant difference between 30 min and 48 hr of recovery in the PLCB group. **: Significant difference in the recovery period between 30 min and 24 hr and 48 hr in the BCAA group.
Blood parameters
For blood lactate, this study analysis unveiled an effect of time [F(3,42) = 98.916, p < .001, η2 partial = .876]. The intervention effect [F(3,44) = 0.558, p = .646, η2 partial = .037] and intervention × time interaction [F(3,44) = 1.731, p = .175, η2 partial = .106] was both found to be non-significant (Figure 4A).
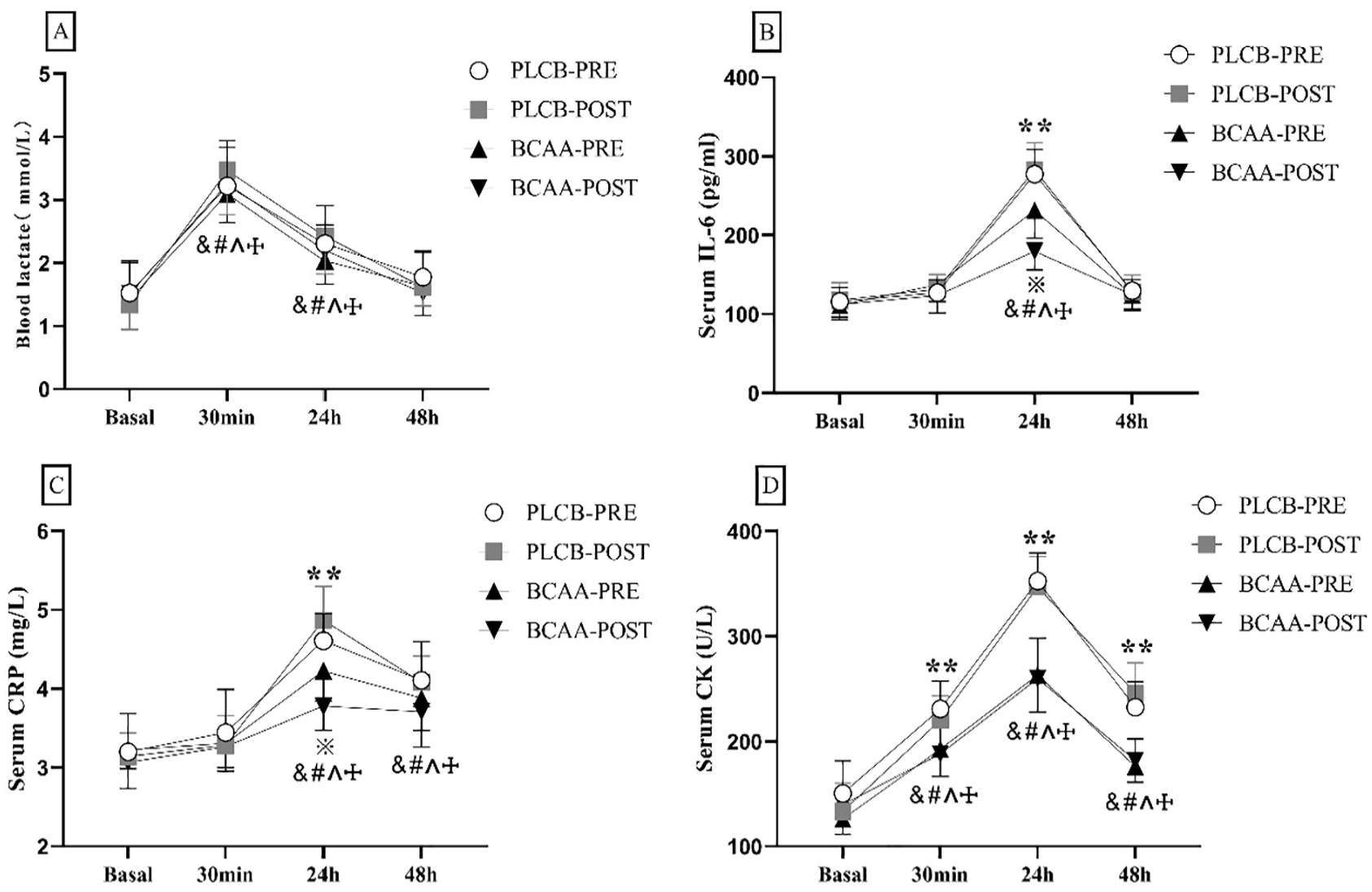
The changes in blood parameters after resistance training in different BCAA supplement timing. (A) Blood lactate, (B) serum IL-6, (C) serum CRP, (D) serum CK. &: significant differences from baseline in Group PLCB-PRE. #: significant differences from baseline in Group PLCB-POST. ^: significant differences from baseline in Group BCAA-PRE. †: significant differences from baseline in Group BCAA-POST. **: significant differences between PLAC and BCAA. *: significant differences between BCAA-PRE and BCAA-POST.
For serum IL-6, this study analysis unveiled an effect of time [F(3,42) = 207.128, p < .001, η2 partial = .937], interventions [F(3,44) = 17.202, p < .001, η2 partial = .540], and an interventions × time interaction [F(3,44) = 23.767, p < .001, η2 partial = .618]. At 24 hr of recovery, serum IL-6 was lower in both groups BCAA-PRE (231.56 ± 35.09; p = .005) and BCAA-POST (179.79 ± 23.81; p < .001) than in the PLCB (pre, post) group. Group BCAA-POST was significantly lower than group BCAA-PRE in the recovery period of 24 hr (p = .001) (Figure 4B).
For serum CRP, this study analysis unveiled an effect of time [F(3,41) = 134.910, p < .001, η2 partial = .908], intervention [F(3,43) = 5.556, p = .003, η2 partial = .279], and an intervention × time interaction [F(3,43) = 12.852, p < .001, η2 partial = .473]. At 24 hr of recovery, serum CRP was lower in both groups BCAA-PRE (4.225 ± 0.382; p < .001) and BCAA-POST (3.783 ± 0.310; p < .001) than in the PLCB group. And, group BCAA-POST was significantly lower than group BCAA-PRE in the recovery period of 24 hr (p = .032) (Figure 4C).
For serum CK, this study analysis unveiled an effect of time [F(3,42) = 248.231, p < .001, η2 partial = .947], intervention [F(3,44) = 54.459, p < .001, η2 partial = .788], and an intervention × time interaction [F(3,44) = 18.493, p < .001, η2 partial = .558]. Serum CK was significantly lower in group BCAA than in group PLCB at 30 min (p < .05), 24 hr (p < .001), and 48 hr (p < .001) of the recovery period (Figure 4D).
Discussion
It is well established that BCAA supplementation effectively reduces DOMS symptoms, eliminates muscle inflammation, and promotes muscle recovery following resistance training (Fedewa et al., 2019; Hormoznejad et al., 2019; Lee et al., 2017). The effects of BCAA supplementation have varied in previous studies due to differences in the athletic populations, exercise intensities, exercise environments, and supplementation protocols. In particular, the timing of BCAA supplementation (pre-/post-exercise) remains highly controversial. The present study was conducted to investigate this issue.
The timing of nutritional supplementation to optimize training outcomes has been supported by empirical evidence (Kerksick et al., 2017; Smith & Betts, 2022). The majority of research on the effects of nutritional supplementation after exercise has been conducted with regard to the post-exercise “anabolic window.” Namely, adequate protein supplementation during the post-exercise window (generally from the immediate post-exercise period to 2 hr post-exercise) promotes myofibrillar protein synthesis in the human body even more (Aragon & Schoenfeld, 2013). Some studies have also questioned whether there is a post-exercise window when adequate protein is provided (Kephart et al., 2016; Kerksick et al., 2017; Tipton et al., 2009). In practice, not everyone has to participate in fasting training before comparing the effect of BCAA, especially for untrained healthy men. The present study strictly controlled the participants’ total calorie intake and dietary ratios throughout the experiment to avoid the influence of a high-protein diet on the experimental results.
VAS is considered the most practical tool for assessing muscle damage (Begum & Hossain, 2019). The present study found that the VAS was significantly lower in the BCAA-POST group than in the placebo group at 48 hr after resistance training. Previous studies have shown that subjects’ VAS scores appeared significantly plus 24 hr after a one-time high-intensity centrifugal exercise and peaked 48 to 36 hr after exercise (Yin et al., 2022). Therefore, the above phenomenon that appears after exercise is reasonable. Consistent with previous studies, a meta-analysis by Michael et al. demonstrated that supplementation with BCAA reduced DOMS late in exercise and that the incidence of DOMS was substantially reduced with BCAA supplementation after exercise compared to placebo (Fedewa et al., 2019).
In addition, Nosaka showed that amino acid intake during the recovery period effectively reduced DOMS and muscle injury by comparing the effects of different amino acid supplementation regimens on elbow endurance exercise (Nosaka et al., 2006).
High-intensity centrifugal exercise results in damage to skeletal muscle fibers, which is characterized by decreased strength, inflammation, and elevated blood levels of muscle-specific proteins such as CK (Bessa et al., 2016). Leakage of CK into the bloodstream is also widely used as an indirect marker of muscle damage (Brancaccio et al., 2010). The present study found that CK in the BCAA and placebo groups after resistance training was significantly elevated to peak at 30 min to 24 hr, followed by a gradual decrease. And it was significantly lower in the BCAA group than in the placebo group. This is consistent with previous studies. Kenji Doma et al. summarized 25 studies to determine the effect of BCAA on markers of muscle injury after intensive exercise and showed that BCAA was effective in reducing levels of biomarkers of muscle injury post-exercise and muscle soreness (Doma et al., 2021). In addition, Emily et al. investigated the effects of BCAA supplementation on indirect markers of muscle injury after endurance exercise (Greer et al., 2007), this study showed that CK activity was significantly lower in the BCAA group than in the placebo group at 4, 24, and 48 hr post-exercise. Although the supplementation protocols in the aforementioned studies differed from the present study (both pre- and post-exercise supplementation), the present study did not find a significant effect of pre- and post-exercise BCAA supplementation on CK.
CRP is an acute-phase protein that is produced in response to inflammatory stimuli, such as tissue injury (Peake et al., 2017). The cytokine IL-6 is a significant factor in the acute phase response, and both are often regarded as messenger molecules associated with inflammatory and immune responses (Miles et al., 2008). The inflammatory response in muscles after resistance training is specifically characterized by elevated levels of inflammatory factors such as CRP and IL-6 (Cerqueira et al., 2020; Ihalainen et al., 2017). The present study revealed that both serum CRP and IL-6 reached their peak 24 hr after resistance training, followed by a gradual recovery. Consistent with a previous study, a study has shown that muscle microstructure damage resulting in inflammation, CRP begins to rise after a few hours, peaks after about 24 to 48 hr, and gradually decreases with the elimination of inflammation (Pumpa et al., 2014). In addition, this study found that CRP was significantly lower in the BCAA-POST group than the BCAA-PRE group 24 hr after resistance training. Some studies have found a strong correlation between the degree of muscle damage and CRP (Xia et al., 2017). It can be speculated that post-exercise BCAA supplementation is more effective in terms of preventing muscle damage.
No significant differences were observed between the four groups in blood lactate levels during the 48-hr recovery period following resistance training. The results of current studies on the effects of BCAA supplementation on blood lactate levels after exercise are inconsistent. The precise mechanism and effect of BCAA on blood lactate remain uncertain and may vary depending on individual differences, type of exercise, dosage, and timing of supplementation (Jafari et al., 2016; Matsumoto et al., 2009a, 2009b). Further studies are necessary to elucidate these effects.
CMJ is a more practical method for evaluating the state of neuromuscular fatigue in the lower limbs, which is related to the adaptation and recovery state of the training (Abian-Vicen et al., 2014). No significant differences were found between the 4 groups of CMJ height during the 48 hr after resistance training. José Maria et al. investigated that BCAA supplementation during two resistance training recovery periods (CMJ) had no effect on muscle function in untrained young adults (Estoche et al., 2019). Although some studies have demonstrated that BCAA supplementation is effective in attenuating post-exercise muscle strength loss, the diversity of research methodologies has led to a lack of clarity regarding the positive effects of BCAA supplementation on muscle function during exercise recovery (Hormoznejad et al., 2019; Shimomura et al., 2010; VanDusseldorp et al., 2018; Weber et al., 2021). More study is still needed in the future. In addition, the present study found that CMJ height in the BCAA group produced significant differences at 30 min and 24 hr after resistance training. However, the placebo group produced significant differences at 30 min and 48 hr after resistance training. Although this is not sufficient to demonstrate the effect of BCAA supplementation on muscle recovery after exercise, the rate of recovery of muscle function also deserves to be scrutinized.
Some limitations of this study must be noted. First, the present study did not analyze blood levels of amino acids to determine the rate of BCAA uptake. Second, the present study had a shorter washout period between the two experiments compared to other studies. The washout of ingested supplementation can be ensured, but the effect of training adaptations cannot be completely excluded.
In addition, there are still issues to be further studied, which are categorized into the following four main aspects. First, participant demographics: the study’s focus on untrained male college students limits the generalizability of findings to trained individuals and females, necessitating future research with a more diverse participant pool. Second, placebo effects: while the placebo design was careful, participant expectations may have influenced outcomes. Future studies should enhance blinding and include thorough debriefing to better control for these effects. Third, dietary control and the absence of strict control over other macronutrients and micronutrients may introduce confounding variables. Future research should implement comprehensive dietary control to isolate the specific effects of protein intake. Finally, practical applications: The findings on branched-chain amino acids (BCAA) in recovery could be more relevant by discussing their practical applications in training and supplementation, including timing, dosage, and combinations with other nutrients.
Conclusion
Resistance training induced the development of DOMS accompanied by elevated inflammatory factors (CRP and IL-6) and muscle proteins (CK). Compared to pre-exercise BCAA supplementation, post-exercise supplementation was observed to be more effective in alleviating the symptoms of DOMS and reducing inflammatory factors. However, it does not change the state of neuromuscular recovery.
