Abstract
High allostatic load (AL), a measure of physiological dysregulation, has been linked with premature morbidity and mortality. There is a paucity of research assessing AL among non-Hispanic Black (NHB) and non-Hispanic White (NHW) American men of various age groups. This study investigated racial differences in AL among NHB and NHW adult men and assessed whether racial differences in AL varied by age. Data were drawn from NHB (n = 232) and NHW (n = 246) men in the Nashville Stress and Health Study. AL was based on the sum of 10 biomarkers that was dichotomized as high AL (four or more high-risk biomarkers) or low AL (fewer than four high-risk biomarkers). Modified Poisson regression models were estimated to assess race differences in AL, adjusting for age, socioeconomic status (SES), and health behaviors. Interactions assessed whether racial differences in AL varied between young (22–49 years) and older (50–69) men. NHB men had a higher prevalence of being in the high AL group (prevalence ratio [PR] = 1.54, confidence interval [CI] = [1.09, 2.18]), relative to NHW men in the total sample. Among young men ages 22 to 49 years, NHB men had a higher prevalence of being in the high AL group (PR = 2.09, CI = [1.25, 3.49]), relative to NHW men. Among older men ages 50 to 69 years, there were no racial differences in AL. Findings underscore the importance of identifying factors that are associated with high AL, which is critical to mitigate premature morbidity and mortality, among NHB men.
Introduction
Racial differences in health outcomes and mortality between Black and White men persist despite increased availability of health care and attention to health disparities (Manuel, 2018). In fact, Black men in the United States have substantially worse health status when compared with White men (Shikany et al., 2018). Although Black men’s overall relative risk of mortality compared with White men was reduced from 1.36 in 2000 to 1.21 in 2014, it remained relatively high (Pathak, 2018). Moreover, the life expectancy of Black men in 2019 was 71.3 compared with 76.3 for White men (Arias & Xu, 2022). This disparity in mortality between Black and White men has been linked to preventable chronic diseases, which are more prevalent among Black men compared with White men (Alsan et al., 2019). For example, Black men report higher rates of asthma, all cancer types, diabetes, hypertension, obesity, and stroke relative to White men. These chronic conditions also tend to develop earlier in the lives of Black men than White men, which contributes to more adverse health consequences throughout the lifespan (Geronimus et al., 2007; Shikany et al., 2018; Thorpe et al., 2016, 2019). Understanding this earlier onset of chronic conditions and premature mortality among Black men compared with White men is key to advancing how the lives of Black men can be improved.
To clarify the biopsychosocial mechanisms contributing to persistent health disparities, scholars have increasingly investigated racial differences in allostatic load (AL), a biological indicator of wear and tear due to repeated adaptation to chronic, ongoing stress across the life course (Karlamangla et al., 2006; McEwen & Seeman, 1999; Seeman et al., 2001). AL provides an integrative framework for understanding the accumulated physiological burden of adaptation to chronic or prolonged states of stress (McEwen, 2000). The concept and framework for the term “allostatic load” was proposed by McEwen and Stellar (1993). The stressful states described in the AL model not only result from negative life experiences, but also “genetic load, individual lifestyle habits reflecting items such as diet, exercise, and substance abuse, and developmental experiences” (Seeman et al., 2001). These stressful states lead to physiological dysregulation of multiple bodily systems and can trigger a cascade of neuro-hormonal activation which leads to adverse health outcomes. Over time, this framework of AL leads to impaired cardiovascular, immune, and metabolic systems, as well as premature mortality (Das, 2013).
AL is used in health disparities research to elucidate group differences in individuals’ physiological responses to stressors, as well as subsequent negative health outcomes observed among different populations (Thomas Tobin, Gutierrez, Bell, & Thorpe, 2022). The cumulative effect of stressors associated with racism and socioeconomic marginalization may influence AL and, in turn, premature mortality (Li et al., 2019). In the United States, the link between AL and premature mortality is not homogeneous across racial and ethnic groups. Several studies have shown the connection between AL and premature all-cause and cause-specific mortality (Borrell et al., 2010; Duru et al., 2012; Seeman et al., 2001). In addition, previous research has explored differences in gender, race, ethnicity, and nativity in AL (Doamekpor & Dinwiddie, 2015; Geronimus et al., 2006; Williams, 1999; Yellow Horse & Santos-Lozada, 2019).
The observed differences in morbidity and mortality risk among Black men are likely driven by an array of complex, interconnected factors, including systemic gendered racism and persistent socioeconomic inequality (Thomas Tobin, Gutierrez, Erving, et al., 2022). This population faces unique challenges due to the intersection of racial and gender-based marginalization that results in elevated exposure to psychosocial stressors across the life course (Thomas Tobin, Gutierrez, Erving, et al., 2022). These stressors, often rooted in historical and contemporary racism, manifest through structural inequalities such as racial residential segregation, limited access to employment opportunities and other resources, as well as greater exposure to structural and interpersonal discrimination (Griffith et al., 2013; Hargrove & Brown, 2015). Black men are also disproportionately represented within the U.S. criminal justice system and face greater risk of incarceration compared with other racial and gender groups in this country (Patterson & Wildeman, 2015). This disproportionate exposure to chronic stressors across the life course faced by Black men may contribute to worse physical health and elevated AL. In fact, numerous studies have suggested that greater exposure to chronic stressors among Black adults compared with White adults (Thorpe et al., 2016; Thomas Tobin, Gutierrez, Erving, et al., 2022) may contribute to the earlier onset of chronic conditions and other adverse health outcomes, including higher AL, observed in this population (Geronimus et al., 2006; LaVeist et al., 2000; Thorpe et al., 2016). These patterns may be especially relevant for understanding AL disparities among men.
While there have been several studies investigating the impact of AL on Black women in the United States, particularly among Black women of reproductive age (Allen et al., 2019; Li et al., 2019; Tan et al., 2017; Williams, 1999), there is a paucity of research focusing on AL and physiological dysregulation among Black men or evaluating disparities between White and Black men across age groups (Gilmore et al., 2022; Tavares et al., 2022). Nevertheless, Duru and colleagues (2012) found that Black, middle-aged men had a higher AL compared with White middle-aged men in the National Health and Nutrition Examination Survey (NHANES III; 1988–1994). Conversely, prior research by Geronimus and colleagues (2006) demonstrated that while Black men had persistently higher AL scores than White men across the life course, these differences were negligible among men below age 30. They also observed similar patterns after accounting for differences in poverty-to-income ratio (Geronimus et al., 2006). Taken together, these findings underscore the need to clarify race and age patterns in AL among men.
To clarify the nature of AL disparities among Black and White men, the present study assessed potential racial differences in AL and determined whether such differences are distinct among young (ages 18–49) and older (ages 50+) men.
Method
Study Design and Sample
The Nashville Stress and Health Study (NSAHS) is a population-based sample of Black and White adults ages 21 to 69 drawn from the city of Nashville and surrounding areas within Davidson County, Tennessee. A random sample was obtained using a multistage, stratified sampling approach. Although Black households were oversampled, sampling weights allowed for generalizability to the county population. Between 2011 and 2014, 1,252 respondents, including 297 Black men, provided information about their personal and family backgrounds, stress and coping experiences, and health histories during 3-hour computer-assisted, race-matched interviews. Interviews also included in-home clinician visits the following day. Clinicians arrived before breakfast to retrieve 12-hour urine samples and collect blood samples; measure blood pressure; take waist, hip, height, and weight measurements; and document prescription medication usage. The NSAHS and all study procedures were approved by the Vanderbilt University Institutional Review Board and described in detail elsewhere (Brown et al., 2016). All participants provided informed consent. The present analyses included 478 men (246 White, 232 Black) for whom complete data were available; less than 1% of the sample was missing sociodemographic or biological data (due to difficulty in drawing sufficient blood, specimen contamination, or clinician visit refusal).
Measures
Allostatic load (AL), our dependent variable, was based on the sum of 10 biomarkers, including primary mediators and secondary mediators. Primary mediators refer to the substances released by the body in response to stress, such as norepinephrine, epinephrine, cortisol, dehydroepiandrosterone sulfate (DHEA-S). Secondary mediators are the effects resulting from the actions of primary mediators, including systolic and diastolic blood pressure, total cholesterol, high-density lipids (HDL), glycated hemoglobin, and waist-to-hip ratio (Geronimus et al., 2006). Based on guidelines established by the MacArthur studies (Karlamangla et al., 2006), each biomarker was designated as low-risk or high-risk based on established clinical risk levels (McEwen & Seeman, 1999; Turner et al., 2016); individuals taking blood pressure or cholesterol medication were also counted as “high-risk” for those biomarkers. Total AL scores were based on a count of these high-risk biomarkers and ranged from 0 to 10, with higher scores indicating greater physiological dysregulation across bodily systems. Consistent with prior studies (Geronimus et al., 2006; Thomas Tobin, Gutierrez, Bell, & Thorpe, 2022), we created a binary AL variable to identify individuals with high AL (1 = four or more high-risk biomarkers) to individuals with low AL (0 = fewer than 4\four high-risk biomarkers). Race, our independent variable, was measured with a dichotomous variable indicating that respondents self-identified as non-Hispanic White (reference category) or non-Hispanic Black (coded 1). Age, measured continuously in years, was also classified in two categories: young men (22–49 years; reference category) and older men (50–69 years).
Based on prior research (Geronimus et al., 2006), we considered socioeconomic status (SES) and other health factors (e.g., smoking, alcohol consumption, physical activity) associated with AL as covariates. Socioeconomic status was assessed using a standardized index of educational attainment (0–20 years of education), annual household income (0 = US$20,000; 1 = US$20,000– US$34,999; 2 = US$35,000– US$54,999; 3 = US$55,000–US$74,999; 4 = US$75,000– US$94,999; 5 = US$95,000+), and occupational prestige (0–100) based on the 2000 Nam-Powers-Boyd occupational scores (Nam & Boyd, 2004). Consistent with prior work using these data (Thomas Tobin, Gutierrez, Bell, & Thorpe, 2022), an SES score for each respondent was calculated by standardizing the three SES variables, summing the scores, and dividing by the number of SES variables for which data were available. This approach equally weighted education, income, and occupational prestige; as such, this affords the opportunity to captures individuals’ placement within a social hierarchy and provides a comprehensive assessment of SES while minimizing preserving data on individuals (Erving & Thomas, 2018; Thomas Tobin, Gutierrez, Bell, & Thorpe, 2022). In this study, continuous SES scores represent the number of standard deviations above or below the sample’s mean SES score, such that scores above zero corresponded with above-average SES.
Health behaviors included smoking, alcohol consumption, and physical activity. These variables were included in our models because of the prior work including health behaviors in their relation to AL (Doamekpor & Dinwiddie, 2015; Geronimus et al., 2006; Hickson et al., 2012). Smoking was measured with a single item: “Are you a current smoker, ex-smoker, or have you never been a smoker?” Former and non-smokers were categorized together (0 = non-smoker, 1 = current smoker). Alcohol consumption was measured with the item: “On the days you drank [alcohol] in the past 12 months, about how many [alcoholic] drinks did you usually have per day?” Based on federal Dietary Guidelines for Americans, 2020-2025 (U.S. Department of Agriculture & U.S. Department of Health and Human Services, 2020), numeric responses were categorized as: (0) None, (1) Moderate Consumption (1–2 drinks daily), (2) Excessive Consumption (3+ drinks daily). Physical activity was also based on federal guidelines (U.S. Department of Health and Human Services, 2018), comparing individuals who engaged in fewer than 75 minutes of vigorous activity per week to those who engaged in at least 150 minutes of moderate activity or 75 to 150 minutes of vigorous activity per week (0 = inactive, 1 = active).
Analytic Strategy
Frequencies and means and standard deviations were used to summarize the total sample of men. Student’s t tests for continuous variables and chi-square tests for categorical variables were used to evaluate the mean and proportional differences by age group for the demographic measures, SES, and health-related characteristics. The prevalence of our outcome variable, high AL, was greater than 10%. Therefore, modified Poisson regression models were estimated (Thorpe et al., 2017; Zou, 2004). An interaction term between age and race was included in the fully adjusted model (Table 2, Model 1). A statistically significant age by race interaction term (p = .0293) provided scientific justification for the analyses to be stratified by age group (Table 2, Models 2 and 3); age (assessed continuously) was included as a covariate within the age-stratified models to account for the wide age range among the “Young Men” and “Older Men” groups. All tests were two-tailed, and p values < .05 were considered statistically significant. Analyses were conducted using STATA 17.0 (StataCorp LP, College Station, TX, USA).
Results
The distribution of select characteristics among men for the total sample and by age and race in the NSAHS is shown in Table 1. In the total sample, NHB men had a below average mean SES score, were more likely to be a current smoker and to report high AL but were less likely to consume excessive amounts of alcohol relative to NHW men. There were no observed differences between NHB and NHW men as it relates to their age and physical activity. Among young men, NHB men had a below average mean SES score, were more likely to be a current smoker and report high AL yet were less likely to consume excessive amounts of alcohol relative to NHW men. There were no observed differences between young NHB and NHW men as it relates to their age or physical activity. Older NHB men had a below average mean SES score, were more likely to be current smokers and report high AL, but less likely to consume excessive alcohol or be physically active relative to their NHW peers. There were no observed differences between old NHB and NHW men as it relates to their age.
Distribution of Sample Characteristics Among Men for the Total Sample by Age and Race, Nashville Stress and Health Study (2011–2014)

For continuous variables, weighted means with standard deviation (SD) and variable range are shown; weighted percentages are presented for categorical variables.
The association between race and AL among men in the full sample and by age group in NSAHS is shown in Table 2. Adjusting for age, SES, smoking, alcohol consumption, and physical activity, in the full sample that contains the interaction term between race and age, NHB men had a higher prevalence of being in the high AL group (prevalence ratio [PR] = 2.00, 95% confidence interval [CI] = [1.19, 3.35]) compared with NHW men. Similarly, among young men, NHB men had a higher prevalence of being in the high AL group (PR = 2.09, 95% CI = [1.25, 3.49]) compared with NHW men. Among older men, there was no observed association by race between race and AL.
Prevalence Ratio for High Allostatic Load Among White and Black Men, by Age, Nashville Stress and Health Study (2011-2014)
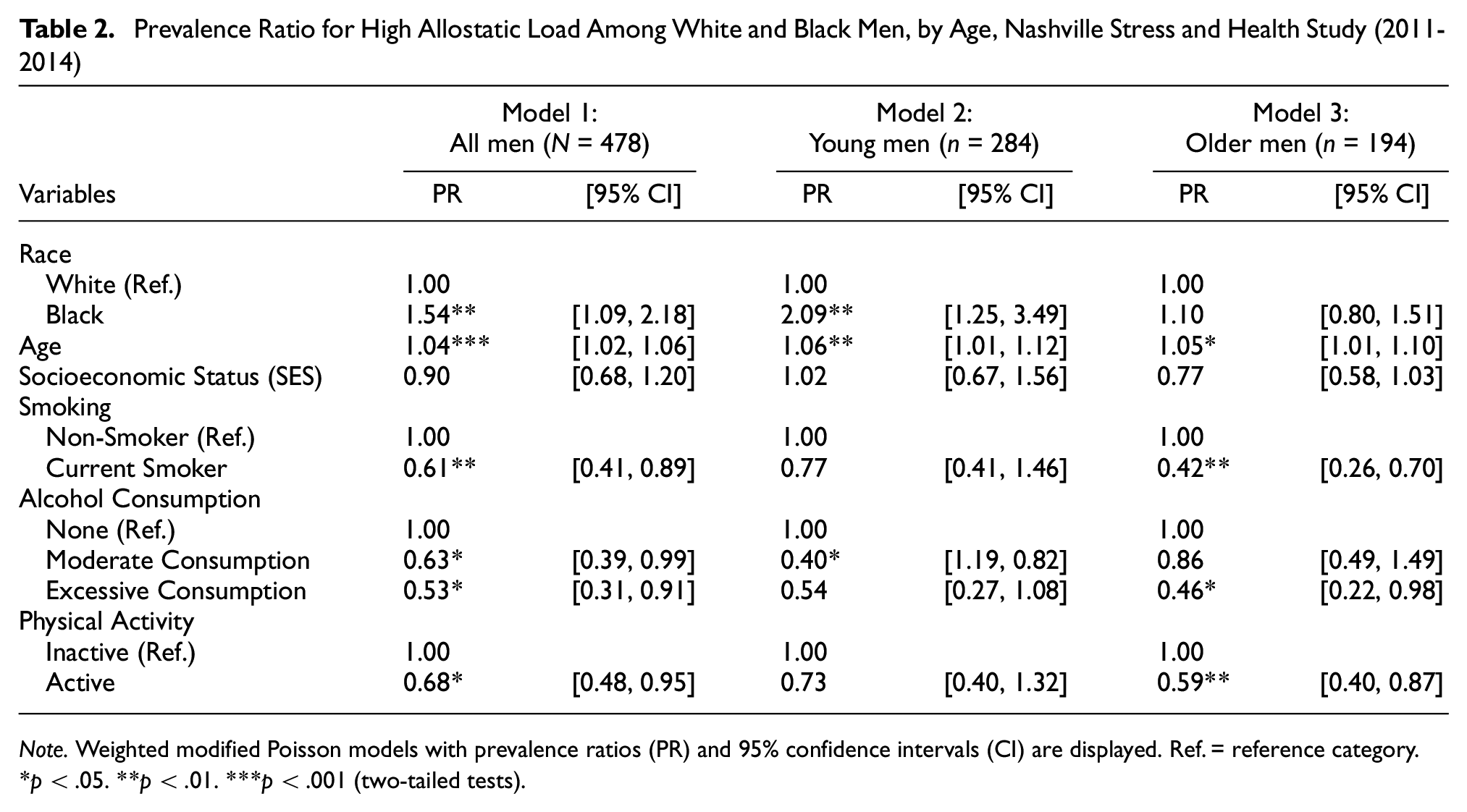
Note. Weighted modified Poisson models with prevalence ratios (PR) and 95% confidence intervals (CI) are displayed. Ref. = reference category.
p < .05. **p < .01. ***p < .001 (two-tailed tests).
Discussion
In this study, we sought to determine whether there were race differences in AL among adult men in the United States and to identify whether such differences varied by age. NHB men had a higher prevalence of being in the high AL group relative to NHW men in the total sample. This association remained for young NHB men compared with young NHW men. However, there was no race difference in AL observed between older NHB and NHW men. Findings underscore the importance of identifying factors that are associated with high AL, which is critical to mitigate premature morbidity and mortality, among NHB men.
We found that NHB men had a higher prevalence of being in the high AL group relative to NHW men. While this work is consistent with prior studies indicating that NHB adults have higher AL than their NHW counterparts (Borrell et al., 2010; Duru et al., 2012; Seeman et al., 2001), it extends previous findings to men specifically. Thus, our findings highlight that a larger proportion of NHB men have physiological dysregulation compared with NHW men. Several explanations might explicate this finding. First, NHB men are more often discriminated against and endure more stressors than NHW. This discrimination has been linked to poor health outcomes among Black men (Brown & Hargrove, 2018; Hargrove & Brown, 2015; Thorpe et al., 2017). Second, structural racism is likely a strong reason that NHB men have more physiological dysregulation than NHW men. Historically, NHB men’s experiences with structural racism likely have impacted their social and environmental conditions, leading to biological imprinting of persistent social and economic disadvantage (Thorpe et al., 2019). There has been some research focusing on proxies of structural racism, namely, residential segregation or integration, and its impact on the health of NHB men (Baxter et al., 2021; Thorpe, Bell, et al., 2015; Thorpe, Kelley, et al., 2015; Thorpe, Kennedy-Hendricks, et al., 2015). Additional research should seek to better understand how direct measures of structural racism impact physiological dysregulation and health outcomes.
In addition to documenting that NHB men had a higher proportion of AL than NHW men, we also demonstrated that this relationship was observed for younger NHB men—but not older NHB men—compared with younger and older NHW men, respectively. There is one plausible explanation for this finding. Younger NHB men may not have the economic or psychosocial resources to deal with stressors compared with younger NHW men. Younger NHB may experience a higher level of stress related to neighborhood exposures as well as discrimination in education and the workplace compared with NHW men. This finding emphasizes the role that age has on the relation between race and AL among men. Based on the descriptive results of this study, NHB and NHW men varied in their exposure to certain risk factors (e.g., lower SES, and current smoking), which may contribute to the greater prevalence of high AL among NHB men compared with NHW men. In particular, findings indicated that high AL was more prevalent among younger NHB men, suggesting a need for interventions aimed at reducing chronic stress and AL earlier in the life course for NHB men. As such, Black–White morbidity and mortality disparities among men may be reduced.
In our study, we observed racial differences in AL among younger men below age 50, but these differences were not apparent among men aged 50 and older. While this finding could potentially be explained by habituation or increased resilience among older Black men due to lifetime exposure to stressors (Assari & Lankarani, 2016; Barnes et al., 2004; Brondolo et al., 2009), alternative explanations warrant consideration. One crucial factor to consider is potential selection bias due to premature mortality among Black men. The older Black men in this study who survived to later ages and were able to participate may represent a relatively robust group, potentially masking population-level disparities. It is also worth noting that while the prevalence ratio for the race variable among older men was in the expected direction, the wide confidence interval suggests a potential sample size issue. This underscores the critical need for targeted efforts to recruit older Black men in future studies to better elucidate these patterns. The nonsignificant finding among older Black men points to the complexity of this issue and the need for multifaceted explanations. While habituation may play a role in stress response over time, it does not negate the cumulative negative impacts of chronic stress experienced earlier in life. Additional research is needed to empirically assess these various hypotheses specifically among men, to disentangle the effects of survival bias from potential stress adaptation, and to better understand the mechanisms underlying these observed age-related differences in AL.
The findings from this study should be interpreted in the context of the following aspects. First, the cross-sectional design of the study precludes us from drawing temporal ordering, or causal inferences. Second, we only focused on the relationship between NHB and NHW men and AL. It is unclear whether these same findings are true for other racial/ethnic or SES populations. Second, these data were collected on a sample of adults in an urban area in Tennessee. Future research should draw on a nationally representative sample. Third, future research should consider the ways that racism impacts AL. Racism operates through distinct mechanisms, such as societal policies and norms, discrimination, and unfair treatment (Gee et al., 2019). A focus on understanding the pathways that racism impacts physiologic regulation for NHB is key to reducing the stark NHB-NHW disparities in health outcomes such as obesity, diabetes, hypertension, and mortality. Finally, additional research is needed to evaluate the importance of stress exposure and psychological resources for shaping NHB men’s resilience.
Notwithstanding the limitations of this work, our study has some important strengths.
One strength of this study is the use of a comprehensive AL measure that includes both primary and secondary mediators of physiological dysregulation. Furthermore, this study indicates that high AL is differently distributed among both younger and older adult NHB and NHW men. Another strength is the use of age-stratified models to predict prevalence of high AL while accounting for socioeconomic and behavioral factors. This analytic strategy allowed us to further elucidate whether younger and older NHB men have a higher prevalence of high AL compared with NHW men. Thus, this research contributes new information regarding the prevalence of AL, and thus potentially increased disease risk, among NHB and NHW men at the intersection of race and age.
These data provided an opportunity to examine race disparities in AL among NHB and NHW men and to determine whether this relation varied by age. Findings from this study indicate that NHB men have higher AL than NHW men. When stratified by age, younger but not older NHB men also have higher AL compared with NHW men. These findings add to the burgeoning line of men’s health research. Furthermore, these findings underscore the importance of identifying factors that are associated with high AL, which is critical to mitigate premature morbidity and mortality among NHB men.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883251317095 – Supplemental material for Race, Age, and Allostatic Load Among Men in the Nashville Stress and Health Study
Supplemental material, sj-docx-1-jmh-10.1177_15579883251317095 for Race, Age, and Allostatic Load Among Men in the Nashville Stress and Health Study by Roland J. Thorpe, Ángela Gutiérrez, Paul Archibald, Amy D. Thierry, Marino Bruce, Corina Mills, Keith Norris and Courtney S. Thomas Tobin in American Journal of Men's Health
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by NIH grants R01AG034067, K02AG059140- 02S1, UL1TR000124, P30AG021684 ), K02AG059140, and U54MD000214. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
