Abstract
This study aimed to investigate the relationship between psychosocial factors and andropause symptoms among Japanese men in an internet-based cross-sectional study. We conducted an internet-based cross-sectional study in 2024, named Japanese Men’s health and Andropause Related Symptoms (J-MARS) study. Data on demographics, medical history, andropause symptoms, lifestyles (physical activity, insomnia, height, weight, and smoking and drinking habits), and psychological distress were collected from 1,489 men aged 20–69 years who participated in the J-MARS study. Andropause symptoms were measured using the Aging Males’ Symptoms (AMS) rating scale, which was included in the analysis. We conducted a multivariate logistic regression analysis, and estimated odds ratios and 95% confidence intervals to examine the associated factors with severe andropause symptoms. Severe andropause symptoms according to the AMS score were noted in 125 men (8.4%). Severe andropause symptoms were associated with former smoking, current smoking, strength training (≥1.0 hr/day), probable insomnia, moderate psychological distress, and severe psychological distress. After excluding 289 participants with cardiovascular disease, cerebrovascular disease, cancer, or depression, severe andropause symptoms were associated with age, absence of a spouse, and suspected insomnia in addition to the above factors. Age, insomnia, and psychological distress were also significantly associated with all AMS subdomains (somatic, psychological, and sexual symptoms). Psychological stress, insomnia, and smoking habits are strongly associated with severe andropause symptoms. These findings may be useful for the prevention of andropause and for patient education.
Introduction
Men face higher risks of conditions such as cardiovascular disease (CVD) than women and have shorter life expectancy and healthy life expectancy (Harris, 2023; Tharakan et al., 2019; World Health Organization, 2018). Consequently, the World Health Organization promotes the enhancement of men’s health and welfare (World Health Organization, 2018). Recently, andropause has attracted more attention within men’s health (Tsujimura, 2013). Andropause, also known as age-related testosterone decline or late-onset hypogonadism (LOH), encompasses a range of symptoms: general fatigue, decreased libido, reduced motivation, erectile dysfunction, decreased concentration, insomnia, irritability, shoulder stiffness, urinary issues, head heaviness, reduced early morning erections, depression, and cognitive decline (Ide, 2023; Tsujimura, 2013). Andropause is also associated with metabolic syndrome, diabetes, and CVD (Hirokawa et al., 2020; Ide, 2023; Tsujimura, 2013), which likely impacts men’s healthy lifespan and quality of life. Therefore, preventing the onset and progression of andropause is essential for improving, maintaining, and promoting men’s health.
Compared to menopause in women, andropause onset in men varies widely as testosterone levels gradually decline with age, with individual variability in timing and symptom manifestation (Ide, 2023). Testosterone levels are also influenced by psychological distress and lifestyle factors (Liu et al., 2021; Odetayo et al., 2023; Peate, 2003), which may contribute to andropause onset and deterioration. Symptoms can occur even when testosterone levels remain stable (Emmelot-Vonk et al., 2011; Hirokawa et al., 2020), indicating the need for a comprehensive assessment.
Health care providers, including physicians and nurses working with andropause patients, strive to prevent symptom worsening by advising on exercise, social support, alcohol intake, smoking, sleep, and diet (Liu et al., 2021; Peate, 2003). However, opinions differ on whether these factors effectively prevent or delay andropause onset and progression. Some studies suggest that exercise raises testosterone levels, thereby potentially protecting against andropause (Peate, 2003), while others report no such effect (Hayes et al., 2015). In addition, since sleep disorders may arise as symptoms of andropause, support for sleep management is essential (Ide, 2023; Peate, 2003). However, the role of sleep disorders in the onset or progression of andropause remains unclear. Evidence-based lifestyle guidance for andropause patients is essential, but comprehensive epidemiological studies exploring the association between psychosocial factors and andropause are limited.
This study aimed to investigate the association between psychosocial factors, including lifestyle habits and living environments, and andropause symptoms in Japanese men. We examined both overall andropause symptoms and those specific to physical, psychological, and sexual domains.
Materials and Method
Study Design and Data Collection
We launched the study project named “Japanese Men’s health and Andropause Related Symptoms (J-MARS) study” in September 2024 to investigate the factors associated with the incidence and deterioration of andropause symptoms and nursing care perspectives. The J-MARS study is a longitudinal study with yearly follow-up from September 2024. We conducted an anonymous, cross-sectional, internet-based baseline survey in Japan from September 24, 2024 to September 26, 2024. Among the survey panels managed by a major nationwide internet research agency, there were 940,817 men aged 20 to 69 years registered. Of these, 49,052 men received an email invitation from the research agency. Among those who received the email, the first 1,500 individuals who provided information on gender and age and passed the attention check were recruited for this study. They agreed to participate in the 2024 survey and yearly follow-up survey with online-based written consent. All survey items were mandatory, and there were no missing data. Survey respondents were awarded web points specified by the research agency. This study was approved by the Institutional Review Board of the Akita University Medical School (approval number: 3209).
The 2024 survey included the participants’ demographic data, medical histories, andropause symptoms, and lifestyles, such as physical activity, insomnia, height, weight, smoking and drinking habits, and psychological distress.
Variables and Measurements
We collected demographic data on age, marital status (with or without spouse), educational level (less than university, university/graduate school graduates, others), and subjective economic status (good: very good or good; poor: very poor or poor), and data on self-reported medical history (cardiovascular or cerebrovascular diseases, respiratory diseases, prostate cancer, cancer without prostate, immunological disease, blood disease, endocrine diseases, kidney diseases, prostate enlargement, depression, and psychiatric disorders other than depression). Age, marital status, subjective economic status, and medical history (cardiovascular or cerebrovascular diseases, cancer without prostate, or depression) were included as exposure variables.
We set the following factors, reported in a previous study to be associated with andropause symptom (Peate, 2003), as exposure variables: smoking habit (never, former, current: 1–9 cigarettes/day, current: ≥10 cigarettes/day), drinking habit (never, a few days or less/month, 2 or 3 days/week, or ≥4 days/week), body mass index (BMI, <18.5, 18.5–24.9, or ≥25.0 kg/m2), strength training (<0.5, 0.5, or ≥1.0 hr/day), insomnia (normal, suspected, or probable), and psychological distress (normal, moderate, or severe). Insomnia was measured using the Athens Insomnia Scale (AIS; Okajima et al., 2013; Soldatos et al., 2000). The AIS consists of eight questions, with a total score ranging from 0 to 24. Participants were classified into three groups based on their AIS score: “normal” (≤3 points); “suspected insomnia” (4–5 points); and “probable insomnia” (≥6 points; Okajima et al., 2013). Psychological distress was measured by the Kessler Psychological Distress Scale (K6; Furukawa et al., 2008; Kessler et al., 2002). The K6 consists of six questions, with a total score ranging from 0 to 24. Results were classified into three groups: “normal” (≤4 points); “moderate” (5–12 points); and “severe” (≥13 points; Tanji & Kodama, 2021).
The outcome variable was the severity of andropause symptoms. Andropause symptoms were measured by the Japanese version of the Aging Males’ Symptoms (AMS) rating scale (Heinemann et al., 1999; Kobayashi et al., 2008). The validity of the Japanese version has been validated (Kobayashi et al., 2008). The AMS rating scale consists of 17 questions evaluated on a 5-point scale (1 = none, 5 = extremely severe). The 17 questions are distributed over three subdomains: somatic (seven questions), psychological (five questions), and sexual (five questions). The severity levels of andropause symptoms (total score of AMS rating scale) were classified as follows: “no” (17–26 points), “mild” (27–36 points), “moderate” (37–49 points), and “severe” (≥50 points; Daig et al., 2003). Each severity of three subdomains was defined as the following: somatic symptoms subdomain, “no” (≤8 points), “mild” (9–12 points), “moderate” (13–18 points), and “severe” (≥19 points). Psychological symptoms subdomain was defined as follows: “no” (≤5 points), “mild” (6–8 points), “moderate” (9–12 points), and “severe” (≥13 points). Sexual symptoms subdomain was defined as follows: “no” (≤5 points), “mild” (6–7 points), “moderate” (8–10 points), and “severe” (≥11 points; Heinemann et al., 1999).
Data Analysis
In total, 1,489 participants were included in the analysis, excluding 11 participants with a history of prostate cancer. For the baseline characteristics, we determined the distribution of participants based on demographic data, medical history, smoking and drinking habits, BMI, strength training, insomnia, and psychological distress. In addition, the distribution of participants was based on the severity of andropause symptoms (total score and each subdomain).
To examine the associated factors with severity based on the total AMS score, we conducted a multivariate logistic regression analysis and estimated odds ratios (ORs) and 95% confidence intervals (CIs). The following two models were employed. In Model 1, adjustment variables included age, marital status, subjective economic status, smoking habit, drinking habit, BMI, strength training, insomnia, and psychological distress among all participants. Model 2 was created after excluding 289 participants with CVD, cerebrovascular disease, cancer, or depression in Model 1 (n = 1,200). In addition, for Model 2, we examined the associated factors with the severity of each subdomain. As a post hoc analysis, we conducted a multiple regression analysis for Model 2 using the AMS score as a continuous variable.
The AMS score has been reported to have high sensitivity but low specificity for LOH diagnosis (Lunenfeld et al., 2021). On the other hand, some studies have reported that the sexual subdomain of AMS was correlated with total and free testosterone in blood and used to diagnose LOH (Chen et al., 2013; Wu et al., 2010). In Japan, a total score of 10 or more points on the four items of the AMS rating scale predicts suspected blood testosterone deficiency status (i.e., LOH; Akehi et al., 2022). Based on the four AMS items described above, the severity of andropause symptoms was assessed using the “testosterone deficiency score” (TD score), which was defined as follows: “nonsevere” (≤9 points) and “severe” (≥10 points). A sensitivity analysis was also conducted to examine the factors associated with andropause symptoms using the TD score.
All statistical analyses were performed using the IBM SPSS Statistics version 28 (IBM SPSS Software Group, Chicago, IL, USA). All statistical tests were two-sided analyses, and differences were considered significant at p < .05.
Results
In total, 1,500 responses were received, and 11 participants with a history of prostate cancer were excluded. Finally, the responses of 1,489 men were included in the analysis.
The baseline characteristics of the participants are detailed in Table 1. The average age (standard deviation) of 1,489 participants was 49.8 years (12.3). Andropause was more prevalent in men aged 40 to 60 years old, accounting for approximately 80% of all respondents. More than half of the respondents were married, had graduated from a university or graduate school, and reported having good economic status. About a quarter of the respondents were current smokers, while less than half reported drinking alcohol at least 2 or 3 days a week. In total, 5.3% of the respondents had a BMI of less than 18 kg/m2, while 25.8% had a BMI of 25.0 kg/m2 or higher. About 30% of the respondents engaged in strength training for more than 30 min a day. Moreover, 46.5% of the respondents experienced probable insomnia, 28.3% reported moderate psychological distress, and 7.7% reported severe psychological distress.
Characteristics of Study Participants (n = 1,489)
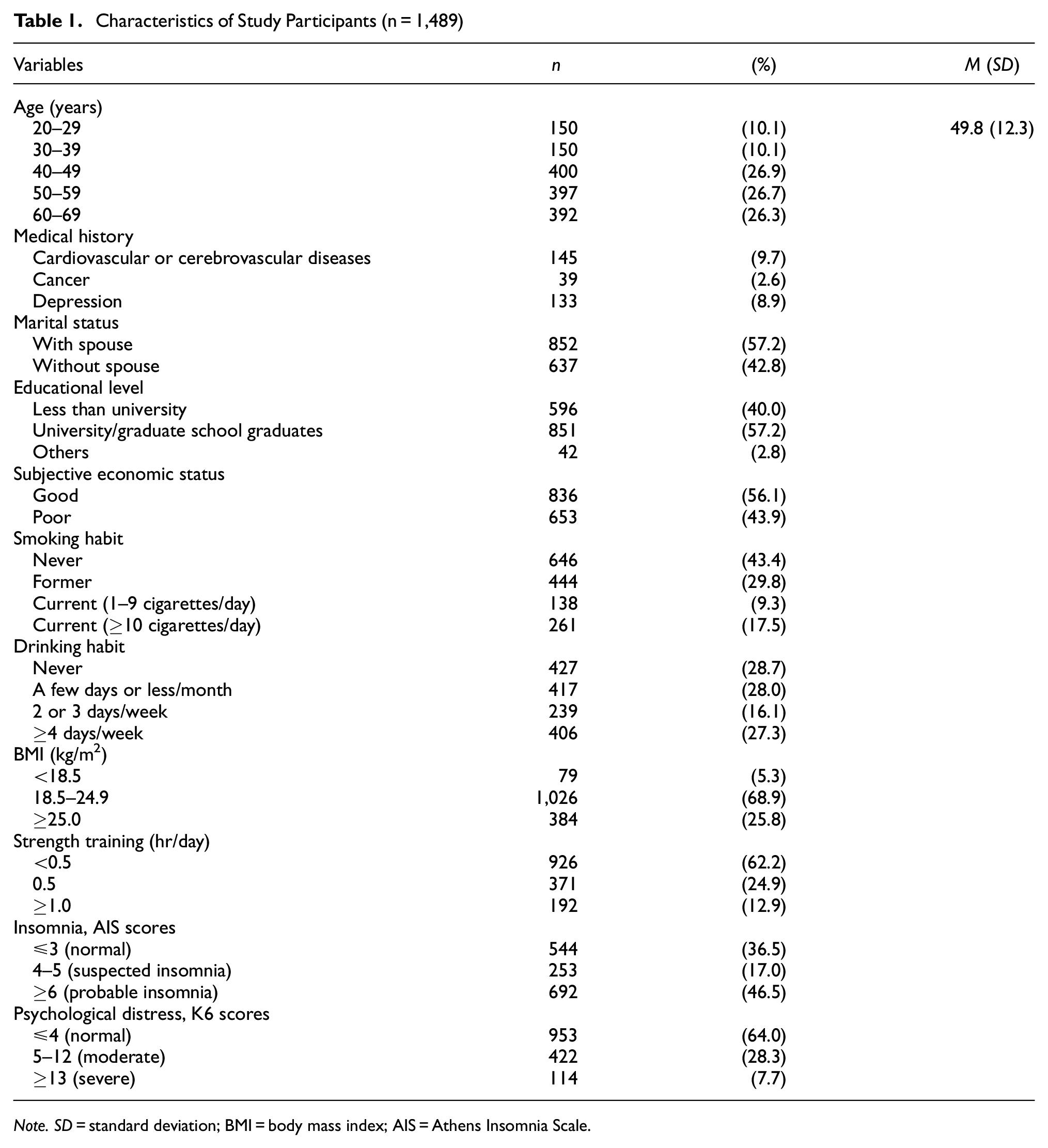
Note. SD = standard deviation; BMI = body mass index; AIS = Athens Insomnia Scale.
Figure 1 shows the prevalence and severity of andropause symptoms based on the total AMS score and three subdomains. For the severity based on the total AMS score, there were 634 (42.6%), 439 (29.5%), 291 (19.5%), and 125 (8.4%) men with no (≤26), mild (27–36), moderate (37–49), and severe (≥50) symptoms, respectively. For the somatic symptoms subdomain, there were 337 (22.6%), 478 (32.1%), 487 (32.7%), and 187 (12.6%) men with no (≤8), mild (9–12), moderate (13–18), and severe (≥19) symptoms, respectively. For psychological symptoms subdomain, there were 491 (33.0%), 412 (27.7%), 325 (21.8%), and 261 (17.5%) men with no (≤5), mild (6–8), moderate (9–12), and severe (≥13) symptoms, respectively. For sexual symptoms subdomain, there were 315 (21.2%), 246 (16.5%), 369 (24.8%), and 559 (37.5%) men with no (≤5), mild (6–7), moderate (8–10), and severe (≥11) symptoms, respectively.
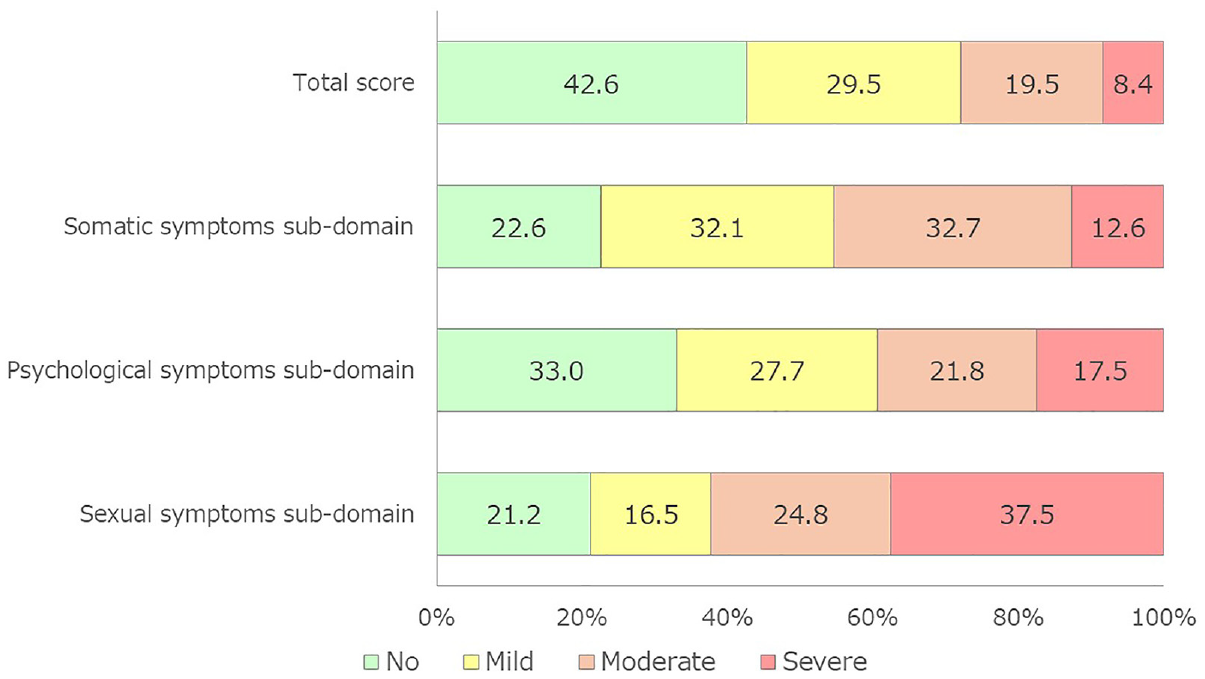
Prevalence of Andropause Symptoms
Table 2 shows the associated factors with severe andropause symptoms. In Model 1, severe andropause symptoms showed a significant positive association with former smoking (OR = 2.71, 95% CI = 1.53–4.78), current smoking (≥10 cigarettes/day; OR = 3.01, 95% CI = 1.59–5.72), strength training ≥1.0 hr/day (OR = 1.85, 95% CI = 1.02–3.33), probable insomnia (OR = 6.59, 95% CI = 2.50–17.41), moderate psychological distress (OR = 5.04, 95% CI = 2.64–9.62), and severe psychological distress (OR = 27.91; 95% CI = 13.31–58.56). Even after excluding 289 participants with CVD, cerebrovascular disease, cancer, or depression, these significant positive associations remained unchanged (Model 2). In Model 2, severe andropause symptoms also have a positive association with age (OR = 1.04, 95% CI = 1.01–1.07), absence of a spouse (OR = 1.83, 95% CI = 1.06–3.15), and suspected insomnia (OR = 4.91, 95% CI = 1.21–19.90). The multiple regression analysis for Model 2 showed similar results to those of Model 2, and it supported the findings in Table 2 (Supplemental Table 1).
Associated Factors for Severe Andropause Symptoms by Total Score of AMS Scale (n = 1,489)
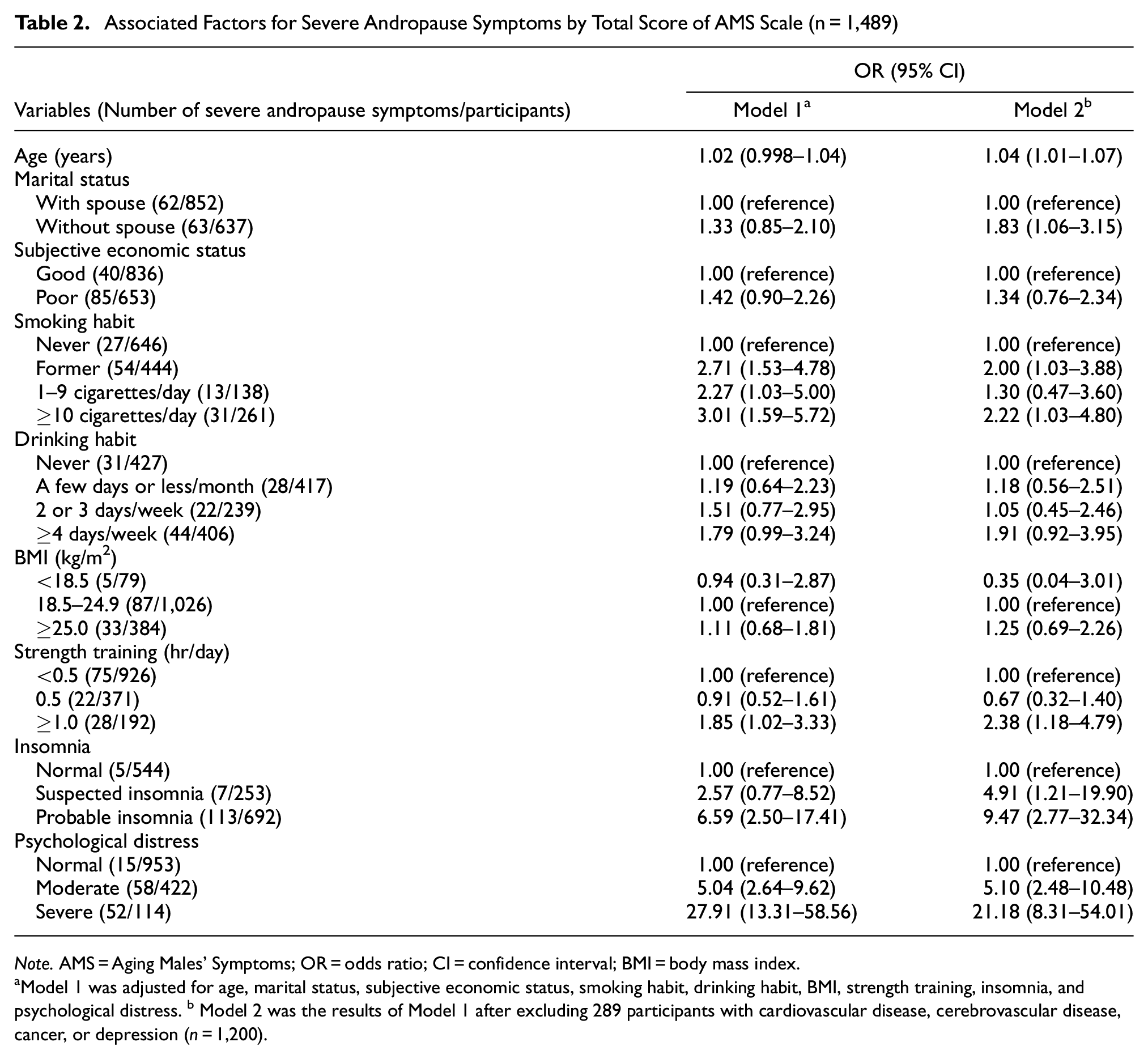
Note. AMS = Aging Males’ Symptoms; OR = odds ratio; CI = confidence interval; BMI = body mass index.
Model 1 was adjusted for age, marital status, subjective economic status, smoking habit, drinking habit, BMI, strength training, insomnia, and psychological distress. b Model 2 was the results of Model 1 after excluding 289 participants with cardiovascular disease, cerebrovascular disease, cancer, or depression (n = 1,200).
Table 3 details the factors associated with severe symptoms according to the AMS subdomains. Age, moderate and severe psychological distress, and probable insomnia were significantly positively associated with severe symptoms across all subdomains. The absence of a spouse was significantly positively associated only with severe symptoms in the psychological subdomain. Former and current smoking (≥10 cigarettes/day) were significantly positively associated with severe symptoms in the somatic subdomain, and former smoking was significantly positively associated with severe symptoms in the sexual subdomain. Engaging in strength training for 0.5 hr/day was significantly negatively associated only with severe symptoms in the somatic subdomain.
Associated Factors for Somatic, Psychological, and Sexual Symptoms by AMS Subscale (n = 1,200)
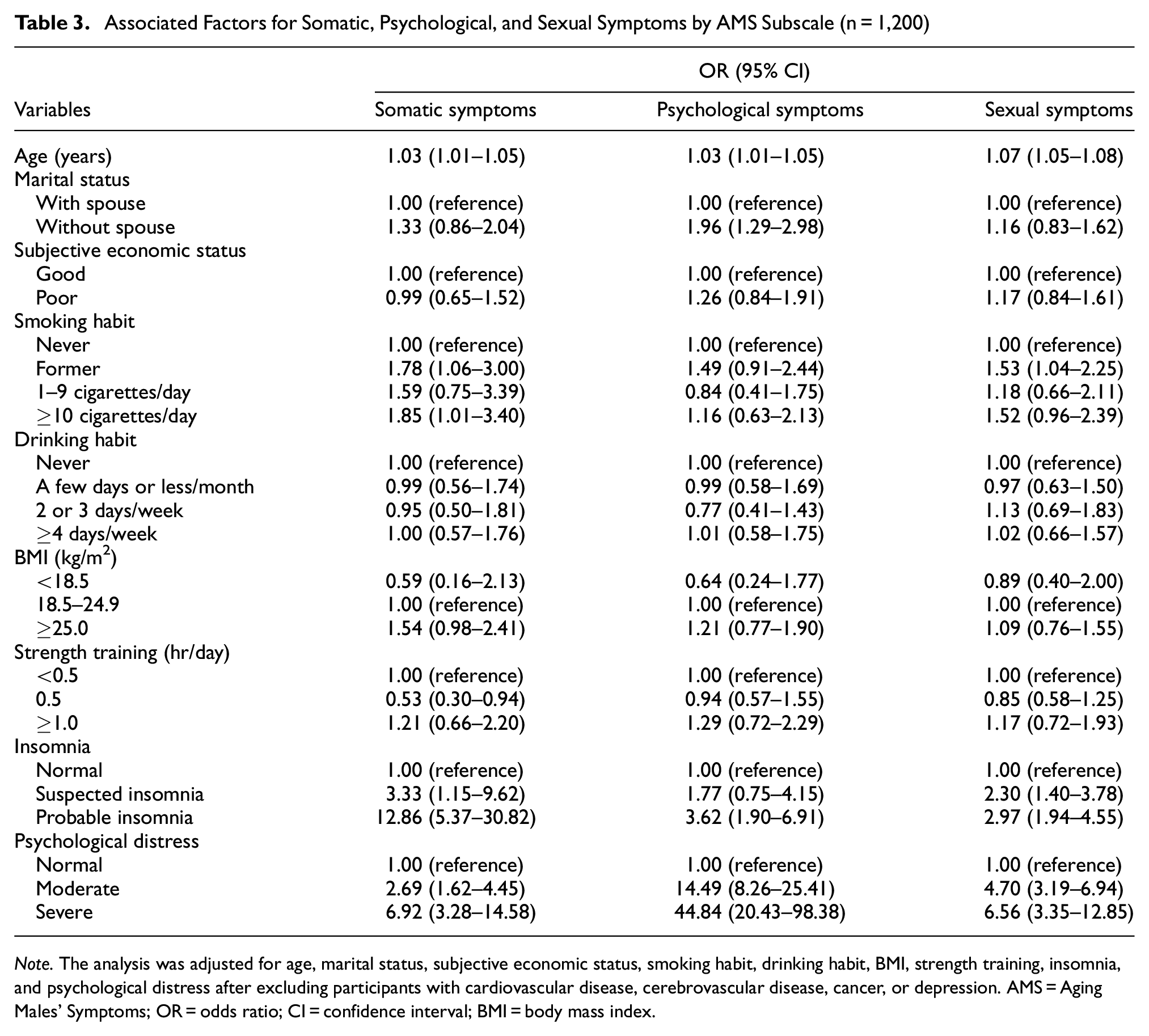
Note. The analysis was adjusted for age, marital status, subjective economic status, smoking habit, drinking habit, BMI, strength training, insomnia, and psychological distress after excluding participants with cardiovascular disease, cerebrovascular disease, cancer, or depression. AMS = Aging Males’ Symptoms; OR = odds ratio; CI = confidence interval; BMI = body mass index.
Table 4 shows the results of the sensitivity analysis examining the factors associated with the severity of andropause symptoms using the TD score. The results were similar to those of the sexual symptoms in Table 3, showing that age (OR = 1.07, 95% CI = 1.05–1.08), former smoking (OR = 1.52, 95% CI = 1.07–2.16), current smoking (≥10 cigarettes/day; OR = 1.66, 95% CI = 1.10–2.50), suspected insomnia (OR = 2.56, 95% CI = 1.67–3.93), probable insomnia (OR = 2.34, 95% CI = 1.60–3.41), moderate psychological distress (OR = 3.74, 95% CI = 2.59–5.40), and severe psychological distress (OR = 4.24, 95% CI = 2.20–8.19) were positively associated with the severity of andropause symptoms.
Sensitivity Analysis About the Associated Factors With Andropause Symptoms Using the Four AMS Items (n = 1,200)

Note. The analysis was adjusted for age, marital status, subjective economic status, smoking habit, drinking habit, BMI, strength training, insomnia, and psychological distress after excluding participants with cardiovascular disease, cerebrovascular disease, cancer, or depression. AMS = Aging Males’ Symptoms; OR = odds ratio; CI = confidence interval; BMI = body mass index.
Discussion
Our study results showed a positive association between moderate and severe psychological distress, insomnia, smoking habits (former and current smoking [≥10 cigarettes/day]), and andropause symptoms. These findings were consistent with the total score on the AMS rating scale and across all subdomains. The AMS score has been reported to have high sensitivity but low specificity for LOH diagnosis (Lunenfeld et al., 2021); the high AMS total score may not reflect lower blood testosterone levels. Therefore, we performed a sensitivity analysis using the TD score to predict blood testosterone deficiency status, which did not differ from the results described above. In contrast, drinking habits and BMI were not associated with andropause symptoms.
Chronic stress, such as psychological distress, is considered to contribute to the onset and deterioration of andropause by reducing testosterone levels through the hypothalamic–pituitary–gonadal axis (Odetayo et al., 2023). Testosterone decline and andropause are associated with depressive symptoms (Kische et al., 2018; Zitzmann et al., 2006), suggesting a mutual relationship between psychological distress and testosterone levels. The results of this study align with these findings. However, some reports deny the association between low testosterone and depression (Asselmann et al., 2019; Kische et al., 2018). One factor contributing to this discrepancy may be the lack of a strict correlation between testosterone levels and andropause symptoms (Emmelot-Vonk et al., 2011). However, in this study, sensitivity analysis was conducted using the TD score, which indicates suspected testosterone deficiency status, showing a positive association with moderate to severe psychological distress. Second, there is the possibility of an association with chronic stress more broadly rather than with depression alone. In this study, even after excluding individuals with self-reported depression, psychological distress remained associated with andropause symptoms. This suggests the need to assess mental health more broadly rather than focusing solely on depression. On the other hand, the fact that the items of the AMS scale, particularly the psychological symptoms subdomain, share content with the K6 scale may influence the association between psychological distress and severe andropause symptoms. However, in Table 3, psychological distress was strongly associated not only with the psychological symptoms subdomain but also with the somatic and sexual symptoms subdomains. These results suggest that while there may be some potential content overlap, it is not solely attributable to that. Third, a previous longitudinal study has reported that changes in testosterone levels may play a role in the onset of depression (Kische et al., 2018). Further longitudinal studies are needed to consider changes in psychological distress, testosterone levels, and andropause symptoms.
This study showed a positive association between insomnia and andropause symptoms, consistent with previous findings (Afsharnia et al., 2020; Hirokawa et al., 2012; Zitzmann et al., 2006). These studies have reported that this association can be explained by changes in sleep patterns and quality due to decreased testosterone levels, as well as the decline in sleep quality through psychological stress linked to lower testosterone. In this study, insomnia was not only associated with the somatic subdomain, which includes sleep-related questions, but also with the total AMS score and psychological and sexual subdomains. This suggests that insomnia may not only be a symptom of andropause but could also contribute to its onset and deterioration.
In this study, former and current smoking (≥10 cigarettes/day) showed a positive association with andropause symptoms, particularly with somatic and sexual symptoms, consistent with previous research findings (Afsharnia et al., 2020; Liu et al., 2021). Conversely, some reports indicate that smokers have higher blood testosterone levels compared to nonsmokers (Lotti et al., 2015; Svartberg & Jorde, 2007). Liu et al. explained this discrepancy by suggesting that the appearance of andropause symptoms due to smoking may outweigh the protective effect of higher testosterone levels (Liu et al., 2021). In this study, even in a sensitivity analysis using the TD score, which indicates suspected testosterone deficiency status, smoking habits showed a positive association. Cultural and genetic influences may also play a role. Future studies need to examine the association between andropause symptoms and testosterone levels.
Exercise, such as strength training, is said to increase testosterone levels, and exercise therapy is often prescribed for patients with andropause (Peate, 2003). However, there are also reports showing no association between exercise, testosterone levels, and andropause symptoms, indicating some debate on this relationship (Hayes et al., 2015). In this study, a J-curve or U-curve trend was observed between the three groups of daily strength training duration (<0.5, 0.5, and ≥1 hr) and andropause symptoms. Specifically, a positive association was found between the AMS total score and daily strength training of ≥1 hr, while a negative association was observed between the somatic subdomain and 0.5 hr of daily strength training. These results remained similar even when substituting daily strength training duration with daily walking or running time (data not shown). This may suggest that individuals with severe andropause symptoms, such as those receiving clinical care, are implementing exercise therapy as prescribed by their physician. Another reason could be that testosterone levels and andropause symptoms are influenced by exercise intensity and combination (e.g., aerobic and resistance exercise; Vieira et al., 2018). Considering this, our study’s results may suggest that moderate exercise has a protective effect on andropause symptoms, while vigorous exercise could be counterproductive. Future studies should examine the association with andropause symptoms, considering exercise intensity, type, and combination.
This study showed a positive association between the absence of a spouse and severe andropause symptoms, particularly psychological symptoms. It has been suggested that having a supportive person is beneficial for patients with andropause (Peate, 2003). The results of this study may indicate that the presence of a supportive individual plays a protective role in mental health.
This study has several limitations. First, being a cross-sectional study, it cannot establish causality. Therefore, we cannot determine whether the examined factors contribute to the onset or deterioration of andropause or simply reflect its symptoms. Second, andropause in this study was defined by the AMS rating scale, which does not always correlate severity scores with testosterone levels; hence, it is not definitive that our definitions reflect LOH syndrome. However, we conducted a sensitivity analysis using the TD score, which indicates suspected testosterone deficiency status in Japanese men, and found results consistent with the main analysis. Third, as an internet-based survey, the study is likely affected by volunteer bias, given that individuals interested in andropause may have been more likely to participate. This may limit generalizability to the overall Japanese male population. Finally, andropause symptoms and testosterone level variations may be influenced by cultural and genetic backgrounds, suggesting that country-specific verification may be necessary.
Conclusion
Psychological stress, insomnia, and smoking habits are strongly associated with andropause symptoms. These findings may be useful for the prevention of andropause and for patient education.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883241312836 – Supplemental material for Psychosocial Factors and Andropause Symptoms Among Japanese Men
Supplemental material, sj-docx-1-jmh-10.1177_15579883241312836 for Psychosocial Factors and Andropause Symptoms Among Japanese Men by Fumiya Tanji, Hirohito Nanbu, Daisaku Nishimoto and Maiko Kawajiri in American Journal of Men's Health
Footnotes
Acknowledgements
The authors thank all the participants in the database they used in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI [grant number JP24K13853].
Ethical Approval
This study was approved by the Institutional Review Board of the Akita University Medical School (approval number: 3209).
Informed Consent
Informed consent was obtained from the participants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
