Abstract
African American/Black (henceforth Black) men face disproportionate risks of morbidity and mortality from both cardiovascular disease (CVD) and colorectal cancer (CRC). The American Heart Association’s Life’s Simple 7 (LS7) tool was designed to examine predictors of CVD with included behaviors also linked to CRC risk (i.e., smoking status, weight, diet, and physical activity). However, no studies have combined LS7 assessment alongside CRC screening history, which serves as a proxy for assessing CRC risk, in Black men. In this study, Black men aged 45–75 participating in annual community wellness events were screened for 6 of 7 LS7 measures (excluding diet, LS6) and self-reported CRC screening. Analyses conducted using R 4.0.5. revealed that Black men (N = 680), with an average age of 57.3 years (SD = 7.5), reported poor (39.7%), intermediate (34.6%), or ideal (25.7%) LS6 scores with higher scores corresponding to lower risk for CVD. However, for every 1-point increase in LS6 scores (0–6), there was a 26% lower odds of reported CRC screening (p = .001). In the fully adjusted model, men with 4+ ideal LS6 behaviors had a 60% lower odds of self-reported CRC screening compared to those with two ideal LS6 behaviors (p < .001). These findings underscore the need for culturally relevant interventions for Black men across all levels of cardiovascular health (CVH) to increase CRC screening uptake.
Keywords
Introduction
Cardiovascular disease (CVD) and cancer are the leading causes of death in the United States (Centers for Disease Control and Prevention, 2021; Martin et al., 2024; National Cancer Institute [NCI], 2022a; Shiels et al., 2022). Colorectal cancer (CRC) is the second leading cause of cancer-related deaths in the United States (NIH, 2023a). CVD and CRC share several risk factors including obesity, hypertension, diabetes, and tobacco use and a common pathophysiology, related to chronic inflammation, oxidative stress, and altered telomere length (Gaitanidis et al., 2019; Sakhuja et al., 2021; J. Zhang et al., 2022). However, CVD and CRC are preventable with lifestyle modifications—including CRC screening to prevent CRC-specific incidence and mortality (U.S. Preventive Services Task Force, 2021b).
Unfortunately, the burden of both diseases is most prevalent across minoritized communities in the United States with African American/Black (henceforth Black) men at the highest risk for earlier morbidity and mortality from both CVD and CRC (Graham, 2015; Lawrence et al., 2022; NIH, 2022b; Rogers et al., 2017; Tsao et al., 2022). Studies have identified Black men at 30% higher risk of dying from CVD compared to non-Hispanic White (henceforth White) men (Health and Human Services, 2022; Murphy et al., 2021). Relatedly, a significantly lower percentage of Black men report ideal cardiovascular health (CVH) in comparison to White men (Brewer et al., 2018), which may partially explain the shorter life expectancy of Black men (Joseph et al., 2022). Similarly, Black men have a 40% to 50% higher risk of dying from CRC and the lowest 5-year survival rates in the United States when compared to any other racial group (American Cancer Society, 2020b; Giaquinto et al., 2022). Partial effects of low survival from CRC can be attributed to low CRC screening utilization rates and diagnosis at later stages of disease (Kwaan & Jones-Webb, 2018; NIH, 2023b; Xie et al., 2022). Black men are in need of multibehavioral, evidence-based interventions to address lifestyle-related risk and protective factors associated with the leading causes of death in the United States (Kwaan & Jones-Webb, 2018; Lawrence et al., 2022; Tsao et al., 2022).
Notably, ideal CVH is associated with both a lower incidence of CVD and lower incidence of CRC (Rasmussen-Torvik et al., 2013; Wang et al., 2021; J. Zhang et al., 2022). The American Heart Association (AHA) established the Life’s Simple 7 framework including four health behaviors (smoking, diet, physical activity, and body mass index) and three health factors (blood pressure, total cholesterol, and fasting plasma glucose) that can be used to stratify CVH into three main categories: ideal, intermediate, and poor (Brewer et al., 2018). The tool serves as a comprehensive guide for individuals to assess and improve their CVH through lifestyle modifications and preventive measures. Previous studies have examined CVH metrics, temporal trends, and associations with various CVD and non-CVD-related conditions including CRC among adults in the United Kingdom (Younus et al., 2016; J. Zhang et al., 2022). In this study, we aimed to explore the association of ideal CVH using six of the Life’s Simple 7 components (smoking, physical activity, body mass index, blood pressure, total cholesterol, and fasting plasma glucose [LS6]) and self-reported CRC screening among Black men aged 45 to 75 years recruited in a U.S. community–based setting. We assessed the hypothesis that a higher LS6 composite score, after adjusting for traditional sociodemographic factors, would be associated with higher odds of self-reported CRC screening. Analyzing the overall risk of CVD and CRC in Black men recruited from a community-based setting could impact the development of multibehavioral interventions tailored to address diverse risk profiles among Black men at highest risk for CVD and CRC.
Materials and Methods
Study Design and Population
Existing data, collected for nonresearch purposes with attendees of an annual, community-based African American Male Wellness Walk (AAMWW), were analyzed retrospectively for this study. Standardized health questionnaires were completed by participants of the 2018, 2019, and 2021 annual AAMWW. The “Walk,” as it is most referred to by community members, was first started in a large Midwest city in 2004 and has spread to more than 16 cities across the United States. The Walk provides a festival-like atmosphere with vendors, concert performers, health screenings, and an optional wellness 5K walk/run. The family-style event is a huge attraction to Black men, women, and children within the community and surrounding areas. During the event, standardized health screening questionnaires were administered to Black men to obtain sociodemographic information (i.e., age, race, gender, etc.), medical history, CRC screening history, biometric measurements, current medications, and lifestyle behaviors associated with chronic disease risk, as described previously (Aboagye-Mensah et al., 2020). Additional demographic characteristics collected included education, annual income, employment, and health insurance status. Participants who did not report LS6 components (n = 768), were not between the ages of 45 to 75 (n = 883), did not report CRC screening history (n = 405), education (n = 33), or income (n = 18), and did not self-identify as male (n = 36) and Black (n = 29) were excluded from analyses (n = 2,172), as shown in Figure 1. The study population was de-identified; thus, the study was exempted from approval by an Institutional Review Board.

Flow Diagram for Inclusion and Exclusion Criteria.
Exposure—LS6 Metrics
Information on other health history/behaviors and biometric covariates used in analyses were six of the AHA’s ideal CVH metrics, which included blood pressure, cholesterol, glucose, body mass index (BMI), smoking, and physical activity (Table 1). Blood glucose, cholesterol, blood pressure, and hemoglobin A1c were clinically examined by certified/licensed health care professionals. These metrics were obtained using a similar protocol outlined in prior studies (Aboagye-Mensah et al., 2020; Brewer et al., 2018). All metrics were evaluated separately using poor, intermediate, or ideal categories as defined by AHA guidelines and prior studies (Aboagye-Mensah et al., 2020; Brewer et al., 2018). Scoring details for LS6 metrics are also reported in Table 1.
Ascertainment of Life’s Simple 6 Metrics

Note. Reproduced in part from Brewer et al.; LS6 indicates Life’s Simple 6; CVH, cardiovascular health; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Blood pressure was measured using a sphygmomanometer, which consists of a cuff wrapped around the upper arm and a pressure gauge. Ideal blood pressure was defined as untreated systolic blood pressure (SBP) <120 mmHg and diastolic blood pressure (DBP) <80 mmHg; intermediate blood pressure was defined as SBP <120 to 139 mmHg or DBP <80 to 89 mmHg or if treated to goal (<120 mmHg/<80 mmHg); and poor blood pressure was defined as SBP ≥140 mmHg or DBP ≥90 mmHg (Brewer et al., 2018).
Cholesterol was measured using a blood lipid panel. Ideal total cholesterol was defined as an untreated total cholesterol <200 mg/dL; intermediate total cholesterol defined by a level of 200 to 239 mg/dL or if treated to goal (<200 mg/dL); and poor was defined by a level of ≥240 mg/dL (Brewer et al., 2018).
Blood glucose was measured using a random blood glucose test. Ideal blood glucose was defined as untreated glucose <100 mg/dL; intermediate glucose was defined as 100–125 mg/dL or treated goal (<100 mg/dL); and poor was defined as ≥126 mg/dL (Brewer et al., 2018). Hemoglobin A1c (HbA1c) was also measured and input for 11 participants missing blood glucose. A glycated hemoglobin of <5.7 was considered ideal, 5.7 to <6.5 without diabetes or 5.7 to <7 with diabetes was considered intermediate, and 7+ with diabetes or 6.5+ without diabetes was considered poor. If <5.7 with medication, participants were reclassified to intermediate (National Institute of Diabetes and Digestive and Kidney Diseases [NIDDK], 2018).
Weight was measured on-site using a zeroed scale. Height was self-reported. BMI was calculated by multiplying weight (pounds) by 703, and then dividing by height squared (inch2) (Aboagye-Mensah et al., 2020). BMI <25.0 was considered ideal; 25.0–29.9 was intermediate; and ≥30.0 was poor (Brewer et al., 2018).
Smoking status was assessed by asking participants “Do you smoke?” with response options of “Yes,” “No,” or “Quit in the last year.” Participants who reported not smoking or quitting in the last year were considered ideal. Current smokers were categorized as having poor CVH (Brewer et al., 2018).
Physical activity level was determined based on self-reported days of exercise per week (response options: 0, 1, 2, 3, 4, 5, 6, 7). Men who reported exercise on 3 or more days per week were categorized as having ideal levels of physical activity. All others were categorized as having poor physical activity. No intermediate category was reported (Brewer et al., 2018; King & Powell, 2018).
Outcome—Self-Reported CRC Screening
The outcome variable for the study was CRC screening. History of CRC screening (i.e., exam date) and CRC screening modality (i.e., stool-based, colonoscopy, or other) were self-reported. Self-reported CRC screening among participants aged 45 to 75 was included in analyses based on screening recommendations outlined by the U.S. Preventive Services Task Force (USPSTF; 2021b). Of importance to note, CRC screening at age 45 for Black adults has been suggested by the American College of Gastroenterology, the American Society of Gastrointestinal Endoscopists, and the American Cancer Society since 2016 (Williams et al., 2016).
Statistical Analysis
Descriptive statistics are shown in Table 2 with p values calculated using chi-square or two-sample t-tests. The association of LS6 scores with CRC screening (yes/no) was assessed using logistic regression modeling with continuous and categorical LS6 scores and sequentially added covariates (age groups “45–49,” “50–64,” and “65–75,” year of walk, education, income, and insurance). Based on known disparities in CRC screening utilization, age groups were created and included in analyses (American Cancer Society, 2019, 2020a).
Characteristics of Participants in the African American Male Wellness Walk by Self-Reported Colorectal Cancer Screening
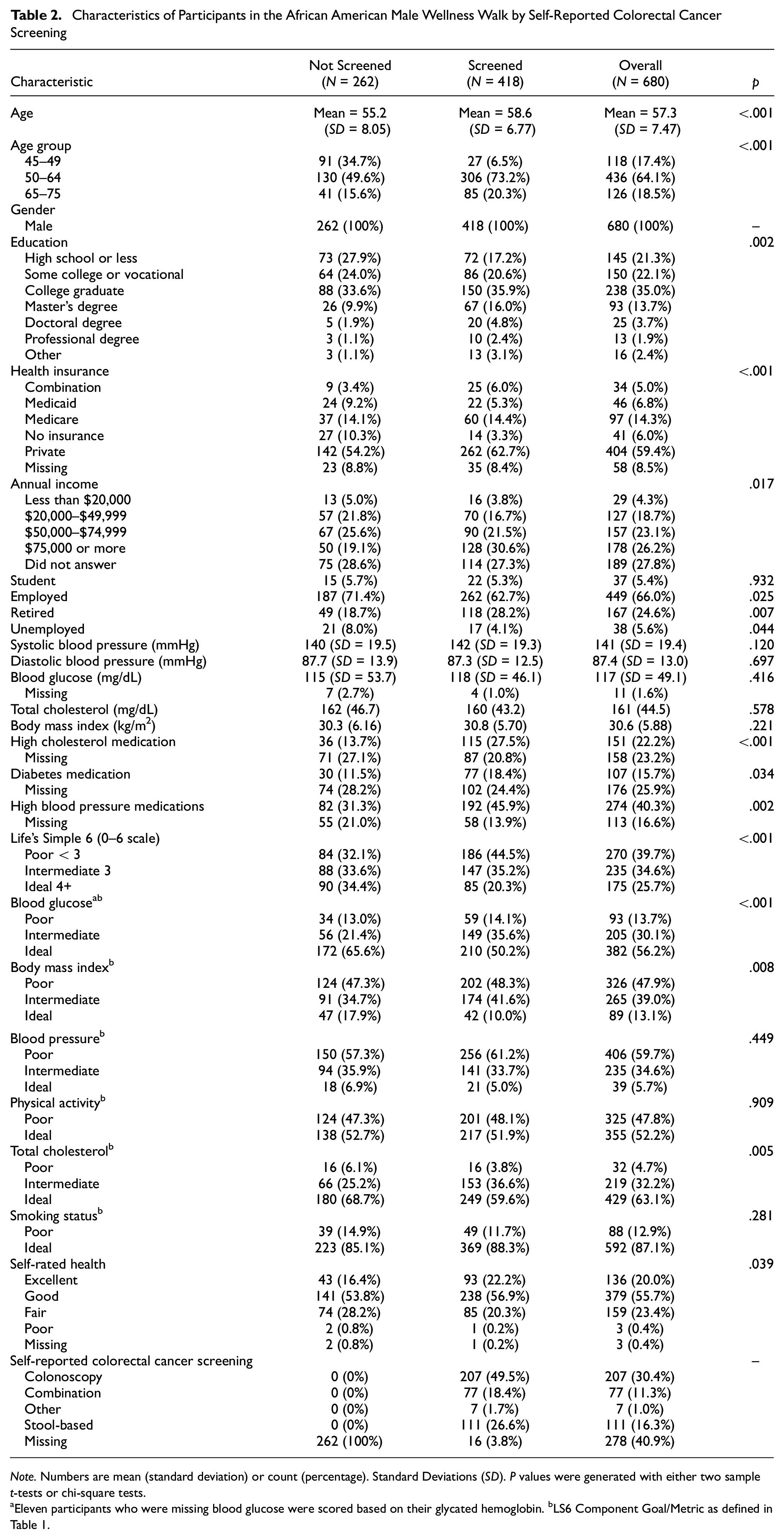
Note. Numbers are mean (standard deviation) or count (percentage). Standard Deviations (SD). P values were generated with either two sample t-tests or chi-square tests.
Eleven participants who were missing blood glucose were scored based on their glycated hemoglobin. bLS6 Component Goal/Metric as defined in Table 1.
For the continuous LS6 (0–6 scale) model, one point was assigned for ideal CVH outcomes and 0 points for intermediate and poor CVH (range: 0–6 points). The categorical LS6 (0–6 scale) cardiovascular outcomes were categorized as ideal if the composite score was ≥4, intermediate if the composite score was 3, and poor if the composite score was <3 to achieve a relatively equal distribution of the sample (Kesireddy et al., 2019; S. Kim et al., 2019). All analyses were conducted using R 4.0.5.
Results
Characteristics of the Analytic Cohort
A total of 680 Black men with an average age of 57.2 (SD 7.48) years completed the survey questionnaire (Table 2). More than half of participants reported private insurance (n = 404, 59%), employment (n = 449, 66%), and education above high school (n = 369, 54%). Participants were categorized as having poor (n = 270, 39.7%), intermediate (n = 235, 34.6%), or ideal (n = 175, 25.7%) CVH.
CRC Screening
Approximately half of participants (n = 418; 61%) self-reported CRC screening via colonoscopy (n = 207; 49.5%) followed by stool-based testing (n = 111; 27%), a combination of both (n = 77; 18.4%), or some other modality (n = 7; 2%) (Table 2). The remaining participants (n = 262; 39%) did not report CRC screening. CRC screening status differed based on age group (i.e., 45–49; 50–64; 65–75; p < .001). Among those who self-reported screening, 63% reported private insurance (n = 262). Irrespective of screening status, more than half of participants were employed (n = 262, 63% of those screened; n = 187, 71% of those not screened, p = .025). Participants who reported CRC screening were more likely to report medication use for blood pressure (n = 192, 46% vs. n = 82, 31%; p = .002), diabetes (n = 77, 18% vs. n = 30, 12%; p = .034), and cholesterol management (n = 115, 28% vs. n = 36, 14%; p < .001) compared to those who had not been screened. In terms of LS6 categorical data, a significantly higher proportion of CRC-screened participants had poor (n = 186, 45% vs. n = 84, 32%) or intermediate scores (n = 147, 35% vs. n = 88, 34%) compared to those who had not been screened for CRC but had ideal CVH (n = 85, 20% vs. n = 90, 34%; p < .001). Supplemental Tables 1 and 2 represent the demographic and health screening breakdown of cohort participants based on the year of the Walk and the percentages based on screening status for LS6 composite score, respectively.
The Association of Ideal CVH Score With Self-Reported CRC Screening
For every 1-point increase in CVH scores (0–6 scale) while adjusting for age, year, education, and income (N = 680), there was a 27.5% lower odds of CRC screening (odds ratio [OR]: 0.725; confidence interval [CI]: 0.614, 0.853; p < .001) (Table 3). Additional adjustment for insurance in the fully adjusted model (n = 622) did not attenuate the association; there was a 26% lower odds of CRC screening (p = .001).
The Continuous Association of Ideal Cardiovascular Health (Continuous LS6 scored 0–6) With Colorectal Cancer Screening

Note. Legend Table 3: The odds ratios describe the odds of colorectal screening per 1-unit increase in LS6 score. N = 680 for all models except model 4 (n = 622). Model 1 is adjusted for age and year. Model 2 is adjusted for age, year, and education. Model 3 is adjusted for age, year, education, and income. Model 4 is adjusted for age, year, education, income, and insurance. Numbers under “LS6 OR” are odds ratios from logistic regression models indicating change in odds of colorectal cancer screening per point change in LS6 Score. Example interpretation: When adjusting for age, year, education, and income, every 1-point higher Life’s Simple 6 score (0–6 scale) is associated with a 28% lower odds of colorectal cancer screening (p < .001).
The categorical LS6 score (0–6 scale) in which intermediate and ideal CVH scores are compared to poor scores are presented in Table 4. In the fully adjusted model (n = 622), there was a 60% lower odds of CRC screening in men with ideal versus poor CVH (OR: 0.403; CI: 0.250, 0.646; p < .001). Similar findings were shown using the 0- to 12-point CVH scoring system (Supplemental Tables 3 and 4).
The Association of Ideal Cardiovascular Health (Categorical LS6 Scored 0–6) With Colorectal Cancer Screening—Categorical Life’s Simple 6 Scored 0–6 Describing Odds of Colorectal Cancer Screening
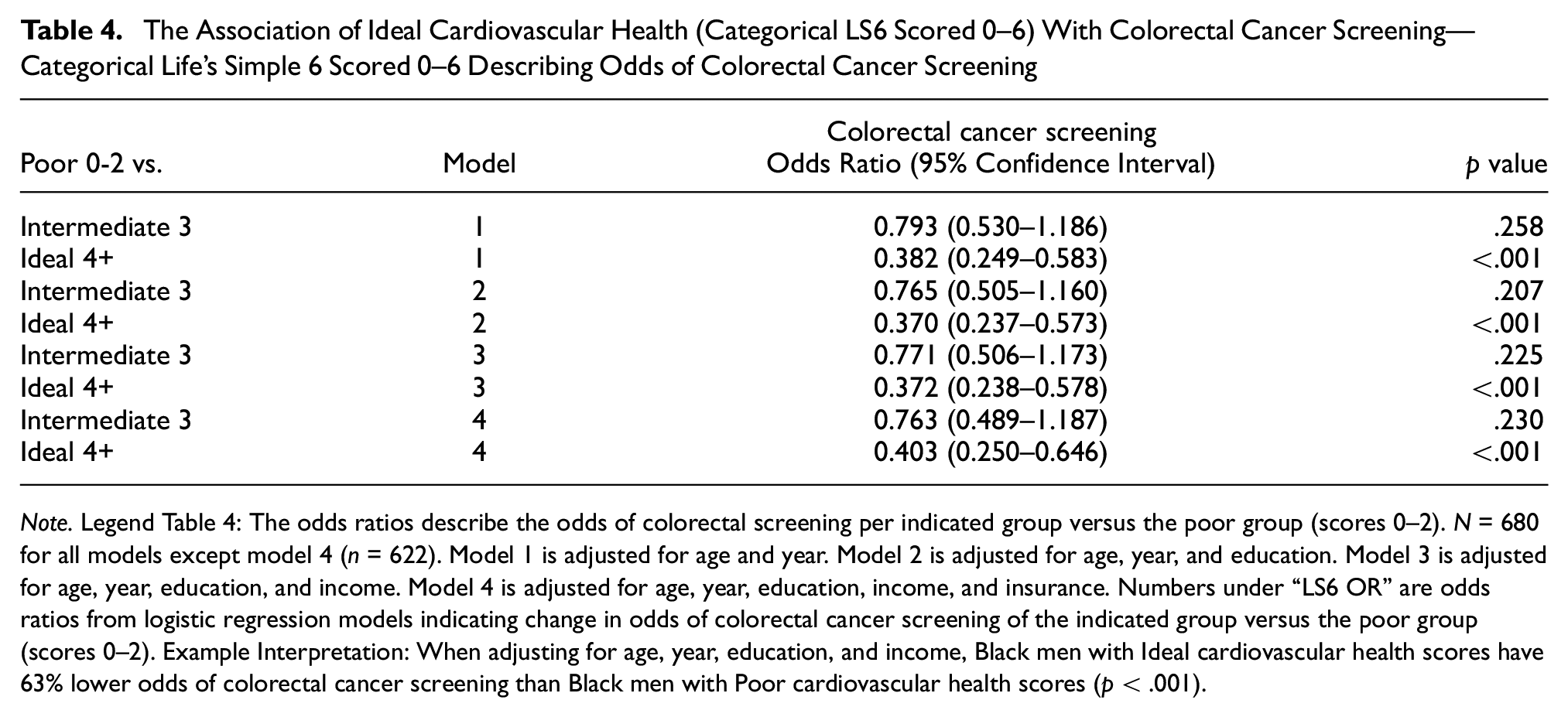
Note. Legend Table 4: The odds ratios describe the odds of colorectal screening per indicated group versus the poor group (scores 0–2). N = 680 for all models except model 4 (n = 622). Model 1 is adjusted for age and year. Model 2 is adjusted for age, year, and education. Model 3 is adjusted for age, year, education, and income. Model 4 is adjusted for age, year, education, income, and insurance. Numbers under “LS6 OR” are odds ratios from logistic regression models indicating change in odds of colorectal cancer screening of the indicated group versus the poor group (scores 0–2). Example Interpretation: When adjusting for age, year, education, and income, Black men with Ideal cardiovascular health scores have 63% lower odds of colorectal cancer screening than Black men with Poor cardiovascular health scores (p < .001).
Discussion
Utilizing the AHA’s LS6 tool, we examined the association of lifestyle-related behaviors and CRC screening in a convenience sample of nearly 700 Black men between the ages of 45 and 75 years. Our findings suggest that higher levels of CVH, in this sample of Black men, were associated with potentially lower odds of CRC screening. In addition, Black men in this study who reported CRC screening were more likely to be older and report use of medications for managing chronic conditions such as high blood pressure, high cholesterol, and diabetes. Our study is among the first to assess these lifestyle-related risk and protective behaviors associated with CVD and CRC among a community sample of Black men in the United States. Our results underscore the necessity for designing and disseminating interventions aimed at informing Black men who embrace multiple, optimal health practices about the crucial role of CRC screening in efficiently mitigating CRC risk.
Previous research has suggested healthy lifestyle-related behaviors (i.e., LS7 metrics) to be associated with lower CRC incidence and mortality (Wang et al., 2021; J. Zhang et al., 2022); however, our study provides evidence to suggest diverse risk profiles among a sample of Black men in the United States. For example, a recent meta-analysis examining the uptake of CRC screenings among men with diabetes reports no difference; however, this study failed to report race/ethnicity and age-based differences known to influence CRC uptake in the general population (Bhatia et al., 2020). Moreover, previous studies primarily recruited European citizens or non-Hispanic White women in the United States, while our study provides insight into the interaction of correlated preventive behaviors within a historically, high-risk group of Black men with low rates of CRC screening (American Cancer Society, 2019). Recent studies reported lower CRC screening rates among residents of the South and Midwest compared to other U.S. geographic regions (Shah et al., 2022). Our study findings can directly inform future work to identify clinically modifiable targets and strategies to address sobering disparities in CRC screening and potentially incidence and mortality among Black men primarily residing in the Midwest (Shah et al., 2022; Wang et al., 2021; J. Zhang et al., 2022).
Our findings demonstrate an increased screening rate in Black men with less-than-ideal CVH. CRC and CVD often coexist due to co-related risk factors; those with fewer ideal CVH risk factors are at an increased risk of CRC and other chronic diseases (Azap et al., 2021; Ogunmoroti et al., 2016; C. Zhang et al., 2021). Having six or more ideal CVH metrics is associated with a 31% lower risk of CRC compared to those meeting only one ideal CVH metric (J. Zhang et al., 2022). Even in comparison to other minoritized groups in the United States, Black adults often have fewer ideal CVH risk factors compared to Hispanic Americans, who often have lower incomes, less education, and are more likely to report being uninsured (Egan et al., 2020). Even with a higher socioeconomic status, Black adults still report poorer overall health outcomes, which could in part be explained through differences in diet, structural racism, and physiologic and persistent life stressors (Azap et al., 2021; Egan et al., 2020).
Our study is among the first to provide CRC screening estimates among Black men aged 45 to 75 considering recent changes to the USPSTF recommended age of CRC screening among average-risk individuals (U.S. Preventive Services Task Force, 2008, 2021a, 2021b). There are very few studies that focus exclusively on Black men and interventions for increasing CRC screening uptake. One study cited that only 5% of such interventions focused solely on Black men (Rogers et al., 2020). Those studies that exclusively focus on Black men often include a very specific subset, such as Black men living with HIV (Kelly et al., 2021).
CRC screening uptake is known to increase with consistent contact with the medical system, yet there are several factors that influence health care utilization in those suffering from multiple chronic diseases including age less than 65, insurance status, immobility, poor subjective health status, lack of alcohol consumption, and physical inactivity (K. Y. Kim et al., 2020; Unanue-Arza et al., 2021; Weber et al., 2008). Furthermore, the management of chronic diseases by providers can influence CRC screening, but there have been mixed reviews to support this. Some studies have shown higher CRC screening rates due to more frequent contact with the health care system, while other studies have shown lower rates of CRC screening due to focus on the management of other chronic conditions (Coronado et al., 2021). One study found that a higher number of co-morbid conditions and higher disease severity resulted in lower rates of CRC screening completion, except for patients with diabetes (Coronado et al., 2021). These findings largely align with the results of our study and suggest a need for more research examining characteristics of patients who use CRC screening as recommended to promote outreach to patients who do not and achieve the 80% in Every Community goal set out by the National Colorectal Cancer Roundtable and Healthy People 2030 (Office of Disease Prevention and Health Promotion, n.d.; Shellnutt, 2020).
Given the low rates of reported CRC screening, disparities in undiagnosed or poorly managed chronic conditions, delay in seeking medical care, and likelihood of inadequate access to health care (Gilbert et al., 2016), health-behavior-change-focused interventions are needed to reduce disparities in CVD- and CRC-related outcomes among Black men. Interventions modeled after the 24-week community-based lifestyle intervention, Black Impact (which demonstrated a positive effect on overall Life’s Simple 7 scores and subcomponents including weight, body mass index, cholesterol, glucose, and diet in Black men), may offer insights toward solutions to such disparities in Black men (Joseph et al., 2022). Other culturally relevant interventions that have demonstrated the potential for promoting behavior change among Black men are inclusive of gender-specific messaging (Lucas et al., 2023). However, there are currently few lifestyle interventions that focus exclusively on Black men and address all LS7 components (Elgazzar et al., 2020).
Community-based interventions can be used to understand and address the injustices that fuel health care inequities, social determinants of health, the mistrust of the health care system, and poor health outcomes in communities of color (Elgazzar et al., 2020; Gray et al., 2022). Unequal access to quality health resources, lack of diversity among health professionals, and racism promoting disease, unhealthy aging, and excess disease burden contribute significantly to health disparities among Black adults. A multifaceted approach that involves patient-centered care, patient-centered outcomes, community-based-interventions, and increased research with minority populations is warranted (Dhaliwal et al., 2022). The successful recruitment of Black men into community-based research and interventions requires a tactical approach that incorporates culturally appropriate strategies. This will require face-to face contact with community members in formal and community settings and health fairs; passive recruitment through media, phone calls, and printed advertisement; and encouraging enrolled participants to spread the word with family and friends (Christy et al., 2016; Davis et al., 2018).
Our study highlights a group of men with elevated levels of CVH and yet lower rates of CRC screening, despite being potentially vulnerable to poorer outcomes from CRC if diagnosed. Future interventions aimed at targeting CVD- and CRC-related risk factors will be paramount to reduce two of the leading causes of death among Black men.
Our study is the first to examine LS6, CVH-related metrics and self-reported CRC screening in Black men. Second, our study included de-identified data from community-based testing contributing to a cohort of Black men with varying socioeconomic status. Third, structured questionnaires were used over the 4-year period. Finally, clinical assessments were performed by trained medical staff.
Despite said study strengths, there are still several limitations to report. One LS7 metric, diet, was excluded in this study due to the lack of an abbreviated measure of the AHA’s guidelines for an ideal diet. However, previous studies reported 0% to 2% of Black adults attain ideal dietary intake (Aboagye-Mensah et al., 2020; Effoe et al., 2017; Joseph et al., 2016; Shay et al., 2012). In addition, daily consumption of fruits and vegetables for many Black men is low (Rogers et al., 2017), which is associated with an increased risk of chronic diseases (Shikany et al., 2018). Nonetheless, a diet high in saturated fats, red and processed meats; and low in fruits, vegetables, and fiber has been linked to both CRC and CVD, the exclusion of diet from our study could have impacted overall CVH scores (Chan et al., 2011; Fan et al., 2017; Handy et al., 2018; Knudsen et al., 2022). More specifically, the intake of sodium and processed meat are highly correlated, but the intake of processed meat is an independent risk factor for CRC. Even though sodium intake is not an independent risk factor for CRC, dietary reduction in sodium is still recommended to improve outcomes for hypertension, metabolic syndrome, and CVDs (Feng et al., 2021).
CRC screening completion was assessed through a self-reported, cross-sectional survey, and assumptions of average-risk. Assessment of CRC screening rates typically rely on self-reporting, which are subject to incongruencies between self-reports and verified medical records (Manning et al., 2016). It is worth noting that in our study, however, CRC screening modalities were grouped together under one screening variable; however, if the modalities were analyzed separately, the study outcome may have been different. Other factors that were omitted from our study included daily alcohol consumption and family and genetic history of CRC. In addition to including those factors, future studies should consider verifying the self-reported surveys with medical records. Owing to the limited representation of Black men in previous studies involving CRC, there are few studies in which we can compare our work. Finally, we report data collected in which 59.4% of participants reported private insurance and 66.0% reported employment. The results may not be generalizable to other communities of Black men.
Conclusion
Future research, of mixed-method approaches, should focus on reducing disparities in CVD and CRC-related lifestyle risk factors by improving CRC screening utilization and disease-related outcomes among Black men. These efforts should focus on increasing CRC screening rates in all Black men, especially considering the increased risk and mortality rates of both CVD and CRC. Interventions should focus on all Black men, but particularly men that are assumed to be healthy with high CVH, given that they may have less contact with the health care system, may not be knowledgeable about recently changed CRC screening recommendations, nor recognize their risk of CRC.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883241249642 – Supplemental material for Cardiovascular Health and Colorectal Cancer Screening in Black Men
Supplemental material, sj-docx-1-jmh-10.1177_15579883241249642 for Cardiovascular Health and Colorectal Cancer Screening in Black Men by Kasey Hornbuckle, Amaris Williams, Joshua J. Joseph, Timiya S. Nolan, Darrell M. Gray, John Gregory and Aldenise P. Ewing in American Journal of Men’s Health
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Preparation of this manuscript and data analysis was supported by The Ohio State University Center for Clinical and Translational Science grant support (National Center for Advancing Translational Sciences, grant no. UL1TR002733). J.J.J. was supported by a grant from the National Institute of Diabetes, Digestive, and Kidney Diseases (grant no. K23DK117041) and the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program Award (rant no. ID# 76236). T.S.N was supported by a grant from the National Cancer Institute (grant no. K08CA245208). The views expressed in this manuscript are those of the authors and do not necessarily represent the views of the National Heart, Lung, and Blood Institute; the NCI; or the U.S. Department of Health and Human Services.
Ethics Approval and Consent to Participate
This project was classified as exempt by The Ohio State University Biomedical Sciences Institutional Review Board.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
