Abstract
The aim of this study was to investigate the potential mechanism through which Yishen Tongluo decoction (YSTL) repairs DNA damage caused by benzo(a)pyrene diol epoxide (BPDE) in mouse spermatocytes (GC-2). The GC-2 cells were divided randomly into the control group, BPDE group, and low-, medium-, and high-dose YSTL groups of YSTL decoction. A comet assay was used to detect the DNA fragment index (DFI) of cells in each group. Based on the DFI results, whole transcriptome sequencing was conducted, followed by trend analysis, gene ontology (GO) enrichment analysis, kyoto encyclopedia of genes and genomes (KEGG) enrichment analysis, and ceRNA network analysis. Compared with the control group, the BPDE group reported a significant increase in the DNA fragmentation index (DFI) (p < .05). Compared with the BPDE group, the low-, high- and medium-dose YSTL groups had a significantly reduced DFI (p < .05). Whole-transcriptome sequencing revealed seven differentially expressed circRNAs, 203 differentially expressed miRNAs, and 3,662 differentially expressed mRNAs between the control group and the BPDE group. There was a total of 12 differentially expressed circRNAs, 204 miRNAs, and 2150 mRNAs between the BPDE group and the traditional Chinese medicine group. The pathways involved include DNA repair pathway, nucleotide excision repair pathway, base excision repair pathway, etc. The ceRNA network reported that Hmga2 was the core protein involved, novel_cir_000117 and mmu-miR-466c-3p were located upstream of Hmga2, and they were regulatory factors associated with Hmga2. Finally, we conclude that YSTL decoction may repair sperm DNA damage caused by BPDE through the novel_cir_000117-mmu-miR-466c-3p-Hmga2 pathway.
Keywords
Introduction
According to a study conducted by the World Health Organization between 1982 and 1985, approximately 15% of couples of childbearing age worldwide suffer from infertility, of which approximately 50% are caused by male factors (World Health Organization, 1987). Semen analysis is a routine examination to assess sperm fertilization capacity, which includes parameters such as sperm concentration, motility, viability, and morphology (Radwan et al., 2016). However, in infertile patients, 50% of them may have normal results in semen analysis (Le et al., 2019). Therefore, attention has shifted toward more in-depth research on the sperm DNA fragmentation index (DFI), as elevated levels of DFI have been identified as an important cause of male infertility, recurrent miscarriages in females, and failure of assisted reproductive techniques (Lewis & Simon, 2010; Wiweko & Utami, 2017).
The reasons for the increase in the sperm DFI are complex and diverse, including factors such as age, environmental pollution, abstinence time, oxidative stress, and unhealthy lifestyle habits (Lu, 2015; Radwan et al., 2016). With the rapid development of modern industrialization and changes in people’s lifestyles, environmental pollution has become one of the main causes of sperm DNA damage (Vecoli et al., 2016). Benzo[a]pyrene (Bap) is mainly derived from the exhaust gases generated by the combustion of fossil fuels such as coal, petroleum, and coal tar, including automobile exhaust, barbecuing, and smoking, among others (Jeng et al., 2015; Ji et al., 2018). According to reports, benzo(a)pyrene can form dihydrodiol epoxide benzo(a)pyrene-DNA adducts with DNA in sperm, thereby damaging the integrity of sperm DNA (Cui et al., 2023) and subsequently affecting semen quality.
In 2003, a study reported that there was no recognized treatment for sperm DNA damage because of its diverse etiology and complex mechanisms. The characteristics of traditional Chinese medicine, including multiple components, multiple targets, and multiple pathways, can minimize inflammatory reactions, prevent oxidative stress, reduce DFI, and regulate the proliferation and apoptosis of reproductive cells (Zhou et al., 2019). The Yishen Tongluo decoction (YSTL) consists of Dodder Seed, Epimedium, Achyranthes Bidentata, Astragalus, Salvia Miltiorrhiza, Rehmannia Glutinosa, and Hirudo Medicinalis. It is an empirical herbal formula that has been clinically used by the research team for over 20 years. Earlier research has suggested that this concoction has the capacity to decrease DFI (Zhang et al., 2022). The Yishen Tongluo Decoction has the ability to repair DNA damage in sperm from rats (Zhang et al., 2021, 2022, 2023). The role of preventing abnormal methylation of sperm DNA (Sun et al., 2019). However, the mechanism of action of this decoction is still unclear. This study aims to investigate the key genes and pathways involved in the repair of sperm DNA damage by YSTL through the establishment of a sperm DNA damage cell benzo(a)pyrene diol epoxide (BPDE) and the screening of differential mRNA and ncRNA expression profiles in mouse germ cells (GC-2) using whole-transcriptome sequencing. Integrated analysis methods and ceRNA regulatory network analysis will be employed to explore the potential mechanisms underlying the repair of sperm DNA damage by YSTL.
Materials and Methods
Cell Lines and Animals
Ten specific pathogen-free (SPF) male Wistar rats weighing 250 ± 20 g were purchased from Henan Skbes Biotechnology Company (Production License No: SCXK [Yu] 2020-005). The rats were housed under the following conditions: temperature of 20 to 25°C, humidity of 40% to 70%, a 12-hour light-dark cycle, standard feed, ad libitum access to water, and regular replacement of feed and bedding. The animal experimental protocol was reviewed and approved by the Ethics Committee of Henan Provincial Hospital of Traditional Chinese Medicine (Approval No: YFYDW2023009). The GC-2spd mouse spermatogonia cell line was purchased from Shanghai Meilian Biological Research Institute (Catalog No: C476).
Reagent
Dulbecco’s modified eagle medium (DMEM) high glucose medium (item number: PM150210, Procell); fetal bovine serum (item number: 164210, Procell); DNA damage analysis kit by KTA3040 comet assay (item number: KTA3040, Abbkine); Phosphate Buffered Saline (PBS) (item number: PB180327, Procell); NaCl powder (item number: 7647-14-5, Hengxing reagent); NaOH granule (item number: 82001, Hengxing reagent); anhydrous ethanol (item number: 64-17-5, Hengxing reagent); Tris-borate-EDTA (TBE) electrophoresis buffer (5×)(item number: PS104S, Ya Enzymes Biology); Dihydroxy epoxy benzopyrene (item number: B287550, Canadian TRC).
Instrument
311CO2 incubator (Thermo Company, Germany); DNA damage analysis kit by KTA3040 comet assay (Abbkine Company, USA); ICX4I inverted microscope (SOPTOP Company, China); ICX4I fluorescence microscope (SOPTOP Company, China); HH-4A digital display constant-temperature water bath pot (Guohua Instrument Company, China); PURELABChorus1 ultrapure water meter (ELGA Company, UK).
Preparation of Drug-Containing Serum
YSTL was a combination of 20 g of Dodder, 20 g of Epimedium, 10 g of Rehmanniae Radix Preparata, 30 g of Radix Salviae Miltiorrhizae, 20 g of Astragalus membranaceus, 6 g of leeches, and 10 g of raw Achyranthes bidentata. All traditional Chinese medicines were purchased from the pharmacy of Henan Province Hospital of Traditional Chinese Medicine. The concentrated decoction of traditional Chinese medicine with a concentration of 2.4 g/mL was prepared by the pharmacy and refrigerated at 4°C. The rats were given intragastric administration once a day for 7 days, and after the last intragastric administration for 1 hour, blood was collected from the abdominal aorta after anesthesia, 30 min was placed in the sterile tube, 2,000 r/min was placed in the sterile tube, the supernatant was taken after centrifugation for 10 min, 30 min was placed in a 56°C water bath to inactivate, and a 0.22-μm microporous filter was used to remove bacteria and stored in a −20°C refrigerator.
Cell Culture
After resuscitation, GC-2 cells were cultured in high glucose DMEM containing 10% fetal bovine serum and 1% double antibodies. The cells were placed in a CO2 incubator at 37°C and 5% CO2 for cultivation. When the cells reached 80% to 90% confluence, they were passaged by transferring one-fourth of the cells to a new culture medium for further cultivation.
Grouping and Administration
The GC-2 cells were randomly divided into five groups: the control group (10% control serum), the BPDE group (1-µm BPDE), the low-dose YSTL group (1-µm BPDE + 8% YSTL medicated serum), the medium-dose YSTL group (1-µm BPDE + 10% YSTL medicated serum), and the high-dose YSTL group (1-µm BPDE + 15% YSTL medicated serum). After 24 hours of BPDE administration, different concentrations of YSTL-medicated serum were added to each group. After 24 hours, the cells were collected for experiments.
The Comet Assay is Used to Detect DNA Damage in Sperm
The DNA damage level of mouse spermatocytes induced by BPDE and the DNA repair level of mouse spermatocytes induced by YSTL drug-containing serum were detected by comet assay. The cell sample was combined with agarose at a ratio of 1:10, and the 75-μL mixture was immediately transferred to the top of the agarose base layer to cover completely, avoiding light at 4°C for 15 min. The glass slides were transferred to 1× cleavage buffer equipped with precooling, 30 to 60 min was cracked away from light, 1 × cleavage buffer was removed and replaced with precooled alkaline solution, and 30 min was unrotated at 4°C to prevent light from unspinning DNA. Transfer the slide to a horizontal electrophoresis tank and apply a voltage of 10 to 15 min to the chamber at 1 V/cm. Transfer the slides from the electrophoresis room to a clean container containing precooled deionized water, wash 2 min, repeat 3 times, absorb ionized water, add precooled 70% ethanol, wash 5 min, remove glass slides from 70% ethanol and air dry. Then, 50 μL/well of 1 × PI dye was added, and the samples were dyed for 10 min at room temperature and observed under a fluorescence microscope. Each slide was magnified 200 times with a fluorescence microscope, and three nonoverlapping comet images were randomly selected for each group. Then, analyze it with comet assay software project (CASP).
Full Transcriptome Sequencing
Gene Denovo Biotechnology Co., Ltd. conducted the complete transcriptome sequencing. Total RNA was extracted with TRIzol reagent, and its quality was evaluated using an Agilent 4200 Bioanalyzer. Small RNA libraries were prepared, and their quality and yield were assessed with Agilent 2100 and ABI Reverse Transcription-Polymerase Chain Reaction (RT-PCR) systems. To ensure data quality, raw data were filtered to eliminate invalid data before analysis. Fastp was used to carry out quality control on the raw reads obtained from sequencing, resulting in clean reads. The cDNA libraries were sequenced on the Illumina HiSeq high-throughput sequencing platform.
Difference Analysis
For mRNA, DESeq2 software was used to perform differential expression analysis on two compared groups of mRNAs. Based on the differential analysis results, mRNAs with false discovery rate (FDR) < .05 and|log2FC| > 1 were selected as significantly differentially expressed mRNAs. For miRNA and circRNA, edgeR software was used to conduct differential analysis on miRNA and circRNA. In the samples and comparison groups, circRNAs and miRNAs with fold change ≥2 and p < .05 were considered significantly differentially expressed circRNAs and miRNAs.
Analyzing Trends and Pathways in Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG)
According to the selected differentially expressed genes, a trend analysis method was employed to screen potential targets of YSTL action. A total of eight spectral clustering patterns with different change trends were obtained. Then, GO analysis and KEGG pathway enrichment analysis were performed on the differentially expressed genes that were downregulated in the BPDE group and restored after YSTL intervention, as well as those upregulated in the BPDE group and restored after YSTL intervention.
Construction of the Coexpression Network
First, circRNAs and mRNAs with significant differences were screened, with a difference threshold of FDR < .05 and |log2FC| > 1. The threshold for screening significantly different miRNAs was set at p < .05 and |log2FC| > 1. Then, potential ceRNAs with mutual interactions were screened, and after obtaining the ceRNA interaction information, a ceRNA regulatory network diagram was constructed using Cytoscape.
Statistical Analysis
Statistical analysis of the data was carried out using SPSS 26.0 software. The data are presented as the mean plus standard deviation (SD). The normality of the distribution was assessed using the Shapiro–Wilk test, and the equality of variances was examined using Levene’s test. If the data exhibited a normal distribution and equal variances, intergroup differences were analyzed using one-way analysis of variance (ANOVA), and pairwise comparisons were carried out using least significant difference (LSD). Spearman correlation analysis was employed, and a two-tailed test was conducted with a significance level of .05. p < .05 denoted significance.
Results
Comet Assay
The comet images of the control group, BPDE group, and YSTL low-, medium-, and high-dose groups are shown in Figure 1. The GC-2 cell DNA of the control group was intact, with uniform cell nucleus size and circular fluorescence, uniform fluorescence intensity, smooth edges, and no comet tail phenomenon, and the DNA was not significantly damaged (Figure 1A). The GC-2 cell DNA of the BPDE group was severely damaged, with a tail resembling a comet in a broom-like shape, similar to a comet (Figure 1B). With the increase in concentration in the YSTL low-, medium-, and high-dose groups, the GC-2 cell nucleus gradually increased in size, the comet tail length gradually shortened, the tail fluorescence intensity gradually weakened, and the diameter of the comet head lengthened (Figure 1C–E).

Comet Assay Images of Mouse Germ Cells. (A) Control Group. (B) BPDE Group. (C) YSTL Low-Dose Group. (D) YSTL Medium-Dose Group. (E) YSTL High-Dose Group. (F) Histogram of Comet Assay Results.
Effect of YSTL on DNA Damage and Repair in Mouse Spermatocytes
The effects of BPDE and YSTL-containing serum at different concentrations are reported in Table 1. TailDNA%, Comet tail length, and Olive tail moment in the BPDE group were significantly higher than those in the control group (p < .001, p < .001, p < .001). TailDNA% in the YSTL medium- and high-dose serum groups was significantly lower than that in the BPDE group (p = .003, p < .001). The comet tail length in the YSTL low-, medium-, and high-dose serum groups was significantly lower than that in the BPDE group (p = .012, p < .001, p = .001). The Olive tail moment in the YSTL low-, medium-, and high-dose serum groups was lower than that in the BPDE group (p < .001, p = .001, p < .001). TailDNA%, comet tail length, and Olive tail moment decreased with increasing concentrations of YSTL-containing serum. TailDNA%, comet tail length, and Olive tail moment were significantly negatively correlated with the concentration of YSTL-containing serum (r = −.896, p < .001; r = −.9, p < .001; r = −.843, p < .001). Based on the experimental results, YSTL high-dose serum was used for subsequent whole-transcriptome sequencing (Table 2).
Experimental Results of the Repair Effect of Different Concentrations of YSTL Decoction Containing Serum on DNA Damage in Mouse Spermatocytes.

Note. All groups were compared with the BPDE group, *** indicates statistical significance (p < .001).
Correlation Analysis of Drug Concentration, Tail DNA%, Comet Tail Length (TL), and Olive Tail Moment (OTM).

Note. ** indicates statistical significance (p < .01).
Difference Analysis
When FC ≥ 2.0 and the p-value < .05 threshold, there were seven differentially expressed circRNA genes, 203 differentially expressed miRNA genes, and 3,662 differentially expressed mRNA genes between the control group and the BPDE group. Between the BPDE group and the drug group, there were 12 differentially expressed circRNA genes, 204 differentially expressed miRNA genes, and 2,150 differentially expressed mRNA genes (Figures 2–4).

Differentially Expressed circRNAs Analysis of the Control Group, BPDE Group and YSTL Decoction Group. (A, B) Volcano Plot of DEcircRNAs. (C, D) Histogram of DEcircRNAs. (E) Heatmaps of DEcircRNAs by Three Groups.

Differentially Expressed miRNAs Analysis of the Control Group, BPDE Group and YSTL Decoction Group. (A, B) Volcano Plot of DEmiRNAs. (C, D) Histogram of DEmiRNAs. (E) Heatmaps of DEmiRNAs by Three Groups.

Differentially Expressed mRNAs Analysis of the Control Group, BPDE Group and YSTL Decoction Group. (A, B) Volcano Plot of DEmRNAs. (C, D) Histogram of DEmRNAs. (E) Heatmaps of DEmRNAs by Three Groups.
Trend Analysis
The total gene expression levels of the control group, BPDE group, and YSTL group were compared pairwise. There were 3,662 differentially expressed mRNAs between the control group and the BPDE group and 2,150 differentially expressed mRNA genes between the BPDE group and the YSTL group. Trend analysis was performed on these differentially expressed mRNAs. Based on the different trends observed in the two comparative groups, they were classified into eight types of trends, namely, Profile 0 to Profile 7. Among them, Profile 2 represents genes that were downregulated in the BPDE group and restored after YSTL intervention. Profile 5 represents genes that were upregulated in the BPDE group and restored after YSTL intervention. In Profile 2, 593 differentially expressed mRNAs were downregulated in the BPDE group and restored after YSTL intervention (p < .05); in Profile 5, 628 differentially expressed mRNAs were upregulated in the BPDE group and restored after YSTL intervention (p < .05) (Figure 5).
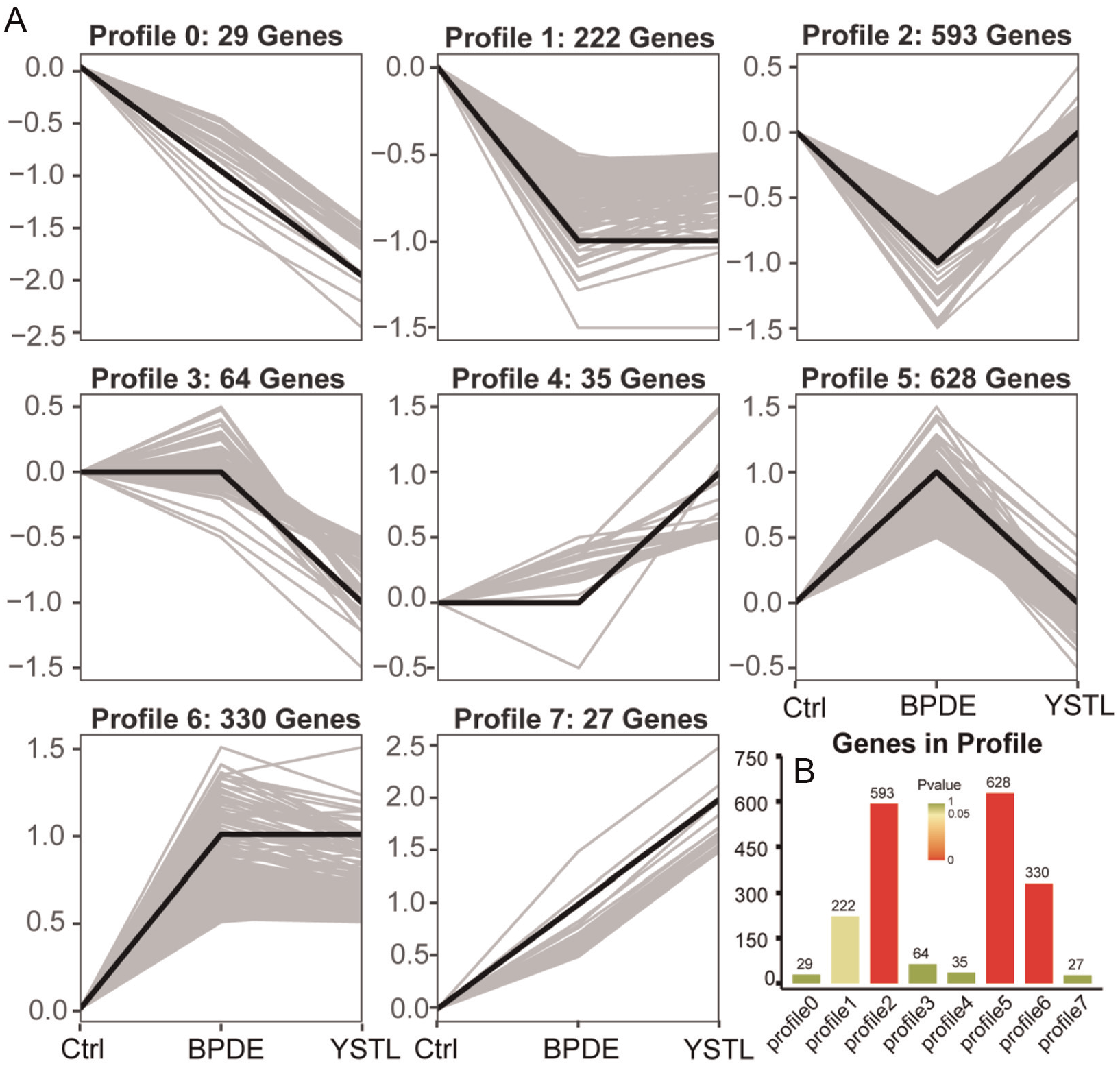
Trend Analysis of Differential Expression of miRNAs. (A) The Trend Pattern Chart: Based on the Trend Normalization of Genetic Data, the Trend Pattern Chart is Plotted, Where the Black Line Represents the Trend Line, and the Gray Line Represents Each Gene. (B) Histogram of the Number of Trend Genes and p-values. The Height of Each Bar Represents the Number of Genes, While the Color of the Bars Represents the p-value.
GO Enrichment Analysis
The differentially expressed genes in Profile 2 and Profile 5 were analyzed by GO. In the category of biological processes, there are pathways related to or repairing sperm DNA damage: DNA repair, nucleotide-excision repair, base-excision repair, mismatch repair, postreplication repair, and double-strand break repair (Figure 6A).

Functional Enrichment Analysis of Differentially Expressed mRNAs. (A) GO Enrichment Analysis of DEmRNAs Between Profile 2 and Profile 5. (B) KEGG Enrichment Analysis of DEmRNAs Between Profile 2 and Profile 5.
KEGG Enrichment Analysis
The genes in Profile 2 and Profile 5 were analyzed by KEGG. In the category of biological processes, there are pathways related to or repairing sperm DNA damage: cAMP signaling pathway, apoptosis, mTOR signaling pathway, PI3K-Akt signaling pathway, JAK-STAT signaling pathway, P53 signaling pathway, and nucleotide excision repair (Figure 6B).
Constructing a ceRNA Regulatory Network
We used mRNA as the core and searched for circRNAs and miRNAs related to regulating mRNA based on ceRNA theory. Hmga2 was upregulated in the control group and BPDE group and downregulated in the BPDE group and YSTL group. In the control group and BPDE group, mmu-miR-466c-3p was located upstream of Hmga2 and negatively correlated with Hmga2 expression. novel_cir_000117 was located upstream of mmu-miR-466c-3p and negatively correlated with mmu-miR-466c-3p expression. It was upregulated in the control group and BPDE group and downregulated in the BPDE group and YSTL group. We constructed the novel_cir_000117-mmu-miR-466c-3p-Hmga2 pathway (Figure 7).

CeRNA Network. (A) ceRNA Network of the Control Group and BPDE Group. (B) CeRNA Network of the BPDE Group and YSTL Decoction Group.
Discussion
Sperm DFI plays a crucial role in normal pregnancy and is widely utilized as an indicator for evaluating male fertility in clinical practice (Lewis & Simon, 2010; Zeqiraj et al., 2018). Research has demonstrated that when sperm DNA damage exceeds 27%, the fertility rate significantly decreases, leading to infertility or miscarriage (Corona et al., 2019). The mechanisms underlying sperm DNA damage include abnormal spermatogenesis, oxidative stress-induced injury, and abnormal sperm apoptosis (Aitken & Koppers, 2011; Muratori et al., 2019). Modern medicine often employs antioxidant, anti-infection, and traditional Chinese medicine treatments to address sperm DNA damage (Abad et al., 2013; Rabaça et al., 2020; Zhai et al., 2018). In 2020, there have been many studies reporting that both traditional Chinese herbal medicine and Chinese patent medicine can enhance sperm quality, specifically in terms of sperm motility and concentration (X. Chen et al., 2020; Zhang et al., 2020). There are also studies reporting that traditional Chinese medicine has the potential to repair sperm DNA damage (W. Q. Chen et al., 2018; Li et al., 2021, 2022). However, there is a limited quantity of research available and a lack of studies investigating the underlying mechanisms of action. The extract of Shengjing capsules can inhibit testicular oxidative damage induced by chromium oxidation, protect sperm DNA, and reduce the DFI (Zhou et al., 2016). The Shengjing capsule contains 19 traditional Chinese medicines, including Cuscuta chinensis, Epimedium, and Astragalus (Wang et al., 2022). These ingredients are similar to the components of YSTL in this research, suggesting that both traditional Chinese medicines can reduce the sperm DFI. This study confirms the role of YSTL in improving sperm DNA damage and elucidates the mechanism of YSTL in repairing sperm DNA damage through whole-transcriptome sequencing.
Full transcriptome sequencing refers to the acquisition of the complete set of RNA molecules, including both coding and noncoding RNAs, produced by a specific species or particular cells under a specific functional state (Costa et al., 2010). Full transcription sequencing technology has the advantages of integrity, systematicness, individual tissue differences, time independence and so on, which can better clarify the principle of differentiation and treatment of traditional Chinese medicine and the action mechanism of prescription and medicine at the gene level. The gene expression profiles were screened by the full transcriptome. In the control group and BPDE group, a total of 3662 genes were screened, while 2150 genes were screened in the BPDE group and YSTL group. Enrichment analysis of mRNA in Profiles 2 and 5 revealed enrichment of pathways related to sperm DNA damage and repair, including DNA repair, the NER signaling pathway, the BER signaling pathway, the MMR signaling pathway, the postreplication repair pathway, the DSB signaling pathway and other signaling pathways. DNA replication is a precise and strictly regulated process. However, DNA damage occurs continuously within cells. In mammals, cells generate approximately 105 DNA damages per day due to spontaneous replication errors and cellular metabolism (Hoeijmakers, 2009). To ensure the integrity and stability of genetic material, DNA damage repair mechanisms play a crucial role. Nucleotide excision repair, base excision repair, mismatch repair, double-strand break repair, and postreplication repair are the repair pathways for DNA damage. These mechanisms can recognize and repair DNA damage caused by various factors.
KEGG is mainly concentrated in the pathways of sperm DNA damage and repair: p53 signaling pathway, cAMP signaling pathway, apoptosis, mTOR signaling pathway, PI3K-Akt signaling pathway, and NER. There were significant differences in the p53 signaling pathway, apoptosis and JAK-STAT signaling pathway. Sperm DNA damage mainly comes from reactive oxygen species (ROS), sperm chromatin packaging, and apoptosis (Gunes et al., 2015; Tamburrino et al., 2012). Studies have demonstrated that an overabundance of ROS can cause DNA damage due to oxidative stress (Tamburrino et al., 2012). One of the main cellular pathways activated by the accumulation of intracellular ROS is the JAK/STAT signaling pathway. During spermatogenesis, apoptosis maintains the balance between germ cells and supporting cells in terms of quantity (Aitken & Baker, 2013; Leduc et al., 2008). Maintaining balance in terms of quantity, morphology, and functionality of sperm is crucial. If apoptosis is abnormal and damaged sperm with DNA cannot be cleared in a timely manner, it can lead to a decline in sperm quality, impaired fertilization function, and abnormal embryo development. When sperm DNA is exposed to toxic substances, it stimulates the synthesis of p53 protein. The p53 protein triggers cell sensors to initiate apoptosis by increasing the production of the pro-apoptotic proteins Bax and Bak and stimulating the synthesis of the pro-apoptotic proteins in the Bcl-2 family (Aitken & Baker, 2013; Jana et al., 2010; Lu et al., 2023).
Bap is a mutagenic and carcinogenic environmental pollutant. Several Studies have reported that Bap can be used as a recognized method to induce sperm DNA damage (Yang et al., 2019), which can cause DNA damage in male sperm and this conclusion has been validated in subsequent animal cell experiments (Mohamed et al., 2010; O’Brien et al., 2016). However, the mechanism by which Bap induces sperm DNA damage is not yet clear. Bap can lead to cancer and lung diseases, and its mechanism of action may be related to ncRNA. Jin et al. found that Bap can cause lung diseases, and their subsequent experiments reported that lung diseases caused by Bap exposure may be related to the dysregulation of miRNA-3173-5p, miR-629-3p, miR-9-5p, miR-1343-3p, and miR-219a-1-3p (Jin et al., 2022). Jiang et al. conducted high-throughput sequencing on the human bronchial epithelial cell line BEAS-2B intervened by BaP, and found that miRNA, lncRNA, and CircRNA play significant regulatory roles in the transformation of human bronchial epithelial cells induced by BaP (Jiang et al., 2018). Xiao et al. performed high-throughput sequencing on the miRNA expression profile in the plasma of lung cancer patients exposed to Bap occupationally and found that the mechanism of Bap-mediated lung cancer in plasma may be related to the dysregulation of miRNAs (Xiao et al., 2024). The above studies confirmed that circRNA, miRNA, and other non-coding RNAs are closely related to the pathogenic mechanism of Bap.
Several studies have reported that traditional Chinese medicine may treat male infertility by regulating ncRNA expression levels. Curcumin, an effective ingredient in traditional Chinese medicine turmeric, was found to reduce testicular damage and germ cell apoptosis caused by heat stress, thereby treating male infertility. Its mechanism of action may be related to the upregulation of miRNA-21 and circRNA0001518 (Paskeh et al., 2024). Astragalus is another traditional Chinese medicine commonly used to treat male infertility, and its main active ingredient is astragalus polysaccharide. Wu et al. (2017) found that dietary supplementation with astragalus polysaccharide improved testicular enzyme activity and function, which may be achieved through modulation of testicular nutritional metabolism and cytotoxic pathways mediated by differential miRNAs. Astragalus, as one of the main components of YSTL formula, indicates that YSTL formula may also improve reproductive function, and its mechanism of action may involve miRNAs.
To further explore the mechanism of YSTL in repairing sperm DNA damage, we conducted ncRNA expression profiling and constructed a circRNA-miRNA–expl regulatory network based on the ceRNA theory. YSTL can downregulate the overexpression of high mobility group protein A2 (Hmga2) induced by BPDE, and Hmga2 is located in the base excision repair pathway, which can inhibit the activation of the DNA repair system, including binding to the promoter of the nucleotide repair gene ERCC1 and inhibiting its activity (Pierantoni et al., 2003), and directly inhibiting the activation of the repair signaling pathway of DNA double-strand breaks (Andrieux et al., 2004; Santulli et al., 2000), thereby affecting the repair of sperm DNA damage. BPDE can downregulate the expression of mmu-miR-466c-3p, which has binding sites for Hmga2. According to the regulatory relationship between circRNA and miRNA, we analyzed the upstream and downstream of mmu-miR-466c-3p and found that novel_cir_000117 is located upstream of mmu-miR-466c-3p and has binding sites with mmu-miR-466c-3p. In the BPDE group, novel_cir_000117 was negatively correlated with mmu-miR-466c-3p, while it was positively correlated with Hmga2. After BPDE treatment, novel_cir_000117 was upregulated in the expression profile, but it was downregulated after YSTL intervention. Since the results of whole transcriptome sequencing can only be used as a predictive tool, the mutual regulatory relationship between molecules has not been verified, and the small sample size may affect the reliability of the results, mmu-miR-466c-3p was not differentially expressed in M-vs.-T, and we mainly constructed the ceRNA network based on mRNA. We concluded that YSTL may improve sperm DNA damage through the novel_cir_000117-mmu-miR-466c-3p-Hmga2 pathway.
Conclusion
In conclusion, this study indicated that BPDE poisoning could lead to DNA damage in mouse spermatogonia cells (GC-2), and YSTL could repair the DNA damage caused by BPDE in mouse spermatogonia cells (GC-2). The mechanism of action may be related to the circ_000117-miR-466c-Hmga2 pathway. This study provides theoretical and experimental basis for later animal cell-level experimental verification.
Footnotes
Author Contributions
W.W., C.Z., and Z.W. contributed to the conception, participated in the study design and reviewed the manuscript. W.W., C.Z., and Z.W. wrote the manuscript and conducted the literature search. Z.W., C.Z., Q.Z., Z.H., N.L., M.M., and H.S. revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 82174377 and No. 81603632), the China Postdoctoral Science Foundation (No. 2023T160201 and No. 2022M721068), the Science and Technology Program of Henan Province (No. 232102311199), the Key Scientific Research Project of Colleges and Universities in Henan Province (No.23A360005), the Special Scientific Research Project of the National TCM Clinical Research Base (No. 2021JDZX2056). All the authors of the manuscript are immensely grateful to the foundations for their valuable support.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
