Abstract
The thalamus and the mesencephalon have a complex blood supply. The artery of Percheron (AOP) is a rare anatomical variant. Occlusion of this artery may lead to bithalamic stroke with or without midbrain involvement. Given its broad spectrum of clinical features, AOP stroke is often misdiagnosed. Usually, it manifests with the triad of vertical gaze palsy, memory impairment, and coma. In this article, we report three cases of bilateral thalamic strokes whose clinical presentations were dominated by a sudden onset of hypersomnia. We also reviewed last 5 years’ publications related to the AOP strokes in males presenting sleepiness or equivalent terms as a delayed complication. The AOP stroke may present a diagnostic challenge for clinicians which should be considered in the differential diagnosis of hypersomnia.
The thalamus is an egg-shaped structure of gray matter on the human diencephalon (Khanni et al., 2018). Its vascular supply is complex and presents multiple anatomical variants. The artery of Percheron (AOP), which is a rare anatomical variant, is characterized by a solitary common arterial trunk arising from the posterior cerebral artery (Lamboley et al., 2011). Occlusion of this artery is rare and may lead to a bilateral paramedian thalamic infarction with or without midbrain involvement (Lamot et al., 2015). This uncommon ischemic stroke syndrome, with its broad spectrum of clinical presentations, is often misdiagnosed and therefore represents a diagnostic challenge for physicians. Acute hypersomnia is an unusual complication of this stroke (Bollu et al., 2017). Here, we report three cases of bithalamic stroke whose clinical presentations were dominated by altered mental status, mainly consistent with hypersomnia. We also briefly report the results of the published cases/case series (2015–2020 period) of male patients presenting an AOP stroke.
Cases
The Ethics Committee of Farhat Hached Teaching hospital waived the need for ethics approval for the publication of the case series’ retrospectively obtained and anonymized data. However, written informed consents were obtained from the three patients for the publication of their cases.
Case 1
A previously healthy 73-year-old man presented to the emergency department with altered consciousness. On admission, his signs were within normal range. On the neurological examination, the patient had a fluctuant consciousness state fully reversible under pain stimuli, excessive daytime sleepiness, and nocturnal agitation. He had neither sensitive nor motor focal signs. All blood tests (e.g., electrolytes test, complete blood count [CBC], and liver function tests) were normal. A head computed tomography (CT) performed 2 hr after the symptoms onset was normal. This situation misled to an initial diagnosis of a coma secondary to viral encephalitis. Forty-eight hours after admission to the intensive care unit, a new brain CT scan showed bilateral thalamic hypodensities in the paramedian territory (Figure 1). Further evaluation aiming at identifying the stroke etiology revealed a rapid atrial fibrillation at electrocardiogram without abnormalities on Doppler ultrasound and transthoracic echocardiography, findings consistent with an embolic mechanism or a heart-brain syndrome. Treatment with anticoagulation (enoxaparin) and amiodarone was started. Two weeks later, there was a marked improvement in the clinical condition (somnolence and behavioral impairment). The patient was transferred to the cardiology department.

Head computed tomography scan illustrates bilateral thalamic hypodensities (arrows) compatible with an ischemic stroke.
Case 2
A 43-year-old man with a past medical history of hypertension was brought to the emergency department after 20-hr of unarousable sleepiness. There was no recent history of drug abuse, head injury, trauma, or seizure. On the physical examination, the patient was afebrile and his blood pressures were high at 230/120 mmHg. On the neurological examination, the patient had a Glasgow coma scale of 7 points (2, 2, and 3 points for ocular, verbal, and motor components) requiring invasive mechanical ventilation. No other abnormalities were noted (neither meningeal syndrome nor focal neurological deficit). On admission, the infectious, toxic, and metabolic encephalopathy tests (e.g., CBC, blood urea nitrogen, creatinine, plasma glucose, liver function tests, and urine drug screen) were normal. Initial CT scan was normal. Twenty-four hours later, a new brain CT scan revealed bilateral paramedian thalamic hypodensities, which were not present on the first CT examination (Figure 2). A 9-lead electrocardiogram showed a right bundle-branch block. A transthoracic and transesophageal echocardiogram, carotid, and vertebral Doppler ultrasound revealed no abnormalities. There was a gradual improvement in the patient’s clinical state. A detailed neurological examination after extubation revealed fluctuating arousal, complete vertical gaze palsy, thalamic aphasia, and behavior changes with agitation and increased impulsivity. The aforementioned psychiatric disorders were managed with haloperidol. Two weeks after hospitalization, the patient was discharged and referred to a neurologist.

Head computed tomography on an axial view illustrates an ischemic stroke (arrows) in the paramedian artery territory of both thalami.
Case 3
An 82-year-old man with a past medical history of hypertension, diabetes mellitus, and myocardial infarction presented to the emergency department with a brief loss of consciousness, followed by drowsiness episodes which were initially neglected and then worsened in a crescendo pattern. On the physical examination, the patient was sleepy, apathetic, and presented no other abnormalities. Results of the routine blood tests (e.g., electrolytes test, blood urea nitrogen, creatinine, plasma glucose, and CBC) were within the normal range. During the hospital course, the patient presented many periods of irrepressible need for sleep, and he was unable to stay awake and alerted during the major daytime waking episodes. Brain magnetic resonance imaging (MRI) showed bilateral paramedian thalamic infarcts (Figure 3). Doppler ultrasound and transthoracic echocardiography were unremarkable. A significant improvement in hypersomnolence during the hospital stay was noted and the patient was discharged on day six following the stroke.

Axial T2-weighted magnetic resonance imaging of the brain illustrates hyperintense bilateral lesions of the paramedian thalami (arrows) compatible with an acute ischemic stroke.
Discussion
The AOP was first described by Gerard Percheron in 1973 (Agarwal et al., 2014; Caruso et al., 2017). This artery arises from the proximal posterior cerebral arteries and supplies both the paramedian thalami and the mesencephalon (Figure 4). AOP is a rare anatomical variant that is present in 4%–12% of the population (Arauz et al., 2014; Kaya et al., 2010; Kocaeli et al., 2013; Uz, 2007). The prevalence of bilateral thalamic infarction caused by AOP occlusion is unknown since it is often misdiagnosed (Arauz et al., 2014). It “seems” that AOP stroke represents 0.1% to 2.0% of ischemic strokes and 4% to 18% of thalamic infarcts (Agarwal et al., 2014; Caruso et al., 2017; Garcia-Grimshaw et al., 2018; Lamot et al., 2015). There is no predilection as regards sex, race-ethnicity, and age in the reported cases of AOP stroke in literature. According to Garcia-Grimshaw et al. (2018), the aforementioned data depend on the etiology of the AOP stroke. Firstly, the latter can occur at any age, but it is particularly common in patients after their 30s mostly between 60 and 70 years (Lin et al., 2018). Secondly, there is a slight male predominance with a male to female ratio of 3:2 (Hermann et al., 2008; Stamm et al., 2018; Suzuki et al., 2016).
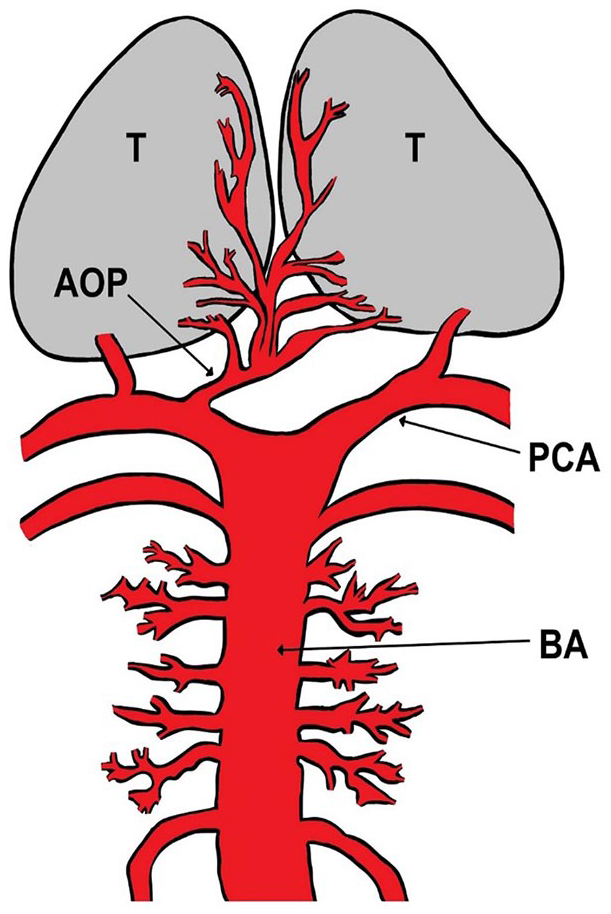
Artery of Percheron (AOP) illustration. BA = basilar artery; PCA = posterior cerebral artery; T = thalamus.
On May 27, 2020, a personal literature review based on a 2015–2020 PubMed search [key items: “artery of Percheron” AND (“case report” OR “case-series”)] found 73 papers published in English language (30, 37, and 6 papers included females, males, and both sexes, respectively). Among the 37 studies including only males, 13 reported sleepiness or equivalent terms (i.e., hypersomnolence, drowsiness, or excessive daytime sleepiness) as a delayed complication of an AOP stroke (Table 1; Afana et al., 2019; Aryan et al., 2016; Bailey & Khadjooi, 2016; Goico & Mikesell, 2018; Harisuthan et al., 2018; Ince & Asan, 2018; Kamasak et al., 2015; Khanni et al., 2018; Oliveira et al., 2018; Shah & Ali, 2018; Vasconcellos et al., 2016; Zelante et al., 2015; Zhou et al., 2015).
Results of the 13 Cases/Case-Series, Published During the 2015–2020 Period, Including Males Suffering From an Artery of Percheron (AOP) Stroke and Reporting Sleepiness or Equivalent Terms (Such as Hypersomnolence, Drowsiness, or Excessive Daytime Sleepiness) as a Delayed Complication.

Note. CT = computed tomography; CTA = CT angiography; DWI = diffusion-weighted imaging; MRA = magnetic resonance angiography; MRI = magnetic resonance imaging; n = number; Yrs = years.
The shape of the infarct can deduce the presence of AOP stroke because this anatomical variant typically causes bilateral paramedian thalamic-mesencephalic infarction (Agarwal et al., 2014; Lopez-Serna et al., 2009). The risk factors of AOP stroke are similar to those of ischemic ones. The two most frequent risk factors are microangiopathy and cardiac embolism (Arauz et al., 2014; Garcia-Grimshaw et al., 2018). The clinical presentations of AOP stroke are extremely variable. The most common clinical features are bilateral vertical gaze palsy (65%), memory impairment (anterograde and retrograde amnesia) (58%), and coma (42%; Garcia-Grimshaw et al., 2018; Lamboley et al., 2011; Lamot et al., 2015). The remaining clinical features reported in the literature include hypersomnolence (29%), akinetic mutism, and behavioral disorders (apathy, agitation, and aggressiveness; Agarwal et al., 2014; Caruso et al., 2017; Garcia-Grimshaw et al., 2018; Lamboley et al., 2011; Lamot et al., 2015; Lazzaro et al., 2010).
In the three reported cases, we found some of the clinical features reported in the previous series, including hypersomnia, coma, vertical gaze palsy, and neuropsychiatric disorders (Table 1). Altered mental status is a classic manifestation of AOP stroke and can range from hypersomnia to coma (Table 1). However, hypersomnia is rarely reported as a first complaint (Cases 1 and 3) since it is usually misdiagnosed as coma or it is lately diagnosed after recovery from coma (Case 2; Bassetti et al., 1996; Hammersley et al., 2017; Hermann et al., 2008; Oliveira et al., 2018). Hypersomnolence (i.e., excessive daytime sleepiness and/or prolonged sleep) in bithalamic stroke is explained by the crucial role of the thalamus in sleep regulation and in maintaining arousal. Hypersomnolence has been attributed to the interruption of noradrenergic and dopaminergic impulses from the ascending reticular activating system to the thalamus (Bassetti et al., 1996; Bollu et al., 2017; Goyal et al., 2012). Sleep-wake disturbances are more pronounced in bilateral than in left-sided or right-sided thalamic infarcts (Hermann et al., 2008). Sleep needs have been reported to remain increased after this stroke (Goyal et al., 2012; Hermann et al., 2008). Unfortunately, we have no follow-up information about the three patients after their discharge.
The complexity and polymorphism of AOP stroke semiology explain why bithalamic infarction is often misdiagnosed, lately detected, or even not detected. It is a real diagnostic challenge for clinicians to detect this condition in a timely fashion. Its diagnosis and treatment may be delayed because of the wide spectrum of its clinical features. Moreover, the initial CT scan can be normal (Khanni et al., 2018; Xu et al., 2017). Diagnosis of AOP stroke is often made retrospectively beyond the thrombolysis or endovascular treatment window. MRI is the neuroimaging of choice to be conducted when AOP stroke is highly suspected (Lamboley et al., 2011). MRI typically shows a hyperintense signal in the region supplied by AOP (Song et al., 2017).
In the three case series, the patients recovered completely. This is in line with previous studies which reported good prognosis of patients with AOP stroke (Arauz et al., 2014; Kichloo et al., 2019; Song et al., 2017).
In conclusion, these case series highlighted the difficulties of recognizing the AOP stroke in a timely fashion. These observations also illustrated the importance of considering this stroke syndrome as one of the differential diagnoses of hypersomnolence.
Footnotes
Acknowledgements
Authors wish to thank Professor Samir Boukattaya for his invaluable contribution to the improvement of the quality of the writing in the present article.
Authors’ Contributions
I.B.S., H.B.S., M.Z., and M.B. substantially contributed to the conception and design, acquisition of data, or analysis and interpretation of data. I.B.S., H.B.S., M.Z., E.E., R.T., and M.B. drafted the article or revised it critically for intellectual content. I.B.S., H.B.S., M.Z., E.E., R.T., and M.B. gave final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.B.S. reports personal fees from AstraZeneca, Saiph, Teriak, Hikma, and Chiesi. The remaining authors declare that they have no conflicts of interest concerning this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
