Abstract
No previous study has raised the effects of RIF on lung function data of chronic obstructive pulmonary disease (COPD) patients. The objective of the present study was to assess the effects of RIF on spirometric data measured in male patients with a stable COPD. Sixteen patients with stable COPD (mean ± SD of age: 64 ± 7 years) who fasted during Ramadan volunteered to the study. Three sessions (Before-R, End-R, and After-R) were selected for spirometry tests that were consistently performed 2.5–4.5 hr before fasting break. Assessment sessions comprised: forced vital capacity (FVC), 1st s forced expiratory volume (FEV1), FEV1/FVC, peak expiratory flow (PEF), maximal mid-expiratory flow (MMEF), and forced expiratory flow rate at the x% of FVC to be exhaled (FEFx%). A reversibility test was performed only during the Before-Ramadan session. Spirometric data were expressed in percentages of local reference values. Findings were analyzed by applying repeated measures analysis of variance. The mean ± SD of the postbronchodilator FEV1/FVC ratio and the FEV1 were, respectively, 0.52 ± 0.14 and 48 ± 19%. The mean ± SD of FEV1 (Before-R: 47 ± 19, End-R: 45 ± 18, After-R: 44 ± 19%), FVC (Before-R: 73 ± 18, End-R: 71 ± 16, After-R: 69 ± 17%), FEV1/FVC (Before-R: 67 ± 16, End-R: 66 ± 16, After-R: 65 ± 16%), PEF (Before-R: 46 ± 19, End-R: 47 ± 22, After-R: 45 ± 21%), MMEF (Before-R: 19 ± 10, End-R: 18 ± 8, After-R: 18 ± 9%), FEF25% (Before-R: 16 ± 6, End-R: 16 ± 5, After-R: 15 ± 5%), FEF50% (Before-R: 21 ± 14, End-R: 20 ± 12, After-R: 20 ± 12%) and FEF75% (Before-R: 27 ± 19, End-R: 27 ± 19, After-R: 27 ± 19%) were not significantly influenced by RIF. RIF did not bring about any significant changes in the spirometric data of stable COPD male patients fasting the 2016 holy month of Ramadan.
Ramadan intermittent fasting (RIF) consists of alternating fasting and feasting periods (Bragazzi, 2015a). It is not only the abstinence (from dawn to sunset) from all types of liquid and solid nutrient intake, but also from smoking, medication, and sexual intercourse (Bragazzi, 2015a). This restriction also includes medications given via oral and parenteral route, but not drugs used via inhalation route (Official Web site of The Presidency of Religious Affairs of the Republic of Turkey, n.d.). The fasting is broken, taking two traditional meals, pre-dawn (suhoor) and after-sunset (iftar) meals. Ramadan, the ninth month of the Muslim lunar calendar, which retreats 11 calendar days each year, is particular for the long mean of abstinence from food (Bragazzi, 2015a). The RIF duration depends on the geographical area, season and country, end of spring, beginning of summer, the tropics and temperate locations being longer (Fenneni et al., 2014; Haouari et al., 2008). The fasting mean duration is usually 12–14 hr but can last up to 18 or even 22 hr in the summer in extreme latitude regions (Fenneni et al., 2014; Haouari et al., 2008).
Fasting is not compulsory for pre-pubescent children, frail elderly, travelers, menstruating, pregnant and breastfeeding women (Athar, 1990). Patients are also exempted from fasting because of ongoing disease conditions (Athar, 1990). In practice, several patients with chronic conditions (Adeli, Aghaali, & Nasab, 2015; Al-Dubeikil & Abdul-Lateef, 2003; Askari, Alavinezhad, & Boskabady, 2016; Aydin et al., 2014; Babineaux et al., 2015; Bener, Colakoglu, Mobayed, & Sabbah, 2006; Bragazzi, 2015b; Chamsi-Pasha & Chamsi-Pasha, 2016; Erkekol et al., 2006; Herrag, Lahmiti, & Yazidi, 2010; Nematy et al., 2015; Norouzy et al., 2013; Tas et al., 2014) do not accept these exemptions and prefer fasting (and/or want to fast and/or insist on fasting) during Ramadan. For example, 67% of Muslim asthmatics do not consider asthma to be a drawback to fasting, and continue fasting by rearranging their medication times to iftar and suhoor (Erkekol et al., 2006). Almost 3% of Muslim patients with lung cancer (Tas et al., 2014) and 93% of Turkish chronic obstructive pulmonary disease (COPD) patients (Aydin et al., 2014) are Ramadan fasters. Although the relationship between religion rituals and health, and particularly the effects of RIF on chronic diseases, has been an interesting issue for physicians nearly for 30 years, this issue was only tackled in a few studies presenting controversial findings (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Askari et al., 2016; Aydin et al., 2014; Bener et al., 2006; Duncan, Husain, Raman, Cheah, & Ch’ng, 1990; Erkekol et al., 2006; Latiri et al., 2017; Moosavi, Kabir, Moghimi, Chehrei, & Rad, 2007; Nematy et al., 2015; Norouzy et al., 2013; Siddiqui, Sabir, & Subhan, 2005; Singha Roy & Bandyopadhyay, 2016; Soori, Mohaghegh, Hajain, & Moraadi, 2016; Subhan, Siddiqui, Khan, & Sabir, 2006). Moreover, the opinions of health-care professionals and scholarly societies, concerning the effects of RIF on health, are divided. Some societies, like the England National Health Service (Information for health-care professionals on fasting and medicines during Ramadan, n.d.), consider that RIF is unsafe for patients with asthma and/or COPD, since they may experience complications. Others, like the international COPD coalition (Role of the International COPD, n.d.), judge it to be safe and advise patients to rearrange their medication times to iftar and suhoor and to break the fasting during exacerbation. For COPD patients, the recent international guidelines (Celli et al., 2015; Vogelmeier et al., 2017) suggested orderly use of the symptom-relieving treatment as well as the disease-development prevention, such as the monitoring and education of patients as well as the avoidance of risk factors (Askari et al., 2016). However, the effects of religious beliefs and behaviors haven’t been addressed and no recommendation has been advanced for COPD patients who prefer fasting during Ramadan.
The spirometry test is a useful investigation tool for diagnosing and monitoring a variety of adults’ respiratory diseases (Miller et al., 2005). During the month of Ramadan, spirometric tests continue to be normally performed on patients with COPD who want to fast. The main question remains: how to interpret in these patients, any possible lung function data changes? Are they caused by the likely effects of fasting on the lung or by medication-use (in case of improvement) or clinical worsening (in case or deterioration)? Several studies have observed the effects of RIF on body functions in patients with chronic conditions (Adeli et al., 2015; Al-Dubeikil & Abdul-Lateef, 2003; Askari et al., 2016; Aydin et al., 2014; Babineaux et al., 2015; Bener et al., 2006; Bragazzi, 2015b; M. Chamsi-Pasha & H. Chamsi-Pasha, 2016; Erkekol et al., 2006; Herrag et al., 2010; Nematy et al., 2015; Norouzy et al., 2013; Tas et al., 2014). However, studies examining the RIF effects on lung function data, are scarce, included only healthy adults or asthmatic adult patients and presented some discrepancies in their findings (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Norouzy et al., 2013; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Subhan et al., 2006; Soori et al., 2016). In view of the above considerations, the aim of the present pilot study was to assess the effects of RIF on spirometric data measured in male patients with a stable COPD. As seen in healthy Tunisian adults (Latiri et al., 2017), the null hypothesis was that RIF did not bring about any significant changes in the spirometric values of patients with a stable COPD.
Population and Methods
Study Design
The present study was designed as a pilot cross-sectional and experimental study. It was performed at the Farhat HACHED University Hospital, Sousse (Tunisia). Approval for the study was obtained from the Ethical Committee of the local Hospital (approval number 2602/2015) and oral consent was obtained from all patients. Participants were informed they were free to leave the study at any time. Patients were not charged any fees for the spirometry test.
The study was performed during summer of 2016. During the study, the elapsed time from dawn to sunset was ~16.5 hr during the whole month of Ramadan (June 6 to July 5). The ambient temperature and humidity means ± standard deviation (SD) during the 50-day study period (June 1 to July 20, 2016) were, respectively, 26.5 ± 2.4 °C and 62.0 ± 7.5%.
Sample Size
The sample size was estimated according to a predictive formula (Kang, Ragan, & Park, 2008) largely detailed in the Appendix. Fifteen patients were needed.
Study Population
Participants were male COPD patients aged 40–80 years. They were recruited in two ways. First, the folders and files of COPD patients followed in the above two departments were verified. Second, some patients were directly addressed by two pulmonologists from the Basic Health Group of Sousse. At the beginning of the study, a letter of information containing details about the study and visit dates was given to the patients. Noninclusion criteria were history of RIF for less than 20 years; narghile-use; actual cigarette smoking; smoking history less than 10 pack-years; tobacco cessation less than 6 months; non-COPD patient; neuromuscular disease; diabetes-mellitus; congestive heart failure; malignancy; vertebral column or thoracic cage abnormalities; oral corticosteroid treatment; and lack of cooperation during spirometry test. Female patients were not included because Muslim laws forbid them to fast during their menses and because lung function is fairly lowered during menses (Cotes, Chinn, & Reed, 1997). Absence during a testing session was applied as an exclusion criterion. To avoid confounding effects, patients with unstable respiratory state (e.g., respiratory tract infection or COPD exacerbation) within 4 weeks prior to the spirometry test were excluded. Patients were instructed to avoid using short-acting bronchodilators 6 to 8 hr prior to the spirometry test. Sampling was made based on a convenience method.
Experimental Design
The experimental design consisted in three sessions: 5 days before Ramadan (Before-R: June 1–4), 3 days at the end of Ramadan (End-R: June 28–30), and 14 days after Ramadan (After-R: July 19–20). During the Before-R session, all patients had to answer a medical questionnaire. Anthropometric data were measured and/or noted and the spirometry test was performed. During the second and the third sessions, only the anthropometric and the spirometric data were determined.
Collected Data and Applied Definitions
The patients were asked whether they fasted during Ramadan. If the answer was “yes,” they were kindly requested to answer further questions related to their socioeconomic and schooling levels, personal medical or surgical histories, chronic medication-use, dyspnea, and smoking. The questions were gathered from the American thoracic society medical questionnaire (Ferris, 1978). It was composed of questions asked in local Arabic dialect by the same-trained operator. This non-validated questionnaire was used to assess several subject characteristics. The patients’ history of COPD exacerbation including hospitalizations was recorded. During the three sessions, patients were systematically asked about the times of their bronchodilator-use, latest meal, and sleep duration. Meal-duration [time (hr) between the last meal and the spirometry test] and sleep-duration [time (hr) of sleeping, including night sleep and naps, before the spirometry test] were noted. Cigarette-smoking (pack-years) and narghile-use were evaluated. Dyspnoea was evaluated according to the modified medical research council (mMRC) scale (Fletcher, Elmes, Fairbairn, & Wood, 1959). Two dyspnoea levels were defined (mMRC < 2 and mMRC ≥ 2). COPD exacerbation was defined as an acute worsening of respiratory symptoms that result in an additional therapy (Vogelmeier et al., 2017). Two schooling [low (illiterate, primary education) and high (secondary and university education)] and two socioeconomic [low (e.g., unskilled worker, jobless) and high (e.g., skilled worker, farmer, manager)] levels were arbitrarily defined. Decimal age (accuracy to 0.1 years) was taken as the number of complete years from birth to the date of the study. Height (±1 cm) and weight (±1 kg) were measured with a mechanical scale (Seca deutschland; Medical Measuring Systems and Scales, seca gmbh & co. kg Hammer Steindamm 3-25 22089 Hamburg, Germany). Body mass index (BMI, kg/m2) was calculated and the following definitions were adopted (Tsai & Wadden, 2013): underweight (BMI < 18.5), normal weight (18.5 to 24.9), overweight (25.0 to 29.9), and obesity (BMI ≥ 30.0).
Spirometry Measurements, COPD Diagnosis and Classification
All spirometric tests were performed at the same time of the day (between 15h00 and 17h00), approximately 279 to 159 min before iftar (mean fasting hours = 16:59 hr). Spirometry was carried out in a standing position. All tests were performed by one qualified person (IL in the authors list) and one portable spirometer (SpirobankG MIR via del Maggiolino 12500155 Roma, Italy) was used. The turbine of the spirometer was calibrated daily with a 3-L syringe. Spirometry was performed according to international guidelines (Miller et al., 2005). The spirometric data [forced vital capacity (FVC, L); 1st s forced expiratory volume (FEV1, L); forced expiratory flow when x% of FVC has been exhaled (FEFx%; L/s), maximal mid-expiratory flow (MMEF, L/s), peak expiratory flow (PEF, L/s), and FEV1/FVC ratio (absolute value)] were expressed in absolute values at “body temperature, barometric pressure saturated with water vapor” (Miller et al., 2005). The measured spirometric data were also expressed as percentages of predicted local reference values (Ben Saad et al., 2013). The FVC maneuver was previously described (Latiri et al., 2017). The reversibility test, largely described elsewhere (Ben Saad, Prefaut, Tabka, Zbidi, & Hayot, 2008), was performed only during the Before-R session.
COPD diagnosis was retained in front of a postbronchodilator FEV1/FVC ratio <0.70 (Celli et al., 2015; Vogelmeier et al., 2017). The severity of airflow limitation, based on the postbronchodilator FEV1, was classified as follows: mild (FEV1 ≥80%); moderate (50% ≤ FEV1 < 80%); severe (30% ≤ FEV1 < 50%), and very severe (FEV1 <30%) (Celli et al., 2015; Vogelmeier et al., 2017). The refined “ABCD” assessment tool derived from the patients’ symptoms [i.e., dyspnea (Fletcher et al., 1959)] and their exacerbation history (including prior hospitalizations) was applied (Vogelmeier et al., 2017), and four groups (A, B, C, and D) were categorized.
Statistical Analysis
The analysis of variable distribution was performed using the Kolmogorov–Smirnov test. When the distribution was normal and variances were equal, the results were expressed as mean ± SD (95% confidence interval). Otherwise, the results were expressed by their medians (lower-upper quartiles). Qualitative data were expressed by relative frequency. Comparisons of the anthropometric and the spirometric data, sleep-, and meal-durations and time of last-use of bronchodilator were made between the three sessions (Before-R, End-R, and After-R). The results were obtained by applying repeated measures analysis of variance. When appropriate, significant differences between means were tested using the Tukey post hoc test. Analyses were carried out using Statistica software (Statistica Kernel version 6; StatSoft, Paris, France). Significance was set at the 0.05 level.
Results
Among the 25 examined patients, only 16 stable COPD patients completed the three sessions. The reasons for dropout were postbronchodilator FEV1/FVC > 0.70 (n = 1) and absence during the second (n = 4) or the third (n = 4) sessions.
Eight and ten patients had, respectively, low socioeconomic and low schooling levels. The median (lower-upper quartiles) of the patients Ramadan experience was 42 years (40–46). The mean ± SD (95% confidence interval) of their tobacco history was 36 ± 27 (22–50) pack-years. The median (lower-upper quartiles) of their smoking cessation duration was 5.0 years (3.0–15.5). Only one COPD patient wasn’t under respiratory treatment. Twelve, fourteen, and six patients were treated by bronchodilators, inhaled corticoid and anti-cholinergic, respectively. Seven COPD patients were mMRC ≥ 2; four had cardiovascular diseases (two have a stable hypertension, one had an obliterating arteriopathy of the lower limbs and one had a dilated cardiomyopathy), two were anemic, one had hypothyroid, and one had dyslipidemia.
The airflow limitation was qualified as “mild to moderate” and “severe to very severe,” respectively, in seven and nine patients. The mean ± SD of the postbronchodilator FEV1/FVC, FEV1 and FVC were, respectively, 0.52 ± 0.14%, 48 ± 19%, and 77±16%. The refined ABCD assessment tool identified that eight, six and two patients belonged, respectively, to groups A, B, and D.
The mean ± SD (95% confidence interval) of age and height were, respectively, 63.63 ± 7.19 (59.80 to 67.46) years and 172±5 (169 to 174) cm. Twelve, three, and one patients were, respectively, normal weight, overweight, and obese.
Ambient temperatures and humidity data during the three sessions were presented in Table 1. Ambient temperature was significantly higher during the End-R session compared to the Before-R one. Humidity was significantly lower during the After-R session compared to the Before-R one.
Ambient Temperature, Humidity and some Characteristics of the 16 Stable COPD Patients.
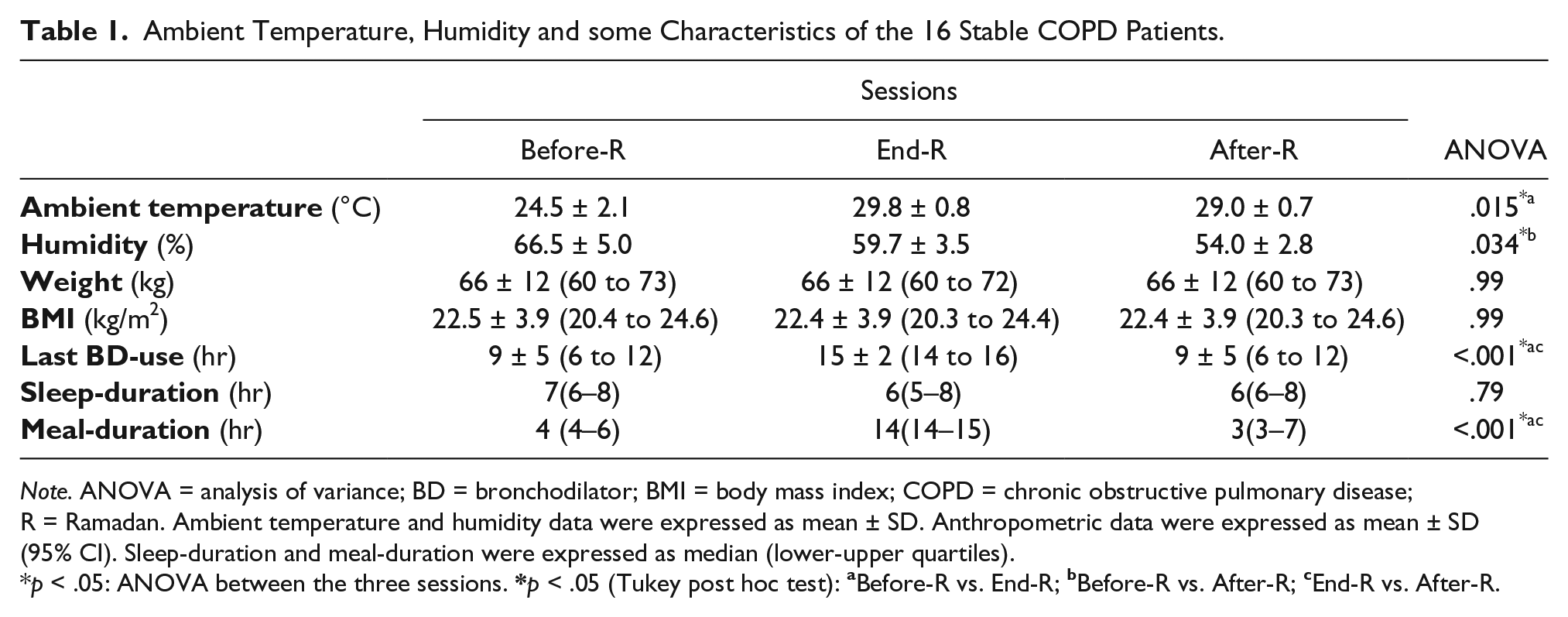
Note. ANOVA = analysis of variance; BD = bronchodilator; BMI = body mass index; COPD = chronic obstructive pulmonary disease; R = Ramadan. Ambient temperature and humidity data were expressed as mean ± SD. Anthropometric data were expressed as mean ± SD (95% CI). Sleep-duration and meal-duration were expressed as median (lower-upper quartiles).
p < .05: ANOVA between the three sessions.
Table 1 reports the patients’ characteristics during the three sessions. Its main conclusions were (a) there was no significant effect of RIF on weight or BMI, (b) there was no significant difference between the three-session sleep-duration, and (c) there was a significant difference between the three-session meal-duration: the End-R meal-duration was significantly higher than those of Before and After-R sessions.
Table 2 reports the COPD patient’s spirometric data expressed in absolute values. RIF had no effect on their spirometric data. Figures 1 and 2 display the patients’ spirometric volumes, ratio and flows expressed as percentages of predicted values. The mean ± SD of FEV1 (Before-R: 47 ± 19, End-R: 45 ± 18, After-R: 44 ± 19%), FVC (Before-R: 73 ± 18, End-R: 71 ± 16, After-R: 69 ± 17%), FEV1/FVC (Before-R: 67 ± 16, End-R: 66 ± 16, After-R: 65 ± 16%), PEF (Before-R: 46 ± 19, End-R: 47 ± 22, After-R: 45 ± 21%), MMEF (Before-R: 19 ± 10, End-R: 18 ± 8, After-R: 18 ± 9%), FEF25% (Before-R: 16 ± 6, End-R: 16 ± 5, After-R: 15 ± 5%), FEF50% (Before-R: 21 ± 14, End-R: 20 ± 12, After-R: 20 ± 12%), and FEF75% (Before-R: 27 ± 19, End-R: 27 ± 19, After-R: 27 ± 19%) were not significantly influenced by RIF.
Spirometric Data of the 16 Stable COPD Patients (n = 16).

Note. ANOVA = analysis of variance between the three sessions; COPD = chronic obstructive pulmonary disease; FEFx%

The effects of Ramadan intermitting fasting on the spirometric volumes and ratio (expressed as percentages of predicted values) of the 16 stable chronic obstructive pulmonary disease patients.

The effect of Ramadan intermitting fasting on the spirometric flows (expressed as percentages of predicted values) of the 16 stable chronic obstructive pulmonary disease patients.
Discussion
The main finding of the present pilot study which addresses the effects of a religious ritual on a group of 16 stable COPD patients identified that RIF did not bring about any significant changes in their spirometric data. The effect of RIF on the respiratory system is neither a simple academic issue nor is it of awareness only just in Muslim countries (Bragazzi, 2015a). It has clinically pertinent inferences. In fact, in a globalized culture, doctors may consult Muslim COPD patients who choose fasting during Ramadan (Al Wakeel et al., 2013). Available proof concerning the health benefits of RIF is limited and greatly controversial (Nematy et al., 2015). Although Islam exempts some patients with chronic diseases from fasting, a lot of them obviously fast, and this can cause their clinical condition to deteriorate due to a persistent gap between current expert knowledge and decisive strong evidence regarding the pathophysiologic and metabolic alterations of fasting (Nematy et al., 2015). To the best of the authors’ knowledge, no previous study has debated the effects of RIF on lung function data of COPD patients. Some studies have examined the impacts of RIF on respiratory mechanics of both healthy and asthmatic adults (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Norouzy et al., 2013; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Soori et al., 2016; Subhan et al., 2006).
Discussion concerning the season and the duration of RIF, the timing of spirometric tests, the number of realized sessions and spirometric measurements is available in the Appendix. Only the sample size, patients’ characteristics and recruitment methods will be discussed in the following sentences.
The calculated sample size (n = 16) is intermediate between those of relative studies including asthmatic patients (Adeli et al., 2015; Askari et al., 2016; Norouzy et al., 2013) [size ranging from 15 (Askari et al., 2016) to 30 (Adeli et al., 2015)] or healthy patients (Abdel-aziz & Ibraheem, 2008; Askari et al., 2016; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Soori et al., 2016; Subhan et al., 2006) [size ranging from 12 (Soori et al., 2016) to 117 (Moosavi et al., 2007)]. However, due to its pilot nature, the present study findings are preliminary, and should be useful in directing researchers seeking to elucidate the effects of RIF on spirometric data of COPD patients.
A minimal 20-year RIF experience was applied as an inclusion criterion. It is possible, as seen in studies assessing the effects of RIF on healthy patients’ exercise performance (Chtourou et al., 2012), that the RIF record of the patients could influence their respiratory adaptations (Ben Saad, 2016; Fenneni et al., 2015). This point was not considered in several related studies including healthy or asthmatic patients (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Askari et al., 2016; Moosavi et al., 2007; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Soori et al., 2016; Subhan et al., 2006; Norouzy et al., 2013). All included COPD patients were ex-smokers. This is a serious point to highlight (Ben Saad, 2016), because smokers compared with ex-smokers, are more irritable during Ramadan (Kadri et al., 2000) and psychological stress has proved to influence pulmonary function (Kang & Fox, 2000). This information was neglected in some relative studies (Adeli et al., 2015; Duncan et al., 1990; Norouzy et al., 2013; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Subhan et al., 2006). The oral medications, injections, ear and nose drops, and suppositories are not allowed for patients who preferred fasting. Use of eye drops and inhalers is accepted as a process that does not nullify the fasting (Official Web site of The Presidency of Religious Affairs of the Republic of Turkey, n.d.). Despite this information, a considerable proportion of COPD patients adjust their medication regimens during RIF without asking their physician (Fazel, 1998). For example, in a Turkish study, the majority of the Muslim COPD patients quit using their medications during Ramadan (Aydin et al., 2014). In the present study, patients were asked about either to keep using their habitual medication or to adjust it in suhoor and iftar times. Only short-acting bronchodilators were stopped 6 to 8 hr before the spirometry test. It seems that all inhaled medications used at iftar and suhoor times are not expected to decrease the efficacy of the drugs and this method “seems” to be suitable for patients who do not want to use their drugs throughout fasting hours (Aydin et al., 2014).
The present study presents four limitations. First, as done in some relative studies (Adeli et al., 2015; Askari et al., 2016; Latiri et al., 2017; Moosavi et al., 2007; Siddiqui et al., 2005; Subhan et al., 2006), the convenience sampling was a confounding factor. For example, all patients were aware of the study aims, which might increase concern about spiritual prejudice and then might stimulate fasters to make additional efforts in lung function testing. Convenience sampling can undermine ability to make generalizations from the present sample to the population being studied. However, its low cost and ease of use make it the preferred choice for a significant number of researchers (Abdel-aziz & Ibraheem, 2008; Askari et al., 2016; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Soori et al., 2016; Subhan et al., 2006). Second, the noninclusion of a control-group of non-fasting COPD patients could be considered as a serious limitation because the internal power of the results from these studies and the changes in the variables assessed cannot be attributed exclusively to RIF (Fenneni et al., 2015; Latiri et al., 2017). In this regard, it has to be underlined that recruiting non-fasting groups in Muslim countries is not effortless, due to religious attitudes (Fenneni et al., 2014, 2015). Indeed, the study of Aydin et al. (2014) reported that patients with asthma or COPD do not feel their diseases to be an inhibitory factor to fast Ramadan. Nevertheless, among the 12 studies (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Norouzy et al., 2013; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Soori et al., 2016; Subhan et al., 2006) demonstrating the “Hawthorne” effects of RIF on the lung function data of healthy and/or asthmatic adults, only two performed in India and in Iran (Askari et al., 2016; Singha Roy and Bandyopadhyay, 2016) included control-groups. The remaining 10 studies conducted in Muslim countries (Iran, Saudi Arabia, Tunisia, Malaysia, Egypt, and Qatar) (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Bener et al., 2006; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Norouzy et al., 2013; Siddiqui et al., 2005; Soori et al., 2016; Subhan et al., 2006) used Before-Ramadan values as control. Third, it was preferable to evaluate the hydration status and the diet regime (especially the medicinal herb/supplement use) of COPD patients for at least two reasons: (a) hypohydration causes a considerable raise in respiratory flow rates because of loss of water from the bronchovascular sheath and airway mucosa potentially reduce the airway resistance (Subhan et al., 2006); and (b) dietary abnormalities unfavorably influence the lung structure and function, the neural control of breathing and the functioning of the respiratory muscles (Riley & Thakker-Varia, 1995). Fourth, it was preferable to assess the impact of RIF on some respiratory symptoms [using e.g., the COPD assessment tool (Vogelmeier et al., 2017)] or the COPD patients physical activity status’ [since it influences the respiratory function (Cheng et al., 2003)] or the static volumes (very useful in the COPD management).
Since RIF did not influence the weight of stable COPD patients, it seems unlikely that in the present study patients were dehydrated. In the studies evaluating the effects of RIF on the lung function data of healthy patients (Abdel-aziz & Ibraheem, 2008; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Soori et al., 2016; Subhan et al., 2006), confusing results, detailed in the Appendix, were noted for weight. Among the relative studies including asthmatic patients (Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Norouzy et al., 2013), only one (Norouzy et al., 2013) looked into weight changes: it was lower at End-R compared to that obtained Before-R. A recent systematic review concluded that RIF could result in a significant weight loss of 1.24 kg (Sadeghirad, Motaghipisheh, Kolahdooz, Zahedi, & Haghdoost, 2014). The heterogeneous findings regarding the effects of RIF on weight may be due to the varying of the patients’ physical activity levels (Nematy et al., 2015).
Relative to Before-R data, there was no significant effect of RIF on spirometry data of stable COPD patients. While a few human studies have been conducted on the effects of RIF on the lung function data of healthy or asthmatic adults (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Norouzy et al., 2013; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Subhan et al., 2006; Soori et al., 2016), or on drug use behavior (Aydin et al., 2014; Wheatly & Shelly, 1993), no previous study has evaluated the effects of RIF on lung function data of COPD patients. Collectively, little consensus exists regarding the effects of RIF on the majority of chronic respiratory diseases (Kazemi et al., 2013). Therefore, making comparisons between literature is not relevant, due to several underlying confounding variables (Kazemi et al., 2013). The following paragraphs will discuss the effect of RIF on lung function data of healthy and asthmatic adults (Abdel-aziz & Ibraheem, 2008; Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Norouzy et al., 2013; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Subhan et al., 2006; Soori et al., 2016) and on drug-use by COPD patients (Aydin et al., 2014; Wheatly & Shelly, 1993).
Results concerning the effects of RIF on lung function data of healthy adults, described elsewhere (Latiri et al., 2017) and described more in the Appendix, are contradictory. Briefly, while some studies reported that RIF doesn’t alter spirometric data (Abdel-aziz & Ibraheem, 2008; Askari et al., 2016; Duncan et al., 1990; Latiri et al., 2017; Moosavi et al., 2007; Siddiqui et al., 2005; Singha Roy & Bandyopadhyay, 2016; Subhan et al., 2006; Soori et al., 2016), others reported that it impacted on some of them (Duncan et al., 1990; Siddiqui et al., 2005; Subhan et al., 2006; Moosavi et al., 2007). Most of the studies performed on asthmatic patients (Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Norouzy et al., 2013) concluded that they can tolerate the RIF without significant alteration of their spirometric flows [PEF (Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Norouzy et al., 2013), MMEF (Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Norouzy et al., 2013), FEF75% (Askari et al., 2016), FEF50% (Askari et al., 2016), FEF25% (Askari et al., 2016)] or volumes [FEV1 (Adeli et al., 2015; Askari et al., 2016; Bener et al., 2006; Norouzy et al., 2013), FVC (Askari et al., 2016; Bener et al., 2006; Norouzy et al., 2013)] or ratios [FEV1/FVC (Adeli et al., 2015; Bener et al., 2006; Norouzy et al., 2013)]. However, one study (Norouzy et al., 2013) identified a significant increase in PEF by an average of 17% during the End-R session compared to the Before-R one. In addition, it seems that mean PEF variability decreased from 13 to 10%, respectively in the first and the fourth weeks of Ramadan (Norouzy et al., 2013). Conclusions regarding the effects of RIF on clinical symptoms of asthmatic patients are also contradictory (Askari et al., 2016; Bener et al., 2006; Herrag et al., 2010; Norouzy et al., 2013). While some studies reported that RIF doesn’t affect either their clinical symptoms (e.g., dyspnoea, cough, wheezing, and chest tightness) (Askari et al., 2016; Norouzy et al., 2013) or their frequency of hospitalization (Bener et al., 2006), others identified a reduction in “objective wheezing” after RIF (Askari et al., 2016) or noted an increase in the rate of emergency consultations for severe exacerbations (Herrag et al., 2010). Moreover, one study (Aydin et al., 2014) concluded that fasting seems to be an important determining factor in medication compliance by modifying the drug-use behaviors in COPD and asthmatic patients. In contrast, a case-series study conducted on two Muslims with chronic reversible respiratory disease, reported that the discontinuation of bronchodilators in Ramadan resulted in an acute exacerbation of their disease and their admission in the intensive care unit (Wheatly & Shelly, 1993). According to these authors (Wheatly & Shelly, 1993), Muslims with COPD should be aware that they put their health at risk when they abstain from taking their drugs for a long period of time.
How Can Changes in Lung Function Data During RIF Be Explained?
In the present study, it may be hypothesized that, the hydration status of the COPD patients was not disturbed and insignificant changes in the weight may have had some physiological impact on preserving the spirometric data during the RIF. In healthy or asthmatic patients, some hypotheses and/or mechanisms, largely described in the Appendix, were advanced to explain the stability of spirometric data during RIF.
Recommendations and Perspectives
Despite the pilot nature of this study, some recommendations for physicians and patients, largely described in the Appendix, may be advanced. They mainly concern the few tips to give to COPD patients, who insist on fasting during Ramadan. Moreover, the present paper is a call for more trials with relevant data from larger control-groups and more regular spirometric measurements throughout Ramadan.
To conclude, the main finding of the present study reported that RIF did not bring about any significant change in the spirometric data of stable COPD male patients fasting the 2016 holy month of Ramadan. This result emphasizes the need for practitioners to take into account the patients’ religious rituals and beliefs when providing medical care.
Footnotes
Acknowledgements
Authors wish to express their sincere gratitude to all participants for their cooperation and for the following two Pulmonologists from the Outpatient Pulmonology. Basic Health Group of Sousse, Tunisia: Raoudha SFAXI and Radhia ZAYANI. They also wish to thank Professor Béchir SAADAOUI for his invaluable contribution in the improvement of the quality of the writing in the present paper.
List of Abbreviations
BMI: body mass index
COPD: chronic obstructive pulmonary disease
FEFx%: forced expiratory flow when X% of FVC has been exhaled
FEV1 : 1st s forced expiratory volume
FVC: forced vital capacity
MMEF: maximal mid-expiratory flow
mMRC: modified medical research council
PEF: peak expiratory flow
R: Ramadan
RIF: Ramadan intermittent fasting
SD: standard deviation
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HBS reports personal fees from AstraZeneca, Boehringer Ingelheim, GSK and Chiesi. The remaining authors declare that they have no conflicts of interest concerning this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
