Abstract
Men continue to bear disproportionate accounts of HIV diagnoses. The Patient Protection and Affordable Care act aims to address health care disparities by recommending preventative services, including HIV screening, expanding community health centers, and increasing the healthcare workforce. This study examined the decision making of physician and primary care health providers to provide HIV screenings. A quasi-experimental design was used to estimate the effects of the Affordable Care Act on provider-initiated HIV screening. The National Ambulatory Medical Care Survey was used to examine HIV screening characteristic from two time periods: 2009 and 2012. Logistic regression indicated that patient and provider characteristics were associated with likelihood of being prescribed HIV screening. Non-Hispanic Black men were more likely to be prescribed HIV screening compared to non-Hispanic White men (odds ratio [OR] = 12.33, 95% confidence interval [CI; 4.42, 34.46]). Men who see primary care providers were more likely to be prescribed HIV screening compared to men not seeing a primary care provider (OR = 5.94, 95% CI [2.15, 16.39]). Men between the ages of 19 and 22 were more likely to be prescribed HIV screening compared to men between the ages of 15 and 18 (OR = 6.59, 95% CI [2.16, 20.14]). Men between the ages of 23 and 25 were more likely to be prescribed HIV screening compared with men between the ages of 15 and 18 (OR = 10.13, 95% CI [3.34, 30.69]). Health education programs identifying men at increased risk for contracting HIV may account for the increased screening rates in certain populations. Future research should examine age disparities surrounding adolescent and young men HIV screening.
Provider-initiated HIV screening continues to be a barrier in the United States, as HIV screening practices among care providers fail to meet federal guidelines and current standards of care. The lack of provider-initiated screening is of significant concern as the Centers for Disease Control and Prevention (CDC; 2015b) reports nearly 20 million newly diagnosed sexually transmitted infections each year. Sexually active adolescents, ages 15 to 19, and young adults, ages 20 to 24, are at an increased risk of both contracting HIV and being unaware of their serostatus. Adolescents and young adults make up only 17% of the population, yet they account for approximately 26% of all new HIV infections in the United States, with more than half of all HIV-infected adolescents believed to have never been tested, and thus unaware of their serostatus (Branson et al., 2006).
Newly diagnosed cases of HIV have recently experienced resurgence. The rise in new HIV diagnosis is particularly alarming among adolescents and young adult men. A recent study reported that between the years 2001 and 2006 there was a 93.1% increase in HIV/AIDS diagnoses among Black men who have sex with other men ages 13 to 24 (Phillips, Wohl, Xavier, Jones, & Hidalgo, 2011). Data from 2005 to 2014 revealed that young adult men who have sex with other men experience the highest percentage increase in newly diagnosed HIV infections of all age-groups (CDC, 2016a). Approximately 26% of newly diagnosed HIV infections occur in young adults and adolescents ages 13 to 24, and four out of five of those diagnosed are males (CDC, 2015b). In 2010, men accounted for 76% of all adults and adolescents living with HIV infection in the United States, and represented 74% of AIDS-related deaths.
Being unaware of individual or partner serostatus continues to be a significant sexual health concern. At the end of 2012, being unaware of individual HIV serostatus was still a significant public health concern with 44% of youth ages 18 to 24 years living with HIV unaware of their positive serostatus (CDC, 2016b). The increased risk of contracting HIV coupled with a high percentage of individuals who are unaware of their positive serostatus place frontline care providers in a unique position to positively affect proactive HIV screening. Studies examining provider behaviors associated with HIV screening has traditionally been limited to urban areas, leading to a dearth of knowledge regarding HIV testing in rural areas (Meyerson, Navale, Gillespie, & Ohmit, 2015). Understanding the characteristics associated with provider-based HIV screening is the first step in changing clinical-based HIV screening. A multilevel shift is needed to alter how the United States views and addresses sexual health. To decrease the growing disparities in sexually transmitted infections, including HIV, a broad sexual health framework is needed to engage patients in more integrated clinical interventions such as STI/HIV discussions and screenings (Satcher, 2001; Satcher, Hook, & Coleman, 2015).
The Patient Protection and Affordable Care Act (ACA) is a federal statute signed into law on March 23, 2010, with most major provisions phased in by January 2014. The ACA aims to improve prevention services, as seen with the provision that most insurance plans must cover HIV screening for all individuals aged 15 to 65 (AIDS.gov, 2015). Extending preventative services to individuals as young as 15, and to populations who are at an increased risk for being infected with HIV, is a significant step in addressing the existing HIV disparity among adolescents and adult men.
Expansion of community health centers (CHCs) is another provision of the ACA to address health disparities in the United States. Because CHCs are important sites for patients to receive STI and HIV screening, the expansion of CHC networks creates a larger capacity and opportunity to provide comprehensive HIV care. Along with expanding CHC capacity, the ACA expands the initiatives to strengthen culturally competent care for all health care providers. Understanding the intersection of identities and health outcomes, such as race, ethnicity, or sexual orientation, is vital to address the continuing HIV epidemic. As of April 2016, only one study has used the National Ambulatory Medical Care Survey (NAMCS) to examine HIV screening patterns among providers, and to our knowledge, no studies have used NAMCS data to examine provider-initiated HIV screening patterns focusing on men (Tai & Merchant, 2014).
Research Design
A quasi-experimental design was chosen with a pretest–posttest no-treatment control group used to examine the early impact of the ACA on primary care provider–initiated HIV screening. Data from the NAMCS were used to examine provider-based HIV screening characteristic of men ages 15 to 25 from the time periods 2009 to 2012. The pretest–posttest no-treatment control group design was used to examine if an intervention produces any effects above and beyond those attributable to the passage of time, concurrent history, or the experience of being assessed (Thyer, 2012, p. 95).
In this study, two levels of provider characteristics were examined as they relate to HIV screening among men ages 15 to 25 before and after implementation of the ACA. The first criterion is primary care role, with the goal being to examine the potential variance between physician- and nurse-initiated HIV screening. To capture data pertaining to providers, other than physicians, the category “Primary Care Provider” was created. This category consisted of nurse practitioners, nurse midwives, registered nurses, licensed practical nurses, and mental health providers. The second criterion is region of the United States.
Conceptual Framework
This study used the Eisenberg model of physician decision making as the theoretical framework to guide variable selection. The Eisenberg model describes clinical decision making in the context of sociological influences (Lin, Erickson, & Balkrishnan, 2011). According to this model, physician decision making is influenced by four interrelated social constructs:
Physician characteristics
Patient characteristics
Physician as a part of the health care system
Physician relationship with the patient
These categories served as the guide to identify variables related to provider-based HIV screening. Because health care providers who are not physicians take an increasingly proactive and independent role in screening patients for HIV, the Eisenberg model was expanded to include mental health providers, Registered nurses, nurse practitioners, and licensed practical nurses as clinical decision makers.
Research Hypotheses
According to the CDC, men continue to bear a disproportionate burden of new HIV infections. Most infections occur within the 25 to 34 age range; however, Black men ages 13 to 24 accounts for 38% of all new HIV infections (CDC, 2016c). Due to the disproportionate number of HIV cases in the youngest age-group, the NCAMS was used to examine clinical decision-making surrounding this age category.
Data Collection
NAMCS 2009-2012 data were used to examine provider HIV screening patterns among men ages 15 to 25. The NAMCS is a national probability survey conducted in conjunction with the Division of Health Care Statistics and the CDC. The primary sampling structure is the physician/patient office visit. Results are based on a sample of visits to non–federal employed office-based physicians who are primarily engaged in direct patient care (CDC, 2015a). NAMCS is a weighted multilevel national survey applicable to all U.S. outpatient visits. The survey exhibits a high invite to complete ratio with no honoraria, suggesting that NAMCS is a credible sample with minimal selection bias.
Measures
The dependent variable, HIV Screening, used information gathered from the patient “Visit Characteristics.” This variable was defined as “HIV Test.”
A modified version of Eisenberg’s model of provider decision making was used to direct variable selection. The conceptual framework depicting application of the Eisenberg model can be found in Figure 1. The first construct of the Eisenberg model is Provider Characteristics. This includes Physician or Primary Care Provider status and practice region. For this study, the definition of “physician” was expanded to include the following NAMCS provider variables: physician, mental health provider, nurse practitioner, and registered nurse/licensed practical nurse. Practice region variables include Northeast, Midwest, South, West, and metropolitan statistical area.

Eisenberg model of provider decision making.
Patient Characteristics is the second construct of the Eisenberg model and includes sex, race, age, education level, income, and payment type. Payment type included Medicaid, private insurance, and self-pay. The NAMCS data regarding “sex” are binary male/female; no further coding was required. Race variables include White, non-Hispanic Black/Black, Asian/Pacific Islander, American Indian/Alaskan Native, and Hispanic or Latino. For purposes of this study, the race/ethnicity variables non-Hispanic White, non-Hispanic Black, Hispanic, and other were created.
Physician’s Relationship With the Health Care System is the third construct of the Eisenberg model and includes information related to practice setting. This study used data collected from the following settings: general and family practice, internal medicine, pediatrics, obstetrics and gynecology, dermatology, and psychiatry.
Physician’s Relationship With the Patient is the fourth construct of the Eisenberg model and focuses on the patients presenting symptoms and/or demands. The NAMCS category “new problem” was used to include patients who may present with symptoms leading to an HIV screening. The CDC, in 2006, provided guidelines for providers to adopt an “opt out” approach instead of an “opt in” directive to HIV screening, meaning HIV screening would be viewed as a standard of care instead of a service being requested. This meant that patients would have to decline HIV screenings if they did not want to participate, and opened the door for other health care providers and services such as pre-/postsurgery to expand basic screening protocols.
Descriptive Statistics
Chi-square tests were used to tests for a significant relationship between the independent and dependent variables.
Regression Models
The statistical model used a logistic regression to examine the factors that influenced health care provider–initiated HIV screening between the years 2009 and 2012. Associations between the dependent variable (HIV screening) and the independent variables were examined as follows: Provider Characteristics (specialty and practice region), Patient Characteristics (sex, race, age, insurance status), Relationship With the Health Care System (practice setting), and Relationship With Patient (reason for visit, new problem). Odds ratios (ORs) were computed to determine the likelihood of receiving HIV screening based on the reason for visit. StataSE Version 14 was used to perform all statistical analysis.
Statistical Results
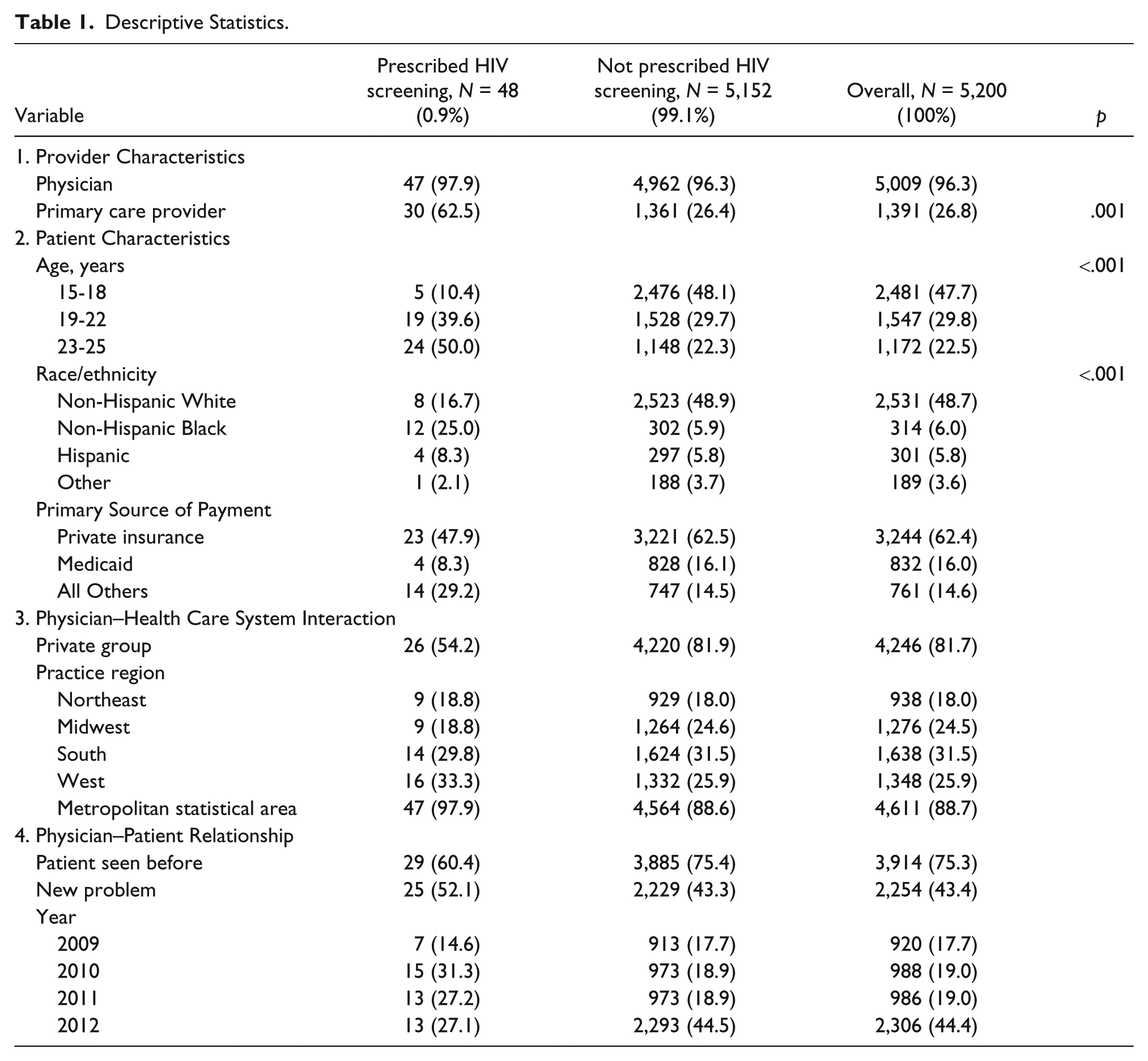
Descriptive statistics of men who were prescribed HIV screening are reported in Table 1. From 2009 to 2012, men without a diagnosed HIV infection and between the ages of 15 and 25 represented 5,200 ambulatory care visits. Among the target study population, 99.1% (n = 5,152) were not prescribed an HIV screening. The majority of the study sample consisted of non-Hispanic White men (75.9%/n = 2,531), with non-Hispanic Black, Hispanic, and men selecting other race making up 9.4% (n = 314), 9% (n = 301), and 5.7% (n = 189), respectively. Almost half of the subject sample (47.7%/n = 2,481) was between the ages of 15 and 18. Men between the ages of 19 and 22 characterized 29.8% (n = 1,547) of the study sample. More than half of the sample (66%/n = 3,244) was covered by private insurance. The majority of subjects were seen in the Southern (31.5%/n = 1,638) region of the United States. Approximately two fifths of visits were reported in year 2012 (44.4%/n = 2306), with 986 (19%) visits in 2011, 988 (19%) in 2010, and 920 (17.7%) reported in 2009.
Descriptive Statistics.
The ORs and 95% confidence intervals (CIs) are reported in Table 2. First, factors that may influence receipt of HIV screening among the sample population were examined. Men who see a primary care provider are 5.9 times more likely to be prescribed HIV screening compared to those men who do not see a primary care provider. Men ages 19 to 22 are 6.6 times more likely to be prescribed HIV screening compared to men between the ages of 15 and 18, and men ages 23 to 25 are 10.1 times more likely to be prescribed HIV screening compared to men between the ages of 15 and 18. Non-Hispanic Black men are 12.3 times more likely to be prescribed HIV screening compared to non-Hispanic White men. Hispanic men are 3.3 times more likely to be prescribed HIV screening compared to non-Hispanic White men. Examination of HIV screening by year did not yield significant results.
Logistic Regression.

Represents significant findings.
Discussion
The first hypothesis of this study, provider-initiated HIV screening will increase among men ages 15 to 25 after implementation of the ACA, did not yield significant results but did demonstrate a positive trend, increasing 12.5% from 2009 to 2012. However, an increase in number of visits was noticed between 2009 and 2012. Men were 1.7 times (OR = 1.69) more likely to be prescribed HIV screening in 2010 compared with 2009. Men were 1.4 (OR = 1.39) times more likely to be prescribed HIV screening in 2011 compared to 2009, and 2.3 (OR = 2.28) times more likely to be prescribed HIV screening in 2012 compared with 2009. This may be attributed in part to the limited amount of time elapsed since enactment of the ACA. Further research is needed to continue to monitor the impact of the ACA on prescription of HIV screening.
Examination of race provided interesting results, reporting non-Hispanic Black men to be 12 times more likely to be prescribed HIV screening compared to non-Hispanic White men (OR = 12.33). The second hypothesis of this study, Black men will demonstrate an increased incidence of HIV screening compared to Caucasian and Latino/Hispanic men, was reported to be true. The CDC, as well as other public health organizations, has developed campaigns focusing on racial and ethnic disparities associated with HIV incidences and care. While it is not possible to ascertain the reason for the increase in HIV screening among race and ethnic categories via the NAMCS database, increased exposure to advertising campaigns and care provider education may account for a change in practice. Because the NAMCS only samples visits to physician offices, many other potential patient contact points are not included. CHCs and state-level clinics that provide sexual health services are not included in this analysis. Men may choose other avenues for sexual health and HIV screening as they may provide a greater level of anonymity as well as discounted or free services.
Examination by age revealed noteworthy results. Men between the ages of 15 and 18 accounted for 48% (n = 2,481) of the subject population yet yielded the lowest percentage of HIV screening (10.4%/n = 5). Comparatively, men between the ages of 19 and 22 are 6.6 times more likely to receive a HIV screening compared with men ages 15 to 18 (OR = 6.59). Extant literature has demonstrated men between the ages of 15 and 25 are at an increased risk of contracting HIV. One way to rationalize this being the majority of the subjects used private insurance to pay for their provider visit, which may also explain the increased number of subjects younger than 19. The majority private insurance plans cover dependent children up to the age of 26. Not surprisingly, men between the ages of 23 and 25 demonstrated the highest number (50%/n = 24) of prescribed HIV screenings. During young adulthood these men may be doing more than simply exploring their sexuality. Ad campaigns such as National Youth HIV & AIDS Day use social and visual media to raise awareness and expose men and women to relevant sexual health issues. Young adulthood brings an increased level of independence as well as an opportunity to seek sexual health services without the consent or knowledge of parents. Removing parental consent might also increase adolescent and young adult self-efficacy to request screenings. Children who are under their parents’ insurance plan might fear their parents seeing “HIV test” on their insurance statement.
This study has limitations. First, the most current NAMCS data are available only up to the year 2012. As a result, only 4 years of data were included to assess the influence of the ACA on the prescription of HIV screening. The ACA became effective in 2010, with most major provisions phased in by January 2014. This period may not be long enough to capture the effects on prescribing HIV screening. Second, the NAMCS is based on self-report of physicians and their staff, creating the potential for recall bias. Third, the NAMCS findings are based on office-based physicians who are primarily engaged in direct patient care. This omits potential provider–subject contact points where HIV screening is offered. Fourth, the NAMCS does not offer state-level identifiers; however, regional data were extracted to examine various regions of the country, including metropolitan statistical areas. As of 2015, 29 states have agreed to Medicaid expansion, leaving 21 “no” states. Because a large percentage of “no” states exist in the southern region of the country, region is an important factor when examining the impact of the ACA. While data were examined based on region of the country, some states that have agreed to Medicaid expansion may be grouped with states that have refused expansion. Finally, the NAMCS data are limiting as a result of the questions asked, specifically, sexual orientation and gender identity (SOGI). SOGI data are important because sexual and gender minority populations experience disproportionate rates of HIV, making SOGI data instrumental in fully understanding the interactions between health care providers and sexual and gender minorities as they relate to sexual health screenings. Altering future NAMCS survey items may provide better insight into the health practices of men.
Despite limitations, these results contribute to the literature by providing evidence of the impact of the ACA on prescription of HIV screening. Study outcomes also indicate that practice guidelines addressing proactive prescription of HIV screening are needed to better address evident age gap in HIV screening among men between the ages of 15 and 18.
Footnotes
Authors’ Note
This study was exempted from institutional board approval at the authors’ institution because the data are publicly available.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
