Abstract
Whether the African American race remains a significant predictor of poorer prostate cancer survival after adjusting for other sociodemographic and treatment-related factors remains unclear. We examined whether disparities in survival among 18,900 African American and Caucasian men diagnosed with prostate cancer in Kentucky remained after adjusting for health insurance (payor source), cancer treatment, cancer stage at diagnosis, prostate-specific antigen (PSA) level, smoking status, and Appalachian region. After adjusting for these predictors, African American men living in Kentucky had poorer prostate cancer survival after 5 years (hazard ratio [HR] = 1.33; 95% confidence interval = 1.11, 1.59) and 10 years (HR = 1.39; 95% CI = 1.18, 1.28) of follow-up, and for the entire follow-up period (HR = 1.41; 95% CI = 1.26, 1.65) compared to their Caucasian counterparts. Thus, health insurance status, cancer treatment, cancer stage at diagnosis, PSA level at diagnosis, smoking status, and geographic location did not explain the racial gap in survival in Kentucky.
Introduction
Overall mortality from prostate cancer (PC) in the United States has declined over the last two decades; however, racial differences in survival after the diagnosis of PC remain an issue of significant public health concern (American Cancer Society, 2012). Evidence from existing literature suggests that racial minority groups, especially African American men, have substantially shorter survival after diagnosis of PC compared with Caucasian men (Brawley, Knopf, & Merrill, 1998; Godley et al., 2003; Jones et al., 2008; Ries et al., 2005; Siegel, Naishadham, & Jemal, 2012). Reasons for these survival disparities remain unclear.
Some population-based studies have reported that racial disparities in survival after the diagnosis of PC can be explained by differences in clinical and sociodemographic factors (Du et al., 2006; Evans, Metcalfe, Ibrahim, Persad, & Ben-Shlomo, 2008; Sridhar, Masho, Adera, Ramakrishnan, & Roberts, 2010; Tewari et al., 2005). However, other studies have also reported that racial survival disparities persist even after adjusting for clinical and sociodemographic factors (Albain, Unger, Crowley, Coltman, & Hershman, 2009; Albano et al., 2007; Du, Lin, Johnson, & Altekruse, 2011; Powell et al., 2002; Robbins, Whittemore, & Thorn, 2000). Thus, there is conflicting evidence on whether survival disparities among race groups can be explained by known predictors of cancer survival.
Few studies have examined this outcome adjusting for health insurance status, cancer stage at diagnosis, prostate-specific antigen (PSA) level at diagnosis, cancer treatment received, smoking status, and geographic location at the same time. To the best of our knowledge, no such study has been conducted in Kentucky, a state with a large Caucasian population (88%; U.S. Census Bureau, 2010). African American’s are the largest minority group (8%) in Kentucky and have the greatest disadvantage in educational attainments, access to health care, poverty, housing, and other sociodemographic parameters (Kentucky Commission on Human Rights, 2011; U.S. Census Bureau, 2010). In this study, we examined whether disparities in survival among African American men and Caucasian men diagnosed with PC remain after adjusting for cancer stage at diagnosis, PSA level at diagnosis, health insurance (payor source), cancer treatment, smoking status, and Appalachian region.
Literature Review
Findings are inconsistent regarding whether racial disparities in PC survival can be explained by known predictors of cancer survival. White, Coker, Du, Eggleston, and Williams (2011) examined racial differences in survival in a cohort of 87,449 men diagnosed with PC and reported to the Texas Central Cancer Registry between 1996 and 2002. These investigators found that after adjusting for age, cancer stage, Gleason grade, socioeconomic status (SES), and rural residence, African American men had a 70% (95% confidence interval [CI] = 1.58, 1.83) increased risk of death after 13 years of follow-up. However, in another population-based study by Robbins, Yin, and Parikh-Patel (2007), the difference in survival among 122,374 African American and Caucasian men diagnosed with PC in California between 1995 and 2004 was completely eliminated after adjusting for age, cancer stage, cancer treatment, Gleason grade, SES, and year of diagnosis (hazard ratio [HR] = 0.99; 95% CI = 0.92, 1.06). Nonetheless, these studies are limited by the potential confounding effects of health insurance and smoking status, which have been reported to have a significant effect on survival after diagnosis of cancer (Gong, Agalliu, Lin, Stanford, & Kristal, 2008; Kenfield, Stampfer, Chan, & Giovannucci, 2011; McDavid, Tucker, Sloggett, & Coleman, 2003; Ward et al., 2004). The current study will fill a gap in the existing literature by controlling for the joint effect of multiple predictors of PC survival including health insurance and smoking status and evaluate racial trends in survival across different follow-up periods.
Materials and Method
Data Source
Incident PC cases were obtained through the Kentucky Cancer Registry (KCR), which is the Central Cancer Registry for the Commonwealth of Kentucky. KCR is a member of the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program and has received annual gold certification awards since 1999 by the North American Association of Central Cancer Registries for the completeness, accuracy, and timeliness of its cancer incidence data. KCR conducts active cancer surveillance, including follow-up information on case status, using linkages with state vital records and the National Death Index. KCR provided the investigators a data file containing the following variables: race, age, date of diagnosis, cancer stage at diagnosis, Gleason score, PSA level at diagnosis, first course treatment, smoking status, insurance payor source, county of residence, Kentucky region (Appalachian/non-Appalachian), date of last contact, vital status and underlying cause of death. A prostate cancer case was defined based on the International Classification of Diseases for Oncology topographic code C61.9 (ICD-O-3). This study was approved by the institutional review board of the University of Kentucky, Lexington.
Study Population and Cancer Attributes
A total of 20,676 PC cases reported to KCR were available for analyses. Only histologically or cytologically confirmed primary site invasive PC cases occurring among African American men and Caucasian men between the ages of 40 to 99 years and reported to KCR from 2001 to 2010, with follow-up through December 31, 2010, were eligible for the study. Cases identified by death certificates or autopsy reports only and those outside the age range 40 to 99 years were already excluded from the data file received from KCR. Additionally, we excluded cases with missing or invalid data on date of diagnosis (n = 76), unknown vital status (n = 49), and those from racial groups other than African Americans or Caucasians (n = 1,651), leaving a total of 18,900 incident PC cases for the final analyses. KCR provided data to characterize cancer staging based on SEER 2000 criteria. In this study, PC stage at diagnosis was categorized as local/regional (referent group), distant, and unstageable/unknown stage. Data on PSA level at diagnosis were recorded in the data file as normal, elevated, or undetermined/unknown. These categories were maintained in the analyses with normal PSA level as the referent group. Information on Gleason score was available for only cases diagnosed between 2004 and 2010 (n = 11,301). Thus, we evaluated the potential confounding effect of Gleason score with sensitivity analyses.
Prostate Cancer Survival Definition
Survival time in months was calculated as the number of completed months between the date of diagnosis and the date of death, date last known to be alive, or the date of last follow-up, whichever came first. PC patients who were still alive at last follow-up (December 31, 2010) were censored at that time. The outcomes of interest were all-cause mortality and PC-specific mortality. All-cause mortality was defined as death from any underlying cause, and PC-specific mortality was defined as deaths where PC is listed as the underlying cause of death. Death rates for all-cause mortality and PC-specific mortality were calculated as the number of deaths divided by the population at risk at each time window.
Treatment
PC treatments were categorized as surgery only (referent group), radiation therapy only, chemotherapy only, other treatments, combination therapy, and no treatment. These treatment categories were created based on information on all disease-directed therapies within 4 months of PC diagnosis and all planned therapies within the first year of diagnosis. The surgery only category consists of surgical interventions such as radical prostatectomy and cryosurgery offered as a sole therapy for PC. This category was used as the referent group because it is a common curative therapy for localized disease and generally associated with better prognosis. Information on radiation type or dose was not available. Hence, all patients who received radiation treatment only as their first course therapy were classified as “radiation therapy.” Patients who received only chemotherapy were placed in the category of “chemotherapy,” and those who received PC-specific treatments other than surgery, radiation, and chemotherapy were categorized as “other treatments.” Patients who received any combination of surgery, radiation, chemotherapy, and other therapies were put in the category of “combination therapy,” and those who did not receive any PC-specific treatment were placed in the category of “no treatment.” The data file received from KCR did not distinguish watchful waiting from those who did not seek treatment or those who refused treatment. Hence, “no treatment” is a broad category consisting of those on watchful waiting, those who refused treatment, and those who did not seek treatment. To evaluate the effect of treatment on PC survival in the multivariate survival models, new categories of “treatment” versus “no treatment” were created. The “treatment” group consisting of all those who received any type of treatment and those in the “no treatment” group represent those who did not received any disease-directed therapy. Analyses were conducted within stage at diagnosis to better address the effect of race on survival adjusting for treatment that is contingent on cancer stage at diagnosis.
Other Study Variables
Information on health insurance was also obtained through KCR. Health insurance coverage (payor source) was categorized as private insurance (managed care/HMO/PPO and other private insurance), Medicare, Medicaid, government insurance (Military/Veterans/Indian/Public health service), unknown insurance status, and no insurance/self pay. The various categories of insurance were compared to private insurance.
Tobacco exposure was categorized as nonsmokers (referent group), smokers, and unknown. Data on previous tobacco exposure, smoking duration, or dose as in pack years were not available. Because tobacco exposure status was unknown for 5,422 (28%) of the cases, this large proportion of cases were retained and their smoking status was classified as “unknown.”
Kentucky region was categorized as either Appalachian or non-Appalachian based on the Appalachian Regional Commission’s designation. Like other Appalachian regions in the United States, the Appalachian regions of Kentucky are characterized by high rates of poverty and health disparities resulting from low-income jobs, high unemployment rates, geographic isolation, less public transportation, fewer physicians, clinics, and hospitals (Behringer & Friedell, 2006). Several studies have found higher cancer mortality rates in Appalachian regions than the non-Appalachian regions of the United States (Fisher et al., 2008; Lengerich et al., 2004; Wingo et al., 2008). Thus, to measure the effect of geographic location on survival, the Appalachian region of Kentucky was compared with the non-Appalachian region (referent group).
Statistical Analysis
Differences in the distribution of demographic and cancer attributes between African Americans and Caucasians were determined and tested using contingency tables and chi-square test or t-test when appropriate. Cox proportional hazard models were used to estimate the relative rate of dying across follow-up. These multivariate survival models also were used to examine the risk of death in African Americans relative to Caucasians with adjustment of age, PSA level at diagnosis, cancer stage, smoking status, cancer treatment, health insurance status, and Appalachian region. Hazard ratios were estimated for 5-year survival, 10-year survival, and overall survival across follow-up. Kaplan–Meier survival curves were used to describe survival disparity by race. The proportionality assumption was considered satisfied when the log-log survival curves were parallel and did not intersect (Kleinbaum & Klein, 2005). The categories of local and regional cancer stage at diagnosis did not satisfy the proportional hazards assumption, and for this reason these two categories were combined into one group, local/regional stage. The proportionality assumption was met once the categories were combined. All statistical analyses were done using Version 9.3 of SAS®, with statistical significance set at α = .05 (two-tailed).
Results
Demographic and cancer attributes of Caucasian men and African American men diagnosed with PC in Kentucky are presented in Table 1. Of the 18,900 men in the cohort, 17,251 (91.3%) were Caucasians and 1,649 (8.7%) were African Americans. Rates of all-cause mortality were significantly higher in African American men than Caucasian men (27.8% vs. 22.1%, respectively, p < .001). Similarly, the proportion of African American men who had died from PC was significantly higher than that of Caucasian men (10.9% vs. 7.0%, respectively, p < .001). Compared with Caucasian men, African American men were more likely to be diagnosed with PC at a younger age (average age = 63.9 ± 10.0 years vs. 66.9 ± 9.8 years, p < .001), have elevated PSA levels at diagnosis (66.6% vs. 64.4%, p < .001), presented with advance staged disease (distant stage = 6.5% vs. 4.3%, p < .001), report tobacco use (smokers = 44.7% vs. 41.4%, p < .001), and to have no health insurance (3.5% vs. 1.4%, p < .001). African American men with PC were less likely than Caucasian men to reside in Appalachian Kentucky (6.6% vs. 28.2%, p < .001) but more likely to have surgery only as treatment for PC (35.2% vs. 29.6%, p < .001). There were no differences in Gleason score between the two race groups (average Gleason score = African Americans: 6.8 ± 1.0 vs. Caucasians: 6.7 ± 1.0, p = .2527).
Demographic and Cancer Attributes Among Caucasians and African Americans Diagnosed With Incident Prostate Cancer in Kentucky, 2001-2010 (N = 18,900).

Testing for differences between African Americans and Caucasians was done using t test for continuous variable and chi-square test for categorical variables.
Information on Gleason score was only available for cases diagnosed between 2004 and 2010.
Table 2 presents results of the unadjusted and adjusted proportional hazard models for all-cause mortality. Relative to Caucasian men, African American men had a 24% increased risk of all-cause mortality after the diagnosis of PC (HR = 1.24; 95% CI = 1.12, 1.36). In the adjusted models, sensitivity analyses were conducted to assess whether Gleason score might confound the results of the study given that a large proportion of the study population had missing data on Gleason score. Results of the sensitivity analyses indicated that Gleason score had no effect on the PC outcomes in this study population. Thus, Gleason score was not included in the adjusted models. After adjusting for age, PSA level, cancer stage, smoking status, health insurance status, cancer treatment, and Appalachian region, African American men had a 31% increased risk of all-cause mortality after the diagnosis of PC (HR = 1.31; 95% CI = 1.19, 1.45). This finding paralleled the Kaplan–Meier survival curves presented in Figure 1.
Unadjusted and Adjusted Probability of All-Cause Mortality Among Caucasians and African Americans Diagnosed With Incident Prostate Cancer in Kentucky, 2001-2010.

Note. HR = hazard ratio; CI = confidence interval.
The proportional hazard model adjusted for the variables listed in the table.

Kaplan–Meier survival curves for all-cause mortality among African Americans and Caucasians diagnosed with incident prostate cancer in Kentucky, 2001-2010.
For PC specific mortality, African American men had a 53% crude hazard risk of death from PC compared to Caucasian men (HR = 1.53; 95% CI = 1.31, 1.79; Table 3). The adjusted hazard rates of PC-specific mortality by race were estimated for 5 years, 10 years, and overall survival with adjustment of age, PSA level, cancer stage, smoking status, health insurance status, cancer treatment, and Appalachian region (Table 4). After 5 years of follow-up, the hazard risk of PC specific mortality was 33% (HR = 1.33; 95% CI = 1.11, 1.59) higher in African American men than Caucasian men. A similar trend of poorer survival for African American men was observed after 10 years of follow-up and for the entire follow-up period, where African American men had a 39% (HR = 1.39; 95% CI, 1.18, 1.63) and 41% (HR = 1.41; 95% CI = 1.26, 1.65) increased risk of death, respectively (Table 4). These findings also paralleled the Kaplan–Meier survival curves presented in Figures 2, 3, and 4. When stratified by stage at diagnosis (Table 5), being an African American male was associated with a 2.06 times higher risk of death from PC for the localized/regional stage over the entire follow-up period (HR = 2.06; 95% CI = 1.63, 2.58), but not the distant stage (HR = 0.99; 95% CI = 0.75, 1.30).
Unadjusted Probability of Prostate Cancer–Specific Mortality Among Caucasian and African American Men Diagnosed With Incident Prostate Cancer in Kentucky, 2001-2010.
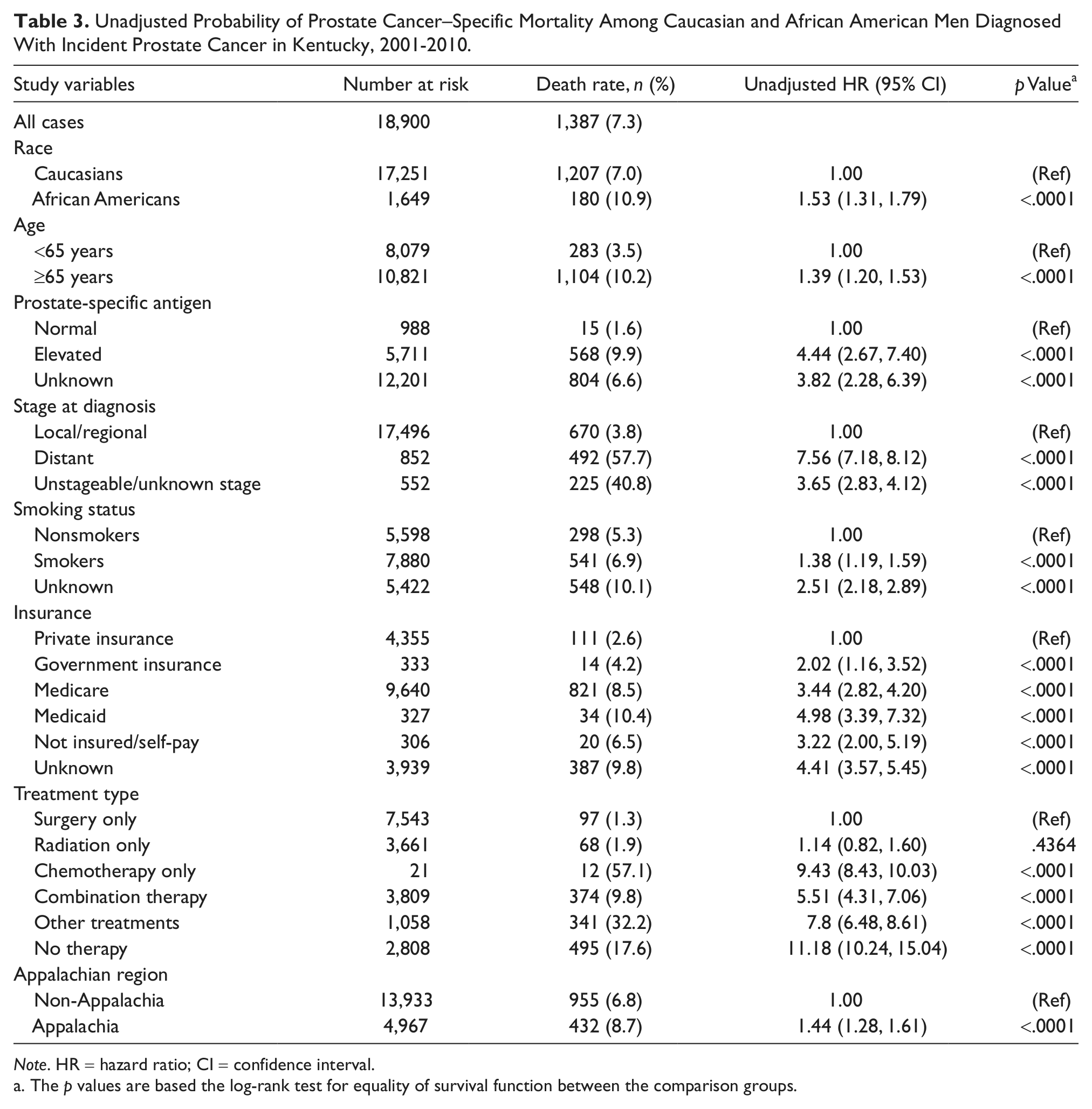
Note. HR = hazard ratio; CI = confidence interval.
The p values are based the log-rank test for equality of survival function between the comparison groups.
Adjusted 5-Year, 10-Year, and Overall Probability of Prostate Cancer–Specific Mortality Among African American and Caucasian Men Diagnosed With Incident Prostate Cancer in Kentucky, 2001-2010.

Note. HR = hazard ratio; CI = confidence interval.
Overall survival comprises cases diagnosed between January 1, 2001, and December 31, 2012, a total of 10 years and 11 months of follow-up.

Kaplan–Meier survival curves for prostate cancer–specific mortality after 5-years of follow-up among African Americans and Caucasians diagnosed with incident prostate cancer in Kentucky.

Kaplan–Meier survival curves for prostate cancer–specific mortality after 10-years of follow-up among African Americans and Caucasians diagnosed with incident prostate cancer in Kentucky.
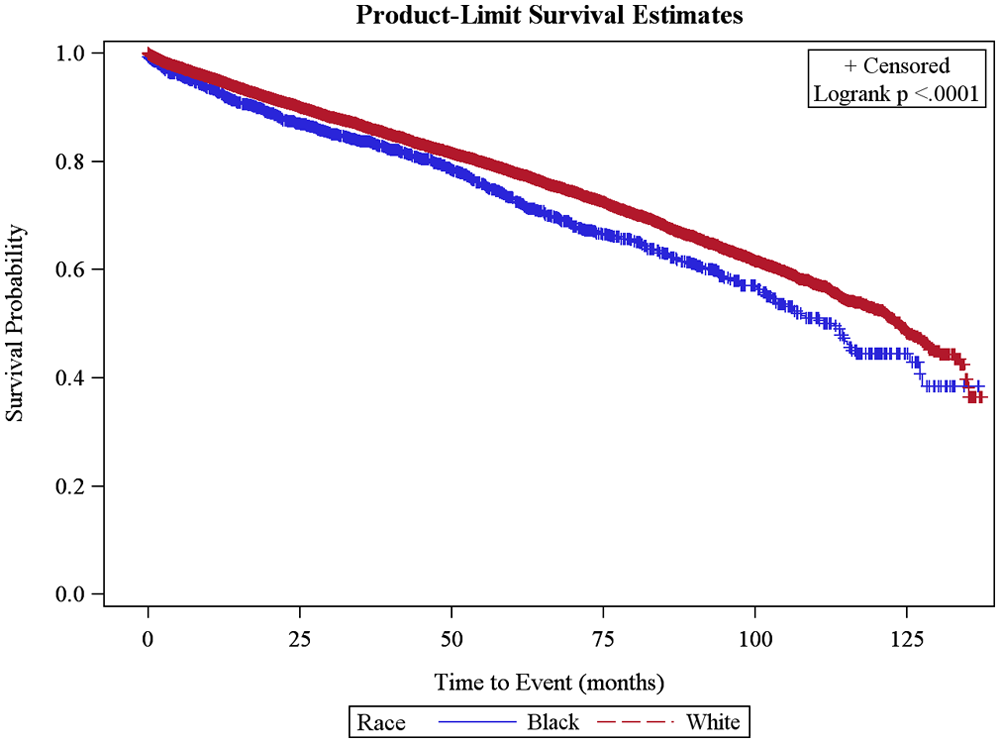
Kaplan–Meier survival curves for overall prostate cancer–specific mortality among African Americans and Caucasians diagnosed with incident prostate cancer in Kentucky.
Stratified Probability of Prostate Cancer–Specific Mortality by Stage at Diagnosis Among African American and Caucasian Men Diagnosed With Incident Prostate Cancer in Kentucky, 2001-2010. a

Note. HR = hazard ratio; CI = confidence interval.
Prostate cancers that were unstageable/unknown stage (n = 552) were not included in these analyses.
Although the multivariate models controlled for the confounding effect of health insurance (a proxy for access to care), additional analyses (data not represented) were performed for patients on Medicare and private insurance separately. Relative to Caucasian men, African American men who were on Medicare had a 68% higher risk of death from PC after adjusting for age, PSA level, cancer stage, cancer treatment, smoking status, and Appalachian region (HR = 1.68; 95% CI = 1.52, 1.93). Similarly, African American men with private insurance had a 20% higher risk of death from PC compared with their Caucasian counterparts after adjusting for the above list of variable (HR = 1.20; 95% CI = 1.16, 1.34).
Discussion
The current study investigated whether disparities in survival among African American men and Caucasian men diagnosed with PC remained after adjusting for health insurance status, cancer stage at diagnosis, PSA level at diagnosis, cancer treatment, smoking status, and geographic location. Results of the study show that African American men diagnosed with PC in Kentucky have shorter survival than their Caucasian counterparts. The survival disadvantage for African American men remained after 5 years and 10 years of follow-up, as well as for the entire follow-up period with adjustment of the predictor variables. The African American race was also associated with higher PC-specific mortality at the local/regional stage at diagnosis. The racial survival disparity observed in this study is unlikely to be due to differences in health insurance status along racial lines since African American men had excess risk of PC-specific mortality among those with the same health insurance. Similarly, sensitivity analyses indicated that Gleason score could not have confounded the results of the study.
Findings from this study are consistent with those reported by recent studies. Du et al. (2011); White et al. (2011); Taksler, Keating, and Cutler (2012); and Singh, Williams, Siahpush, and Mulhollen (2011) have all reported shorter survival in African American men after the diagnosis of PC. In these studies, the racial gap in survival remained after adjusting for known predictors of cancer survival. Also, recent studies of PC mortality-to-incidence ratio in South Carolina and Georgia have reported 58% and 55% higher PC mortality-given incidence, respectively, among African Americans than Caucasians (Hébert et al., 2009; Wagner et al., 2012). However, few studies have reported negative association between race and survival after diagnosis with PC. Using data from the Department of Defense Cancer Registry, Optenberg et al. (1995) observed similar survival outcomes for African American men and Caucasian men diagnosed with PC. This negative finding can be attributed to the relatively small sample size (n = 1,606), strict criteria used to recruit healthy men into the military, or access to similar medical care due to Department of Defense health insurance. In a meta-analysis, Bach et al. (2002) also observed similar survival outcomes for African Americans and Caucasians diagnosed with PC, yet this study was limited by the combination of cases diagnosed before and after 1986, which is not advisable due to dramatic changes in the incidence, staging, and grading of PC since the widespread use of PSA test was introduced for screening in 1986 (Adami, Hunter, & Trichopoulos, 2008).
The current analyses clearly show that African American men have poorer survival after diagnosis of PC than Caucasian men. Reasons for this survival disparity are not well understood. However, the survival inequality is likely multifactorial including differences in patterns of behavior, structures, and dynamics of society as well as inequalities in access to equitable standards of care. For example, physicians who treat African Americans tend to have less clinical training and have reported greater difficulties in obtaining access to clinical resources for their patients compared with physicians who treat Caucasians (Bach, Pham, Schrag, Tate, & Hartgrave, 2004).
Strengths and Limitations
The strengths of our study include the ability to adjust for multiple predictors of cancer survival including medical insurance coverage (an imperfect but widely accepted proxy for access to care), smoking status (a known risk factor for a range of chronic diseases), and a larger sample size. The evaluation of both all-cause mortality and PC-specific mortality also adds to the strengths of this study. This study confirms that factors such as cancer stage at diagnosis, smoking status, health insurance status, cancer treatment, and geographic region are not contributors to the racial disparity in PC survival between African Americans and Caucasians. Identifying factors that do not contribute to the racial differences in PC survival helps clarify direction for future studies in this area of health disparity. Additionally, the mortality differences observed in this study population is particularly important because Kentucky has unique population attributes and sociodemographic characteristics from that of other parts of the country where these health disparities have been reported. Our study was limited by restricting the study sample to PC cases diagnosed in Kentucky only, which limits generalizability to this population. Also, there is the possibility that the observed association between race and mortality after the diagnosis of PC could be due to the effects of other unmeasured factors such as comorbid conditions, SES, and stage-specific treatment guidelines adherence between the two race groups.
Conclusion
African American men living in Kentucky have poorer survival after the diagnosis of PC compared with Caucasian men. Health insurance (payor source), cancer treatment, cancer stage at diagnosis, PSA level at diagnosis, smoking status, and geographic location did not explain the racial gap in survival. Future studies should consider the effects of comorbidities, expanded measures of socioeconomic status, stage-specific treatment guidelines concordance between African Americans and Caucasians, and potential differences in PC biology along racial lines.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
