Abstract
McLaughlin, Kyle, Steve Roy, Marika Falla, Giacomo Strapazzon, Andrew M. Luks, Ken Zafren, Hermann Brugger, Martin Musi, Iztok Tomazin, John Ellerton, Ghan Bahadur Thapa, and Peter Paal. Pharmacological prophylaxis and supplemental oxygen for unacclimatized rescuers at very high altitude: scoping review and 2025 joint recommendations of the International Commission for Mountain Emergency Medicine and the International Society for Mountain Medicine. High Alt Med Biol. 27:60–77, 2026.
Background:
Mountain rescuers and pilots rapidly ascending to altitudes above 3,500 m are exposed to the detrimental effects of hypobaric hypoxia, including cognitive and physical impairment, as well as high-altitude illness (HAI). We conducted a scoping review of oxygen supplementation and pharmacologic measures to improve cognitive and physical performance and prevent HAI in unacclimatized rescuers rapidly ascending above 3,500 m during rescue missions.
Methods:
Following Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines, 723 articles were screened, 133 reviewed and a total of 50 articles were included for data extraction, based on the intervention: 6 on oxygen, 29 on acetazolamide (AZ), 17 on dexamethasone (DEX), 3 on nifedipine, and 5 on phosphodiesterase-5 inhibitors.
Discussion:
Supplemental oxygen improves physical and cognitive performance at high altitude and is recommended for rapid ascent rescues >30 minutes between 3,500 and 4,000 m, and for rescues of any duration above 4,000 m. If oxygen is administered, pharmacological prophylaxis is not required. If oxygen is unavailable, AZ or DEX can be used for rapid ascent rescues above 3,500 m for longer than 3 hours to reduce the incidence and severity of acute mountain sickness. At altitudes above 5,000 m or for rescues requiring prolonged physical work, the use of both AZ and DEX is recommended.
Conclusions:
To enhance the safety and effectiveness of high-altitude rescues, we provide recommendations for the use of supplemental oxygen and pharmacologic prophylaxis to reduce the risk of HAI and improve cognitive and physical performance during rapid ascents to altitudes >3,500 m.
Introduction
Given the large number of people traveling to high altitudes (HAs) annually (Burtscher et al., 2001) and the increasing number of climbers aiming to summit the world’s highest mountains (Global Rescue, 2024; Salisbury et al., 2021), the risk of accidents and the need for mountain rescue operations are rising (Gasser, 2022).
Altitudes ranging from 1,500 to 3,500 m are considered “HA,” 3,500 to 5,500 m as “very high altitude” (VHA), and above 5.500 m as “extreme altitude” (Roach et al., 2016). In this review, “VHA” refers to the altitudes greater than 3,500 m. Rescue operations at VHA present unique challenges beyond typical low-elevation mountain rescue risks. (Milani et al., 2023; Wu, 2011). These rescues often involve rapid ascent, usually by helicopter. They can range from quick patient transports to complex rescues that require prolonged physical ground work in complicated terrain and challenging weather. The modes of travel and patient conditions vary.
Acute exposure to hypobaric hypoxia at VHA can impair cognitive and physical function and increase the risk of developing high-altitude illnesses (HAI) such as acute mountain sickness (AMS), high-altitude cerebral edema (HACE), and high-altitude pulmonary edema (HAPE) (Burtscher et al., 2021; Castellani et al., 2021; Fulco et al., 1998; Grocott et al., 2009; Mallet et al., 2023; Paal et al., 2022; Willmann, 2015; Yan, 2014). The risk of HAI varies depending on several factors, including genetic predisposition, length of time spent at HA, maximum altitude reached, ascent rate, flying to HA, and sleep elevation (Burtscher et al., 2023; Hackett et al., 1976; Hansen et al., 1967; Hillenbrand et al., 2006; Poudel et al., 2025). Cognitive and psychiatric symptoms should also be considered, as neuropsychiatric symptoms can be more subtle than the classical symptoms of HAI (Falla et al., 2024a).
For the general population traveling to HAs, a gradual ascent with time for acclimatization is the most effective way to reduce the risk of HAI (Luks et al., 2024). However, mountain rescue operations do not allow time for a gradual ascent, and many rescuers may not have spent sufficient time at HA prior to a mission to benefit from residual acclimatization effects. Therefore, other measures, including supplemental oxygen and pharmacologic prophylaxis against HAI, must be considered to mitigate the risks of hypobaric hypoxia and ensure safe performance during rescue.
Most published guidelines on HAI prevention using pharmacologic prophylaxis and other measures focus on the general public’s standard ascent profiles (Luks et al., 2024) and do not address the specific challenges of rapid ascent to VHA during rescue missions. Furthermore, these guidelines fail to consider the differences between air and ground rescue or the distinct needs of pilots and rescuers. Protocols for VHA rescues have been established in certain regions, but these vary greatly depending on the altitude, topography, and local political and economic factors (Civil Aviation Authroity of Nepal CAAN, 2013; European Union Aviation Safety Agency EASA, 2007; Federal Aviation Administration, 2021; Government of Canada, 2023; Ollig, 2024; Smith, 2020).
The objective of this scoping review is to address these gaps in knowledge and evaluate the literature on oxygen supplementation and pharmacologic prophylaxis to improve cognitive and physical performance and prevent HAI in unacclimatized rescuers stationed at low altitudes who are swiftly deployed to VHA. The findings will inform recommendations for mountaineers, rescuers, pilots, and rescue organizations to improve the safety of rescue missions above 3,500 m.
Methods
We adhered to the Preferred Reporting Items for Scoping Reviews and Meta-Analysis Protocols (Tricco et al., 2018). Objectives, inclusion and exclusion criteria, and Population-Intervention-Comparator-Outcome (PICO) questions were defined.
A systematic literature search was conducted in PubMed, Cochrane Database, and Embase through Jun 24, 2024. We sequentially evaluated the titles, abstracts, and full texts of all publications identified through our search for potentially relevant publications. A manual search of the eligible studies followed a database search.
Two authors identified studies that utilized supplemental oxygen during rapid ascent to VHA using the search string outlined in Supplementary Data S1. The search strategy was organized according to the Population–Concept–Context mnemonic, as recommended for scoping reviews by the JBI Collaboration (Peters et al., 2020). Data were extracted from the final six studies (Table 1).
Studies of Supplemental Oxygen for Rapid Ascent Above 3,500 m
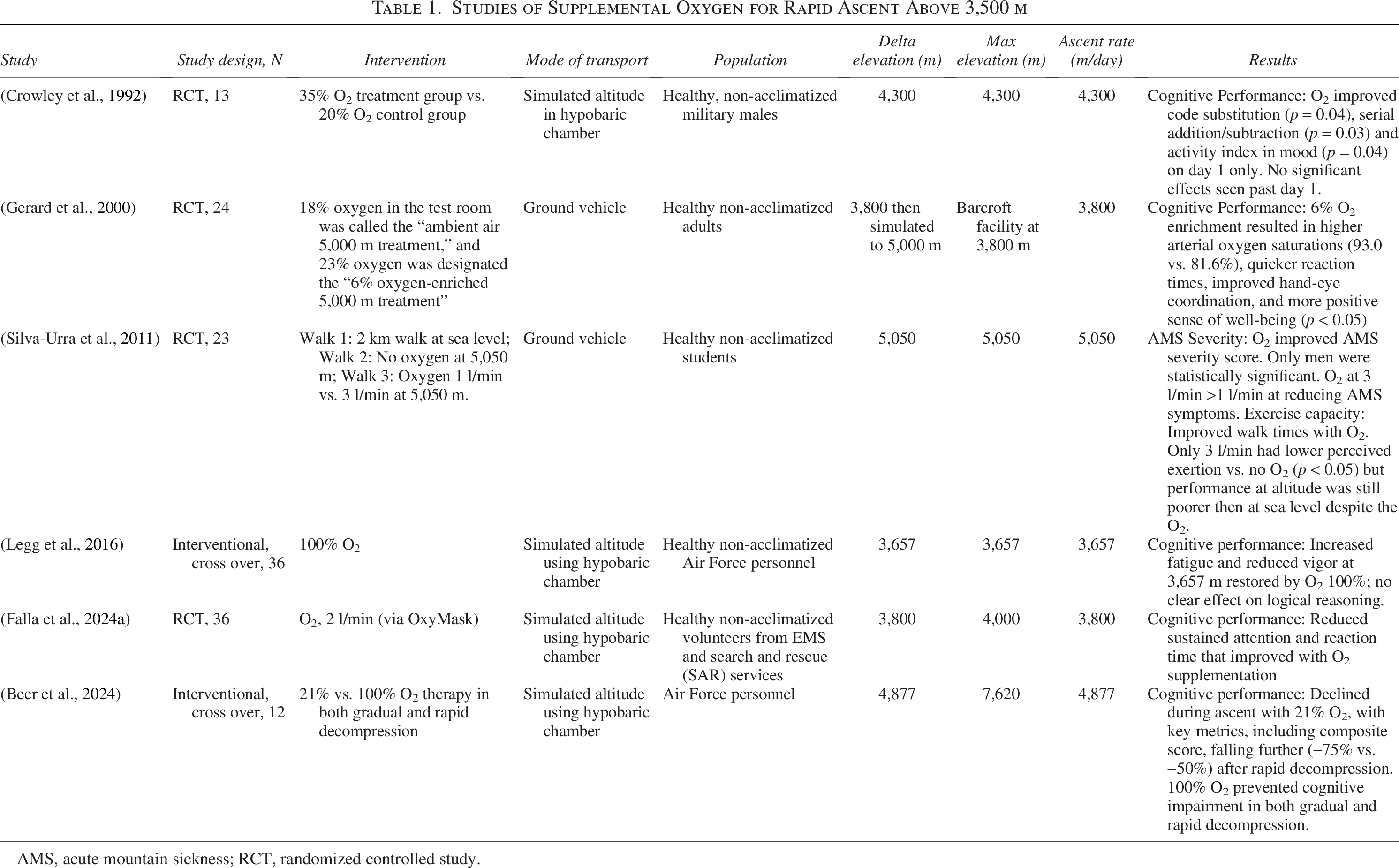
AMS, acute mountain sickness; RCT, randomized controlled study.
Two authors, along with an experienced librarian, developed search strings to identify articles on pharmacological prophylaxis for HAI during rapid ascent (>300 m/day) to VHA (Supplementary Data S2). All publications were independently reviewed and disagreements were resolved by a third author. The final search results were exported to the Covidence software and duplicates were removed. Data from the final 44 studies were evaluated for quality and relevance using the PICO questions (Tables 2–5).
Studies of Acetazolamide for Rapid Ascent Above 3,500 m
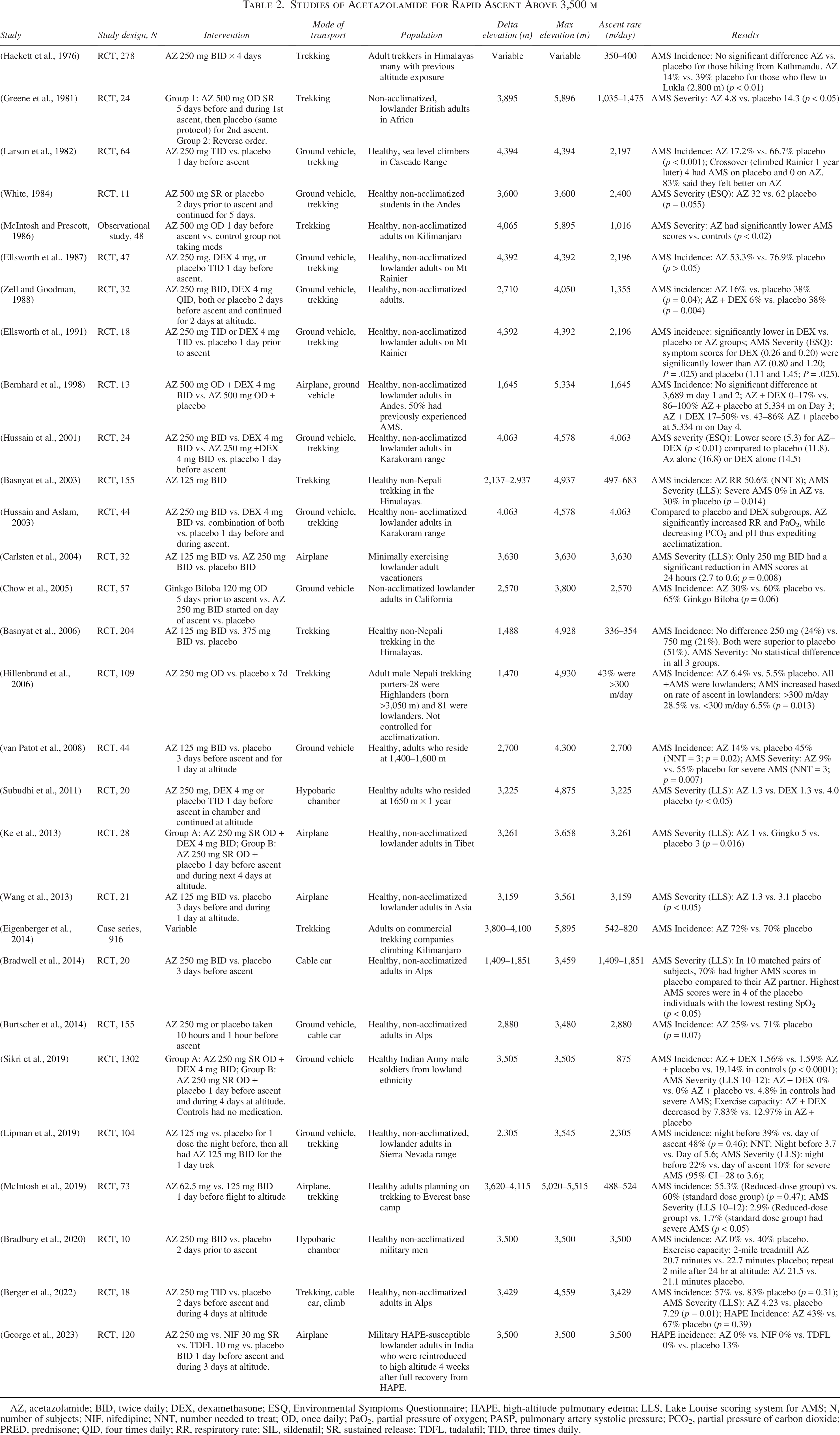
AZ, acetazolamide; BID, twice daily; DEX, dexamethasone; ESQ, Environmental Symptoms Questionnaire; HAPE, high-altitude pulmonary edema; LLS, Lake Louise scoring system for AMS; N, number of subjects; NIF, nifedipine; NNT, number needed to treat; OD, once daily; PaO2, partial pressure of oxygen; PASP, pulmonary artery systolic pressure; PCO2, partial pressure of carbon dioxide; PRED, prednisone; QID, four times daily; RR, respiratory rate; SIL, sildenafil; SR, sustained release; TDFL, tadalafil; TID, three times daily.
Studies of Dexamethasone for Rapid Ascent Above 3,500 m
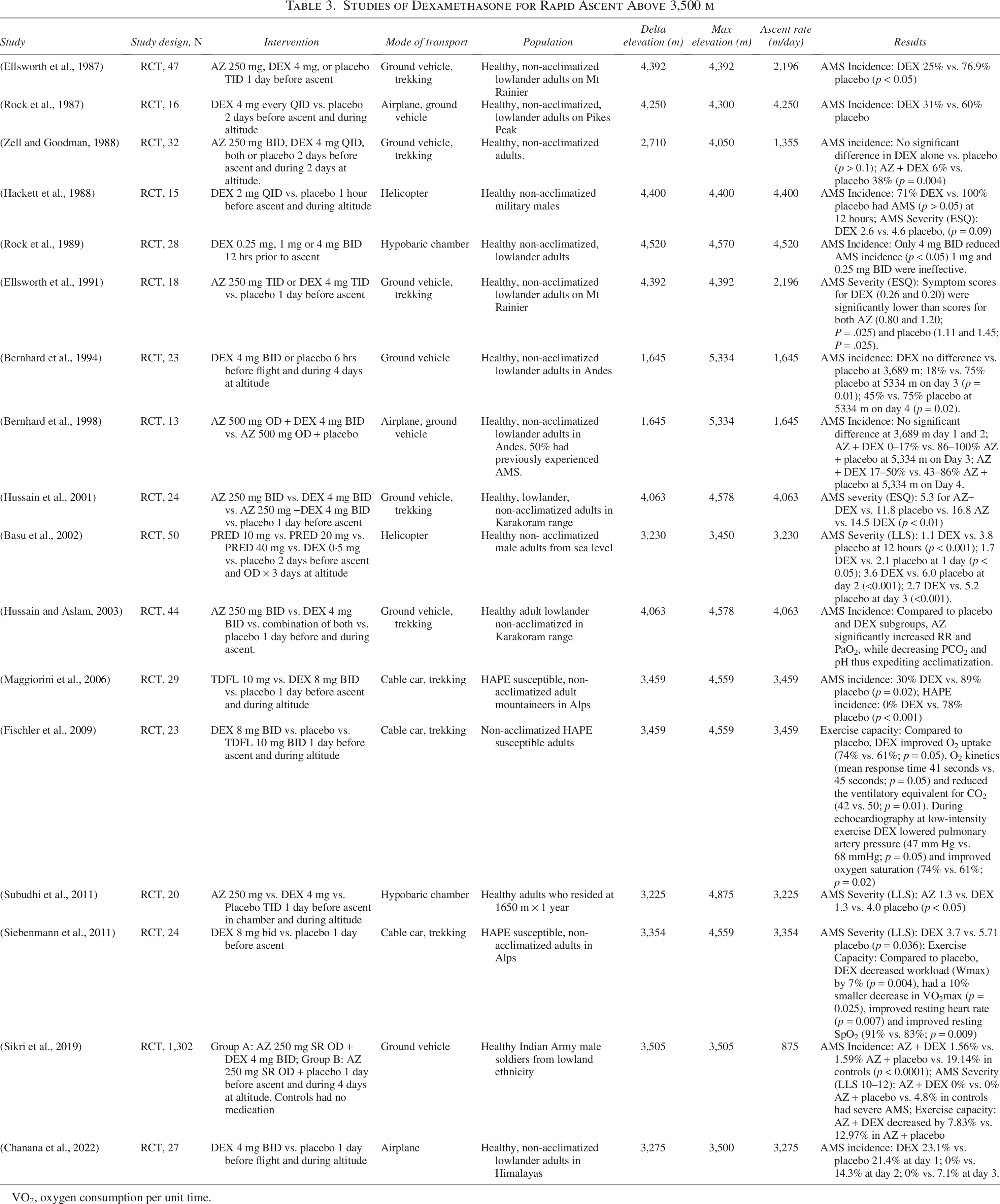
VO2, oxygen consumption per unit time.
Studies of Nifedipine for Rapid Ascent Above 3,500 m

Studies of Phosphodiesterase-5 Inhibitors for Rapid Ascent Above 3,500 m
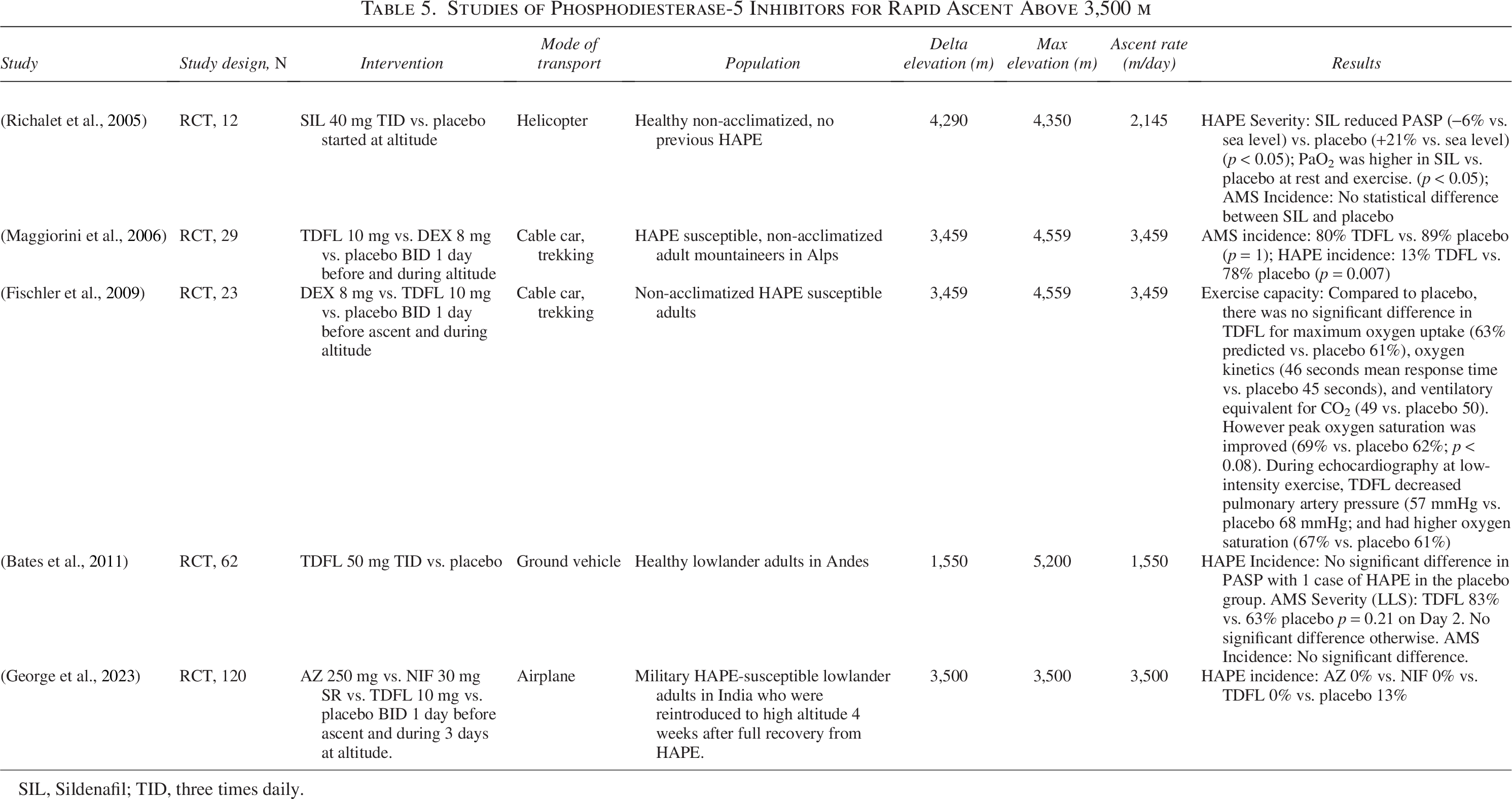
SIL, Sildenafil; TID, three times daily.
Recommendations were developed and graded by consensus based on the strength and quality of evidence using the system of the American College of Chest Physicians (Guyatt et al., 2006). Finally, the article was discussed and accepted by the International Commission for Alpine Rescue Medical commission at the ICAR Congress 2024 in Thessaloniki, Greece and endorsed by the International Society for Mountain Medicine in January 2025.
Results
We screened 723 articles, reviewed the full text of 133 articles, and included 50 studies for data extraction. The final data were categorized according to the interventions, with 6 articles on oxygen, 29 on acetazolamide (AZ), 17 on dexamethasone (DEX), 3 on nifedipine (NIF), and 5 on phosphodiesterase-5 inhibitors (PDE-5I). Eight articles addressed both AZ and DEX, and one article was included in AZ, NIF, and PDE-5I tables (Fig. 1).
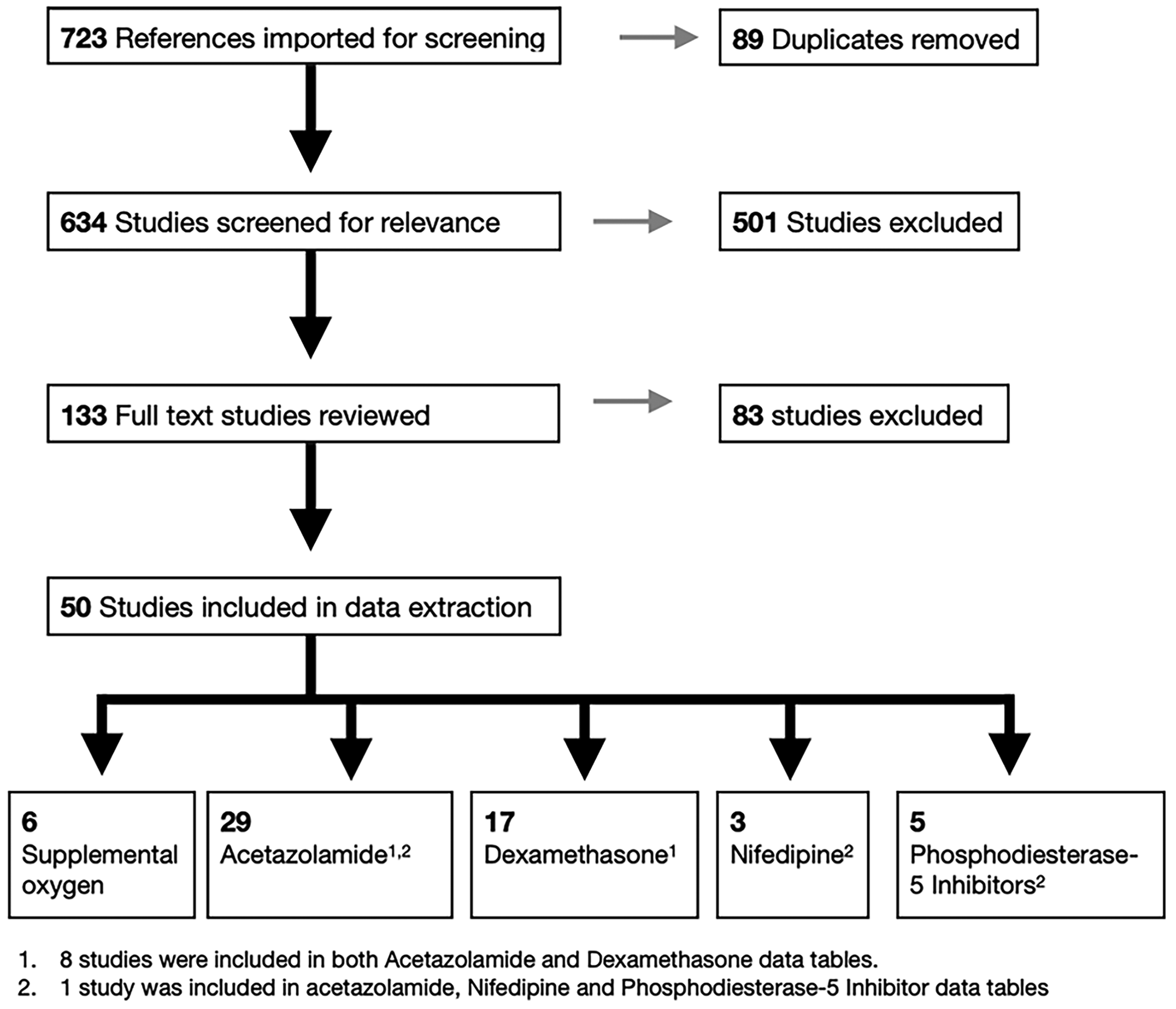
Literature search results for supplemental oxygen and pharmacologic prophylaxis used in rapid ascent to very high and extreme altitude.
Supplemental oxygen for rapid ascent to VHA
Several studies have documented the effects of hypoxia on cognitive performance and mood following ascent to HA in unacclimatized subjects. These studies consistently found evidence of cognitive impairment, including impaired executive function, working memory, grammatical reasoning, and mood alteration (Crowley et al., 1992; Falla et al., 2024a; Falla et al., 2024b). Field studies on aircraft and helicopter operations at altitudes between 3,000 and 5,500 m indicate that crew members experience symptoms of hypoxia, including cognitive impairment or lightheadedness (Cable, 2003), which are exacerbated by physical activity (Smith, 2005; Smith, 2007; Tristan, 2017). Other studies have also shown a reduction in physical performance at HA, which could negatively affect rescue operations (Clebone et al., 2020; Vogele et al., 2021).
Data on oxygen supplementation at HA are limited and primarily derived from chamber studies involving either normobaric or hypobaric hypoxia. Six studies specifically examined the use of supplemental oxygen during rapid ascent (>300 m/day) to VHA (Table 1). Supplemental oxygen mitigates the negative effects of hypoxia on mood (Legg et al., 2016) and various aspects of cognitive function, including memory, arithmetic, and visual and auditory tasks (Beer et al., 2024; Crowley et al., 1992; Gerard et al., 2000; Legg et al., 2016). In the context of rescue operations, a hypobaric chamber study (Falla et al., 2024b) found that oxygen supplementation improved attention and reaction time in helicopter emergency medical services (HEMS) providers during acute exposure to 4,000 m.
Although supplemental oxygen had a consistent positive effect, additional systemic stressors such as dehydration, sleep deprivation, prolonged shifts, fatigue, and rapid ascent may further exacerbate cognitive impairments at VHA. This is especially important for rescue missions that involve complex decision-making and physical activities.
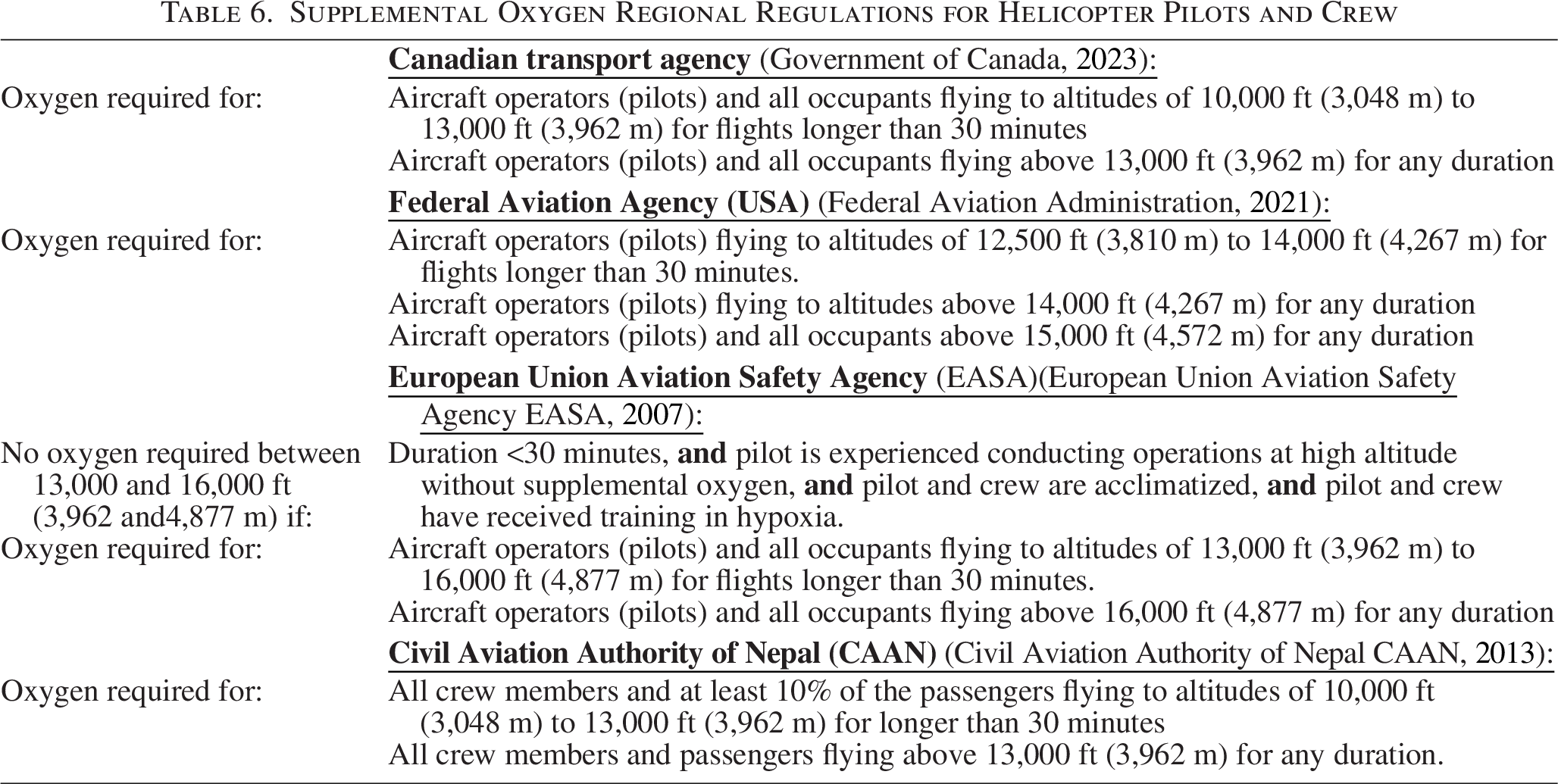
The positive cognitive effect of supplemental oxygen for pilots at VHA has been acknowledged by aviation agencies in Canada, the United States, Nepal and Europe, and is reflected in their aviation regulations (Table 6). However, these regulations do not specifically comment on oxygen supplementation for rescuers inserted into high-altitude terrain purposely or by accident (grounding). Supplemental oxygen is often reserved only for the pilots and not available for the rescuers in the helicopter.
Supplemental Oxygen Regional Regulations for Helicopter Pilots and Crew
Exercise capacity is reduced at HA, including physical activities critical to rescue at VHA, like performance of chest compressions (Clebone et al., 2020; Vogele et al., 2021). Simulated and field studies have shown a reduction in chest compression depth within 60–90 seconds of initiating CPR (Vogele et al., 2021), as well as a decrease in the number and depth of effective compressions (Wang et al., 2014). Supplemental oxygen improved CPR performance at a simulated altitude of 2,438 m (Clebone et al., 2020) and improved the 6-minute walk test at 5,050 m in unacclimatized subjects carrying 4 kg weights (Silva-Urra et al., 2011). Although oxygen is unlikely to have a significant impact on rescuer performance for quick patient transport, these studies suggest that supplemental oxygen would help preserve physical performance during rapid ascents above 3,500 m, particularly during activities such as crevasse rescue, avalanche extrication, and human external cargo (HEC) long-line rescue operations.
Supplemental oxygen improved AMS severity scores at 5,050 m in a dose-dependent manner (Silva-Urra et al., 2011). AMS may occur within 6–12 hours of exposure to hypoxia (Bartsch and Swenson, 2013), suggesting that short helicopter trips to VHA place pilots and rescuers at minimal risk of HAI. However, supplemental oxygen may still be required to mitigate the effects of hypoxia on cognitive function and physical performance. When supplemental oxygen is used, there is likely no added benefit from pharmacological prophylaxis; however, this has not yet been studied in the context of rapid ascent to VHA. If the duration at VHA exceeds the oxygen supply, pharmacological prophylaxis should be administered.
Supplemental oxygen can be administered via nasal prongs, simple oxygen masks, or non-rebreather masks, using continuous or on-demand flow systems. At 5,050 m, a supplemental oxygen flow rate of 3 l/min was superior to 1 l/min in reducing AMS symptoms and perceived exertion (Silva-Urra et al., 2011). At simulated altitudes of 8,100 m, flow rates of 1 l/min at rest and 2 l/min during exercise were sufficient to maintain oxygen saturation at a level that compensates for hypoxemia-induced symptoms (Wakeham et al., 2023). The effective altitude at rest was lowered by 35% and 66% for 1 l/min and 2 l/min, respectively. With exercise, there was no effect with 1 l/min, but the effective altitude was lowered by 21% with 2 l/min and 47% with 4 l/min (Wakeham et al., 2023). However, the optimal mask, delivery system, flow rate, and utility of oxygen saturation monitoring have not been adequately studied. Lighter battery-powered oxygen concentrators may be a viable alternative to heavy oxygen canisters in rescue operations, particularly for terrestrial rescue teams, providing adequate battery power is available (Sakaue et al., 2008).
Although supplemental oxygen is more accessible for helicopter (air) rescue, the cognitive and physical benefits of supplemental oxygen are still applicable to terrestrial (ground) rescue teams, although its practicality may be compromised due to the weight and limited availability of oxygen canisters. Each terrestrial rescue mission should involve risk analysis to determine the utility of oxygen. Genetically adapted high-altitude rescuers may opt to forgo supplemental oxygen, but many rely on it while guiding above 8,000 m to maintain optimal performance. Consequently, withholding supplemental oxygen from this group should be viewed as altitude-dependent.
Recommendations
During flight: Pilots and aircraft crews should use supplemental oxygen in accordance with the regional aviation regulations (Table 6). If no regulation exists, supplemental oxygen should be used based on the following guidelines (strong recommendation, moderate-quality evidence):
Optional: duration <30 minutes flights below 4,000 m. Recommended: duration >30 minutes flights between 3,500 and 4,000 m. Mandatory: any duration above 4,000 m
Rescuers on the ground after helicopter rapid ascent: Unacclimatized rescuers flown to VHA should use supplemental oxygen on the ground and during long-line HEC missions lasting more than 30 minutes at altitudes between 3,500 and 4,000 m and for any duration above 4,000 m (strong recommendation, moderate-quality evidence).
Terrestrial rescuers approaching patients by ground: Terrestrial rescue teams rapidly ascending above 3,500 m should use supplemental oxygen, if it is available and practical (strong recommendation, moderate-quality evidence).
Personnel using supplemental oxygen need not use pharmacological prophylaxis unless the duration of exposure to VHA exceeds the oxygen supply (strong recommendation, low-quality evidence).
Pharmacological Prophylaxis for Rapid Ascent to VHA
Acetazolamide prophylaxis for AMS
AZ enhances altitude acclimatization through carbonic anhydrase inhibition in the kidneys, red blood cells, brain, pulmonary and systemic vasculature, and chemoreceptors (Swenson and Teppema, 2007). Typically, it is administered at a dose of 125 mg every 12 hours, but the optimal dose for rapid ascent to VHA, especially at elevations above 5,000 m, remains unclear. It has been suggested that high-risk groups, such as rescue teams, may benefit from 250 mg every 12 hours although this has not been studied in rapid ascent rescue to VHA (Carlsten et al., 2004; Luks et al., 2024; Toussaint et al., 2021). For teams rapidly ascending to VHA, AZ is discontinued upon descent to the previously acclimatized elevation. However, for those remaining longer at VHA, it should be continued for 2–4 days or until descent, whichever occurs first. (Luks et al., 2024). As AZ has a sulfa moiety, it should not be used in individuals with previous anaphylaxis or Stevens–Johnson Syndrome following sulfonamide administration (Luks et al., 2024).
We identified 29 studies assessing AZ’s efficacy in reducing AMS incidence and severity during rapid ascent to VHA. (Table 2). Although the study designs varied, AZ consistently reduced the incidence and severity of AMS compared with placebo after rapid ascent. One study found a non-significant trend toward lower AMS incidence when AZ was initiated the night before ascent; however, they also found that day-of-ascent initiation lowered average AMS symptom severity scores (Lipman et al., 2019).
AZ was generally well tolerated, with no significant adverse effects in three studies (Bernhard et al., 1998; Ellsworth et al., 1991; McIntosh et al., 2019; Sikri et al., 2019), although these were not powered for adverse events. Among the reported side effects, paresthesia is the most common (35–91% incidence) (Basnyat et al., 2006; Basnyat et al., 2003; Chow et al., 2005), while taste disturbance, polyuria, nausea, fatigue, and mood change have been reported (Ellsworth et al., 1987; Ke et al., 2013; McIntosh and Prescott, 1986; Zell and Goodman, 1988). Most of these were minor, as only 20% of those who experienced paresthesia were likely to miss a dose (Basnyat et al., 2003).
There are conflicting findings on the cognitive effects of AZ, with studies showing no adverse cognitive effects (Ellsworth et al., 1991), while others have found adverse effects on concentration, cognitive processing speed, reaction time, short-term memory, working memory (Wang et al., 2013), and deterioration in psychological and memory testing (White, 1984).
One smaller study found perception of exercise difficulty was higher with AZ than with placebo at 3,459 m (Bradwell et al., 2014) but a more methodologically sound study (Bradbury et al., 2020) showed no significant effect on exercise performance. If AZ affects exercise capacity, the changes are likely to be small and inconsequential for rescue at VHA.
Given the earliest onset of AMS in rapid ascent was within 5 hours (Hackett et al., 1988) and typically requires 6–12 hours for onset (Bartsch and Swenson, 2013), rapid ascent to VHA for durations less than 3 hours, such as in helicopter rescues, presents minimal risk for AMS. However, if the duration at VHA is expected to exceed 3 hours or is uncertain, pharmacological prophylaxis is recommended if supplemental oxygen is not used.
Recommendations
AZ should be used for prophylaxis against AMS in rapid ascent rescues to altitudes between 3,500 and 5,000 m if the rescue is expected to last more than 3 hours and immediate deployment is not required (strong recommendation, high quality evidence).
Start AZ the day before ascent if possible. Day-of-ascent initiation is acceptable and confers a benefit over placebo (strong recommendation, moderate-quality evidence).
AZ should be administered at a dose of 250 mg every 12 hours (strong recommendation, moderate-quality evidence).
After rapid ascent, the time spent at VHA is limited to the minimum required to perform the rescue (strong recommendation, low-quality evidence).
AZ should be discontinued upon descent to the previously acclimatized elevation. If prolonged duration is unavoidable at VHA, AZ should be continued during the rescue for 2–4 days or until descent (strong recommendation, low-quality evidence).
AZ should not be used in rescuers with prior anaphylaxis or Stevens–Johnson syndrome caused by sulfonamides (strong recommendation, low-quality evidence).
DEX prophylaxis for AMS
DEX, a corticosteroid, does not facilitate acclimatization to hypobaric hypoxia but is effective in preventing and treating AMS and HACE. It can be used as an alternative to AZ for individuals at moderate to high risk of AMS (Luks et al., 2024) and because of its quicker onset, it is preferable for immediate deployment situations. Typical dosing for AMS prophylaxis is 2 mg every 6 hours or 4 mg every 12 hours, but higher doses of 4 mg every 6 hours may be beneficial in high-risk situations, such as airlifts to altitudes above 3,500 m with immediate physical activity (Hackett et al., 1988; Luks et al., 2024). If rescuers rapidly ascend to VHA and then immediately descend, DEX can be discontinued upon descent to the previously acclimatized elevation. For longer stays at VHA, DEX should be continued for 2–4 days or until descent (Luks et al., 2024). For use for >7 days, DEX must be tapered to avoid adrenal suppression (Luks et al., 2024).
Seventeen studies evaluated the effects of prophylactic DEX on the incidence or severity of AMS during rapid ascent to VHA (Table 3). The studies consistently showed a statistically significant decrease in AMS incidence and severity compared to placebo within the first 24 hours or in the subsequent 2 days at altitude. Abrupt cessation of DEX can unmask AMS symptoms, which may persist for several days (Rock et al., 1987).
Dosing regimens varied among studies, but only 4 mg every 12 hours showed significant improvement in AMS incidence compared to 1 mg and 0.25 mg every 12 hours (Rock et al., 1989).
The combination of AZ and DEX has shown mixed results. In one study, no difference was observed in AMS incidence nor severity at 3,505 m between patients treated with AZ and those treated with both AZ and DEX; however, exercise capacity was better in the combination group (Sikri et al., 2019). Another study found no difference between AZ + placebo and AZ + DEX for the first 2 days after rapid ascent to 3,989 m, although the AZ + DEX group showed improvement in the subsequent 2 days at 5,334 m (Bernhard et al., 1998). Another study found that AZ + DEX significantly decreased AMS incidence compared to placebo, with no significant difference between DEX monotherapy and placebo (Zell and Goodman, 1988). A small, underpowered study found a statistically significant improvement in AMS incidence with AZ + DEX compared to AZ alone, DEX alone, and placebo (Hussain et al., 2001). Although the evidence favoring concurrent use of AZ and DEX is limited, combination therapy may be beneficial for rapid ascent >5,000 m (Bernhard et al., 1998) or missions with prolonged physical ground work at VHA (Sikri et al., 2019), especially if rapid deployment is required.
Rare but serious side effects like psychosis have been reported with high dose and long-term use of DEX in hospital settings (Janes et al., 2019), but short-term use was generally well tolerated in our review. Four studies found no adverse effects (Basu et al., 2002; Bernhard et al., 1994; Bernhard et al., 1998; Sikri et al., 2019) and another study found that over 50% of subjects reported fatigue, depression, and insomnia after cessation (Zell and Goodman, 1988). There was no difference in multiple tests of cognitive function between DEX, AZ, and placebo groups at altitude (Ellsworth et al., 1991). DEX has been shown to mitigate the hypoxia-related decline in exercise capacity for HAPE susceptible patients after rapid ascent to HA (Fischler et al., 2009; Siebenmann et al., 2011) especially when combined with AZ (Sikri et al., 2019).
Recommendations
DEX 4 mg every 6 hours should be used for AMS prophylaxis in rapid ascent rescues when the duration above 3,500 m is expected to exceed 3 hours, and immediate deployment is required (strong recommendation, moderate-quality evidence).
For rescues above 5,000 m with a duration exceeding 3 hours, or missions with prolonged physical ground work, DEX should be used in conjunction with AZ (Strong recommendation, low-quality evidence).
DEX should be used rather than AZ for AMS prophylaxis in individuals with prior anaphylaxis or Stevens–Johnson syndrome from sulfonamides (strong recommendation, low-quality evidence).
DEX should be tapered slowly rather than abruptly stopped if used for more than 7 days (strong recommendation, high-quality evidence).
For prolonged exposure to VHA, DEX should be used for 2–4 days or until descent to the previously acclimatized elevation (strong recommendation, low-quality evidence).
Nifedipine prophylaxis for HAPE
NIF, a calcium channel blocker, is used to prevent and treat HAPE owing to its ability to dilate the pulmonary vasculature. HAPE is unlikely to occur with short VHA exposure, even for those with a history of HAPE, because it typically takes hours or even days to develop. Rescuers with previous HAPE face a higher risk of recurrence with prolonged exposure from a helicopter malfunction or sudden weather changes. Therefore, we recommend excluding individuals with a history of HAPE from rapid ascent rescue to VHA, especially in situations where prolonged exposure is a possibility. If participation is unavoidable for such individuals, supplemental oxygen should be used, or if that is not possible, NIF should be administered at a dose of 30 mg sustained release (SR) every 12 hours or 20 mg SR every 8 hours. NIF should be continued for 4–7 days if prolonged duration at VHA is necessary, and should be stopped once descent begins (Luks et al., 2024).
Two randomized controlled studies of NIF for HAPE prophylaxis in rapid ascent to HA found statistically significant reductions in HAPE incidence in HAPE-susceptible subjects compared with placebo (Table 4) (Bartsch et al., 1991; George et al., 2023). No study has compared different dosing regimens for NIF.
Recommendations
Rescuers with a history of HAPE should avoid rapid ascent rescues above 3,500 m unless absolutely necessary (strong recommendation, low-quality evidence).
Rescuers with a history of HAPE who must rapidly deploy to altitudes above 3,500 m should use supplemental oxygen, or if unavailable, initiate NIF 30 mg SR every 12 hours or 20 mg SR every 8 hours (strong recommendation, high-quality evidence).
Phosphodiesterase-5 inhibitors prophylaxis for HAPE
PDE-5I, such as sildenafil and tadalafil dilate pulmonary arterioles and blunt hypoxic pulmonary vasoconstriction at HA (Sylvester et al., 2012). Tadalafil 10 mg every 12 hours is recommended for HAPE prophylaxis in patients with a history of HAPE who cannot take NIF (Luks et al., 2024).
Four randomized controlled trials were identified studying PDE-5I for HAPE prophylaxis in subjects with a history of HAPE during rapid ascent to VHA (Table 5). Tadalafil reduced the incidence of HAPE when compared to placebo (George et al., 2023; Maggiorini et al., 2006) and was equivalent to NIF in only one study (George et al., 2023). No study has shown that PDE-5I is superior to NIF for HAPE prevention in this population.
Recommendations
Rescuers with a history of HAPE who must rapidly deploy >3,500 m should only use tadalafil (10 mg every 12 hours) if they are unable to take NIF for HAPE prophylaxis (strong recommendation, moderate quality evidence).
NIF and PDE-5I should not be used concurrently (strong recommendation, high quality evidence).
Scope for further research
Future research should focus on applying pharmacologic prophylaxis and supplemental oxygen to rescuers rapidly ascending to VHA in real-world mountain scenarios. Field studies will provide a better understanding of the actual cognitive and physical performance outcomes, incidence, and severity of HAI for different ascent profiles in both ground and air rescue. Research should include comparative dosing of AZ and DEX alone and in combination, various oxygen flow rates and delivery systems, and evaluate the additive effect of oxygen with pharmacologic agents. Further study is necessary to assess the impact of supplemental oxygen, NIF, and PDE-5I on rescuers with a history of HAPE who rapidly ascend to VHA.
Limitations
The lack of appropriately large randomized controlled trials with well-matched study groups and significant differences in study design, including heterogeneity in AMS assessment scoring, make comparisons between studies difficult. Few studies have specifically investigated the rapid ascent of rescuers to VHA, and data are limited on the distinctions between air and ground transportation. Separate recommendations were made for supplemental oxygen and pharmacologic prophylaxis because no studies have examined them together. Although simulated HA environments have been used in previous research, real rescue environments are typically more chaotic, with numerous variables affecting cognitive and physical functioning. There is a lack of research on the optimal method for administering supplemental oxygen and the potential benefits of monitoring pulse oximetry. Future studies should investigate the delta elevation change and not just absolute altitude, as well as the HAI risk mitigation above 5,000 m.
Conclusions
This scoping review provides recommendations for the use of supplemental oxygen and pharmacological prophylaxis to reduce the risk of HAI and improve cognitive and physical performance in unacclimatized rescuers stationed at low altitudes who are swiftly deployed to VHA. We have adapted these recommendations into an algorithm for rescuers rapidly ascending to VHA (Fig. 2) and have included a recommended survival kit as a contingency plan for unexpected prolonged stays at VHA (Table 7). These recommendations will enhance safety and improve the rescue response while serving as a foundation for international guidelines and organization-specific rescue protocols in VHA environments.
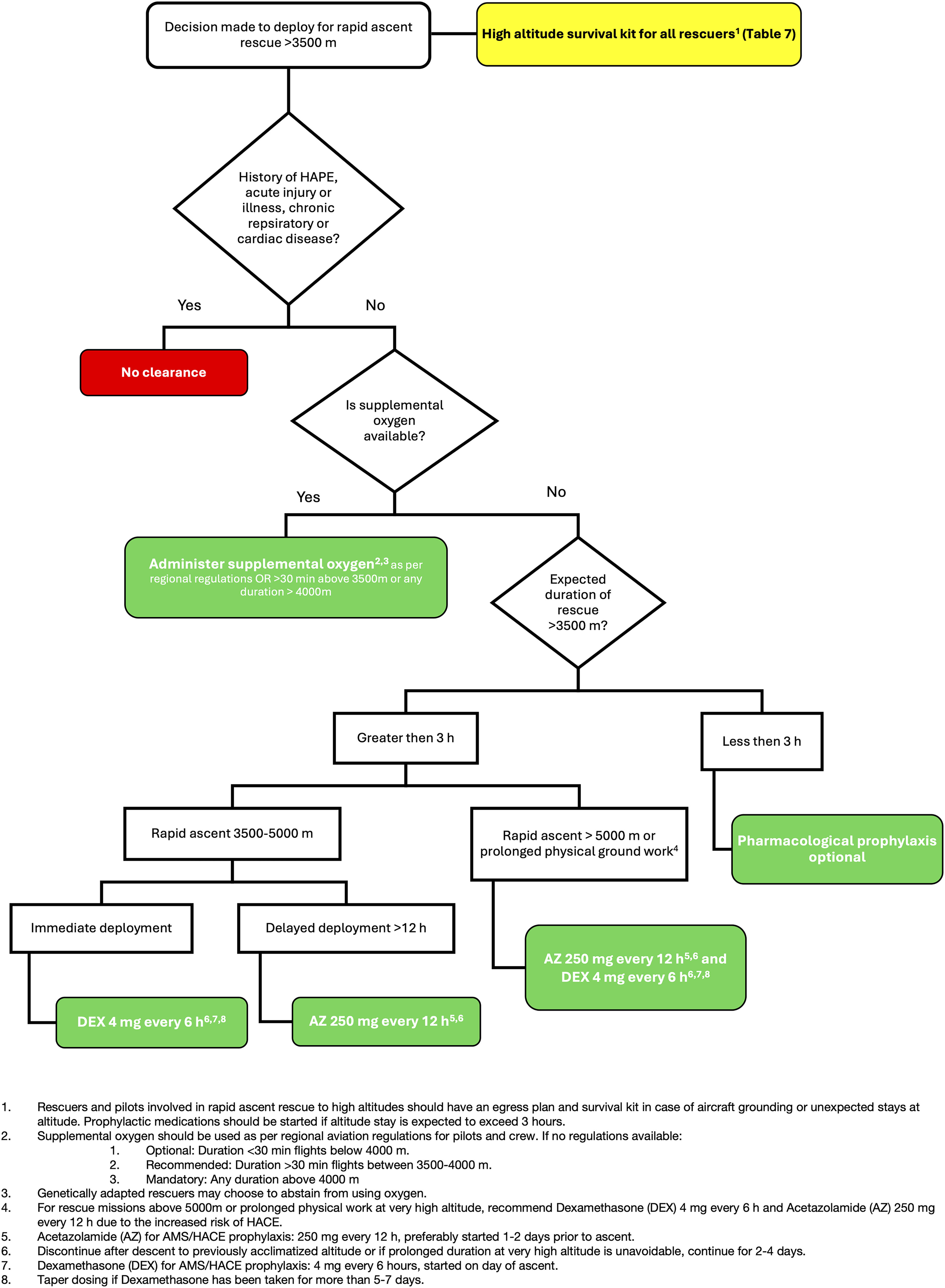
Supplemental oxygen and pharmacologic prophylaxis algorithm for rescuers and pilots engaging in rapid ascent rescue above 3,500 m.
High-Altitude Survival Kit for Rescuers Unable to Descend Following Rapid Ascent >3,500 m

All rescuers and pilots engaging in rapid ascent rescue to very high altitudes should have an egress plan and survival kit in case of aircraft grounding or unexpected stay at altitude. Prophylactic medications should be initiated if the duration at the altitude is expected to be > 3hours.
HACE, high-altitude cerebral edema.
Authors’ Contributions
K.M., S.R., P.P., M.F., and G.S. contributed to the design, implementation of the research, and to the analysis of the results. All the authors contributed to the writing of the article. K.M. and P.P. supervised the project.
Footnotes
Acknowledgment
The authors thank
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The publication as an open-access article was facilitated by funding from ICAR MedCom.
Data Availability Statement
Research data supporting this publication are available in Supplementary Data S1, S2 and ![]() .
.
Supplemental Material
Supplemental Material
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
