Abstract
Objective:
The Bentall procedure is a well-established surgical technique for managing aortic root disease involving the ascending aorta and aortic valve. The use of automated suturing technology may facilitate ergonomic, reliable suture placement, especially in minimally invasive approaches. Here we present the results of a study to evaluate the feasibility of using automated suturing technology for Bentall procedures in an ex vivo porcine model using a passive beating heart simulator.
Methods:
This study included 20 ex vivo porcine hearts, divided into an automated suturing cohort (n = 10) and a manual suturing cohort (n = 10). A Bentall procedure was performed on each heart, with the subject automated suturing technology used in place of manual suturing in the first cohort. After the procedure, each heart was tested in a passive beating heart testing simulator under increasingly challenging hemodynamic conditions (80, 100, and 120 mm Hg); any fluid leakage at the proximal anastomosis was quantified. Data were analyzed using nonparametric statistical tests.
Results:
Overall, leakage from the proximal anastomosis increased with higher pressure and longer duration in both groups (P < 0.001). There was no statistically significant difference in leakage between the automated and manual suture cohorts (P > 0.05), indicating that the study technology appears to be feasible and effective for placing sutures in Bentall procedures. Correlation analysis indicated a moderate positive relationship between aortic pressure and leakage in both groups.
Conclusions:
The subject automated suturing technology demonstrated comparable performance to manual suturing in ex vivo Bentall procedures, with no significantly different leakage across a range of increasing aortic pressures.
Keywords
Central Message
In ex vivo simulated Bentall procedures, the automated suturing technology delivered sutures of similar holding strength to manually placed sutures under elevated pressure conditions. This study demonstrated that this automated technology is feasible for Bentall procedures, warranting further clinical investigation regarding its application.
Introduction
The Bentall procedure is a well-established surgical technique for the management of aortic root disease involving the ascending aorta and aortic valve. It was first described by Bentall and De Bono in 1968. 1 The Bentall procedure typically involves the replacement of the aortic root with either a mechanical or a biological valve conduit or a biological. Earlier approaches such as plication, supracoronary aortic replacement, aneurysm banding, or external support devices were historically used and may still be applicable in selected anatomies that do not meet current guideline thresholds for aortic root replacement. At an aortic root diameter of greater than 5.5 cm, the Bentall procedure is the “gold standard” modality for the management of aortic root disease, as aortic root dilatation with or without aortic valve dysfunction may cause spontaneous dissection and rupture of the aorta.2,3
Despite the broad application, the Bentall procedure is associated with a relatively high rate of reoperation due to bleeding, ranging from 4% to 18.3%, as reported by Zehr et al. and Etz et al.4,5 Moreover, Mataraci et al. highlighted a significant correlation between intraoperative reclamping due to bleeding and higher in-hospital mortality. 6 The introduction of the “button” coronary ostial anastomosis technique has significantly improved outcomes of the Bentall procedure by preventing both early and late coronary distortion or kinking. However, bleeding at the proximal anastomosis site remains a major challenge, particularly when the posterior aortic wall is involved. 7 A possible underlying cause of bleeding is that the sutures between the hard suture ring and the soft and fragile tissue (aortic annulus) may have several small gaps, especially in the case of infective endocarditis or Marfan syndrome. 8
Some cardiac surgeons have adopted the minimally invasive Bentall procedure in the last decade to minimize surgical trauma and reduce complications associated with sternotomy. 9 The RAM® Device (LSI SOLUTIONS®, Victor, NY, USA) for automated suturing can make annular suturing more reliable, with a standardized bite width and depth, and using this technology may reduce suturing times, thereby reducing aortic cross-clamp time and cardiopulmonary bypass duration. Dr Bakhtiary has described his technique for “microinvasive” aortic valve replacement using this automated suturing platform, further supported by the safety, feasibility, and effectiveness of the RAM Device for endoscopic aortic and mitral valve replacement reported by El-Sayed Ahmad et al.10,11 However, a quantitative evaluation of this technology for Bentall procedures, particularly regarding bleeding, has not yet been conducted.
The aim of this study was to evaluate the quality of proximal anastomosis sutures placed using an automated suturing technology (RAM Device) when compared with manually placed sutures during Bentall procedures in an ex vivo porcine model under increasing pressure conditions.
Methods
Heart Preparation
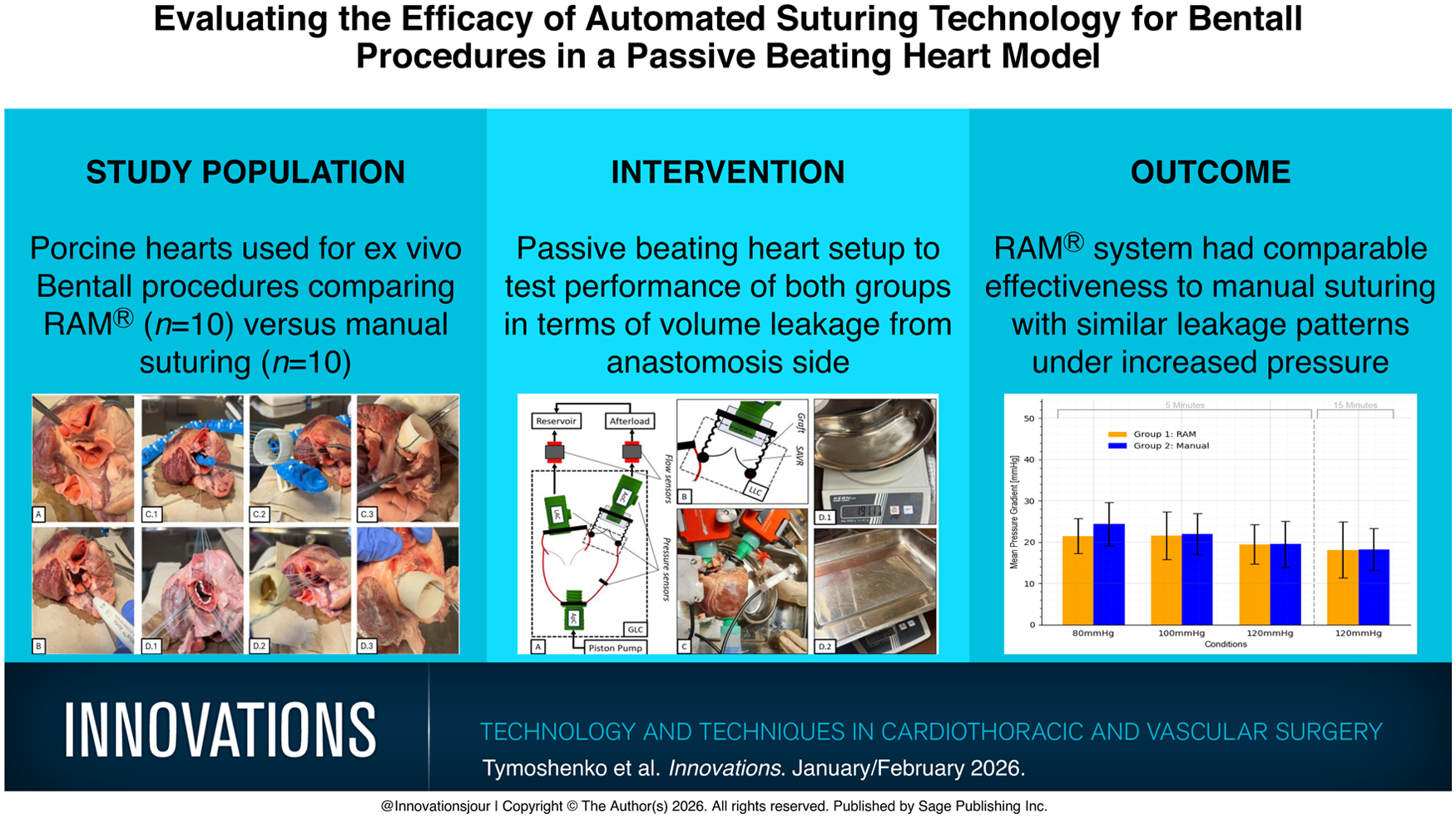
Ex vivo Bentall procedures were performed in 20 porcine hearts. The hearts were divided into an automated suturing cohort (group 1, n = 10) and a control manual suturing cohort (group 2, n = 10). In the automated suturing group, the Bentall was performed using the RAM Device to place sutures; in the manual suturing cohort, all sutures were placed manually using conventional techniques. The porcine hearts were collected from a local abattoir and cleaned from nonrelevant tissue within 12 h from collection. Connective tissue between the aorta and the pulmonary artery was removed to isolate the aortic vessel. After clot removal and inspection of the left ventricle and atrium integrity, the hearts were weighted and finally frozen to −20 °C for 3 days. After defrosting for 12 h, the hearts in both groups were prepared as follows:
The entire aortic arch, including the aortic root, was resected (Fig. 1a).
The coronary buttons were excised and retracted using stay sutures.
The aortic valve leaflets were removed, and the aortic annulus was sized.
A conduit was created from a vascular graft (Terumo Aortic Vascular Grafts size 26, Sunrise, FL, USA) and a bioprosthetic valve (Avalus™ size 19, Medtronic, Dublin, Ireland) using a running 3-0 polypropylene suture.
(a) Heart after aortotomy; (b) valve sizing. (c) Usage of the automated suturing, (d) parachuting of vascular graft, and (e) result of the first group showed. (f) Preparation of manually placed sutures, (g) parachuting of vascular graft, and (h) result of the second group.
Automated Suturing Cohort
Interrupted, noneverted, pledgeted annular mattress sutures were placed using the automated RAM Device, starting at the base of the left cusp, followed by the right coronary cusp and noncoronary cusp. An adjunctive technology, the SEW-EASY® Device (LSI SOLUTIONS), was used to pass the annular sutures through the prosthetic sewing cuff. After suture placement was complete, an automated suture securing technology (COR-KNOT® Device, LSI SOLUTIONS) was used to secure the sutures. Of note, in the automated suturing group, the proximal anastomosis between the graft and the aortic annulus after valve removal (Fig. 1e) was also completed using the second row with automated technology, in the same manner described above.
Manual Suturing Cohort
Similarly, pledgeted annular mattress sutures were placed manually, using conventional techniques, employing a double-armed 2-0 PremiCron® suture (B. Braun, Melsungen, Germany) placed with a shafted needle driver, following the same suturing pattern as in the first group. With a parachute suture, the conduit was implanted. Again, the proximal anastomosis between the graft and the remaining aortic segment (Fig. 1h) was completed using the second row with manual PremiCron sutures.
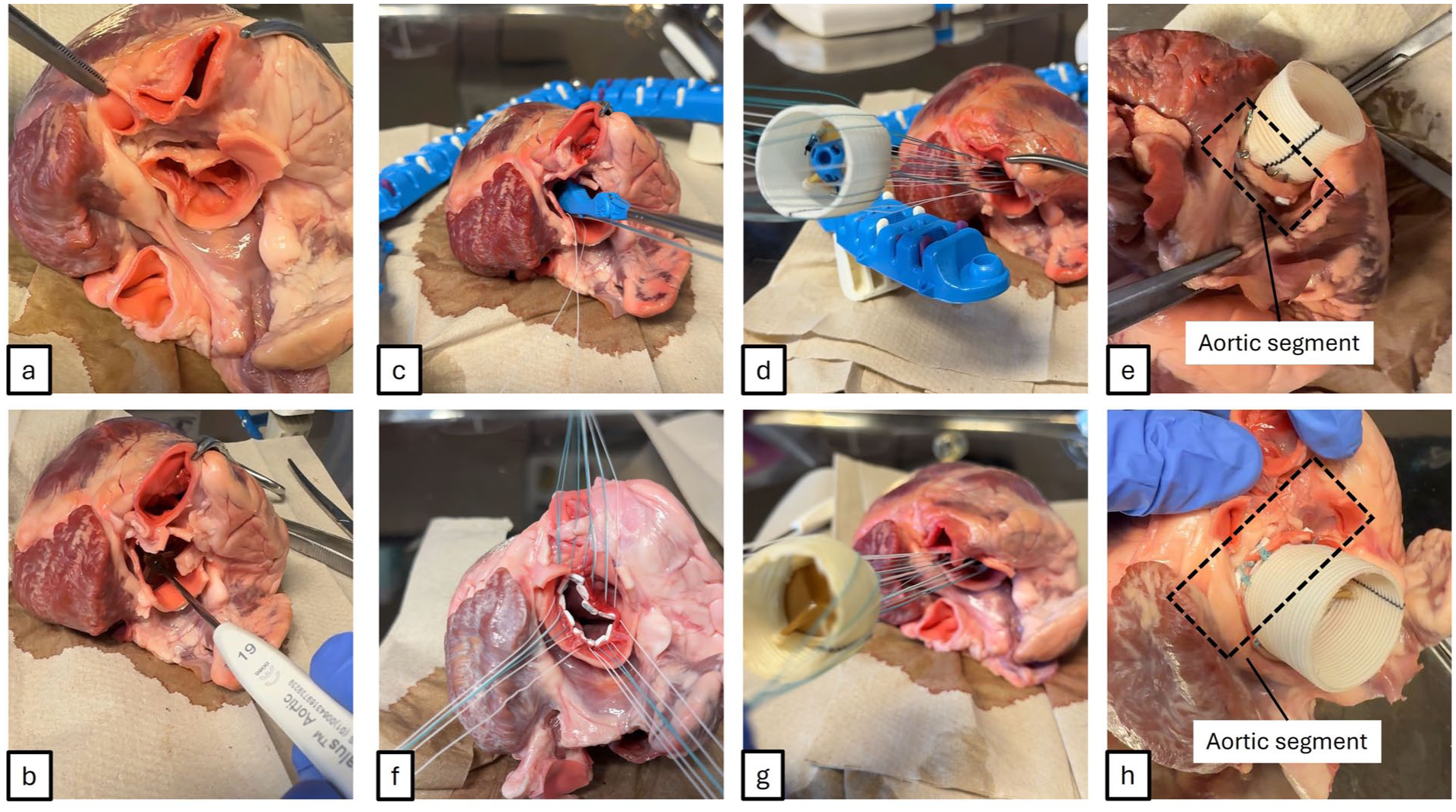
For both groups, after the Bentall procedure was complete, the hearts were prepared to be connected to the passive beating heart test setup. Custom 3-dimensional printed connectors were attached at the distal side of the conduit (aortic connector [AoC]), the left atrium (LAC), and the apex of the left ventricle (ApC), as shown in Figure 2.
(a) General scheme of the test bench used to evaluate the Bentall procedure in passive beating hearts. LAC, AoC, and ApC are the left atrium, aortic, and apical connectors used to connect hydraulically to the heart. (b) Zoom into the anastomosis region where the LLC was used to collect the VL. (c) Photograph of the heart with respective connections, pressure sensors, and flow probes. Leakage measurement for the (d) LLC and (e) GLC. AoC, aortic connector; ApC, apical connector; GLC, general leakage collector; LAC, left atrial connector; LLC, localized leakage collector; VL, volume leakage.
Experimental Setup
A passive beating heart simulator was set up to test the performance of both groups in terms of volume leakage (VL) from the anastomosis side. A localized leakage collector (LLC) was placed just below the proximal anastomosis to collect the VL during experiments. In addition, a general leakage collector (GLC) was also placed under the setup to evaluate the general leakage (GL) due to connector interfaces between the setup and the porcine heart. The AoC, ApC, and LAC were connected to a pulse duplicator (Vivitro Labs, Victoria, BC, Canada), to the afterload, and to the reservoir, respectively (Fig. 2). The linear drive was set to 70 mL/stroke at 70 bpm. The working fluid used was distilled water at 27 °C. Despite the rheological differences with blood (higher viscosity than water), saline solution and distilled water have been established as acceptable working fluids in preclinical passive beating heart studies.12–15 Physiological pressure and flow were recorded using piezo-resistive transducers (Honeywell, Charlotte, NC, USA) and flow probes (Sonotec, Halle, Germany), respectively. Analog signals were recorded with an ADC (National Instruments, Austin, TX, USA) and elaborated using the software LabVIEW (National Instruments).
After a successful connection to the setup, the circuit was primed. The GLC and LLC were used only after deairing and setting the correct actuator conditions. The experiment protocol is described as follows:
Normal condition: mean arterial pressure (MAP) 80 mm Hg, 5 min volume collection
Hypertensive condition: MAP 100 mm Hg, 5 min volume collection
Increased hypertensive condition (IHC): MAP 120 mm Hg, 5 min volume collection
Extended IHC: Additional 10 min volume collection at MAP 120 mm Hg
After each condition, VL and GL were measured using a scale. After each measurement, the GLC and LLC were emptied to continue with the next condition. Care was taken to avoid any leakage before the start of each condition evaluation.
The primary outcome of the study was leakage from proximal anastomosis according to the conditions of perfusion.
Statistical Analysis
Pressure and flow data were processed with a standard sampling frequency of 200 Hz and a low-pass filter (20 Hz). The results are reported using mean and standard deviation. Given the small size of the sample, Shapiro–Wilk was used to test for normal distribution. Because the data were not normally distributed (95% confidence interval), the Mann–Whitney nonparametric test was performed to compare leakage data and pressure data in each testing condition (95% confidence interval). The significance level is reported if P values exceed the standard P value significance of 0.05. Further, a correlation test between average aortic pressure and leakage volume was performed to evaluate a possible dependence between the 2 variables. Due to the non-normality of the data, a Kendall rank test was performed. In addition, linear regression was performed to evaluate the relationship between aortic pressure and anastomosis leakage.
Results
The weight of the test hearts was similar in both groups (447.8 ± 34.8 g in group 1 vs 448.7 ± 44.5 g in group 2, P = 0.976). In all samples, an aortic valve size of 19 and an aortic prosthesis size of 26 were used. The study groups exhibited comparable mean pressure gradients across the various conditions (Fig. 3).
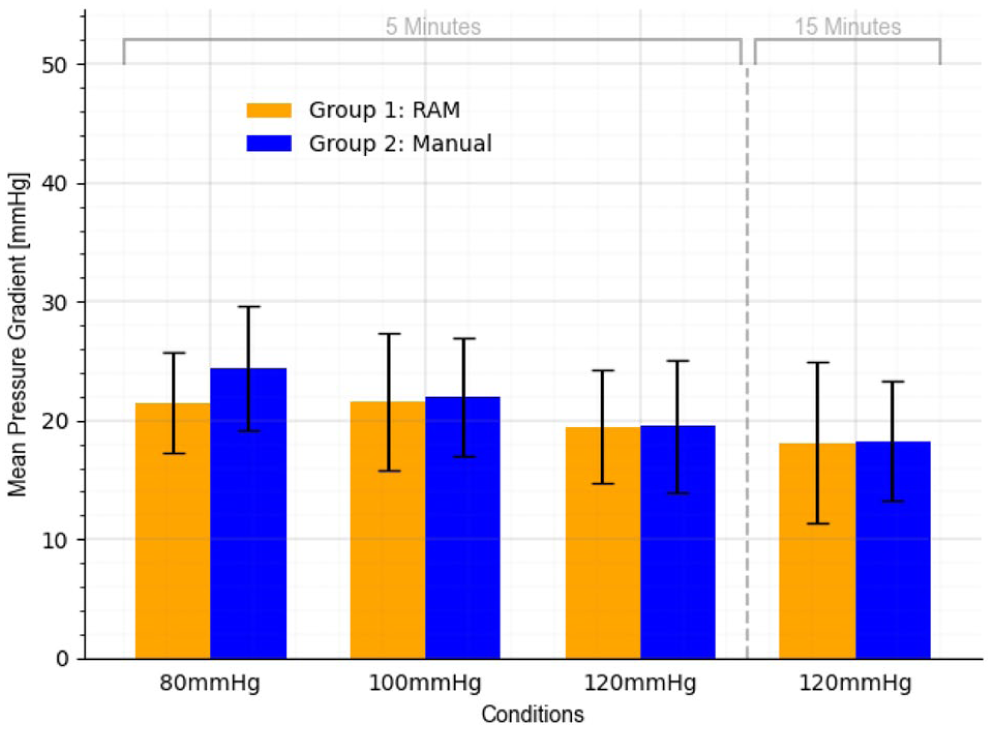
Mean pressure gradient (mm Hg) in both groups at different conditions.
In both groups, the proximal leakage increased with higher pressure and longer duration, as shown in Figure 4. Nevertheless, the study groups were not found to be statistically different according to leakage across different conditions (Table 1).
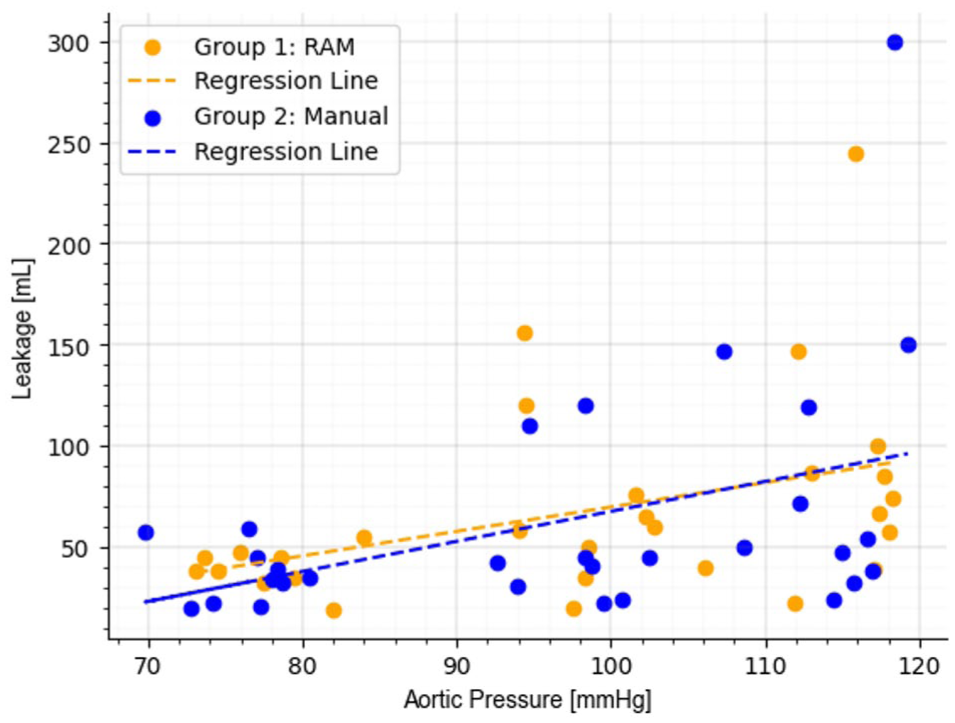
Linear regression analysis between aortic pressure and anastomosis leakage in the study groups. Kendall correlation values were 0.324 in group 1 (RAM) and 0.289 in group 2 (manual).
Leakage Across Different Conditions by Study Group.
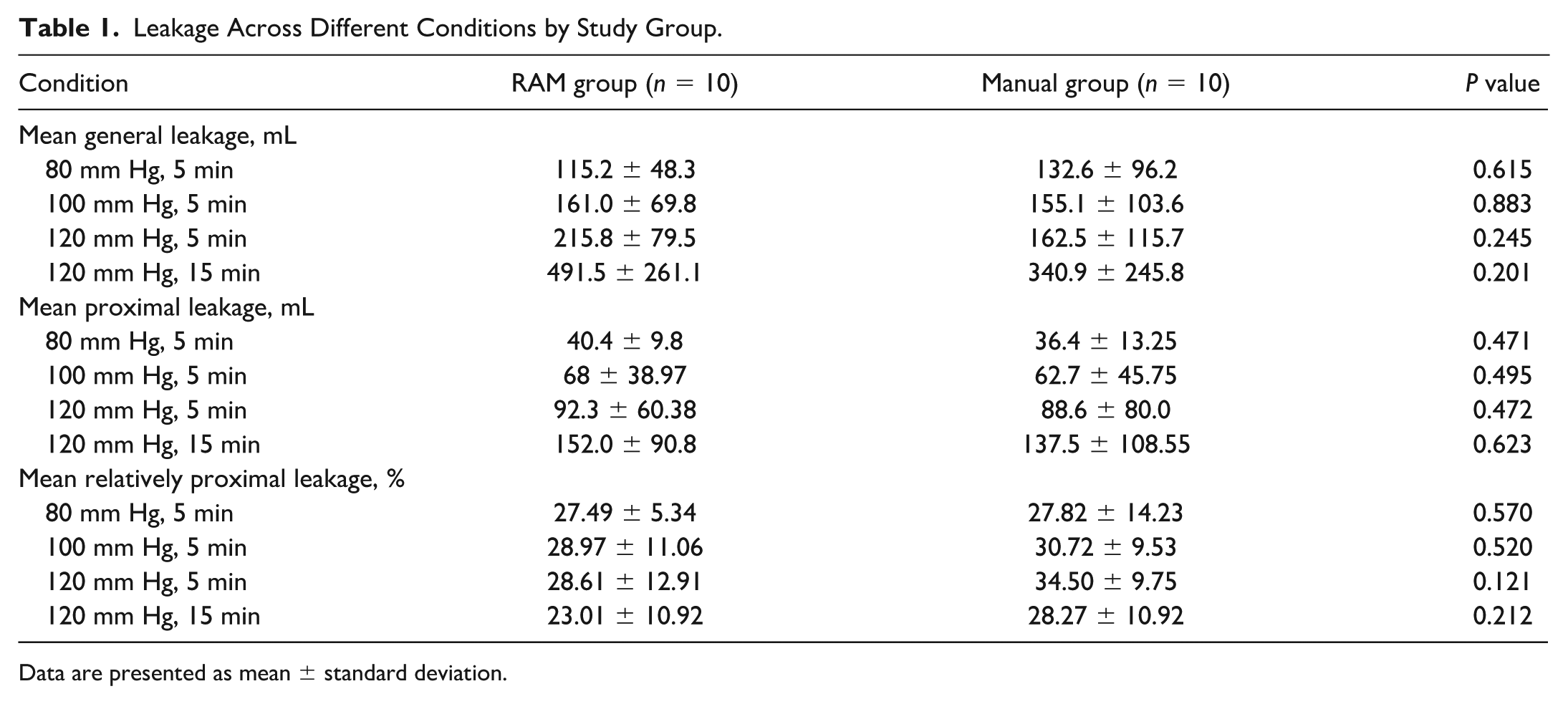
Data are presented as mean ± standard deviation.
GL also increased with higher pressure and longer duration, remaining statistically similar between the groups (Table 1). Moreover, the percentage of relatively proximal leakage (compared with GL) remained consistent between groups across all conditions.
The correlation analysis indicated a moderate positive relationship between aortic pressure and leakage in both groups, with Kendall correlation values of 0.324 in group 1 and 0.289 in group 2 (Fig. 4). Furthermore, linear regression analysis showed that leakage increased by approximately 1.20 mL per 1 mm Hg rise in aortic pressure in group 1 and 1.48 mL per 1 mm Hg in group 2.
Discussion
This ex vivo study quantified and evaluated the feasibility and performance of the RAM suturing device for proximal anastomosis during the Bentall procedure. The results indicate that this automated suturing technology appears to provide hemostatic quality comparable with manual suturing techniques.
Due to the involvement of multiple processes in the Bentall procedure, including aortic valve replacement, coronary anastomosis, and ascending aorta replacement, full sternotomy has become the primary approach for this surgery. 11 Over the past decade, the medical field has increasingly adopted minimally invasive surgical techniques, which offer significant advantages over traditional interventions, such as reduced surgical trauma, shorter recovery times, and improved esthetic outcomes. 16 Numerous studies have begun to introduce minimally invasive techniques for the Bentall procedure.17–20 According to the study by He et al., the Bentall procedure performed through a right anterior minithoracotomy is a safe and effective approach that can be successfully carried out on appropriately selected patients. 18
Nevertheless, the widespread adoption of minimally invasive Bentall surgery is limited by several challenges, including surgeon concerns about the increased technical complexity and the steep learning curve associated with these procedures. 21 In this case, the use of automated suturing devices, such as the RAM Device and adjunctive technology, could offer significant benefits during minimally invasive Bentall surgery.
Our experimental findings indicate that the study technology produces leakage patterns similar to those of the manual suturing technique in ex vivo Bentall procedures. Both groups demonstrated a proportional increase in leakage with rising pressure and extended perfusion duration, which aligns with the expected mechanical response of suture-based anastomoses under hemodynamic stress. Importantly, the leakage detected in this experiment setting was significantly higher than that in the clinical scenario, as distilled water instead of the more viscous blood was used, and no coagulation was present.
Similar leakage between groups suggests that the study technology does not introduce additional leakage risks compared with traditional suturing methods. The slightly lower rate of leakage rise in automated suturing technology may be associated with a more uniform and consistent suture placement, potentially reducing the formation of small gaps between the prosthetic conduit and the native aortic tissue. Although this difference was not statistically significant in this study, it suggests that the study technology may enhance anastomotic integrity in specific clinical scenarios, particularly in challenging tissue conditions such as those seen in Marfan syndrome or infective endocarditis.
Moreover, consistent with research from Zubarevich et al., during the placement of the annular sutures, unnecessary suture tension can be reduced by using automated technology. 22 The automated suturing device can provide deeper and stronger bites than a regular remote manually placed suture, while also preventing needle rotation in the calcified annular plane, if required. 22 The bites placed with this technology are also of standardized width and depth, providing the surgeon with more control during the suturing process.
This study has several limitations. The experimental model used ex vivo porcine hearts, which provide a controlled environment for assessing proximal anastomosis. However, they cannot fully replicate the hemodynamic and biological characteristics of the human aorta. Anatomical differences between the porcine and human aortic roots may influence the results, particularly regarding tissue adaptation to mechanical stress. 23 The study was conducted on a limited sample size (N = 20), which reduces the overall statistical power of the analysis. Although statistical methods were chosen appropriately for the sample size, a larger number of specimens could improve the accuracy of the conclusions. Also, the experimental model did not replicate the physiological processes of hemostasis and tissue remodeling, which are crucial factors in a clinical setting. In real surgical practice, hemostasis, inflammatory response, microvascular blood flow, and fibrosis processes reduce the risk of bleeding and improve anastomotic stability.
Bakhtiary et al. have demonstrated that endoscopic microinvasive aortic valve surgery with automated suturing technology may provide enhanced patient outcomes. 11 In a video presentation contemporaneous with this study, Bakhtiary’s group also highlighted automated suturing technology in a totally endoscopic Bentall procedure. 24 The precise conduit suture placement and excellent patient outcome are consistent with the results of this study, demonstrating this technology’s applicability to a clinical setting. Further studies with larger sample sizes and clinical trials are necessary to confirm the efficacy and safety of this approach.
Conclusions
Our experimental study demonstrated comparable performance of an automated suturing technology to the manual technique in ex vivo Bentall procedures regarding leakage patterns under challenging pressurized conditions. The study technology was demonstrated to be feasible for Bentall procedures in this model, and encouraging results indicate that further investigations in a clinical setting are warranted.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.A. is proctor/consultant/speaker (Edwards, Abbott, Medtronic, Boston, BBraun, and Zoll) and received institutional research grants (Edwards, Abbott, Medtronic, and LSI SOLUTIONS). All other authors declare no conflicts of interest in relation to this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Austrian Federal Ministry of Labour and Economy, the National Foundation for Research, Technology, and Development, and the Christian Doppler Research Association funded part of the research.
