Abstract
Objective:
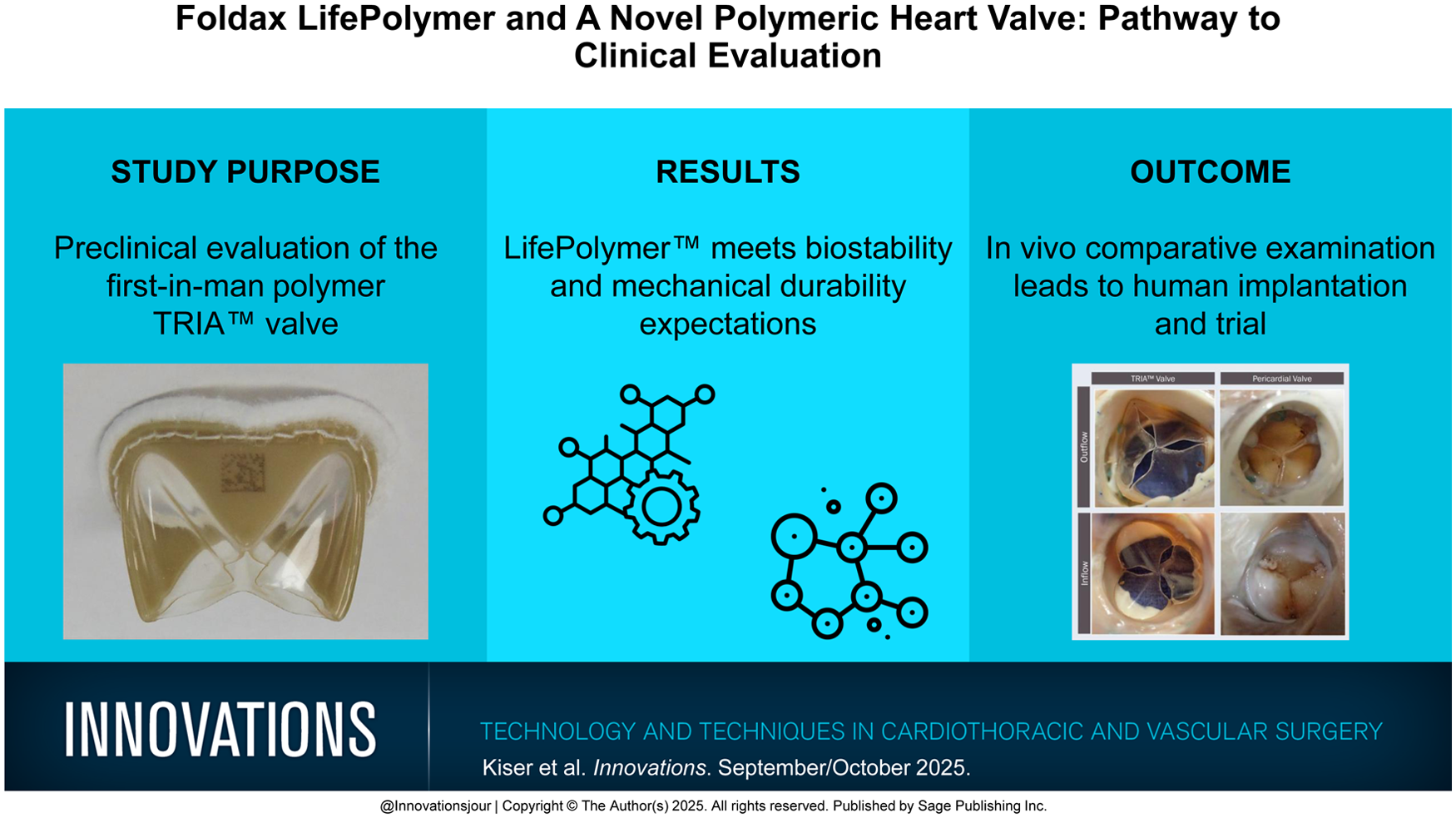
An optimal valve replacement prosthesis demands durable leaflet technology, superior hemodynamic performance, and ease of use. Preclinical evaluation of polymer leaflets has historically demonstrated mechanical failure related to biodegradation. We present the preclinical evaluation of the novel TRIA™ polymer valve (Foldax, Salt Lake City, UT, USA) and a case report of TRIA mitral valve replacement.
Methods:
A uniquely formulated, biostable, and biocompatible polymer (LifePolymer™ [LP], Foldax) has been designed to meet the functional demands of cardiac hemodynamics. Preclinical in vitro evaluation included biocompatibility testing, thrombogenicity testing, and toxicologic assessment followed by evaluation in the arteriovenous shunt of nonhuman primates and in the aortic position in sheep. Clinical evaluation of early human aortic and mitral implantation included computed tomography imaging and echocardiographic examination.
Results:
In vitro studies of LP demonstrated no evidence of toxicity or tissue injury, no cytological injury in cell culture, and no intracutaneous sensitization. LP proved to be nonhemolytic by direct and extract methods, and complement activation was insignificant. Genotoxicity analysis proved LP to be nonmutagenic. All standard toxicologic assessments were within the margin of safety. Biostability was confirmed without polymer degradation or excessive comparative thrombogenicity. Ovine 6-month aortic valve explantation showed no leaflet calcification and minimal fibrinous depositions. An early human case example shows no evidence of leaflet thrombus formation at 6 months and a mean mitral gradient of 3 mm Hg at 12 months.
Conclusions:
LP has met the requirements for a prosthetic polymer human heart valve. The surgical TRIA Mitral Valve has demonstrated promising early human clinical success, potentially facilitating a lifetime valve replacement strategy.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
