Abstract
Objective:
To assess the midterm outcomes and feasibility of using 3-dimensional total endoscopic surgery (3D TES) for complete correction of partial atrioventricular septal defect (PAVSD) in children.
Methods:
A descriptive study from January 2018 to June 2024 involved 20 patients with PAVSD who underwent surgical repair using 3D TES. The average patient age and average weight were 9.1 (range: 4 to 15) years and 25.02 kg, respectively. Mitral valve regurgitation was classified as mild in 2 patients, moderate in 12, and severe in 6, whereas tricuspid valve regurgitation was mild in 13 patients and moderate in 7.
Results:
Peripheral circulation was established in all patients. Cardiopulmonary bypass and aortic cross-clamp times were 86.2 min and 142.2 min, respectively. The mechanical ventilation time averaged 4.9 h. Postoperative echocardiography included complete closure of the atrial septal defect, with mild mitral valve regurgitation in 16 patients and no regurgitation in 4. Tricuspid valve regurgitation was mild in 11 patients, and 9 patients had no regurgitation. During an average postoperative follow-up period of 3.35 years, there were no mortalities or cases requiring reoperation.
Conclusions:
The 3D TES demonstrates feasibility, safety, and efficacy in treating PAVSD in children, with apparent aesthetic advantages.
Central Message
This study of 20 patients found that 3-dimensional total endoscopic cardiac surgery presents a safe and viable alternative for treating partial atrioventricular septal defects in children, offering promising initial outcomes and evident cosmetic benefits.
Introduction
Partial atrioventricular septal defect constitutes approximately 3% of all congenital heart conditions. 1 Typically, patients receive diagnosis and treatment around the age of 1 year, with most undergoing surgery involving a traditional sternotomy approach.2,3 However, in recent years, significant shifts have been witnessed in treatment methodologies, bringing not only favorable outcomes but also notable advantages such as avoiding sternotomy-related complications, minimizing bleeding risks, and reducing sternitis and sternum deformities. 4 These advancements hold particular esthetic significance, especially for female patients. These evolving techniques encompass minimally invasive procedures, including right thoracotomy or axillary incisions, notably total endoscopic surgery. 5 Although several studies have explored endoscopic interventions in treating this condition, most have focused on adult patients. 3 In this article, we present a case series involving pediatric patients afflicted with partial atrioventricular septal defect who underwent treatment via 3-dimensional (3D) total endoscopic surgery.
Methods
From January 2018 to June 2024, 20 patients diagnosed with partial atrioventricular septal defect (9 male patients, 11 female patients) underwent 3D total endoscopic surgery (Fig. 1). Exclusion criteria encompassed cases of complete or intermediate atrioventricular septal defect, inability to administer peripheral extracorporeal circulation, and previous right thoracic surgeries, previous sternotomy, or congenital pectus excavatum. This descriptive study outlines preoperative indicators, intraoperative procedures, and postoperative outcomes.
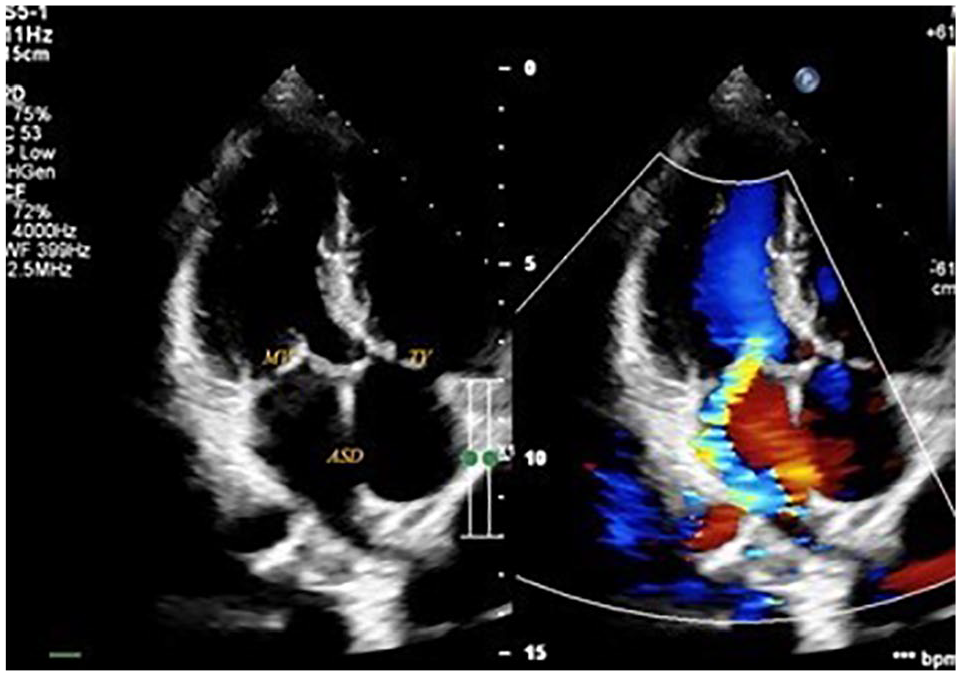
Preoperative echocardiography. ASD, atrial septal defect; MV, moderate mitral valve regurgitation; TV, mild tricuspid valve regurgitation.
Operative Technique
Under endotracheal anesthesia, patients underwent transesophageal echocardiogram setup. Positioned in a 30° left lateral decubitus position, patients were prepared with a 3D endoscopic screen on the left side, while the surgeon operated from the right (Fig. 2a). Cardiopulmonary bypass commenced via femoral arterial cannulation indirectly through the 8 mm Unigraft-Dacron (B. Braun, Melsungen, Germany), with percutaneous jugular and femoral venous cannulation according to the Seldinger method and diameter ranging from 15 Fr to 21 Fr (Medtronic, Dublin, Ireland). We placed the 12 mm trocar (working port) in the sixth intercostal space in the right midclavicular line, the 10 mm trocar (3D camera) in the fifth intercostal space in the midaxillary line, and the 5 mm trocar (instrument port) in the third intercostal space in the anterior axillary line (Fig. 2b).
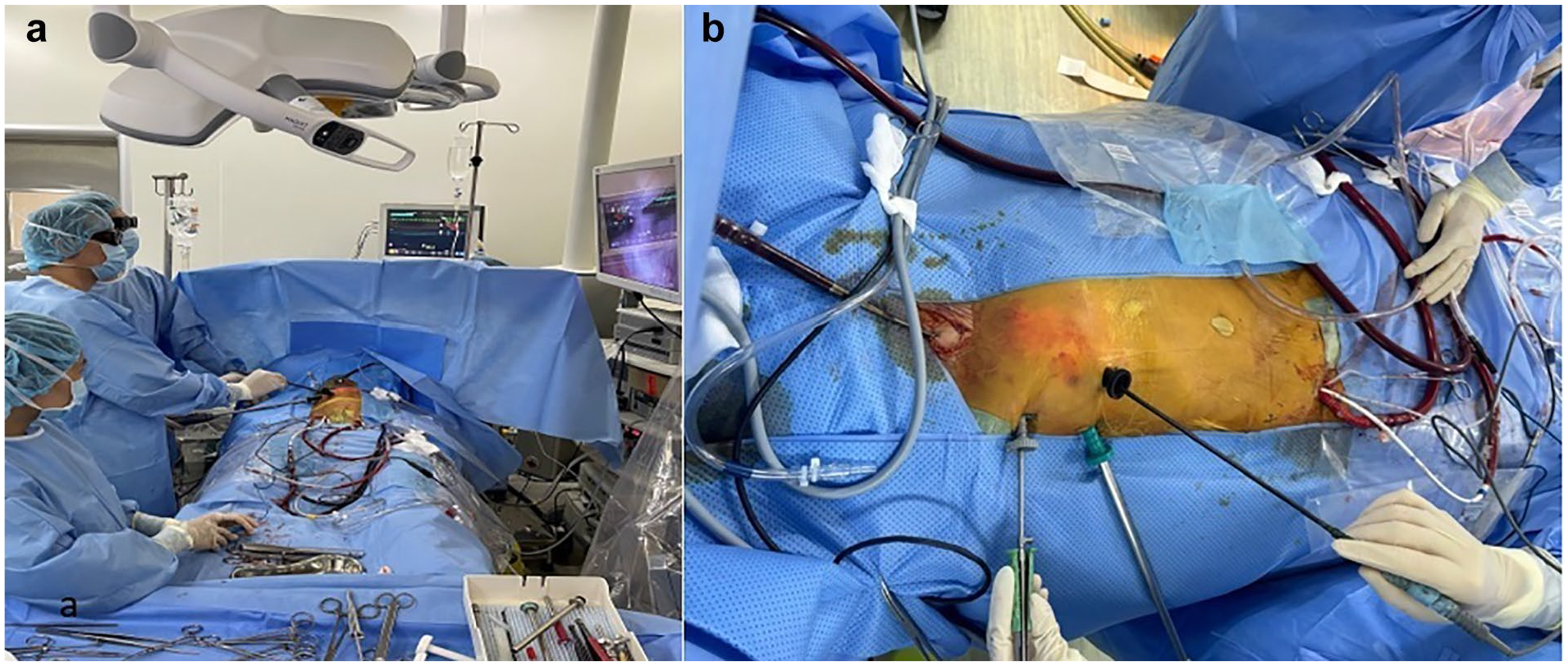
Patient’s position. (a) Three-dimensional endoscopic screen on the patient’s left side. (b) Site of trocar and peripheral cardiopulmonary bypass.
We started cardiopulmonary bypass, stopped the ventilator, and opened the pericardium 2 cm anterior to the phrenic nerve. The aorta was cross-clamped by a Chitwood clamp (Scanlan International, Inc, St Paul, MN, USA), which was inserted in the fourth intercostal space at the midaxillary line, and then antegrade Custodiol cardioplegia (Dr. Franz Köhler Chemie GmbH, Bensheim, Germany) was administered. Right atriotomy was completed. Four stay sutures were placed to secure the edges of the atriotomy to the pericardial edges, exposing the atrial septal defect, mitral valve, and tricuspid valve. The cleft was closed using interrupted 5.0 Prolene sutures from the annulus to the leaflet edge of mitral valve (Fig. 3a). Saline solution was injected into the left ventricle to verify valve competency (Fig. 3b) and mark the boundary to close the atrial septal defect with the XenoSure patch (LeMaitre Vascular, Inc, Burlington, MA, USA; Fig. 3c). We repaired the tricuspid valve by suturing the commissure of the septal and posterior leaflets (Fig. 3d). Then we closed the right atriotomy, evacuated the air, released the aortic cross-clamp, placed electrodes, inserted a pleural drain, closed the pericardial, stopped the cardiopulmonary bypass, removed the cannulas, and neutralized the heparin.
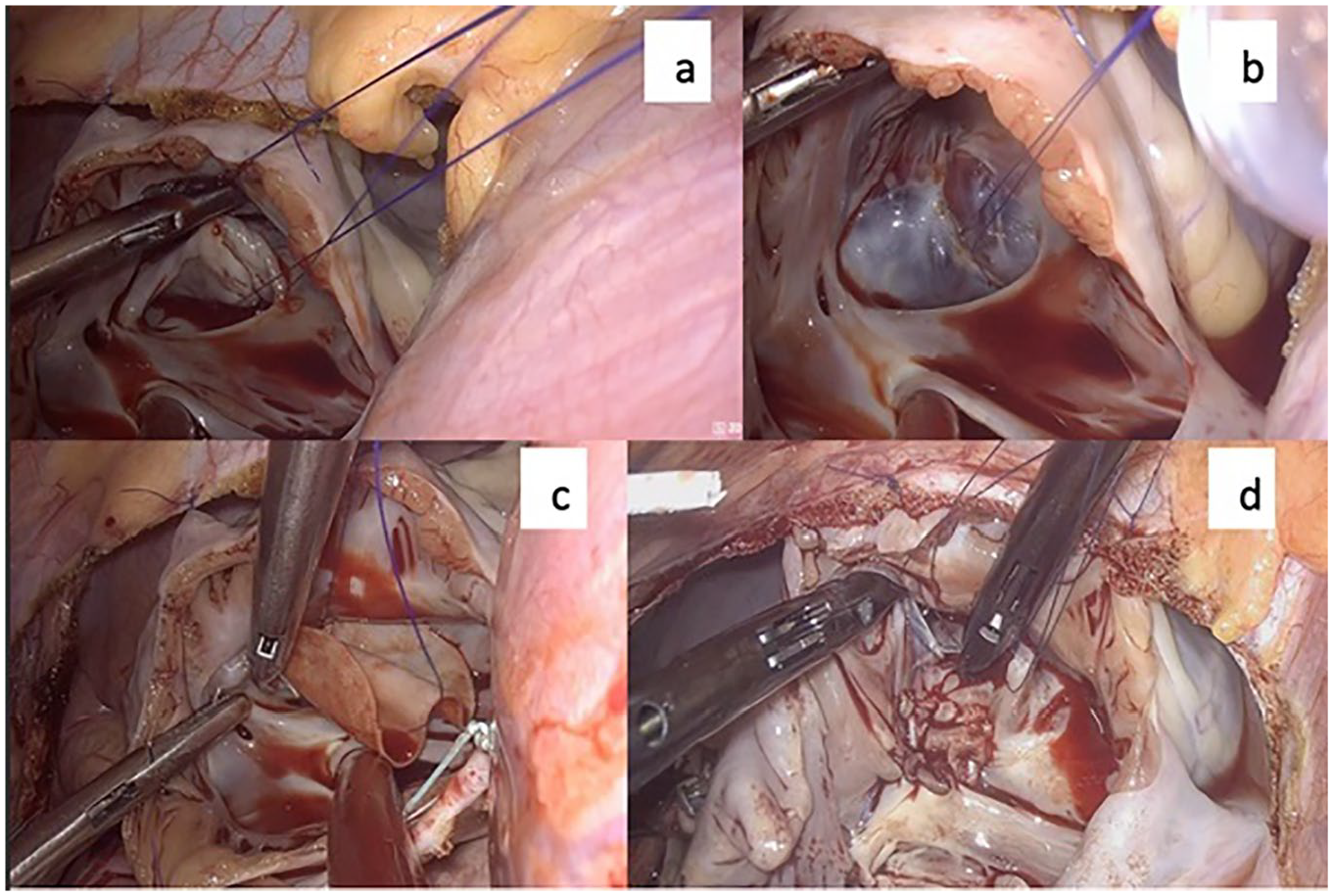
Repairing all defects. (a) Closing mitral valve cleft; (b) testing mitral valve; (c) atrial septal defect patch closure; (d) tricuspid valve repair.
Results
Through studying 20 patients, we have the following results. Preoperative characteristics are reported in Table 1. The average patient age and average weight were 9.1 years (range: 4 to 15 years) and 25.02 kg (range: 15 to 40 kg), respectively. All patients showed signs of heart failure at Ross classification level II or III. Preoperative echocardiogram described ostium premium type of atrial septal defect and mean pulmonary artery pressure of 44 mm Hg (range: 30 to 60 mm Hg). Mitral valve regurgitation was severe in 6 patients (30%; Fig. 1).
Preoperative Characteristics.
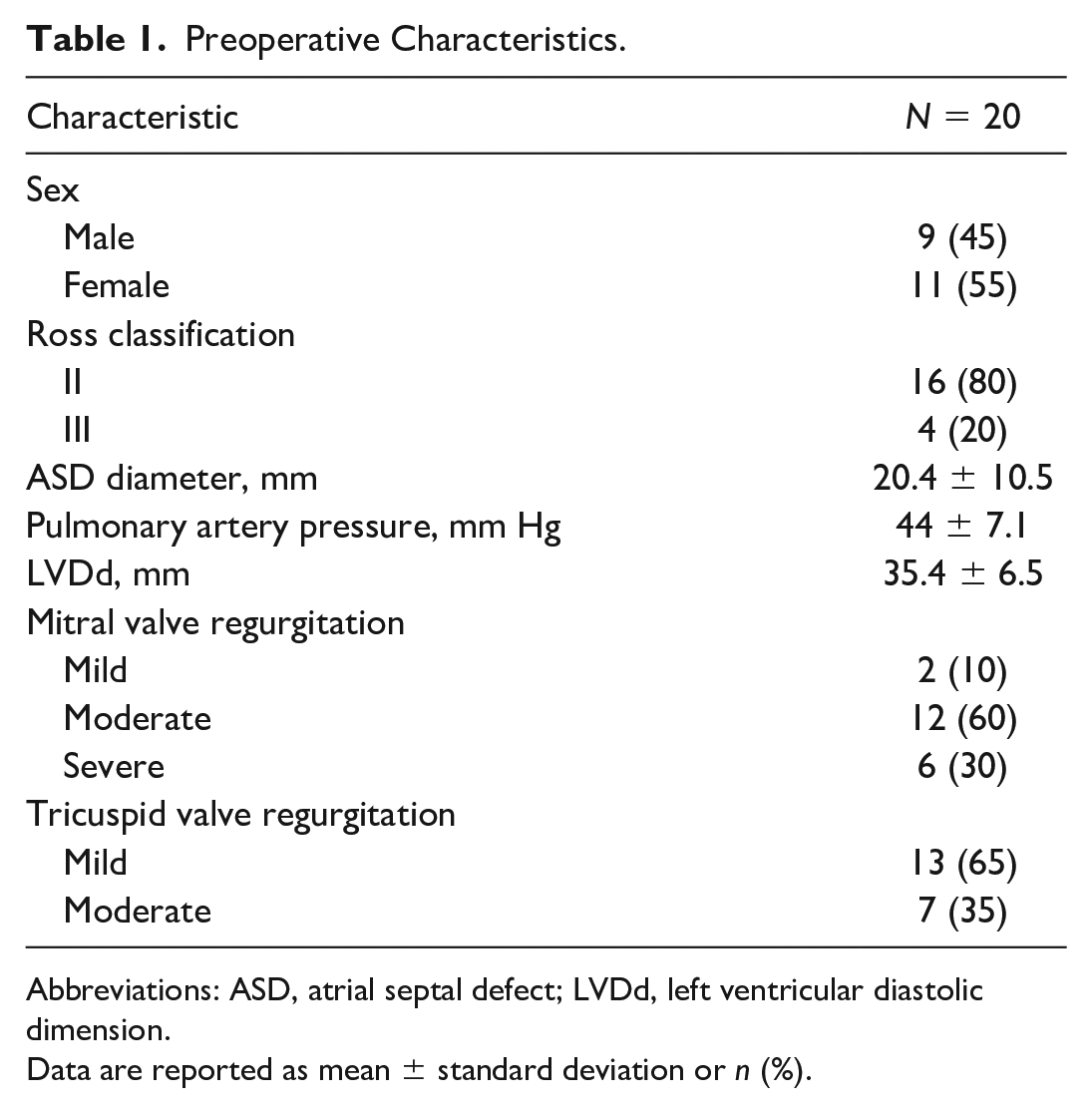
Abbreviations: ASD, atrial septal defect; LVDd, left ventricular diastolic dimension.
Data are reported as mean ± standard deviation or n (%).
Perioperative and postoperative results are reported in Table 2 and Table 3. All surgeries were performed successfully with no cases requiring sternotomy. Surgical techniques included repairing the mitral valve (suturing the cleft anterior leaflet of the mitral valve), patching the atrial septal defect, and repairing the tricuspid valve (suturing the commissure of septal and posterior leaflets). The average postoperative follow-up time was 3.35 ± 2.45 years (range: 1 to 8 years). The postoperative follow-up results are shown in Table 4.
Perioperative and Postoperative Characteristics.
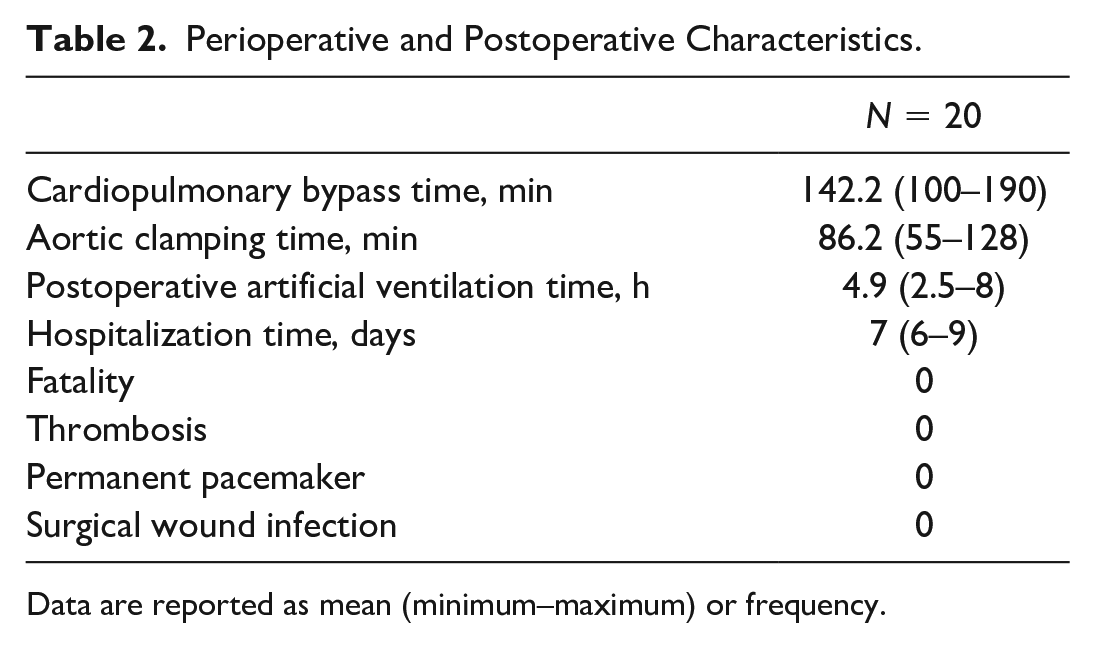
Data are reported as mean (minimum–maximum) or frequency.
Postoperative Echocardiography.
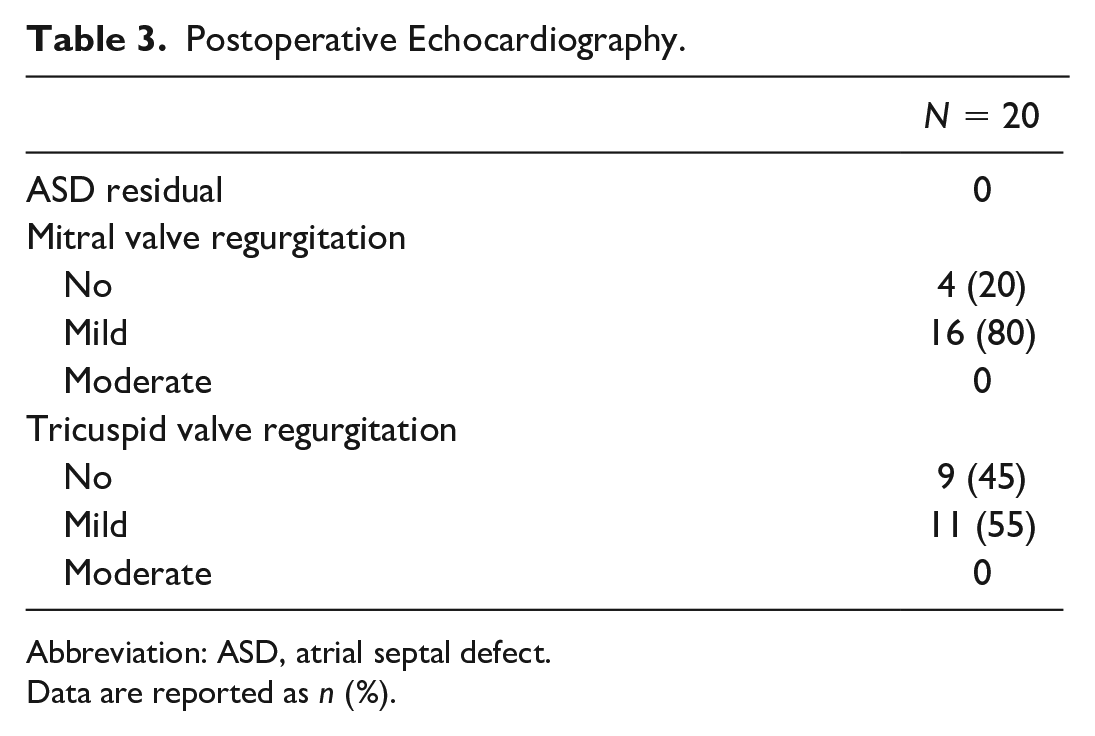
Abbreviation: ASD, atrial septal defect.
Data are reported as n (%).
Postoperative Follow-Up.
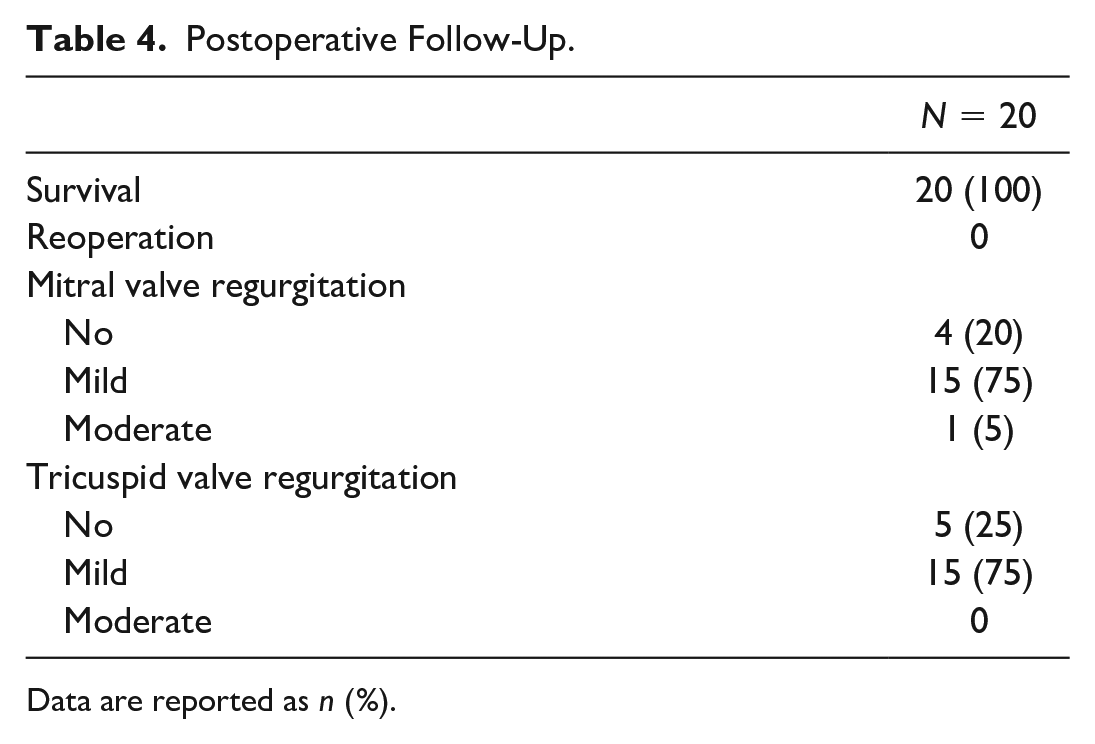
Data are reported as n (%).
Discussion
Partial atrioventricular septal defect represents a prevalent congenital heart disease, typically necessitating surgical intervention around the age of 1 year, contingent upon factors such as the severity of left atrioventricular valve regurgitation, diameter of the primary atrial septal defect, and presence of pulmonary hypertension. 6 Comprehensive surgical correction involves repairing the mitral valve by closing the anterior cleft, closing the ostium premium type of atrial septal defect with an artificial patch, and addressing any issues with the tricuspid valve. 7 Traditionally, these procedures are conducted via a sternotomy, yielding favorable outcomes during long-term follow-up.6,8
In recent times, the adoption of minimally invasive approaches for congenital heart conditions, including partial sternotomy, right thoracotomy, assisted endoscopy, and total endoscopy, with or without robotic assistance, has gained popularity. These advancements have shown promise in enhancing patient recovery. Notably, for children with partial atrioventricular septal defects, the recent literature predominantly discusses approaches via right thoracotomy or right axillary incisions.9–12 At our hospital, minimally invasive surgery is indicated for all patients with partial atrioventricular septal defect. Complete endoscopic surgery is indicated when the patient weighs more than 15 kg and has a common femoral artery diameter on Doppler ultrasound measured more than 5 mm (peripheral circulation can be established). Reports on the use of 3D total endoscopic surgery in pediatric cases are scarce.
For established peripheral circulation, we choose to place the arterial cannula indirectly through the artificial graft to avoid the risk of femoral artery stenosis at the cannula placement site as well as to eliminate the risk of arteriospasm when placing the cannula directly into the lumen, causing increased pressure when functioning the artificial heart-lung machine. In addition, it avoids the risk of damaging the external iliac artery at the tip of the arterial cannula. When placing an indirect arterial cannula through an artificial graft, it also helps to irrigate the lower limb during surgery, preventing the lower limb from ischemia that affects limb function. A possible risk when placing an indirect arterial cannula through an artificial graft is bleeding at the vascular anastomosis site, so when performing the anastomosis, it is necessary to use 6.0 or 7.0 vascular sutures, continuous stitching and paying attention to suturing the entire layer of the artery. If bleeding occurs through the anastomosis but does not affect the cardiopulmonary bypass operation, use a small suction tube to suck the blood back to the extracorporeal circulation machine.
Endoscopic cardiac surgery encompass 2-dimensional (2D) and 3D endoscopy, along with robotic techniques. 13 The 3D systems and robots offer distinct advantages over 2D systems, providing surgeons with a 3D perspective, enhanced depth perception, and detailed anatomical visualization, thereby improving hand-eye coordination.14–16 Although robotic surgery offers similar advantages, it requires extensive training and incurs high costs, posing challenges for implementation in resource-limited settings. 15 The cardiopulmonary bypass time and the aortic cross-clamping time with 3D endoscopy are comparable to those of open surgery and thoracotomy. Evaluation of long-term outcomes following complete surgical repair often hinges on the condition of the mitral and tricuspid valves. The 3D endoscopy facilitates precise visualization of these valve chordae tendineae and suture locations, leading to optimal valve repair outcomes. 17 During our follow-up, none of the patients experienced moderate or severe valve regurgitation necessitating reoperation (Fig. 4).
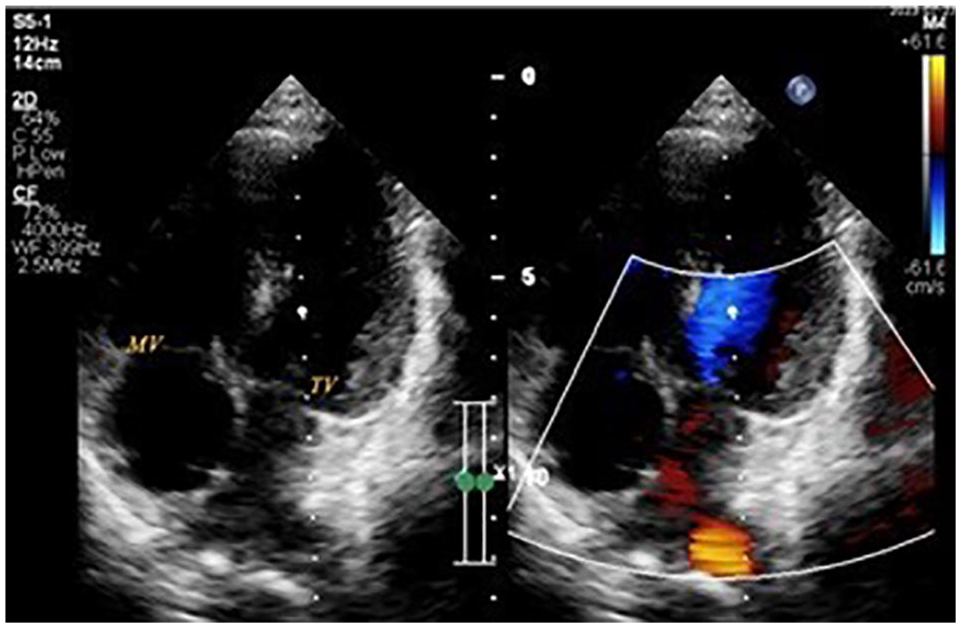
One-year postoperative echocardiography. MV, no mitral valve regurgitation; TV, mild tricuspid valve regurgitation.
Esthetic enhancement emerges as a notable benefit of total endoscopic surgery. By not having to saw the sternum, patients avoid lengthy surgical scars and the risk of chest deformities associated with the bone-healing process. Conversely, minimally invasive procedures via thoracotomy may pose risks of chest deformity and affect the development of the right mammary gland, particularly in female patients, due to potential damage to the vessels supplying the breast. 10 In addition, patients may still bear a visible scar on the chest wall. Total endoscopic surgery mitigates these drawbacks by using small trocar holes (up to 12 mm) that minimally affect mammary gland development and chest wall musculature. Moreover, the resulting surgical scar is inconspicuous, often hidden beneath the inframammary crease (Fig. 5).
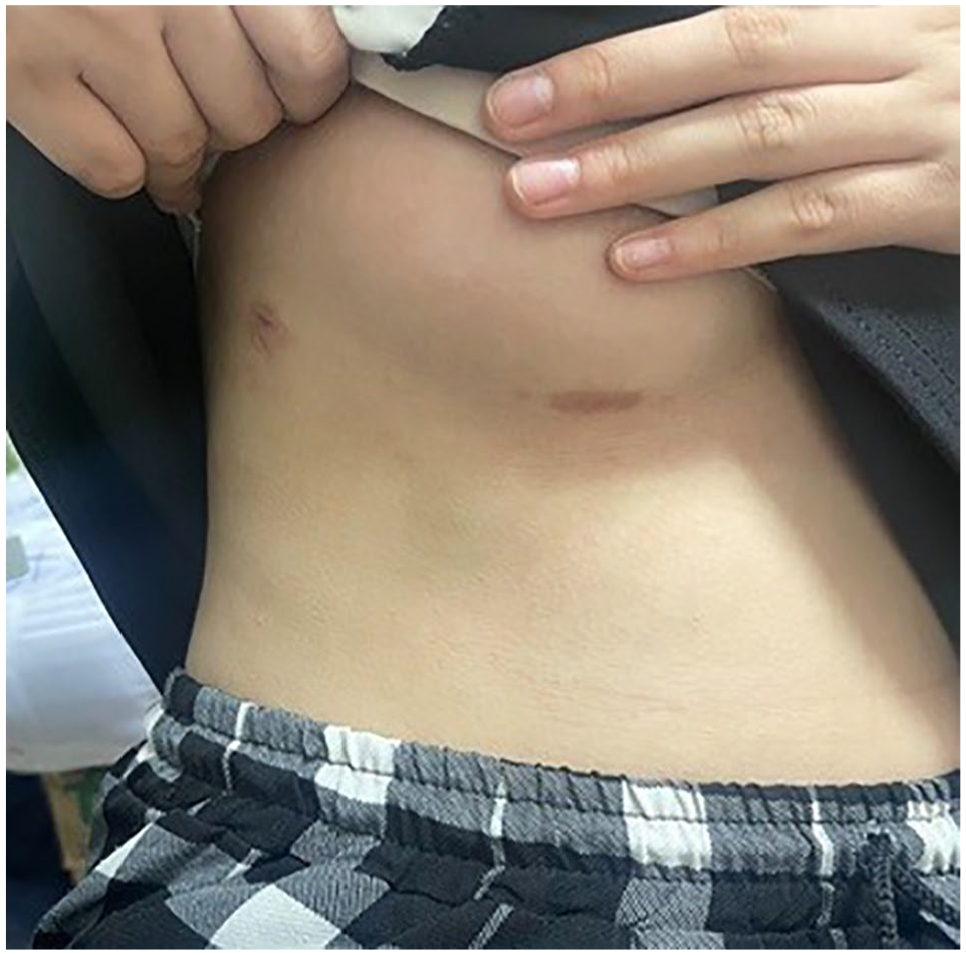
Scars 4 years after operation.
Conclusions
The 3D total endoscopic cardiac surgery technique presents a safe and viable alternative for treating partial atrioventricular septal defects in children, offering promising initial outcomes and evident cosmetic benefits.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
