Abstract
In the evolving landscape of endoscopic aortic valve replacement (AVR), the alignment of instruments is crucial for overcoming difficulties related to the narrow aortic space. Here, we describe a technique of lateral suturing based on the principle of instrument triangulation, which allows perpendicular stiches to the aortic annulus like an open aortic valve procedure. This technique facilitates endoscopic AVR, performed through a 3 cm long right anterolateral mini-thoracotomy without rib retraction using a 3-dimensional endoscope and femoro-femoral cardiopulmonary bypass. Our approach uses a more lateral positioning of the vent and the transthoracic clamp as well as the addition of a single lateral port, which allows optimal angulation of the instruments to the aortic annulus and the ascending aorta. This technique facilitates endoscopic suturing without the use of automated devices through a transverse aortotomy. Annular sutures are placed in a perpendicular way in the aortic annulus by avoiding challenging “hook” stitches. In this case, we use an additional 10 mm port in the fourth right intercostal space through a small periareolar incision. With this technique, it is possible to implant any type of aortic prosthesis, either biological or mechanical.
Keywords
Central Message
We describe the use of an additional single lateral port made over the fourth intercostal space to facilitate endoscopic aortic valve replacement, which allows for implant of any aortic prosthesis type and additional procedures.
Introduction
Totally endoscopic aortic valve replacement (TEAVR) is an evolving technique for the surgical treatment of aortic valve disease. It is facilitated by 3-dimensional (3D) endoscopy using either conventional long-shafted or robotic instruments. Both techniques are based on the principle of transthoracic endoscopic manipulations performed without direct vision through the thoracotomy. A 3D camera is used either in a static way stabilized on a holder or by using a dynamic robotic commandment. 1
Recent single-center retrospective reports demonstrated that TEAVR is a safe and feasible technique for preserving the integrity of the sternum and is associated with less trauma and pain and quicker recovery compared with traditional full sternotomy. 2 Other authors could demonstrate shorter hospital and intensive care stays, mainly due to the minimized surgical trauma. Nevertheless, and as expected for most minimally invasive cardiac procedures, operative times are longer and the learning curve is steep. 3
The anatomical suitability for TEAVR should be determined by preoperative imaging, especially computed tomography (CT). Favorable anatomy includes an aortic root position placed on the right side or centrally behind the sternum, a distance of less than 10 cm between the aortic root and the chest wall at the right second intercostal space, and the absence of calcifications of the ascending aorta and the aortic root. The absence of peripheral vascular disease, severe atherosclerosis of the descending or the abdominal aorta, and other contraindications for peripheral cannulation are important prerequisites for the safe performance of TEAVR. 4
Due to the anterolateral position of the main incision, conventional placement of the aortic annular sutures is cumbersome. The 3D view is commonly obstructed by the needle holder, which uses the same trajectory as the camera to bring the sutures and place the stitches at the annular plane (Supplemental Video 1). To avoid this, Bakhtiary et al. have described the use of automated devices for suture placement, which allow a more perpendicular suturing trajectory to the aortic annulus. Such devices carry 2 needles and allow suture placement by minimizing the number of sutures needed to half of the ones needed by the conventional technique.5,6 On the other hand, the devices are still bulky, obstructing the view of the aortic annulus—a structure that is narrow per se—thus jeopardizing the reproducibility of the procedure, and this requires a learning curve that is quite steep.
In this article, we describe an alternative approach for suturing maneuvers using a typical triangulated endoscopic principle that allows excellent visualization of each and every stitch and perpendicular suturing of the aortic annulus and the ascending aorta.
Surgical Technique
The patient is placed in a 30° left supine position, and the right arm is slightly abducted to expose the right anterolateral chest wall. After establishment of general anesthesia and single-lumen endotracheal intubation, the cardiopulmonary circuit is connected through the groin vessels.
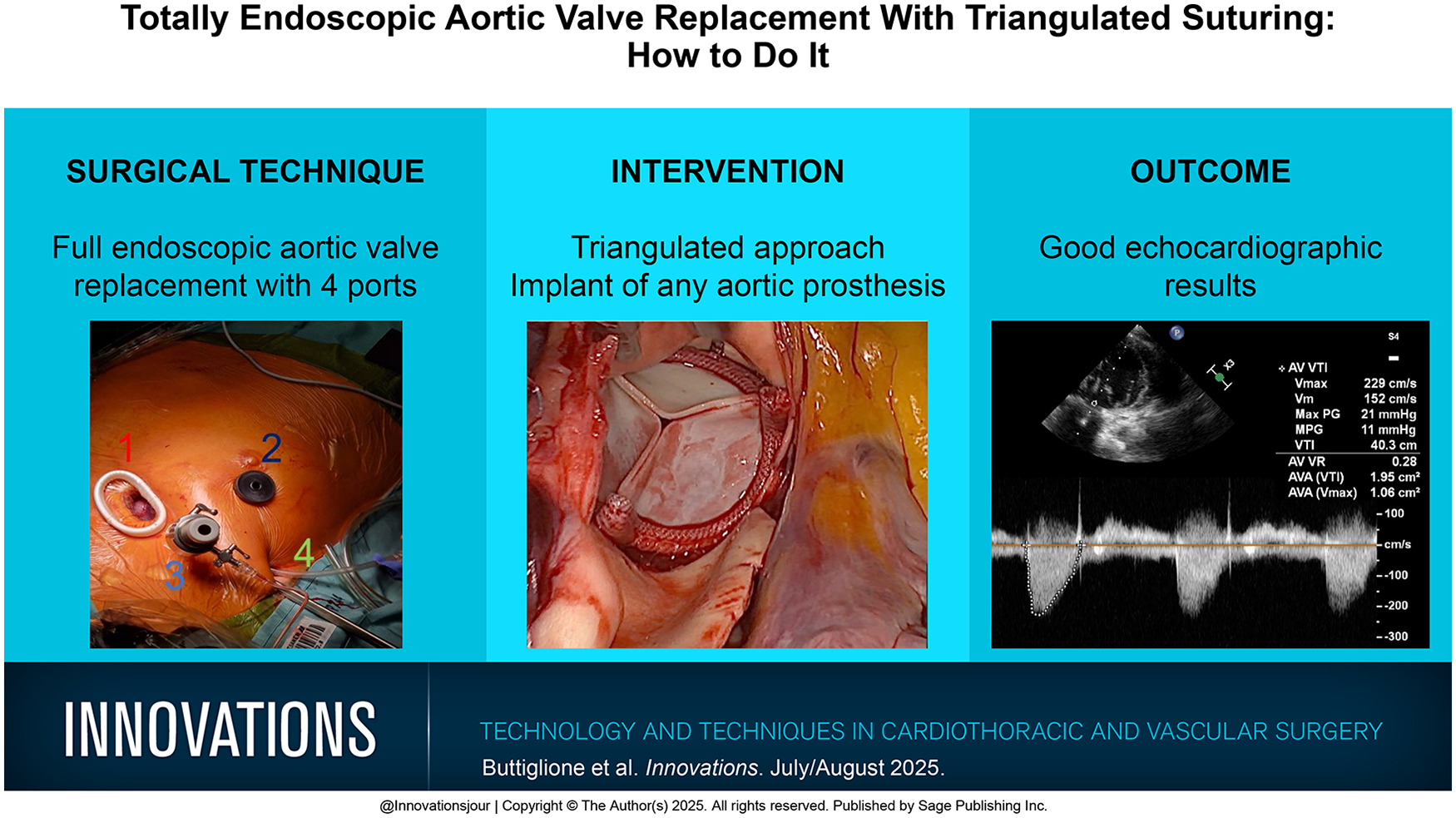
The following ports are used for TEAVR: (1) a 3 cm incision anterolateral minithoracotomy is performed at the midclavicular line above the second intercostal space that is used as the main incision; (2) a working port is placed through a 10 mm right periareolar incision in all male patients or in female patients beyond the reproductive age entering the fourth intercostal space; (3) a stab incision for the endoscopic camera port is made in the second intercostal space, at the level of the right anterior axillary line at least 2 cm laterally to the main incision; (4) a stab incision to accommodate the transthoracic cross-clamp and venting catheter is placed at the level of the right midaxillary line in the third intercostal space. An extra small soft-tissue retractor and a 30° downward 3D camera is used for visualization (Fig. 1). All surgical maneuvers are performed by using the main incision for the left-hand and the periareolar working port for the right-hand instrument in a triangulated fashion to avoid obstruction of the view by surgical instruments of manipulations.
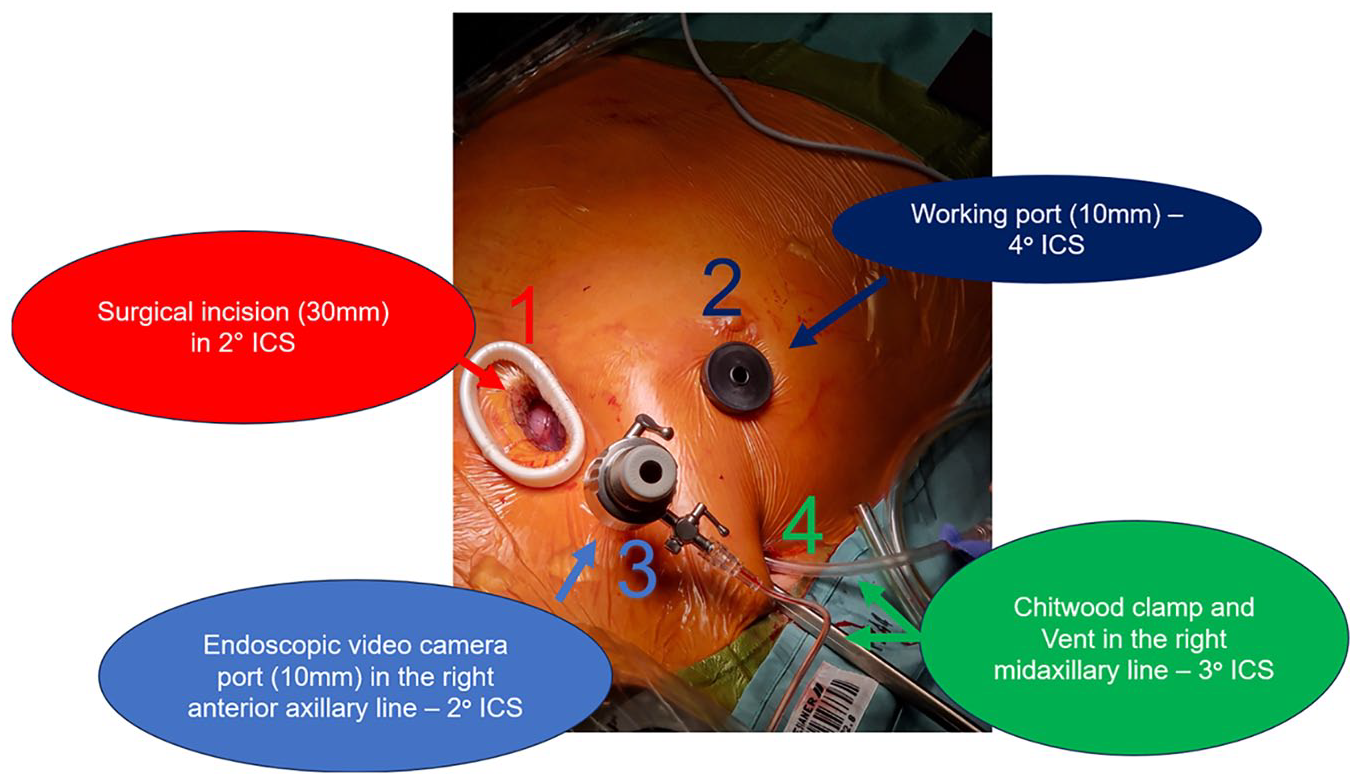
The image shows the 4 ports used for totally endoscopic aortic valve replacement: (1) surgical incision, (2) working port, (3) port for endoscopic video camera, and (4) port for Chitwood clamp and vent. ICS, intercostal space.
The pericardium is opened at least 3 cm parallel to the phrenic nerve using the LigaSure™ vessel-sealing device (Medtronic, Dublin, Ireland) with the Maryland jaws; pericardial traction sutures are brought through the thorax using an Endo Close™ Trocar Site Closure Device (Medtronic). The tissue behind the posterior aortic wall and the pulmonary bifurcation is opened to allow a safe placement of the transthoracic clamp at the distal part of the ascending aorta and avoid injury of the pulmonary artery.
A double purse-string suture is placed at the highest point of the ascending aorta just below the designated cross-clamp area. A purse-string suture is placed to secure a vent in the right upper pulmonary vein to avoid distention of the left ventricle, especially in patients with aortic regurgitation, and allow a clean operative field. The aorta is cross-clamped, and a single-shot antegrade Bretschneider cardioplegia (Custodiol) is administered (Supplemental Video 2).
After cardioplegia administration, a longitudinal aortic incision is performed toward the noncoronary sinus up to the level of the sinotubular junction. The aortic valve is exposed by 2 stay sutures on each side, which are attached to the corresponding pericardial edges, above the phrenic nerve. In this way, an excellent view of the aortic valve and the aortic annulus can be achieved (Supplemental Video 3). Once the aortic valve has been exposed, the aortic leaflets are carefully resected to avoid debris embolization. A dedicated universal sizer is used to determine the size of the valve prosthesis (Supplemental Video 4). A bovine bioprostheses (Avalus 23 mm, Medtronic) is chosen as a valve substitute.
Valve suturing starts on the left-hand side of the left-right commissure and continues clockwise up to the beginning of the noncoronary sinus. To avoid view obstruction of the scope, we continue counterclockwise with the left coronary sinus and finalize the stitches at the noncoronary sinus. The sutures are placed in a triangulated fashion by introducing the needle through the minithoracotomy with the left hand and passing it to the needle holder. The latter is introduced through the working port in the fourth intercostal space, and it is used to pass the needles through the annulus. The sutures are externalized through the thoracotomy with forceps and are secured at the suture organizer. After parachuting the prosthetic valve down into the annulus, we use the COR-KNOT MINI® device (LSI Solutions, Victor, NY, USA) to secure the valve sutures (Supplemental Video 5).
Finally, the aortotomy is closed in 2 rows using a horizontal mattress suture and an over-and-over continuous suture thereafter. After root deairing, the cross-clamp is removed. After weaning from cardiopulmonary bypass, a postoperative transesophageal echocardiogram demonstrates normal function of the bioprosthesis and no signs of paravalvular leak, and continuous-wave Doppler shows a mean pressure gradient of 11 mm Hg (Supplemental Video 6).
Discussion
The described technique is based on the principle of triangulation between the left and the right instrument. 7 The scope is positioned between the 2 instruments to avoid view obstruction by surgical maneuvers. Moreover, inadvertent collisions between the instruments and the scope are avoided in this way. Apart from advantages in visualization, the technique allows a more ergonomic placement of the annular sutures for valve implantation. First, triangulation allows traction and countertraction for tissue dissection. Second, it facilitates perpendicular annular sutures without the need to verticalize the needle to perform the so-called “hook stiches.” Almost all sutures can be placed in a 90° fashion to the opening of the needle holder. Both forehand and backhand sutures are possible. Unlike hook stitches, perpendicular stitches provide better tactile feedback of the stitch depth and the quality of the tissue, which is crucial to avoid tears causing paravalvular leaks. Third, by using traction on placed sutures or on the annular tissue with the forceps, the annulus can be better exposed. The latter facilitates a quick and precise placement of each and every suture around the annulus.
The longitudinal aortotomy is another important component to enhance visualization of the annular plane and increase working space. Using a longitudinal incision, the aorta is opened toward the direction of the scope. The coronary ostia can be easily identified, and the complete aspect of the aortic annulus is exposed. Moreover, aortic stay sutures do not interfere with the movement of the working instruments. Most importantly, avoiding remote areas, such as the left-sided part of the aorta toward the pulmonary artery, reduces the risk of bleeding in an area that is difficult to repair. Closure of the aortotomy is easier due to the direct visualization of the linear cut and the perpendicular position of the needle holder and the forceps in relation to the aortic incision. Although we have begun this operation by using a transverse incision, we have witnessed the limitations of working with the instruments in a funnel built by the aorta as a cylinder. The longitudinal aortotomy allows significantly more freedom of movements within the aortic root and minimizes the risk of iatrogenic injury of the aortic tissue.
Conclusions
The triangulated approach for TEAVR is one strategy for performing endoscopic replacement of the aortic valve through a 3 cm incision and 3 port access using a 3D camera. This technique allows the implant of any type of aortic prosthesis as well as the performance of additional procedures such as annular enlargement or septal myectomy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
Our institution does not require ethical approval for reporting individual cases or case series. Written informed consent was obtained from the individual for the publication of any potentially identifiable images or data included in this article.
Supplemental Material
Supplemental material for this article is available online.
