Abstract
Objective:
Previous learning curve analyses of minimally invasive mitral valve (MV) repair have focused largely on early safety outcomes without including detailed mitral repair quality outcomes. This study investigates the learning curve of minimally invasive MV repair over a 15-year experience, focused on clinical outcomes and evidence-based technical failure endpoints.
Methods:
All MV repair operations were performed by a single surgeon between May 2008 and February 2023. Patient data were stratified into 3 groups of tertiles. Failure endpoints were defined as postrepair residual mitral regurgitation ≥ mild and a 30-day composite outcome. Cumulative log-likelihood curves were constructed for minimally invasive MV repair using the primary outcomes as technical failure endpoints. Control limits were determined using previous analyses of the Society of Thoracic Surgeons database.
Results:
A total of 362 consecutive patients across 15 years were included. Across tertiles, there was a significant trend toward shorter cross-clamp time (P < 0.001), cardiopulmonary bypass time (P < 0.001), and hospital length of stay (P = 0.005). Learning curve analysis demonstrated crossing of the lower threshold at ~60 patients for postrepair mitral regurgitation ≥ mild and ~85 patients for the 30-day composite outcome. The mean adjusted risk scores for both primary outcomes based on a multivariable logistic model demonstrated no significant differences across tertiles.
Conclusions:
The estimated number of operations to achieve optimal repair outcomes and durability is ~60 to 85 patients. These data can improve the design of surgical training competencies, beyond avoidance of complications, and instead focus the learning curve on what is necessary to achieve optimal mitral repair outcomes.
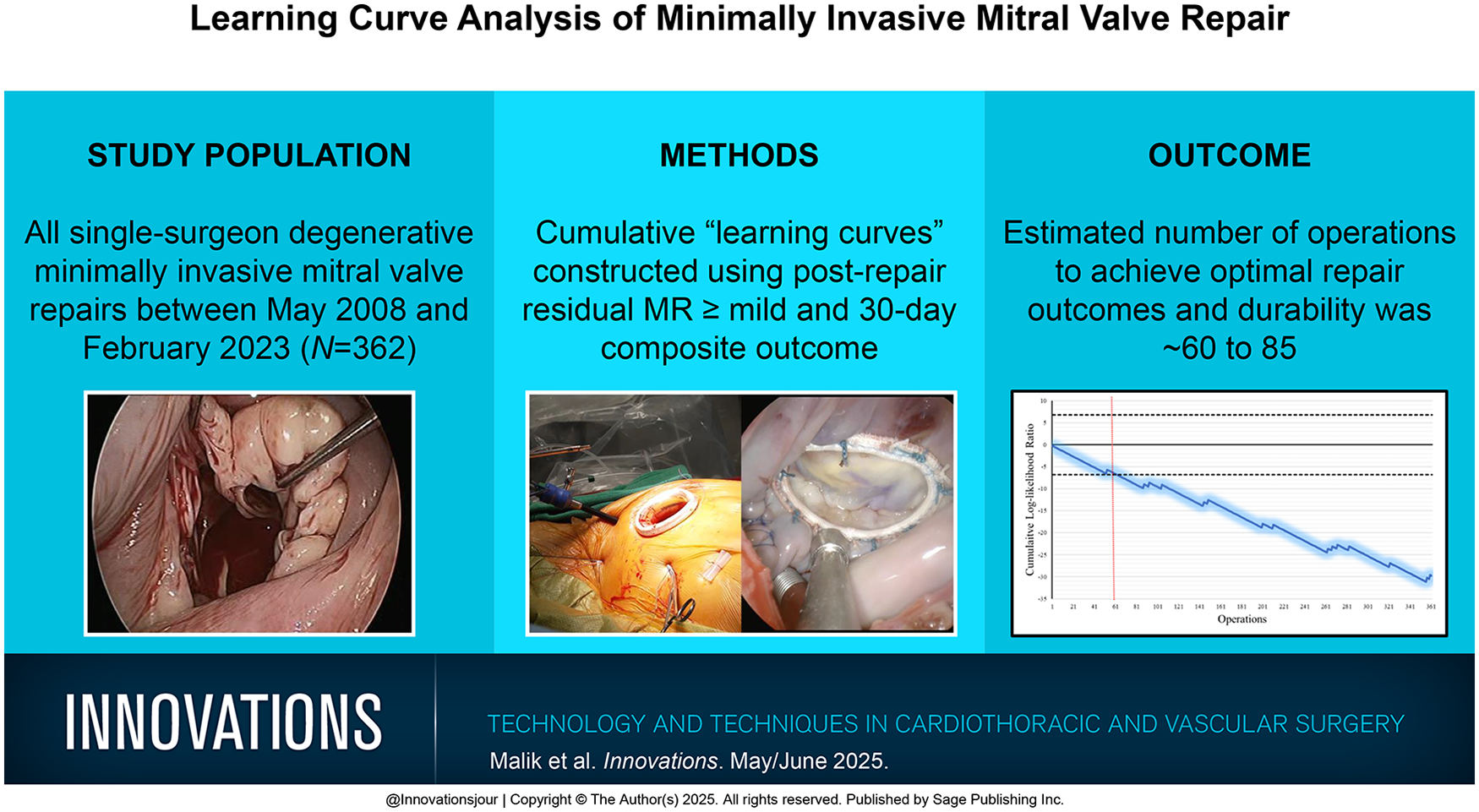
Central Message
Using evidence-based failure endpoints in surgeons learning to perform minimally invasive mitral valve repair, the estimated number of operations to achieve optimal repair outcomes and durability is ~60 to 85. This learning curve was established for a single surgeon in 362 patients over 15 years.
Introduction
Despite ongoing debate, minimally invasive mitral valve repair (MIMVr) has been widely adopted and is associated with excellent short-term and long-term results.1–3 Compared with conventional median sternotomy, MIMVr has associated benefits, including shorter hospital length of stay, faster return to baseline physical function, and improved cosmesis. Importantly, minimally invasive techniques are associated with similar morbidity and mortality as conventional median sternotomy, recently confirmed in an important randomized, clinical trial. 2 However, MIMVr is technically demanding and requires specialized training to obtain operative proficiency with successful outcomes. 3
Similar to other volume-outcome relationships in surgery, there is an inverse relationship between the annual number of MIMVr performed and the expected complication rates. 4 Previous analyses have reported prolonged learning curves for trainees attempting to achieve mastery of MIMVr. Generally, between 75 and 125 procedures were thought to be necessary for a trainee to feel comfortable with MIMVr, depending on their previous level of training.5,6 These learning curve analyses had focused on safety endpoints such as perioperative mortality and complication rates but had not investigated mitral repair quality outcomes.
Our study investigated the learning curve of MIMVr over a 15-year period, using evidence-based failure endpoints. Technical failure endpoints, such as postrepair residual mitral regurgitation (MR) and 30-day composite outcomes (stroke, reoperation, prolonged ventilation, renal failure, and wound infection) were investigated to further our understanding of the learning curve for MIMVr.
Methods
In this study, we performed a retrospective analysis of 362 consecutive patients who underwent MIMVr by a single surgeon between May 2008 and February 2023. The surgeon (M.W.A.C.) had formal fellowship training in minimally invasive and robotic MVr surgery prior to starting independent practice, from which all operations included in this analysis take thereafter. Although all procedures were completed independently by the same surgeon, an experienced surgeon mentor was available for guidance during the early career. Complexity and patient selection were conservative early in practice and expanded with surgeon experience.
An observational learning curve analysis was conducted on patients who underwent a MIMVr via a right minithoracotomy approach (Fig. 1) for degenerative MV disease. Further details of our surgical approach can be found in previous publications.7–12 The inclusion criteria were all patients with degenerative MR who underwent MIMVr with or without concomitant tricuspid valve repair, atrial septal defect (ASD) or patent foramen ovale (PFO) repair, cryoablation, and/or left atrial appendage excision. We excluded patients who underwent sternotomy as a primary approach, had indications for surgery outside of degenerative MR including endocarditis, had rheumatic disease, had functional pathology, and underwent concomitant coronary bypass surgery, aortic valve surgery, or aortic surgery.
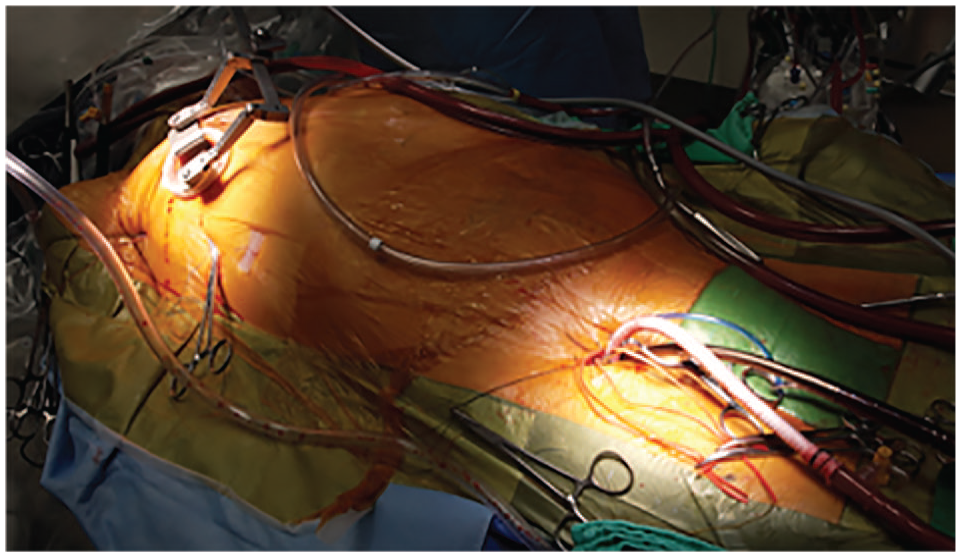
Patient positioning and cannulation setup for a right minithoracotomy approach to repair the mitral valve.
Patient data were stratified into 3 groups of tertiles to undergo univariable comparison over different eras of time to establish a longitudinal trend. Annual surgical volume was graphically plotted for visual context (Supplemental Fig. 1). We decided on 2 failure endpoints based on previously established markers of quality for MVr (1) postrepair residual MR greater than or equal to mild (MR ≥ mild) based on postoperative echocardiogram and (2) a 30-day composite outcome including stroke, reoperation, prolonged mechanical ventilation (>48 h), renal failure, and wound infection.13,14 In addition, we compared stand-alone outcomes across tertiles, including the aforementioned outcomes from the composite, death, myocardial infarction, atrial fibrillation, hospital length of stay, and intensive care unit length of stay. Long-term outcomes were plotted across tertiles, including median clinical follow-up time, need for MV reoperation, New York Heart Association (NYHA) status, degree of MR on most recent echocardiography, and survival. The univariable comparison of patient characteristics and outcomes across tertiles is presented in the tables. The P value and standardized mean difference were also calculated. A P value <0.05 was considered significant.
A multivariable logistic regression was performed using a priori knowledge for covariate selection based on the interaction of the covariates with the primary outcomes. Variables selected for adjustment in the model included age, sex, atrial fibrillation, surgical approach, body mass index, coronary artery disease, congestive heart failure, chronic obstructive pulmonary disease, cerebrovascular disease, peripheral vascular disease, estimated glomerular filtration rate, diabetes mellitus, NYHA class ≥3, leaflet pathology, preoperative ejection fraction, preoperative hemoglobin, myocardial infarction in the last year, urgency, concomitant tricuspid valve repair, concomitant cryoablation, concomitant ASD or PFO closure, commissuroplasty, patch repair, and resection. The mean adjusted risk scores for the primary outcomes were then plotted across tertiles using these logistic models to compare operative complexity over time. A P value was calculated with significance set at <0.05.
Cumulative sum control charts or “learning curves” were then constructed for MIMVr using the primary outcomes as technical failure endpoints. The alpha and beta limits were set at 5%. Based on 2 large retrospective analyses of MVr in the Society of Thoracic Surgeons (STS) database, we determined an acceptable rate of the primary outcomes of postrepair residual MR ≥ mild and 30-day composite outcome to be 10% each.13,14 As such, control limits were built using the rate of the primary outcomes in these analyses of MV from the STS database. Using these control limits, a cumulative log-likelihood ratio chart was plotted with positive and negative horizontal boundary lines. The log-ratio was plotted on the y-axis against the number of operations on the x-axis. If performance is acceptable, the graph will trend downward toward the lower horizontal boundary line, with this line being the threshold for technical competency based on the primary outcomes. This point of intersection provided a rough idea of the number of operations required to achieve a point at which the rate of failure was acceptable in comparison with our prespecified control rate (i.e., rate of primary outcomes in STS database).
Results
A total of 362 patients underwent MIMVr between May 2008 and February 2023 (median age = 63 [55, 72] years, female = 30% [n = 94]). These patients were further divided into 3 equal cohorts or tertiles to compare patient characteristics and outcomes over different eras of time (group 1 = operation 1 to 120, group 2 = operation 121 to 241, group 3 = operation 242 to 362). The univariable comparison of patient baseline characteristics and the intraoperative details are presented in Table 1 and Table 2, respectively.
Baseline Patient Characteristics of Minimally Invasive Mitral Valve Repair for Degenerative Pathology Over 15 Years Grouped by Tertiles of Sequential Operations.
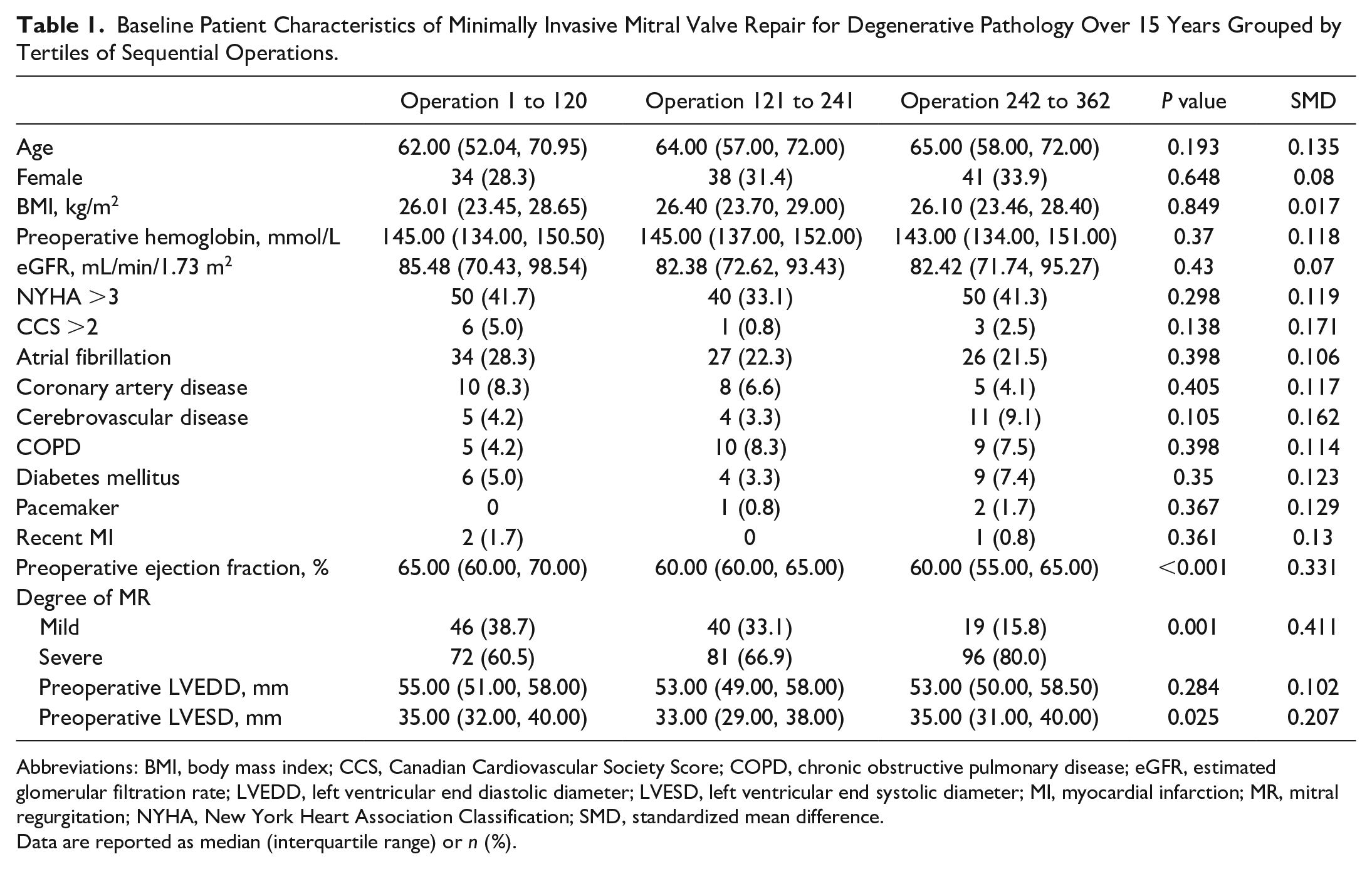
Abbreviations: BMI, body mass index; CCS, Canadian Cardiovascular Society Score; COPD, chronic obstructive pulmonary disease; eGFR, estimated glomerular filtration rate; LVEDD, left ventricular end diastolic diameter; LVESD, left ventricular end systolic diameter; MI, myocardial infarction; MR, mitral regurgitation; NYHA, New York Heart Association Classification; SMD, standardized mean difference.
Data are reported as median (interquartile range) or n (%).
Intraoperative Details of Minimally Invasive Mitral Valve Repair for Degenerative Pathology Over 15 Years, Grouped by Tertiles of Sequential Operations.
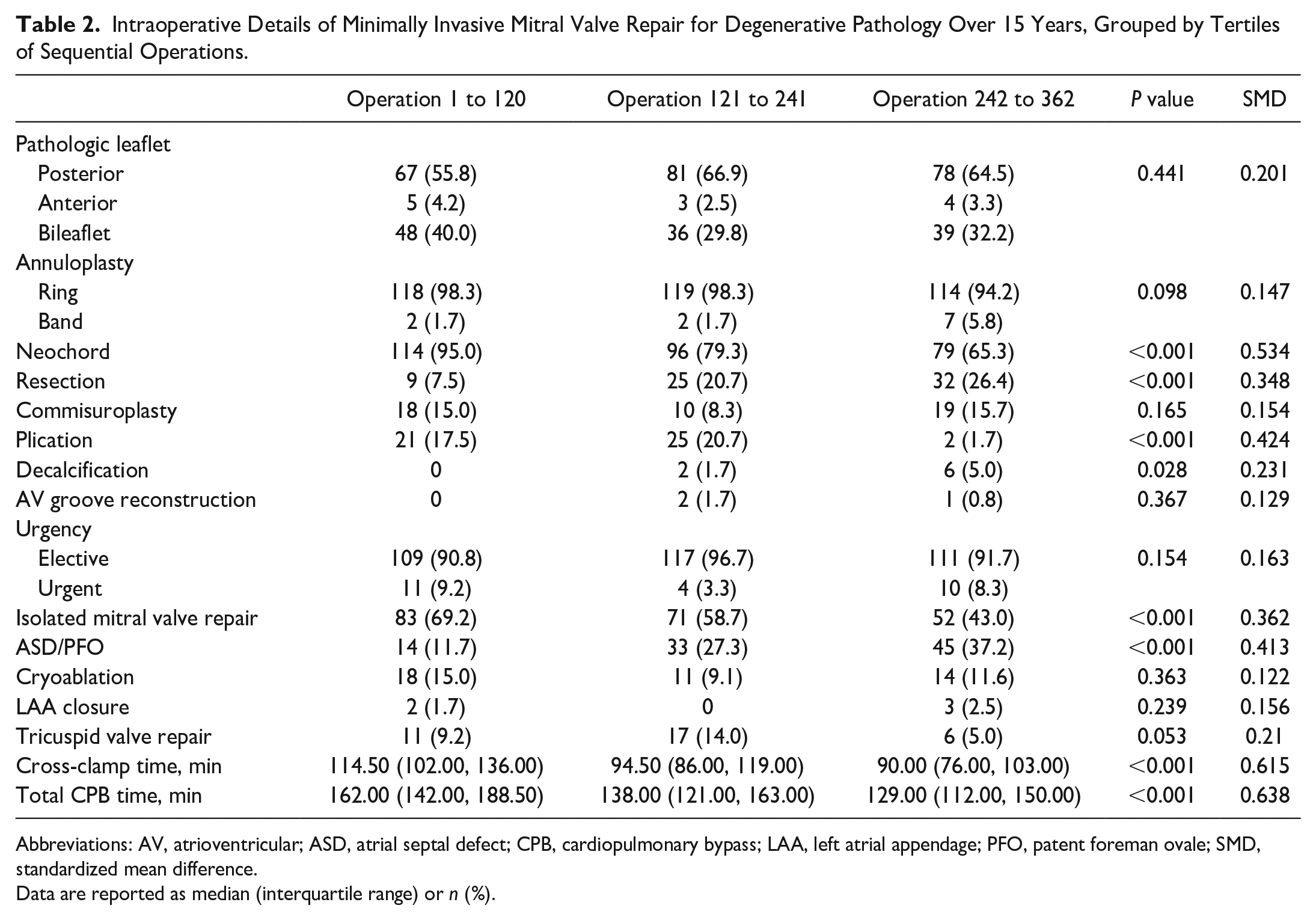
Abbreviations: AV, atrioventricular; ASD, atrial septal defect; CPB, cardiopulmonary bypass; LAA, left atrial appendage; PFO, patent foreman ovale; SMD, standardized mean difference.
Data are reported as median (interquartile range) or n (%).
Across tertiles, baseline characteristics were grossly similar. There was a significant trend over time toward more patients having severe MR (60.5% vs 66.9% vs 80%, P = 0.001), lower ejection fraction (65% vs 60% vs 60%, P < 0.001), and a larger difference in left ventricular end systolic diameter (35 mm vs 33 mm vs 35 mm, P = 0.025).
Intraoperatively, there was a significant difference over time in the relative use of various repair techniques: neochordae repair (95% vs 79.3% vs 65.4%, P < 0.001), resection repair (7.5% vs 20.7% vs 26.4%, P < 0.001), plication (17.5% vs 20.7% vs 1.7%, P < 0.001), and decalcification (0% vs 1.7% vs 5%, P = 0.028). In addition, we found a significant difference in the frequency of concomitant ASD/PFO repairs (11.7% vs 27.3% vs 37.2%, P < 0.001). There was a trend toward shorter cross-clamp time (114.5 min vs 94.5 min vs 90 min, P < 0.001) and cardiopulmonary bypass time (162 min vs 138 min vs 129 min, P < 0.001) across tertiles (Fig. 2).
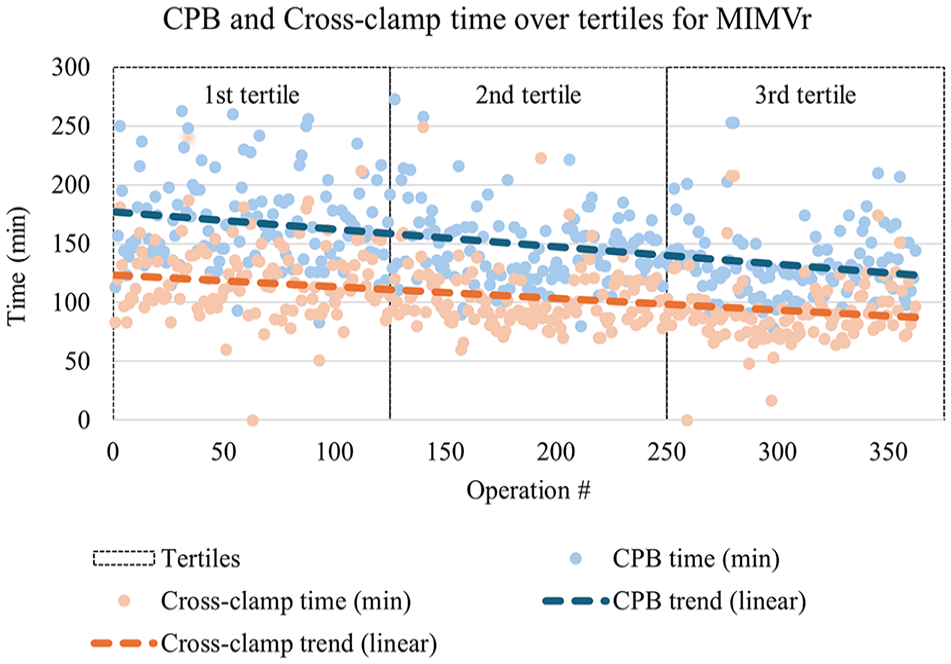
Scatterplot of cross-clamp time and total CPB time over 15 years of MIMVr, grouped by tertiles of sequential operations. CPB, cardiopulmonary bypass; MIMVr, minimally invasive mitral valve repair.
The univariable comparison showed that postoperative outcomes across tertiles were generally similar (Table 3). However, there was a trend toward shorter hospital length of stay (6 days vs 6 days vs 5 days, P = 0.005). The long-term outcomes are shown in Table 4. The median follow-up time was significantly longer for patients in earlier tertiles, and as such, there were higher rates of death and MR recurrence in the earlier tertiles. There was no difference in reoperation on the MV or NYHA status.
Postoperative 30-Day Outcomes After Minimally Invasive Mitral Valve Repair Grouped by Tertiles of Sequential Operations.
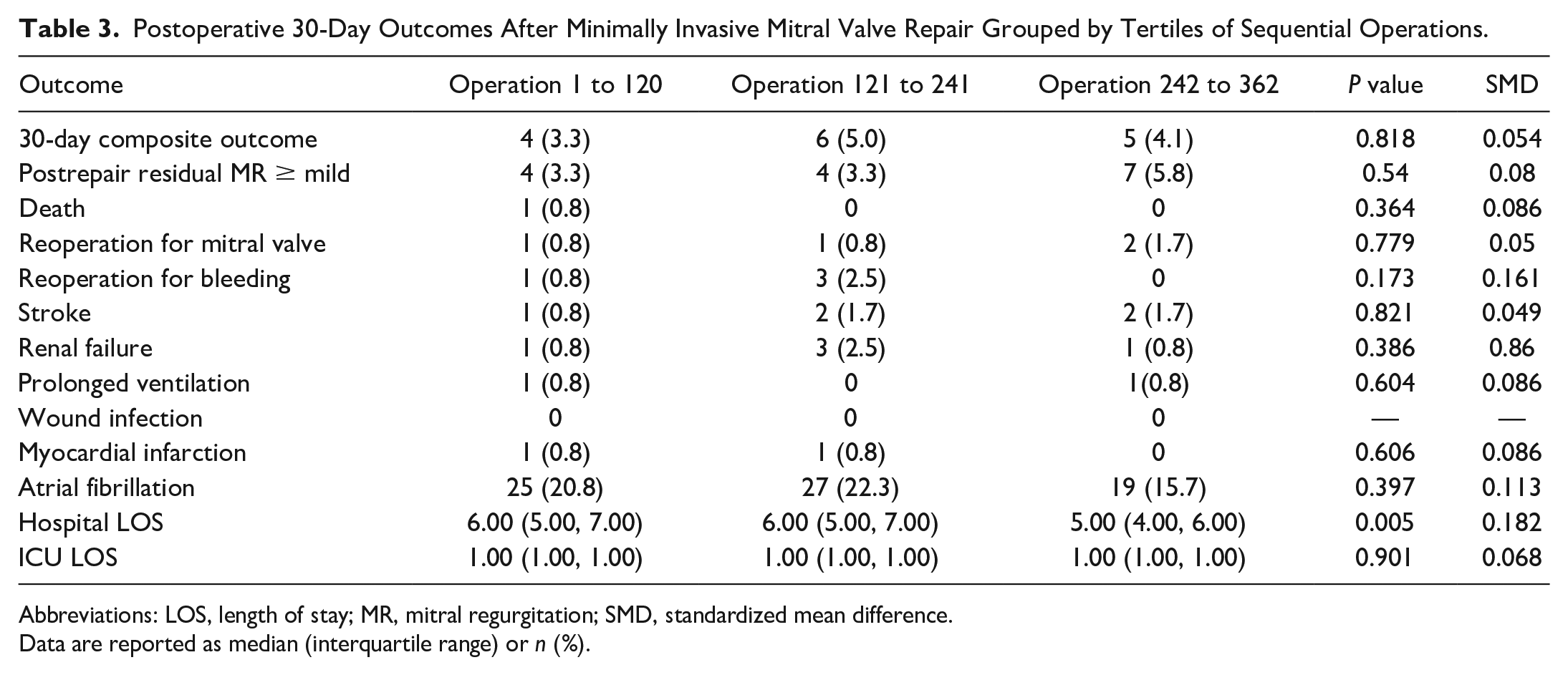
Abbreviations: LOS, length of stay; MR, mitral regurgitation; SMD, standardized mean difference.
Data are reported as median (interquartile range) or n (%).
Long-Term Postoperative Outcomes After Minimally Invasive Mitral Valve Repair Grouped by Tertiles of Sequential Operations.
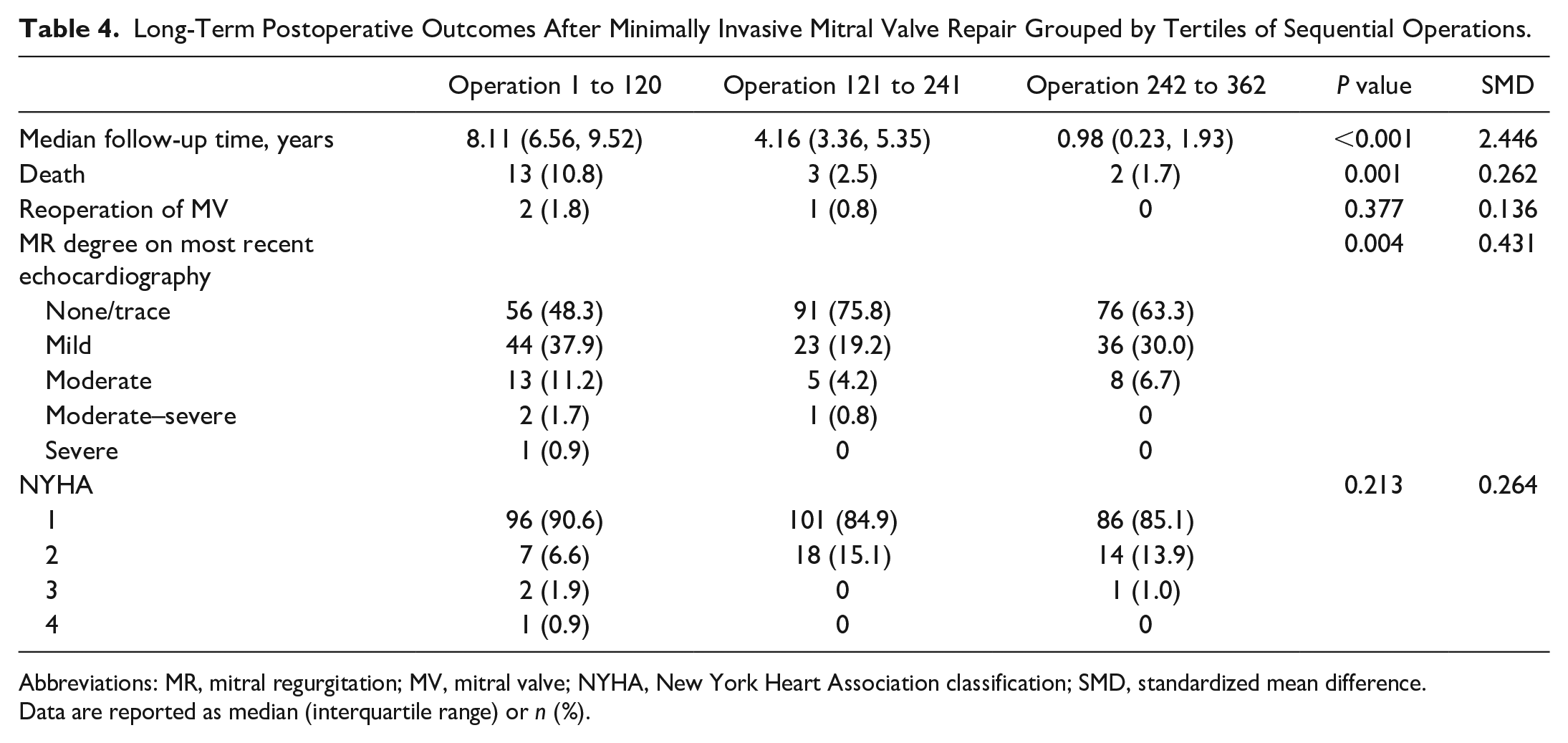
Abbreviations: MR, mitral regurgitation; MV, mitral valve; NYHA, New York Heart Association classification; SMD, standardized mean difference.
Data are reported as median (interquartile range) or n (%).
Cumulative log-likelihood curves or “learning curves” were plotted for both of the primary endpoints of interest: (1) postrepair residual MR ≥ mild based on postoperative echocardiogram and (2) a 30-day composite outcome (Fig. 3, Fig. 4). The learning curves cross the horizontal boundaries established by our prespecified control limits at approximately 60 and 85 operations, respectively.
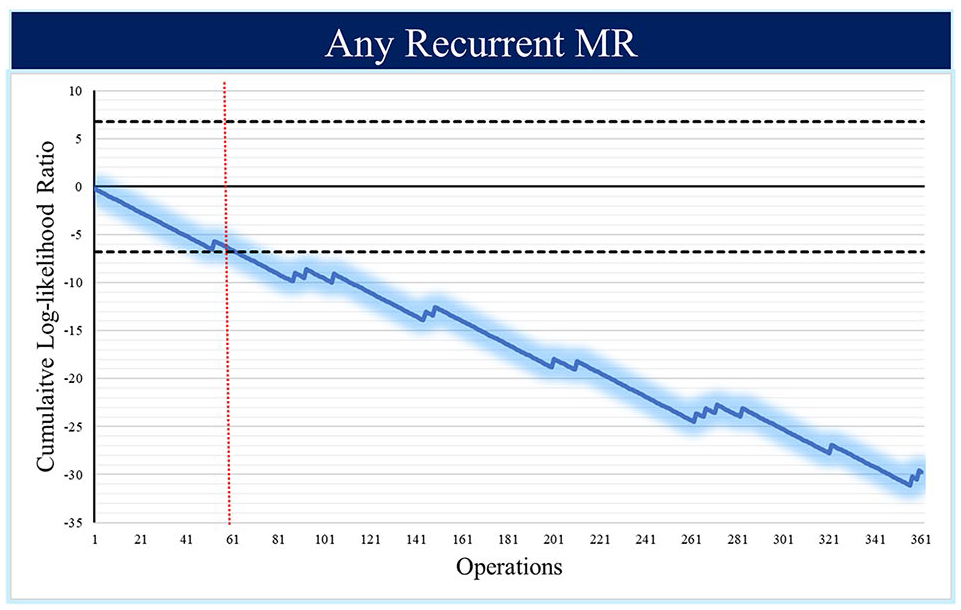
Cumulative log-likelihood ratio curve for postrepair residual greater than or equal to mild. MR, mitral regurgitation.
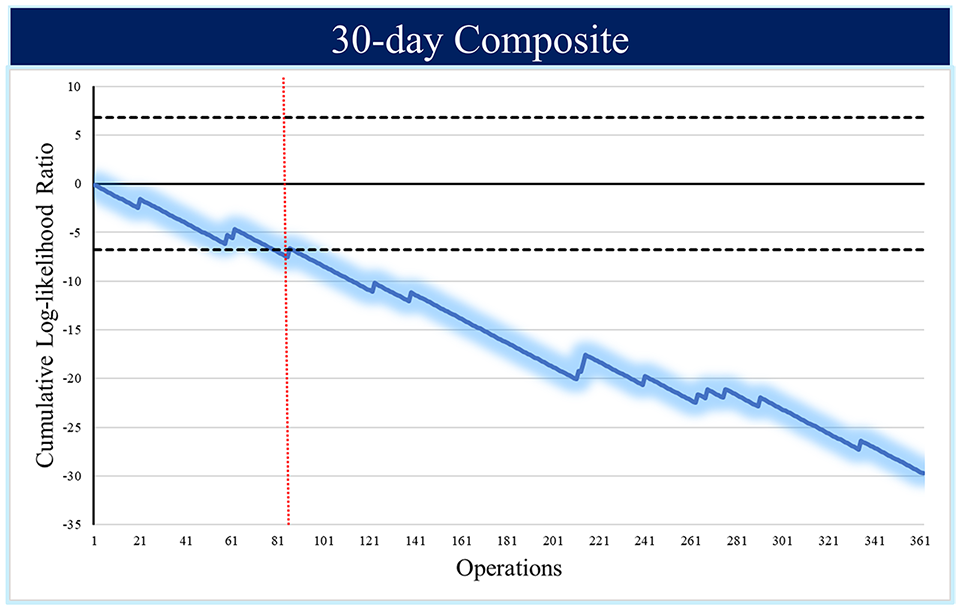
Cumulative log-likelihood ratio curve for postoperative 30-day composite outcome.
Adjusted risk scores were calculated based on our multivariable logistic regression of the primary failure endpoints. There was no significant difference in average risk scores across the 3 cohorts for either of the primary outcomes (Fig. 5).
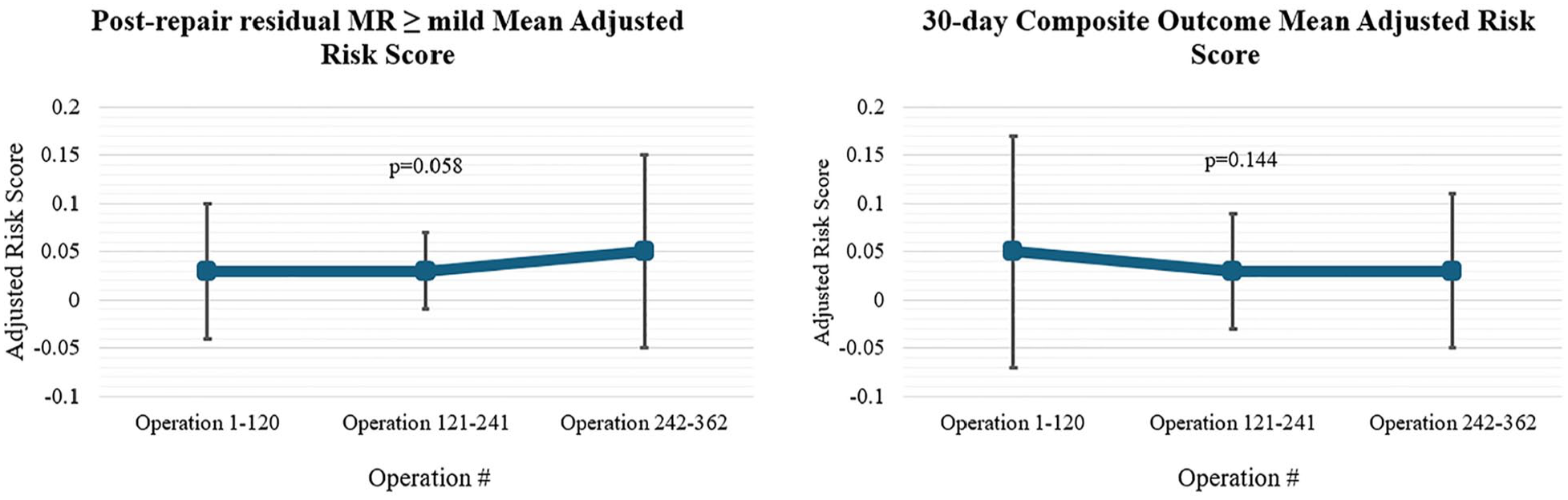
Mean adjusted risk score for the primary outcomes based on the multivariable logistic model, grouped by tertiles of sequential operations. MR, mitral regurgitation.
Discussion
Our single-center, observational, learning curve analysis was completed for all MVr operations performed by a single surgeon over 15 years, stratified into 3 tertiles for comparison over time. Our results indicate that the estimated number of operations required to achieve optimal repair outcomes and durability is approximately 60 to 85.
The benefits of MIMVr have been extensively researched and widely reported. A recent randomized controlled trial showed that MIMVr is as safe and effective as sternotomy for degenerative MR at 1 year after operation. 2 The growing prevalence of MIMVr in North America warrants further exploration of meaningful technical endpoints to define operative success.
To assess training efficacy, several studies have examined the learning curve in MIMVr to determine the average number of cases required to become proficient. These studies focused on morbidity/mortality measures to define technical success and reported a range of 75 to 125 operations for trainees to achieve proficiency. There is also a steeper learning curve for MIMVr versus MV replacement.5,6,15 Previous studies of learning curves have laid a valuable foundation for evaluating minimally invasive training programs. However, we believe that there is a gap in the literature, and evidence-based quality endpoints, such as freedom from mild or worse MR, can be used to aid in developing or refining future and present training program competencies.
One metric is cardiopulmonary and cross-clamp times. Studies have reported prolonged cardiopulmonary bypass and cross-clamp times as disadvantages of MIMVr. 6 Our results demonstrated a significant trend toward shorter cross-clamp time, cardiopulmonary bypass time, and hospital length of stay between the first and third tertiles of operations. This was despite a significant increase in the number of concomitant procedures being performed at the time of repair over tertiles. These results align with the previous literature and suggest that as the surgeon’s minimally invasive operative volume increases, so does their efficiency.
While various endpoints have been used to define operative success in MIMVr, the Mitral Valve Academic Research Consortium defines the procedural success of MV operations as a reduction in MR to optimal or acceptable levels in addition to other safety endpoints.16–19 Our results showed a decrease in the cumulative log-likelihood ratio for postrepair residual MR and 30-day composite outcomes (stroke, reoperation, prolonged ventilation, renal failure, and wound infection) as the number of MIMVr completed by a surgeon increased. Safety endpoints are important, but mitral repair outcomes, such as residual regurgitation, must be monitored as these quality metrics are foundational for the long-term success of any mitral repair program.
Our study had several limitations. This retrospective, observational, cohort study had the inherent limitations of this type of analysis. In addition, this research represents the experience of a single surgeon and center, which may limit the external generalizability of our results. Long-term outcomes are biased by follow-up length, and therefore, we cannot draw any conclusions about long-term durability over the training period. Finally, the choice of surgical approach was not randomized and was biased by preoperative patient characteristics. We aimed to address these limitations in our statistical analysis; however, all biases could not be removed.
Conclusions
MIMVr has become a widely accepted and used operation over the past 20 years. Although previous learning curves have helped to establish safety limits, we believe that our study adds to the evidence that between 60 and 85 operations are required to cross the threshold to achieve optimal mitral repair outcomes as well.
Supplemental Material
sj-pdf-1-inv-10.1177_15569845251337406 – Supplemental material for Learning Curve Analysis of Minimally Invasive Mitral Valve Repair
Supplemental material, sj-pdf-1-inv-10.1177_15569845251337406 for Learning Curve Analysis of Minimally Invasive Mitral Valve Repair by Mohsyn Imran Malik, Brandon Loshusan and Michael W. A. Chu in Innovations
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael W. A. Chu reports a relationship with Medtronic that includes speaking and lecture fees, a relationship with Edwards Lifesciences Corporation that includes speaking and lecture fees, a relationship with Terumo Aortic that includes speaking and lecture fees, and a relationship with Artivion, Inc. that includes speaking and lecture fees. The other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
