Abstract
Objective:
Primary cardiac tumors are uncommon, often benign, but can be potentially life threatening. Minimally invasive endoscopic (ENDO) techniques have been shown to be a feasible alternative for tumor resection compared with conventional sternotomy (CS). This study compared the clinical and surgical outcomes of a small series of patients undergoing cardiac tumor resection operations.
Methods:
Between November 2009 and December 2022, 34 consecutive patients underwent cardiac tumor resection using either ENDO (n = 21) or CS (n = 13) techniques. We compared early perioperative outcomes, echocardiographic outcomes, and long-term clinical and tumor recurrence outcomes.
Results:
Baseline characteristics were similar between groups; however, the ENDO group included younger patients (56 ± 16 vs 62 ± 17 years) and more female patients (83% vs 53%). The tumor was located in the left atrium (n = 19, 56%), right atrium (n = 5, 15%), or either ventricle (n = 4, 12%). In-hospital mortality and stroke frequency were similar for both groups (n = 0). There was no significant difference in cardiopulmonary bypass or cross-clamp times, respiratory or renal failure, or intensive care unit or hospital lengths of stay. At follow-up (ENDO, 42 [2 to 131] months vs CS, 54 [1 to 156] months), there were no deaths in the ENDO group and 2 patients died in the CS group (P = 0.21). No patients in either group experienced tumor recurrence.
Conclusions:
In selected patients, both ENDO and CS approaches to primary cardiac tumor resection were safe, effective, durable, and associated with similarly good early and late results.
Keywords
Central Message
A study of 34 consecutive patients found that minimally invasive endoscopic and conventional sternotomy approaches to the resection of cardiac tumors were associated with similarly good early and late outcomes in selected patients.
Introduction
Primary cardiac tumors are uncommon, with a reported incidence rate of 0.001% to 0.3%. 1 They occur more frequently in women than in men and are commonly benign in approximately 90% of all cases, with most characterized as myxomas or papillary fibroelastomas. Malignant cardiac tumors account for 10% of cases and are most commonly undifferentiated pleomorphic sarcomas, followed by angiosarcomas and leiomyosarcomas. 2 Approximately 80% of myxomas have been reported to occur in the left atrium, and most of these are attached to the interatrial septum. Between 5% and 20% are found in the right atrium, and a small minority are found in both atria or, in some instances, the left or right ventricle. 3 Patients with primary cardiac tumors are often found incidentally or present with a nonspecific triad of symptoms related to obstruction, embolism, or constitutional symptoms.2,4 Echocardiography is often the first imaging modality used, with computed tomography and cardiac magnetic resonance imaging being used for better tumor characterization and preoperative planning.
Primary cardiac tumors often require expedient surgical resection, particularly if obstructive symptoms are present. Delayed treatment can increase the risk of life-threatening complications such as valvular obstruction or cerebral embolization.5,6 The conventional median sternotomy (CS) approach has been the mainstay of treatment. With the rise in minimally invasive endoscopic (ENDO) techniques to treat valve disease, these less invasive techniques have proven to be a feasible alternative to CS.1,3,6–8 Less invasive ENDO techniques are attractive to patients because of the smaller incisions, faster healing, less disability, associated shorter hospital length of stay (LOS), and overall cosmesis. 9 However, concerns in tumor surgery remain adequacy of exposure, completeness of resection, and risk of tumor recurrence. 10 Even in CS approaches to primary cardiac tumor resection, tumor recurrence rates of 5.6% have been reported. 1
We aim to report our 12-year experience with benign and malignant cardiac tumor resection, comparing patients undergoing ENDO and CS approaches. For both approaches, we investigated patient demographics, tumor characteristics, perioperative characteristics, and midterm patient outcomes.
Methods
We performed a retrospective study from November 2009 to December 2022 with a total of 34 consecutive patients undergoing cardiac tumor resection by a single surgeon. Patients either underwent ENDO surgery (n = 21) or CS surgery (n = 13). We reviewed the medical records of all included patients. Their baseline, perioperative, and postoperative data were obtained and analyzed.
Surgical Techniques
Minimally invasive endoscopic surgery
We have previously described our ENDO technique. 11 In brief, we positioned the patient in a 20°, right lateral decubitus position and used double-lumen endotracheal tube intubation. We routinely employed bicaval cannulation with a percutaneous 16 Fr right internal jugular superior vena cava drainage catheter placed above and a 25 Fr multiport venous cannula placed below by transesophageal echocardiography (TEE) guidance to enable tumor resection from any cardiac chamber. Using a bicaval TEE view, extreme care and visualization was given to the advancement of the guidewires to barely cross the cavoatrial junction, thus minimizing any iatrogenic tumor dislodgement in the case of right atrial masses. Femoral arterial cannulation was achieved via an 8 mm Dacron side graft. We used a 3 to 4 cm right anterolateral port access incision (fourth intercostal space for all lesions except aortic valve masses [third intercostal space]) and a 5 mm port for the 0° or 30° endoscope (Fig. 1). We prefer to perform these cases using a transthoracic aortic cross-clamp and cardioplegic arrest with del Nido cardioplegia delivered antegrade through the aortic root. Vacuum assist was commonly used, and the cava were snared only in anticipation of right heart exposure. TEE was used to confirm the anticipated tumor location and to search for additional lesions.
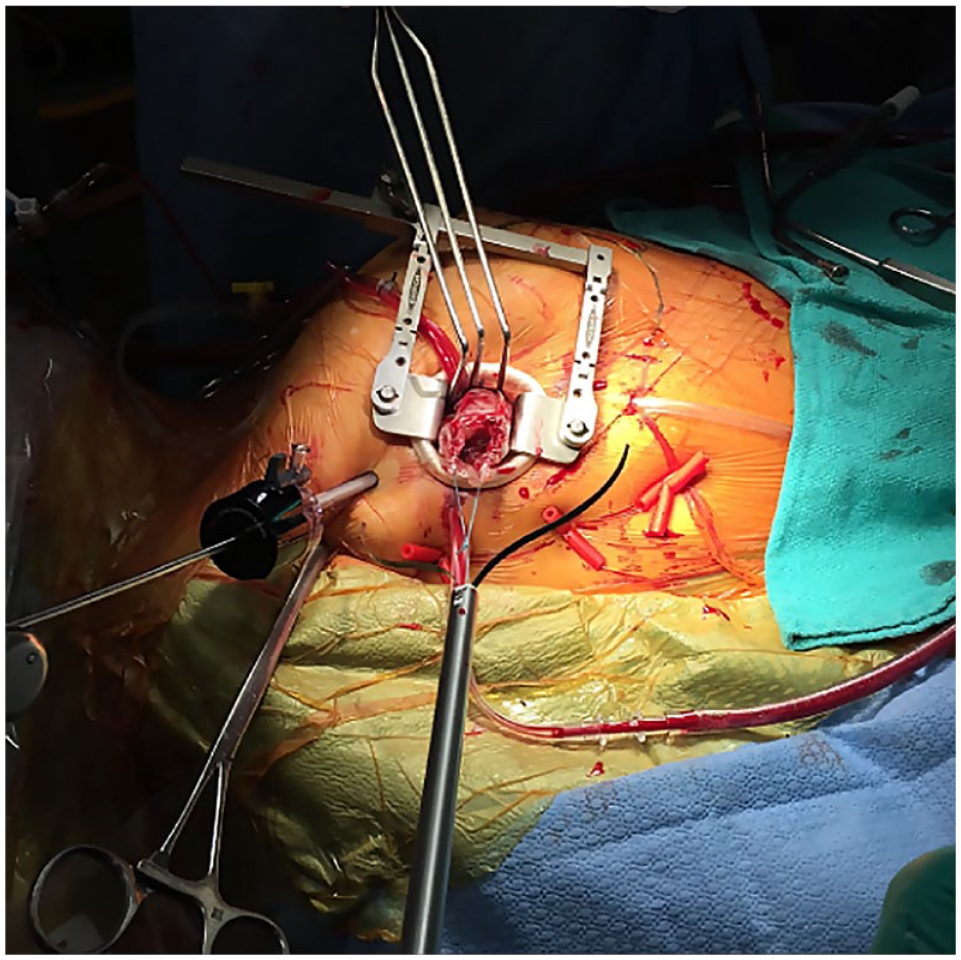
Minimally invasive endoscopic surgery setup.
After cardiac arrest, we generally entered the left atrium just lateral to the tumor stalk, identifying the optimal cardiotomy location by TEE and instrument palpation. If the tumor was present in the left or right ventricle, we entered the appropriate atrium and crossed the atrioventricular valve with the camera to expose the tumor properly. If the tumor was on the aortic valve, we entered through an aortotomy. We carefully held the mass by its stalk and always resected it en bloc, including part of the chamber wall, and worried about reconstruction after. Once the tumor was completely free, we placed it into an endobag and closed the endobag completely before externalizing the tumor across the chest wall port. If the tumor was too large, we could also place it within a second, more robust endobag; remove the soft-tissue retractor; and dilate the port site to enable safe removal of the tumor. After externalizing the tumor, we examined it carefully to look for any signs of tumor fragmentation and, most importantly, examined the stalk base to ensure full and complete resection margins. If there was any concern, we would go back and perform wider resection margins of the base. We ensured that the same en bloc approach to tumor resection was used for benign and malignant tumors. We repaired the base of the chamber wall either directly or with autologous pericardium depending on the size of the defect. The chamber was closed and the remainder of the operation completed in the usual fashion. Careful postoperative TEE was performed to look for any residual tumor fragments and to ensure normal valve function without any residual septal shunts.
Conventional sternotomy
Tumor resections with CS were performed using standardized central cannulation, aortic cross-clamp, antegrade cardioplegia, and similar tumor resection techniques as the ENDO approach. Intraoperative TEE was used to assess for residual tumor, valve competencies, residual interatrial shunt, and global cardiac function.
Perioperative Management and Follow-Up
All tumors were sent for complete pathological analysis. Postoperatively, all patients were transferred to our cardiac surgery recovery unit and progressed to the ward when deemed suitable. Our center used an open and collaborative approach between cardiac surgeons and intensivists for postoperative patient care. Furthermore, the postoperative course was documented, and all our patients received a routine predischarge transthoracic echocardiogram (TTE). Finally, all patients were followed up annually with TTE.
Statistical Analysis
Continuous variables are expressed as mean ± standard deviation or median (interquartile range [IQR]) and compared using Student’s t test or Mann–Whitney U test. Categorical variables are expressed as percentages and were compared between the 2 groups using χ2 tests or Fisher’s exact test. IBM SPSS Statistics, Version 29 (IBM Corp, Armonk, NY, USA) was used to perform statistical analyses. All pairwise comparisons had a Bonferroni correction performed. A P value <0.05 was considered statistically significant.
Results
Early Outcomes
All 34 patients underwent successful surgical resection of their primary cardiac tumor (Fig. 2). Baseline characteristics (Table 1) were similar between the groups with the exception that patients in the ENDO group were younger (56 ± 16 vs 62 ± 17 years) and had more female patients (83% vs 53%). The tumor was located in the left atrium (n = 19, 56%), right atrium (n = 5, 15%), left or right ventricles (n = 4, 12%), or a small number in other locations (Table 1).
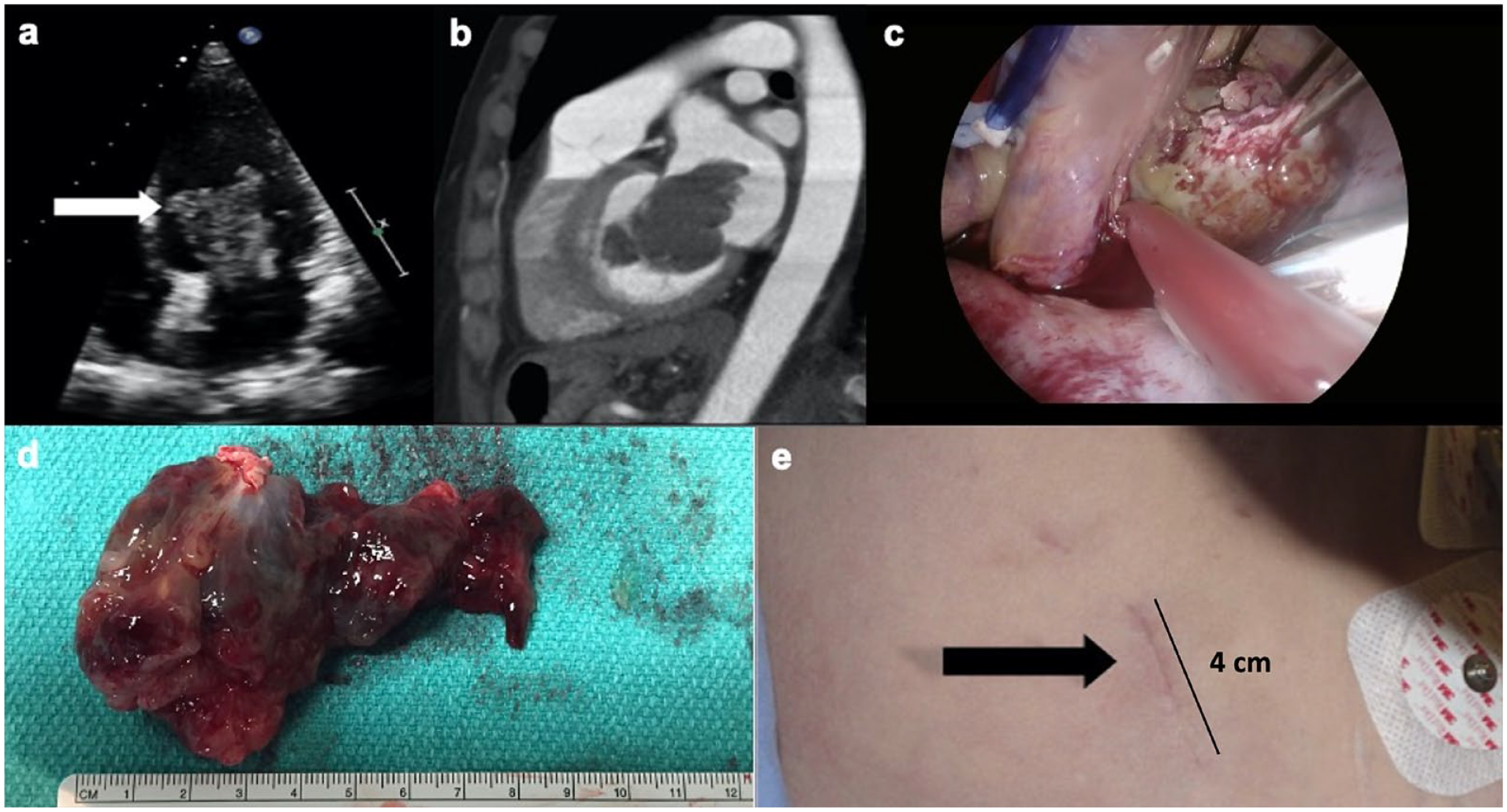
(a) (b) Preoperative transthoracic echocardiography and computed tomography. (c) (d) Endoscopic view and resection of left atrial myxoma (9 cm). (e) Minimally invasive endoscopic surgery incision size.
Baseline Characteristics.

Abbreviations: BMI, body mass index; CCS, Canadian Cardiovascular Society; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; CS, conventional sternotomy; ENDO, minimally invasive endoscopic surgery; IDDM, insulin-dependent diabetes mellitus; NYHA, New York Heart Association; OHA, oral hypoglycemic agent.
Data are reported as mean ± standard deviation or n (%).
There were no patients who experienced in-hospital/30-day mortality or a stroke in either group. In the ENDO and CS groups, the mean cardiopulmonary bypass time was 91 and 94 min (P = 0.80), respectively, and the mean cross-clamp time was 49 and 60 min (P = 0.48), respectively. Most patients were extubated less than 24 h postoperatively. There were no differences in any major complications including respiratory failure, renal failure, new atrial fibrillation, wound infection, or reoperation for bleeding. Very few patients in either group required any blood transfusions.
The median intensive care unit (ICU) LOS was 1 day in both the ENDO and CS groups (P = 0.16), and hospital LOS was 6 versus 7 days (P = 0.57) in the ENDO and CS groups. Complete perioperative details are displayed in Table 2.
Perioperative Characteristics.

Abbreviations: CS, conventional sternotomy; ENDO, minimally invasive endoscopic surgery; ICU, intensive care unit.
Renal failure defined by the Acute Kidney Injury Network as an abrupt rise of creatinine, within 48 h, by more than 200% from baseline. 12
Data are reported as mean ± standard deviation, median (interquartile range), or n (%).
Tumor Characteristics and Late Clinical Outcomes
The pathological findings of the resected cardiac tumors are summarized in Table 3. In summary, most of the resected tumors were benign, comprising myxomas (56%), papillary fibroelastoma (9%), thrombus (9%), and renal tumor caval thrombus (6%). Malignant masses were rare, comprising sarcomas (3%) and follicular thyroid carcinomas (3%). The mean tumor sizes between the ENDO and CS groups were similar (3.9 ± 2.8 × 2.4 ± 1.4 × 1.9 ± 1.2 cm vs 4.7 ± 3.3 × 3.3 ± 2.1 × 3.0 ± 2.1 cm, P = ns).
Tumor Characteristics.

Abbreviations: APLS, antiphospholipid syndrome; CS, conventional sternotomy; ENDO, minimally invasive endoscopic surgery.
Data are reported as mean ± standard deviation or n (%).
The details of the follow-up outcomes are outlined in Table 4. In summary, patients in the ENDO and CS groups had a similar postoperative follow-up duration (42 [IQR 2 to 131] months vs 54 [IQR 1 to 156] months, P = 0.20). At the latest follow-up assessment, neither group had evidence of tumor recurrence, stroke, peripheral embolization, reoperation, or arrhythmia. However, 1 patient in the ENDO group had moderate mitral regurgitation, which was treated with medical optimization and observation. There were 2 deaths during the follow-up period, both from the CS group at 33 days and 101 months, respectively (P = 0.21; Fig. 3). The first patient suffered a sudden cardiac arrest and was unable to be resuscitated. The second patient was a very elderly, frail nonagenarian who was suffering from multiple medical comorbidities and had elected for medical assistance in dying, 8 years after surgery.
Follow-Up Outcomes.

Abbreviations: CS, conventional sternotomy; ENDO, minimally invasive endoscopic surgery; MR, mitral regurgitation.
Data are reported as median (interquartile range) or n (%).
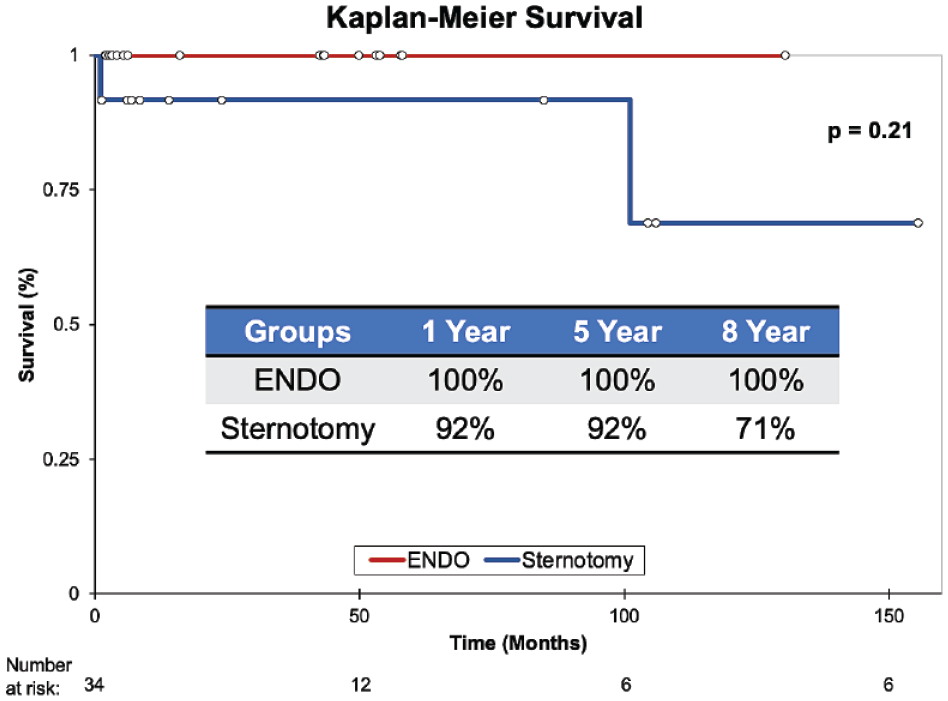
Kaplan–Meier survival graph for ENDO approach (red) compared with conventional sternotomy (blue). ENDO, minimally invasive endoscopic.
Discussion
Our results describe the use of ENDO and CS for successful cardiac tumor resection in 34 patients. We included all-comer patients with cardiac tumors in this study to report early outcomes but more importantly to investigate long-term tumor recurrence risks, specifically with the ENDO approach compared with CS. In our study, most of the patients were female, most cardiac tumors were found in the left atrium, and myxoma was the most common diagnosis, all of which predispose nicely to an ENDO approach, similarly to other previous studies. 13 In our experience, we were able to demonstrate similarly excellent late freedom from recurrent tumor disease, regardless of surgical approach. However, we were unable to demonstrate the usual benefits of the ENDO approach such as shorter hospital LOS, transfusion benefits, and lower complication rates. This could be attributable to the small numbers of patients and inadequate power of the study as well as the relatively low complication rates in either group. Importantly, there was no significant difference in long-term survival or tumor recurrence between the groups, as demonstrated in the Kaplan–Meier curves (Fig. 3).
ENDO techniques have been well developed for valve repair and replacement, atrial septal defect repair, arrhythmia surgery, coronary artery bypass, and other operations. It has been a logical and understandable step forward to apply these advanced techniques to the resection of localized cardiac tumors.6,14 Robot-assisted cardiac tumor resection has also been demonstrated with good success in shortening hospital LOS and postoperative pain benefits. 15 Many studies have demonstrated that minimally invasive approaches for cardiac tumor resection are safe, reduce postoperative pain, decrease ICU and hospital LOS, and decrease hospital resource utilization.9,16 Patients may benefit from minimally invasive approaches with decreased postoperative pain because incisions are smaller and there is less manipulation of the thorax compared with standard CS approaches. Our own results suggest these trends, but failed to reach statistical significance, likely related to the small sample size. Other studies have also demonstrated that minimally invasive approaches to cardiac tumor resection did not influence the size of the tumor removed, the resection margins, or recurrence rates.9,16 This is in keeping with our findings in Table 4.
There are no randomized trials, and most of these studies suffer from small sample sizes with limited follow-up and all the usual limitations of small observational cohort studies. Nonetheless, patients have a strong desire for less invasive approaches without compromising the success of the tumor resection. There is growing evidence of the success of minimally invasive and robotic techniques for the resection of malignant pulmonary tumors without compromising resection margins or cancer-free survival. 17 Thus, it would logically make sense that in selected patients, complete cardiac tumor resection should be feasible with similarly good tumor-free results, especially considering that most cardiac tumors are benign and far more localized than lung tumors. In our experience, most cardiac tumors can be approached with the ENDO technique and will depend on surgeon experience. However, tumors requiring large cardiac chamber resection, multiple chamber exploration and reconstruction, large amounts of ventricular free wall reconstruction, or right ventricular outflow tract and pulmonary artery tumors are likely best treated through a CS approach.
Limitations
This study is limited by the inherent confounding effects associated with a retrospective analysis and the relatively small sample size of our series. In our study, the relatively small absolute sample size may have resulted in the study being underpowered to detect small differences in safety and efficacy. Furthermore, all cases were performed by a single surgeon well experienced in minimally invasive cardiac surgery. Therefore, these results may not be generalizable to other low-volume minimally invasive centers. Finally, there may be unmeasured confounding variables that resulted in the surgeon opting for an ENDO versus CS approach. Despite these early confounders, there did not appear to be a difference between midterm ENDO and sternotomy-based outcomes.
Conclusions
In our 12-year single-center retrospective analysis of cardiac tumor resection, we demonstrated that an ENDO surgical approach for resection of cardiac tumors is a feasible and less invasive approach for tumor resection compared with a CS. Further investigation is warranted.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.W.C. is supported as the Ray and Margaret Elliott Chair in Surgical Innovation and has received Speaker’s honoraria from Medtronic, Edwards, Terumo Aortic, and Artivion.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
