Abstract
Objective:
Safety and sustainability are key elements of a robotic mitral valve (MV) program at any stage of development. Challenges include the positioning of the surgeon at the robotic console, increasing patient complexity, and upstream administrative staffing difficulties. We instituted a systems approach to maximize patient safety and maintain robotic service viability.
Methods:
A single dedicated robotic operating room (OR) was equipped as a microsystem with team training in the operative steps, ergonomics, digital tools, and an explicit culture of safety. Outcomes of all robotic mitral procedures including concomitant procedures in the microsystem OR by a single surgeon were retrospectively reviewed.
Results:
From January 2014 through December 2023, 1,529 consecutive MV patients were operated with an endoscopic robotic approach. Ten patients (0.65%) were converted to conventional approaches. Overall, 1,300 MV repairs (85%) were performed with residual MV regurgitation of none to trace in 1,205 patients (92.7%), mild in 92 patients (7.1%), and moderate in 3 patients (0.23%). MV replacements were performed in 229 patients (15%) with no paravalvular leaks. Mortality was 0.08% in the repair group and 0.87% in the replacement group. No deaths have occurred in the last 38 months. Stroke occurred in 0.31% of repair patients and 1.3% of replacement patients. One patient developed transient renal failure.
Conclusions:
Organization of the robotic OR as a microsystem is associated with surgical efficacy and very low morbidity and mortality. A comparable microsystem approach using all or select components may promote safety and sustainability for robotic MV programs at all levels.
Central Message
A systems safety approach was instituted in a single robotic mitral OR creating a microsystem. A heterogeneous population of mitral patients underwent an endoscopic robotic approach with satisfactory technical outcomes and very low morbidity and mortality.
Introduction
Robotic instrumentation affords the surgeon the ability to perform mitral valve procedures through right chest ports using telemanipulation. While prospective patients typically focus on the temporary benefit of skeletal preservation, robotic surgeons must focus on efficacy and safety. 1 The unique characteristic of robotic surgery that affects these 2 objectives is the repositioning of the surgeon to the robotic console to control the instruments. Efficacy, as defined by the exact replication of conventional mitral procedures, required reengineering of surgical tasks using the assistance of patient-side personnel. 2 These challenges have been overcome, and numerous groups have published robotic mitral series demonstrating efficacy in complex and heterogeneous patient populations.3–5
Although efficacy with robotic instrumentation has been widely reported, the potential compromise to patient safety by the surgeon repositioning to the robotic console has received little scrutiny.6,7 This robotic operating room (OR) paradigm shift can compromise safety in several ways. First, critical communications between the console surgeon and the OR personnel can be compromised. Second, the surgeon, while immersed in the robotic endoscopic visual field, can lose situational awareness of the patient’s status. Third, the surgeon cannot visualize the actions of the assisting team members positioned at the operating table.
Early in our experience, we compensated for these safety challenges by using a minimally invasive surgeon as the patient-side assistant,2,8 limiting robotics to low-risk patients with simple mitral pathologies and few comorbidities, 9 and maintaining a small dedicated robotic team.3,7,10–12 All 3 compensation methods proved to be unsustainable due to surgeon productivity issues, organic growth with more complex patients, and eventual administrative staffing challenges. We evolved to midlevel patient-side assistants and a large pool of robotic team members. Rotating team members, often with less experience and often interchanging during a procedure, became the norm. It seemed unlikely that a complex endeavor like robotic mitral surgery could survive a competency challenge.
In 2014, to maintain safety and ensure the sustainability of the maturing robotic mitral program, we progressively instituted a systems safety approach emulating the practices of high-reliability organizations.7,13–16 A systems approach functions under the premise that any complex activity, such as robotic mitral surgery, performed by humans interacting with technology will routinely experience errors. These errors may be most prevalent when the surgeon is positioned at the console away from the operating table and may result in permanent complications. 6 The systems approach functions to prevent these errors when possible or to provide identification and mitigation when not. The goal is zero permanent complications.
The cardiac surgeon operating at the robotic console and a commercial pilot flying in meteorologic conditions have similar communication and situational awareness requirements, and both function under time constraints. 17 These similarities led to the application of several aviation error-reduction methodologies, nuanced to robotic cardiac surgery, to create a systems approach in a single dedicated OR—in essence, a microsystem.14,18
This microsystem is comprised of 3 components. 19 First, the steps and substeps of the robotic procedure are organized by their potential to injure organs or critical structures if omitted, performed incorrectly, or performed out of sequence. All team members are trained to focus on these steps. Second, the microsystem employs both ergonomic factors and digital information tools. Third, both implicit and explicit efforts are made to create a team culture focused on patient safety.
We have reported the outcomes from the first 12 years of our endoscopic robotic mitral experience.2,3 This report details the endoscopic robotic outcomes of mitral patients operated over the ensuing decade by a single surgeon in an OR specially equipped as a microsystem.
Methods
From January 2014 to December 2023, clinical and operative data from 1,529 consecutive endoscopic robotic mitral valve procedures performed by a single robotic surgeon in the microsystem OR were prospectively collected and merged with the Society of Thoracic Surgeons (STS) institutional database. A retrospective review of this mitral valve database forms the basis for this report. All mitral valve procedures including concomitant left or right atrial procedures with the intent of using robotic instrumentation were included. The study was approved by the Institutional Review Board of Emory University. Patient demographics and outcomes are described as count (percentage) or mean ± standard deviation. The preoperative preparation, technical operative steps, and nontechnical OR environment are described.
Preoperative Steps
All patients had a computerized tomographic angiogram to plan peripheral perfusion, aortic occlusion, and port placement. A preoperative robotic worksheet was scanned into the electronic medical record and made available to the operating team prior to the day of surgery. On the day of surgery, an initial briefing in the preoperative area was performed by the surgeon and the anesthesia team regarding the plan for perfusion.
Technical Operative Steps
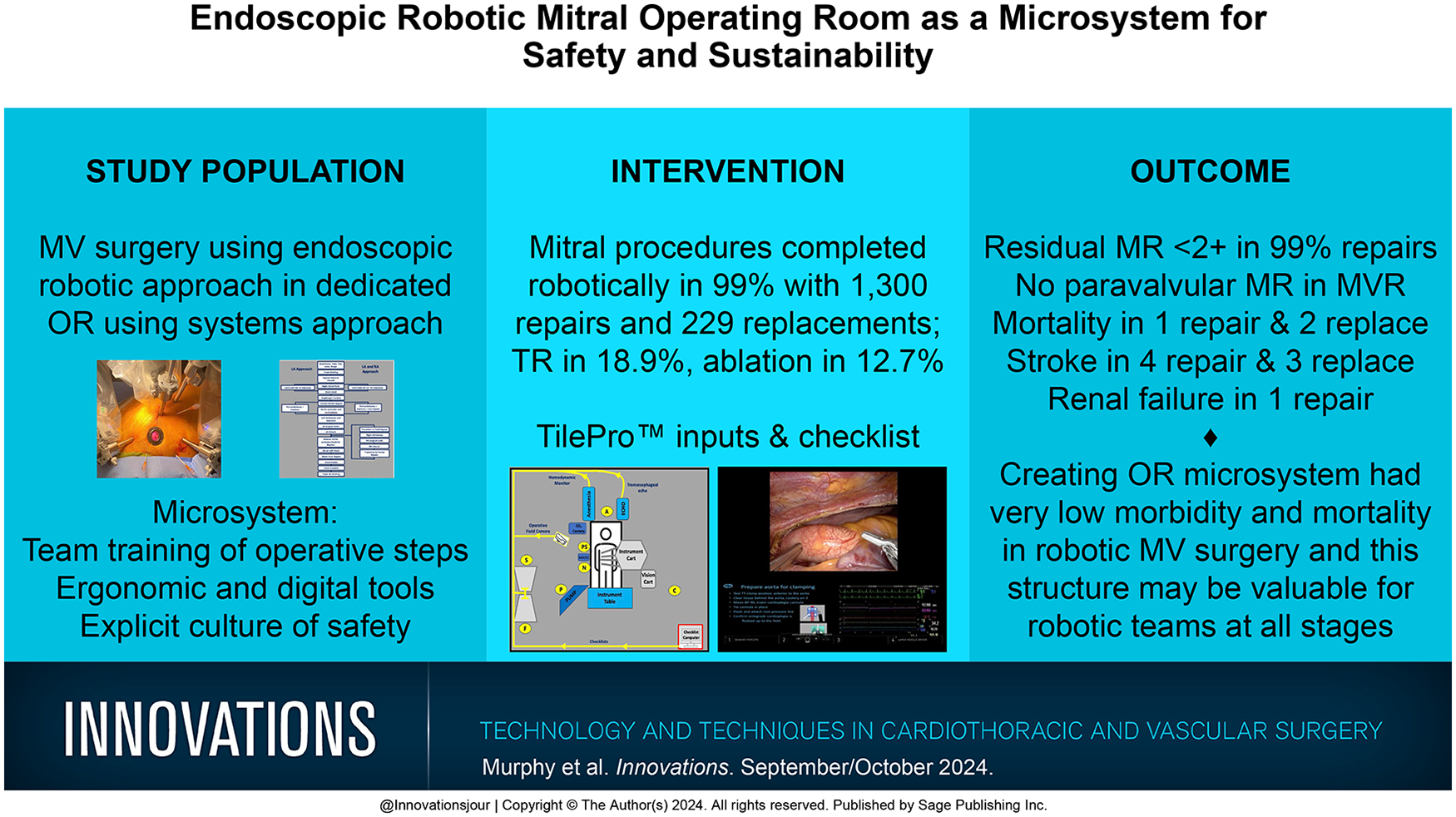
The operative steps of an endoscopic robotic technique for mitral surgery are summarized in Figure 1 as previously described.2,3 Procedures were performed with four 8 mm ports for the robotic instruments and a 15 to 20 mm port for patient-side assistant access. All robotic surgical procedures were replications of known conventional procedures performed with peripheral perfusion, balloon or clamp aortic occlusion, and robotic instruments working in conjunction with shafted instruments controlled by patient-side assistants (Fig. 2).3,20–22
Endoscopic robotic mitral steps. LA, left atrial; RA, right atrial; TEE, transesophageal echocardiography.
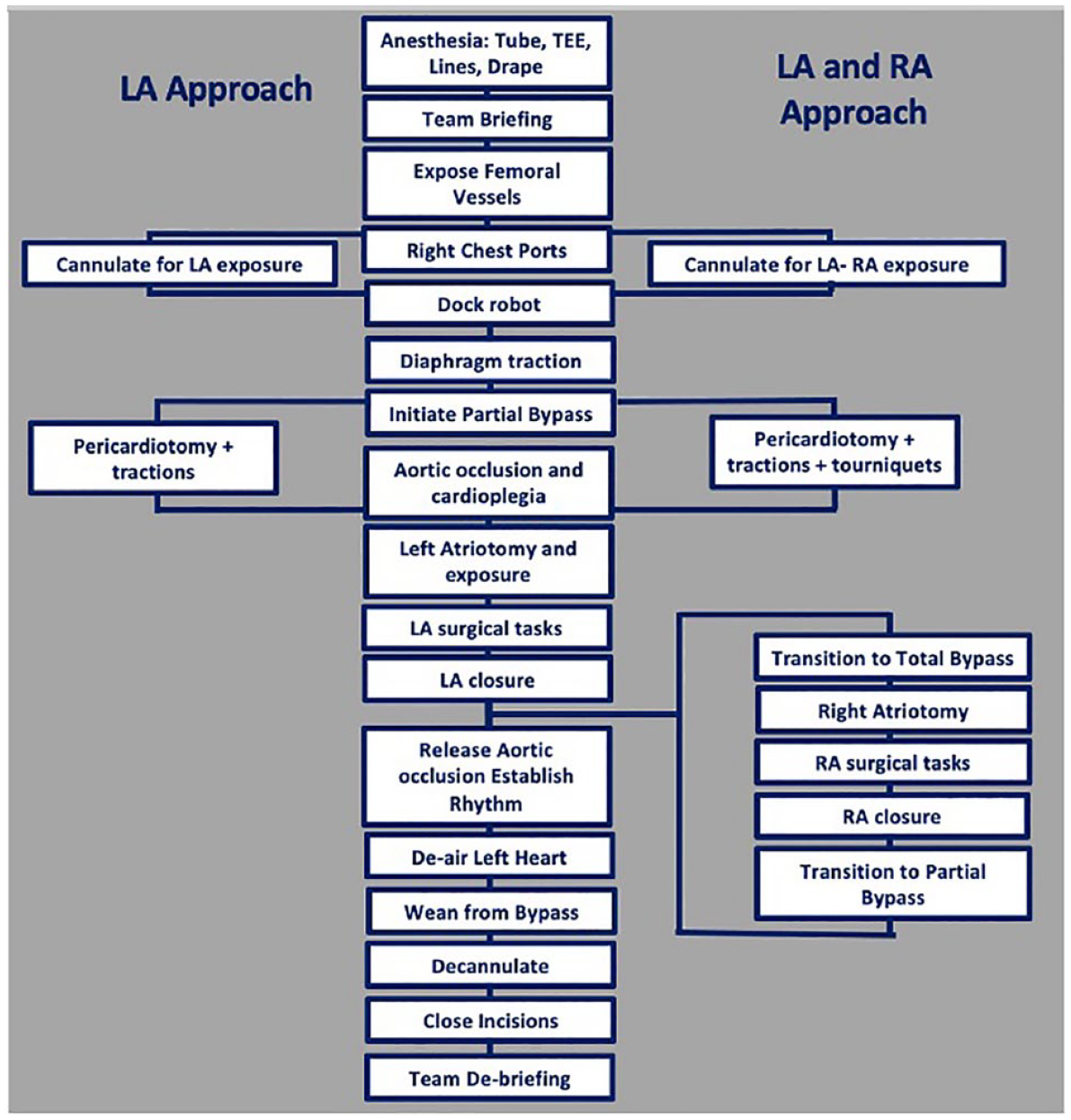
(a) Right chest robotic ports. (b) Robotic instruments collaborating with patient-side assistants.
Nontechnical Environment
Briefing
Following patient identification and prior to incision, the robotic surgeon conducted a briefing summarizing the patient’s cardiac disease, perfusion and port placement plan, and surgical task intentions (Fig. 3). A preoperative photo of the patient was projected on the OR video screen. The briefing was closed with a request to all team members to report to the surgeon any deviations from the projected steps. 23

Pre-incision team briefing checklist. LAA, left atrial appendage; MV, mitral valve; SAM, systolic anterior motion.
Ergonomics
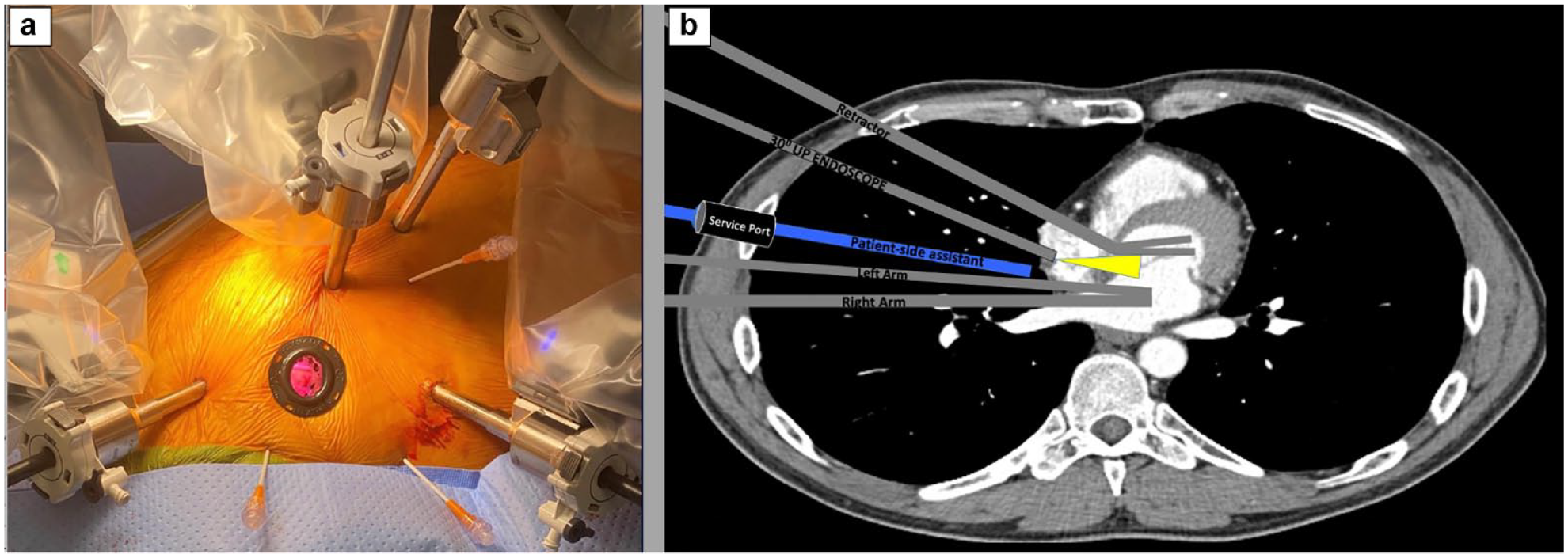
The robotic OR equipment and team members were positioned as depicted in Figure 4a. A pathway was maintained for the scrub nurse from the patient’s left side to the patient’s right side (Fig. 4b). Multiple video screens displaying hemodynamics, robotic console video, and team member checklists were positioned to support both the bedside and console phases of the robotic procedure (Fig. 4c).
Equipment and team member positions (left panel). Pathway for scrub personnel (middle panel). Hemodynamic, robotic, and checklist video screens (right panel).
Checklists
At the initiation of the procedure, the circulating nurse activated the appropriate checklist series based on the perfusion plan, aortic occlusion method, and planned operative tasks. Each team member had a dedicated video screen with operative step checklists specific to their team role. The checklists were customized for each team member, with the console surgeon and patient-side assistant sharing the same checklist content. The checklists with a chime tone alert were advanced by the circulating nurse or the console surgeon.
Robotic console video display
Video information was configured from several digital sources in the OR and input to the TilePro™ (Intuitive Surgical, Inc., Sunnyvale, CA, USA) multi-image display in the robotic console. The TilePro display inputs, selected by the surgeon, include the hemodynamic monitor coupled with transesophageal echocardiography (TEE), right chest operative field camera, operative step checklists, or problem-solving guidelines (Fig. 5).
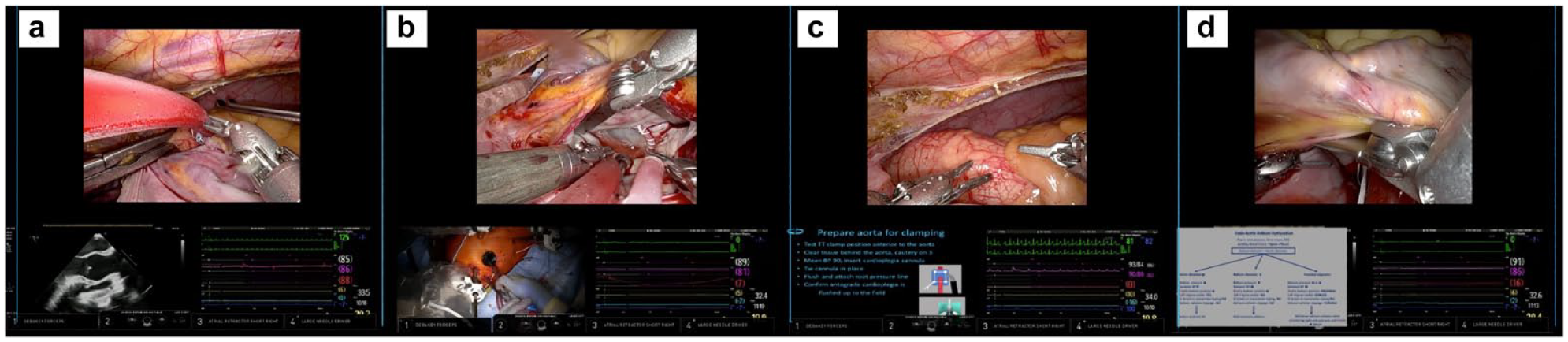
TilePro™ (Intuitive Surgical, Sunnyvale, CA, USA) examples with hemodynamic monitor and (a) transesophageal echocardiography, (b) operative field camera, (c) operative step checklist, and (d) problem-solving algorithm.
Debriefing
During the final technical steps of the procedure with all team members in proximity to the operative table, a debriefing was conducted to review possible modifications of the operative steps. 23 Operative video recordings were available for review.
Results
During the study period, 1,529 patients were operated with the endoscopic robotic approach. The preoperative demographics are presented in Table 1. The surgery was completed robotically in 1,517 patients (99%). Twelve patients (1%) required conversion to a sternotomy approach for mediastinal access, perfusion, or bleeding issues. All converted patients are included in the data.
Preoperative Patient Demographics.
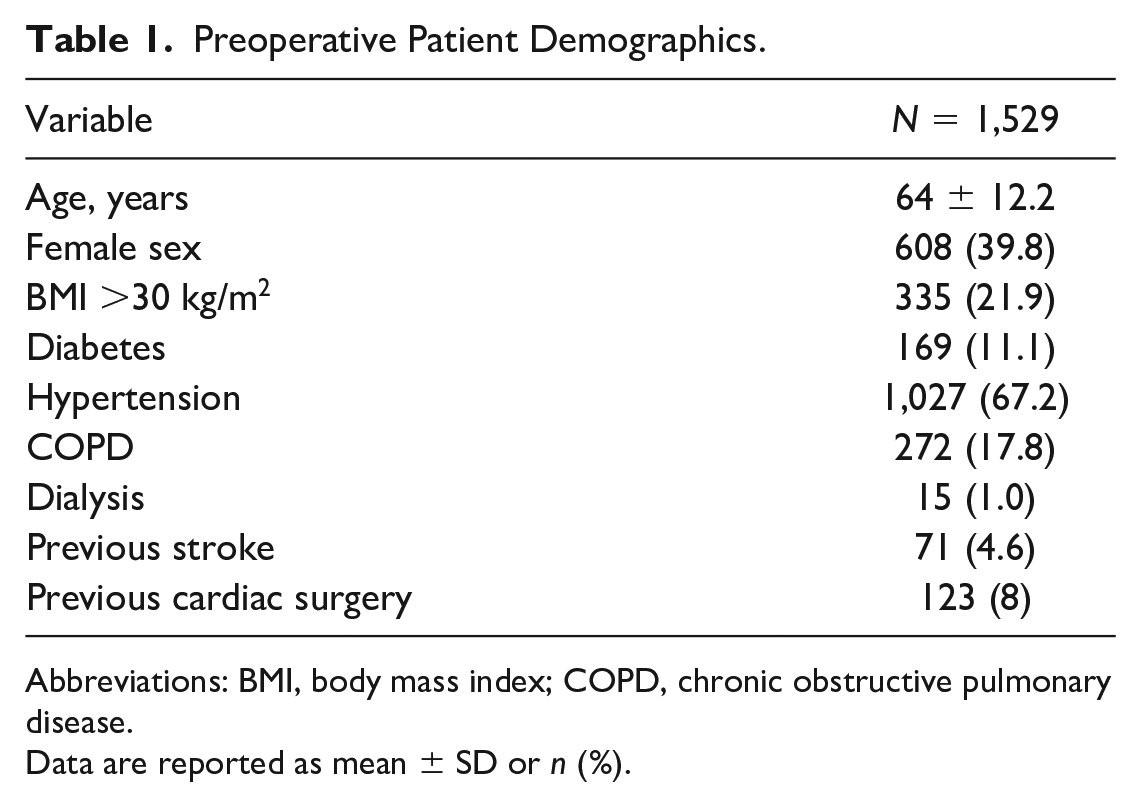
Abbreviations: BMI, body mass index; COPD, chronic obstructive pulmonary disease.
Data are reported as mean ± SD or n (%).
Mitral valve and concomitant procedures are presented in Table 2. Mitral valve repair was achieved in 1,300 patients (85%). Mitral valve replacement was performed in 229 patients (15%) including 179 porcine and 50 mechanical prostheses. No perivalvular leaks were noted on intraoperative echocardiography. No patients required mitral reoperation.
Concomitant Procedures and Operative Times.
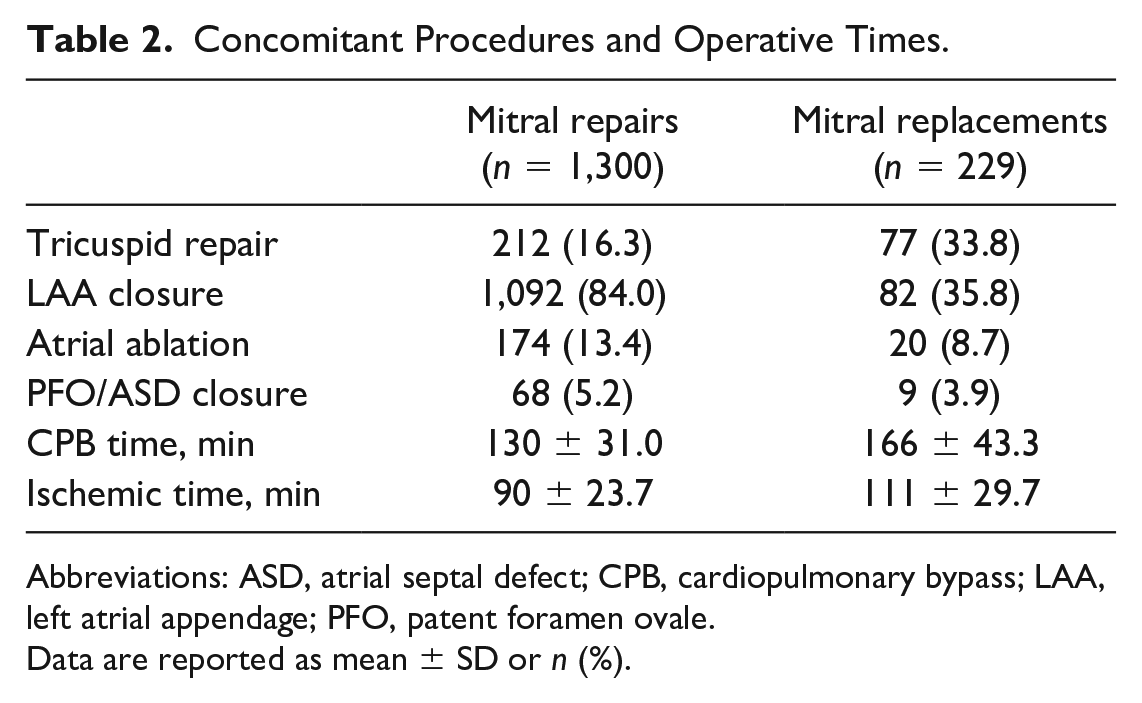
Abbreviations: ASD, atrial septal defect; CPB, cardiopulmonary bypass; LAA, left atrial appendage; PFO, patent foramen ovale.
Data are reported as mean ± SD or n (%).
There were 3 operative mortalities in the series, 1 in the repair group (0.08%) and 2 in the replacement group (0.87%). Postoperative outcomes and the average median STS predicted risks over the 10-year study are presented in Table 3. There were no deaths during the last 38 months of the study period. One episode of transient renal failure occurred in a mitral re-repair patient who has been previously described. 22 Reexploration for bleeding declined to less than 1% in the last 3 years of the study period.
Postoperative Outcomes.
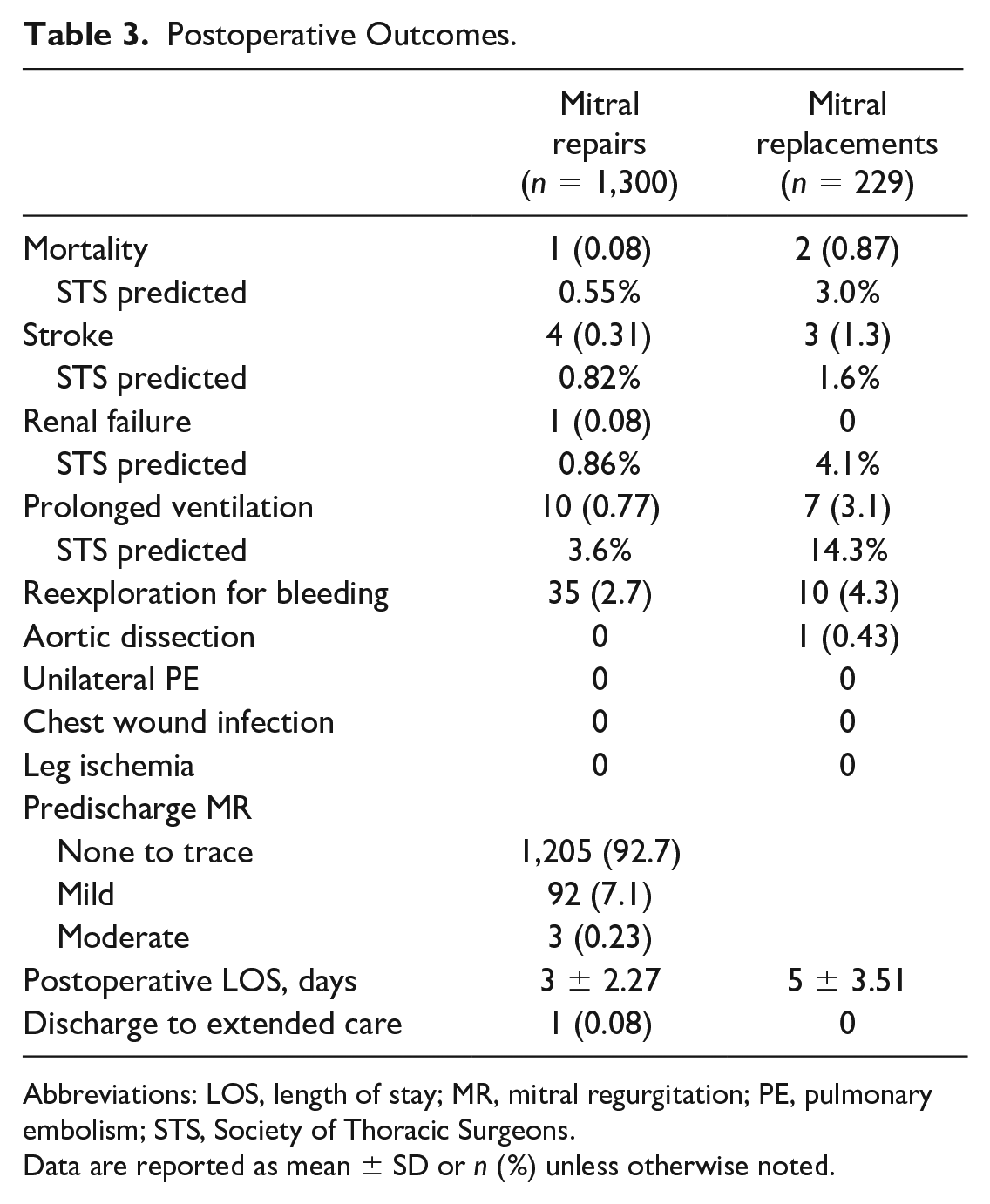
Abbreviations: LOS, length of stay; MR, mitral regurgitation; PE, pulmonary embolism; STS, Society of Thoracic Surgeons.
Data are reported as mean ± SD or n (%) unless otherwise noted.
Discussion
Endoscopic robotic mitral surgery is a tightly coordinated process involving multiple team members engaged in a dynamic organized activity. 24 This retrospective study demonstrates that structuring this organized activity using a systems approach is associated with very low mortality and permanent morbidity in a heterogeneous all-comers patient population, the ultimate desired outcome of any safety intervention. 23 Outcomes of robotic mitral surgery have traditionally been considered a reflection of patient complexity and surgeon ability. 16 However, such a 2-dimensional analysis fails to consider that the technical operative steps, regardless of how meticulously choreographed, are intertwined with the multiple nontechnical elements of the surrounding robotic OR environment.6,16,25,26 The focus of a systems approach for safety is the integration of the technical tasks into a third dimension: the nontechnical environment. Although robotic technology may itself improve patient safety, augmenting this nontechnical environment using a comprehensive error-reduction system may amplify the impact of robotics on patient safety. Applying a systems approach to a single cardiac OR creates a microsystem. 14
Given the complex and dynamic integration of robotic instrumentation with human team members, a microsystem model assumes that errors, missteps, and substep omissions that can potentially permanently injure the patient are likely to occur in nearly every case. Most of these errors and missteps will involve failed team communication or situational awareness lapses.27,28 The design of the microsystem is the prevention of these deviations when possible. Alternatively, when errors or mishaps do occur, their detection is a critical function of the microsystem. This detection affords the robotic team the opportunity to mitigate the problem before permanent injury can occur. While it is not possible to predict what errors or missteps will occur in any given case, the construct of the microsystem is to have a planned response for every known deviation the team has previously experienced or has been reported by other robotic teams.
The endoscopic robotic mitral procedure consists of 3 distinct phases (Fig. 6). Phases 1 and 3, although critical, are performed with the team members in conventional open heart surgery positions. It is in phase 2 when the surgeon relocates to the robotic console that team communications and surgeon situational awareness of patient condition have the greatest potential to be compromised. 6 The systems approach is important for safety at all steps of the procedure but becomes even more critical during the robotic console portion of the procedure.
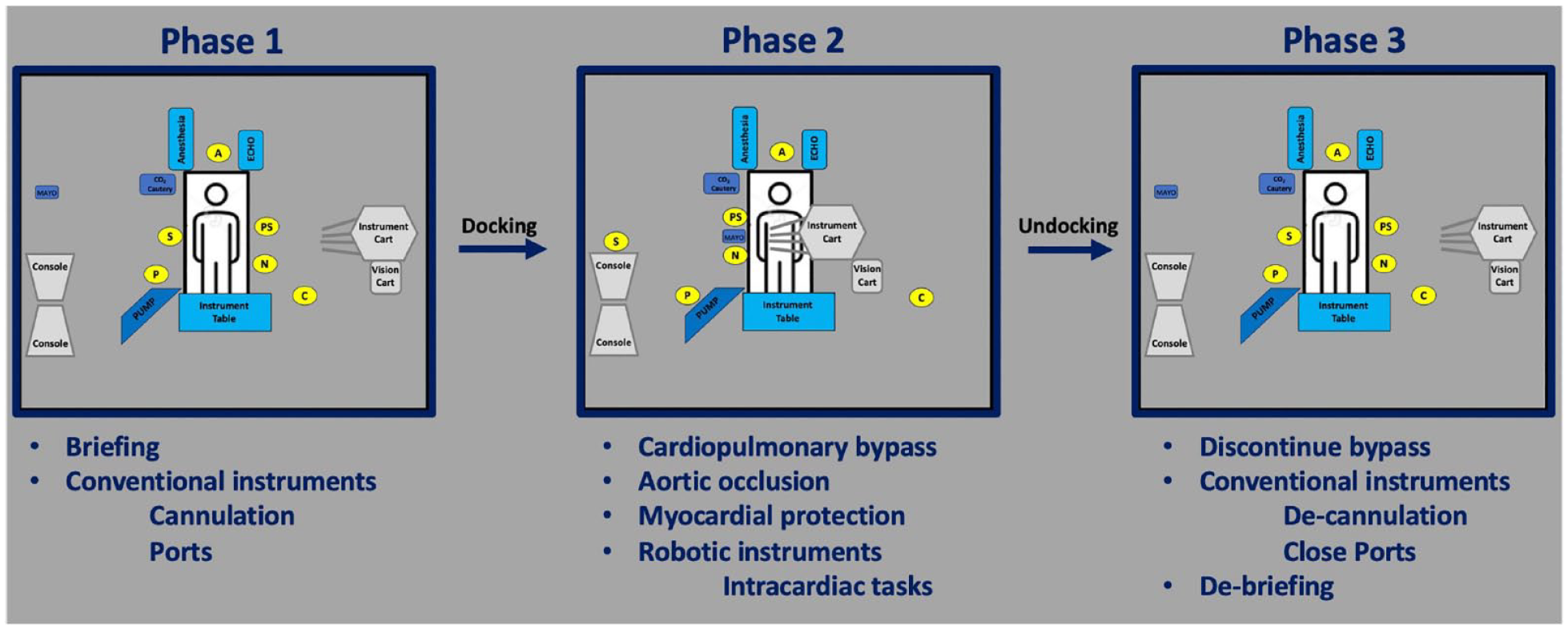
The 3 phases of robotic mitral surgery.
Any compromise in team communication is a known threat to patient safety in cardiac surgery.23,27 While at the console, the surgeon is limited to verbal communication with the team members, either directly or through the robotic sound system. Although the robotic sound system is very effective, it transmits all sounds in the OR to the surgeon. For this reason, the OR is kept as quiet as possible, and all conversations in the room are limited to those that relate to patient care. 29 All team member communications are preceded by the intended receiver’s name and are closed loop in structure. 23 A nomenclature is established for devices and instruments to prevent communication errors. Personnel changes during phase 2 of the procedure are reported to the console surgeon to maintain communication accuracy.
Similarly, the situational awareness data relevant to the patient’s condition that the conventional surgeon normally gleans from viewing the operative field and video monitors is altered when the surgeon moves to the robotic console. The microsystem is designed to restore this critical information to the console surgeon during the console portions of the mitral surgery to reduce errors. 30
The robotic surgeon, similar to the conventional surgeon, has a specific plan of operative steps that they routinely perform from memory. Traditionally, supporting team members learn these steps by repeated exposure through multiple case experiences. With a systems approach, the steps and substeps of the surgeon’s plan are explicitly shared with all team members. This educational process accelerates the team member awareness of the operative plan and is aimed at orchestrating team interactions. 24 The team member education focuses not on how the surgeon technically performs the sequential surgical tasks but rather on how their supportive roles augment efficacy and safety at multiple points in the overall operative procedure architecture. The microsystem then employs numerous tools to complement this distributed cognition to maximize orientation, communication, and situational awareness for the entire robotic team. 19 These tools include ergonomic factors, video images displayed on the TilePro feature of the robotic console, video-displayed team member checklists, and problem-solving guidelines.
A key feature of the room ergonomics is the positioning of the robotic console and the pump adjacent to the right chest operative field, as shown in Figure 4a. This configuration allows the console surgeon to disengage from the console viewer during robotic workflow pauses, directly visualize the right chest operative field, and maintain a view of the hemodynamic monitor and a 2-dimensional robotic video monitor while communicating directly with the patient-side assistants and the perfusionist. We have found these transient pauses to reduce surgeon fatigue on longer cases. This positioning also enhances the critically important communication between the surgeon and perfusionist.
A second ergonomic feature is the positioning of equipment to allow the scrub technician to reposition to participate as the second patient-side assistant during phase 2, reducing staffing and promoting cross training (Fig. 4b).
The third important feature of the room ergonomics is the strategic positioning of 3 types of video monitors (Fig. 4c) around the OR. First, hemodynamic monitors are viewable continuously for all team members. Second, the robotic console video is viewable by all team members during phase 2 of the procedure. Visualizing the robotic operative field orients the team member to the current surgical step and allows them to anticipate what they need to perform on the next step. 31 Third, each team member has a dedicated video screen displaying an operative step checklist customized to their role. Ergonomic details are presented in Supplemental Video 1.
Early in our robotic experience, we created operative step checklists that could be visualized by the surgeon in the TilePro feature of the robotic console. The basic steps of endoscopic robotic mitral surgery have not significantly changed over the past 2 decades, but the substeps have undergone continuous modification to identify threats and improve safety and efficiency. This process has been fueled by lessons identified during the debriefing step. At the beginning of this study period, the operative step checklist model was extended to all team members. These checklists, visible on dedicated team member video screens and console TilePro, are customized for each team member as they relate to the surgeon’s specific robotic technique. These checklists are designed to promote safety in multiple ways. 32 First, they enhance team member situational awareness, orienting any new personnel entering the OR and promoting prospective planning for the next step. 33 Second, they enhance team communication by requesting tasks to be performed at specific operative steps without verbal communication from the surgeon. Third, they instruct team members to report pertinent data at specific operative steps to augment surgeon situational awareness. Fourth, the checklists promote team member reporting to the surgeon of any unusual occurrences or deviations from standard procedure. Such deviations can create workflow interruptions or cause injury to organs or other critical structures. This relationship is comparable to the airplane copilot cross-checking the pilot’s actions to prevent omissions or deviations. Fifth, checklists can employ forcing functions compelling the surgeon to consider all possible causes of a clinical aberration rather than act with hindsight bias. 34 Sixth, the team member checklists have been used extensively in the training of new personnel by established robotic team members. Lastly, team members can also visualize the checklists of other team members promoting mutual knowledge of their respective roles. This mutual cognition can promote cross-checking and supportive activities. Complex activities such as aortic occlusion by clamping or balloon inflation require coordination of multiple team members. Review of step checklists with the entire team prior to these actions can promote this team member coordination. Some error-reduction processes, such as prevention of brain injury or unilateral pulmonary edema, involve multiple substeps that are woven through the checklists of multiple steps and may trigger prospective sensemaking by the surgeon. 31 Examples of checklist function are presented in Supplemental Video 2.
The microsystem expands the TilePro video capability of the robotic console to supplement verbal input from team members and enhance the surgeon’s situational awareness of the patient status. This feature provides video input that mirrors the visual data the surgeon normally acquires by direct vision during conventional surgery. Like the airplane pilot who maintains an instrument scan, the robotic surgeon can maintain a scan of patient status. 35 TilePro allows the surgeon to select 2 video inputs to supplement the robotic endoscope display. The hemodynamic monitor, because of its critical importance, is consistently used as 1 input. The second video input can be the TEE, a camera on the chest operative field, the checklist of the current operative step, or an instructive guideline or algorithm from the OR computer. Examples of the TilePro function are presented in Supplemental Video 3.
The situational awareness data collected from TilePro inputs and the checklist-generated verbal communications provide the console surgeon with a comprehensive mental picture of the patient’s status and may reduce errors. 30 However, the transmission of this situational awareness data to the console surgeon is only the initial step in safe patient management. The surgeon is continually analyzing the data, recognizing deviations, anticipating the patient’s trajectory, and executing remedial action. Although problem-solving guidelines are available on TilePro, it is more practical for the surgeon to have problem-solving solutions as part of stored memory as there is no autopilot function on the robot. 28 The surgeon must analyze the situational awareness data using this stored memory and request more detailed input from team members to resolve a deviation and then revisit the situational awareness data to confirm correction of the problem (Fig. 7). An example of this dynamic is depicted in Supplemental Video 4.
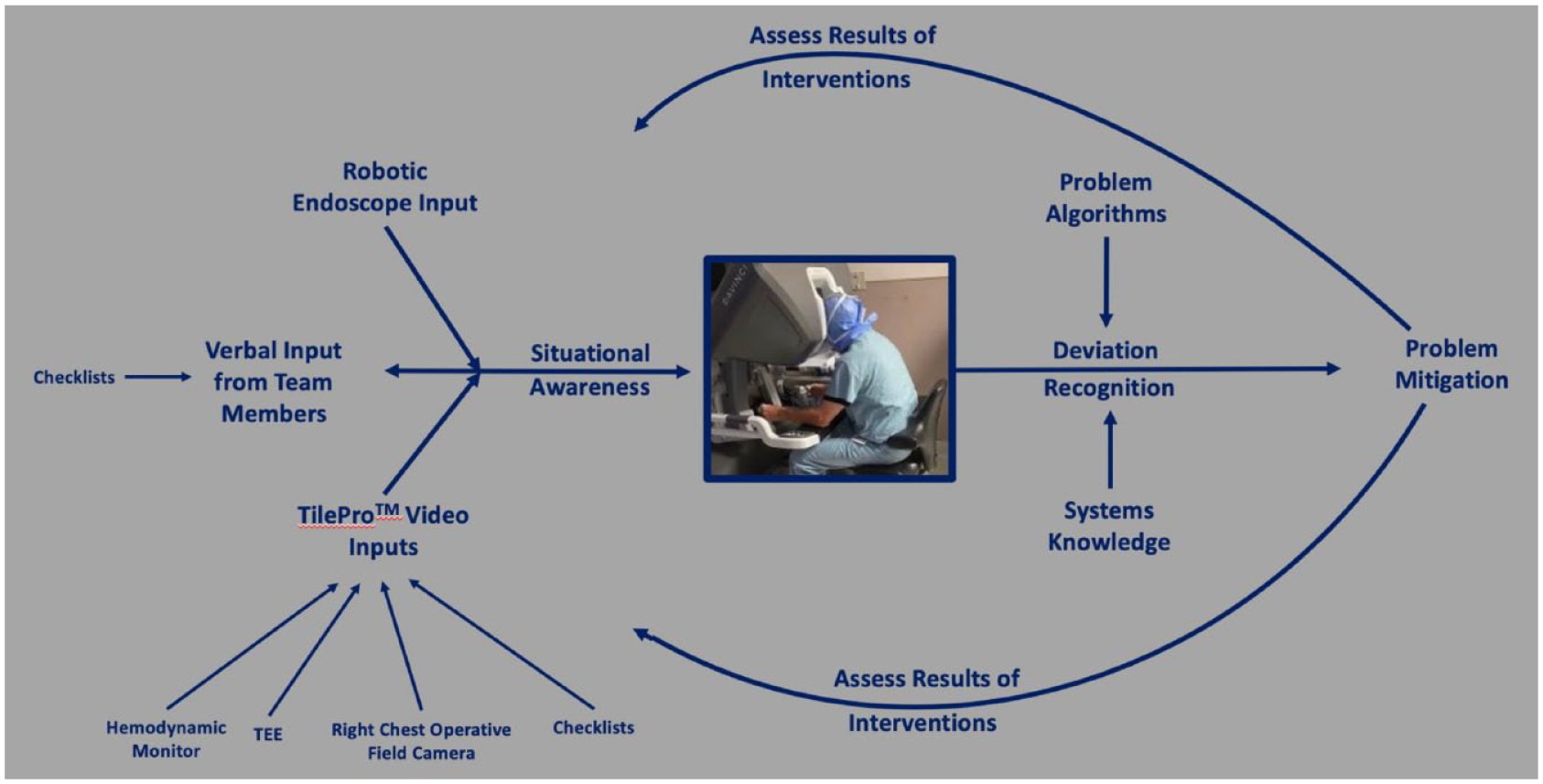
A schematic of situational data analysis, deviation recognition, mitigation, and confirmation of correction. TEE, transesophageal echocardiography.
For the systems approach and the tools of the microsystem to be effective, the OR must have the third key component, which is a culture of safety.19,23 Contributing factors include a quiet OR environment, video screens with checklists, and formal briefings and debriefings. Projecting a patient photo, often with family members, personalizes the priority of safety to the team. However, surgeon demeanor is the most important factor creating this safety-focused environment.23,25 This behavior includes projecting a shallow hierarchy and recognizing all deviations as team errors rather than individual errors. This culture of safety promotes the debriefing step as a critical component in the evolution of the OR microsystem. Forthright team member input from these debriefings is valuable for modifications of the operative steps and incorporation into team member checklists. Using checklists for preservation of the lessons learned, an aviation policy, enhances experience-related learning and disseminates the knowledge to personnel absent during the index cases.36,37
While select modifications are incorporated into the operative technique, microsystems exhibit a reluctance to simplify the operative steps. 14 The problem with simplification is in predicting the patients in whom given substeps become lifesaving. Maintaining a comprehensive operative approach to avoid patient vulnerability is a key feature of the robotic OR microsystem.
While initiating an endoscopic robotic mitral valve program is a significant undertaking, sustaining a robotic mitral program may be an even greater challenge, as evidenced by the failure of nearly half of the programs that make the initial effort. 38 Sustainability is difficult for 2 significant reasons. First, organic growth with more technically complex patients naturally follows initial success.21,22 Organic growth is mandatory for most programs to increase operative volume and enhance proficiency. Second, the surgeon has little influence over the upstream administrative compromises necessary to maintain staffing in the robotic OR. However, the surgeon can influence the creation of a microsystem OR environment to manage more complex patients and compensate for team member variability.14,39
The technical mitral valve surgical tasks in the present study were performed with very low mortality and morbidity. The contribution to this procedural safety generated by the introduction of a systems approach into the study robotic OR is difficult to separate from other possible contributing factors, including referral bias and overall institutional robotic mitral experience. No randomized trials were performed to analyze the relative contributions of the 3 components of the systems approach. We have experienced the tightly linked nature of the systems approach components and have elected to maintain a comprehensive utilization of all components. Broad acceptance by robotic surgeons and team members has motivated the institution to equip a second robotic OR as a microsystem.
Conclusions
Organizing the robotic mitral OR as a microsystem is associated with low mortality and morbidity. A microsystem OR may benefit inaugural programs or lower-volume programs experiencing significant time intervals between procedures. Higher-volume programs facing more complex cases and changing team dynamics may also benefit from a microsystem approach for safety and sustainability.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Douglas A. Murphy is a consultant to Intuitive Surgical, Inc. and Edwards Lifesciences.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
