Abstract
Objective:
We conducted a systematic review of all available evidence on the feasibility and safety of minimally invasive coronary artery bypass grafting (MICS CABG) in patients with multivessel coronary artery disease (CAD).
Methods:
A systematic literature search in PubMed, MEDLINE via Ovid, Embase, Scopus, and Web of Science was performed to identify all relevant studies evaluating outcomes of MICS CABG among patients with multivessel CAD and including at least 15 patients with no restriction on the publication date.
Results:
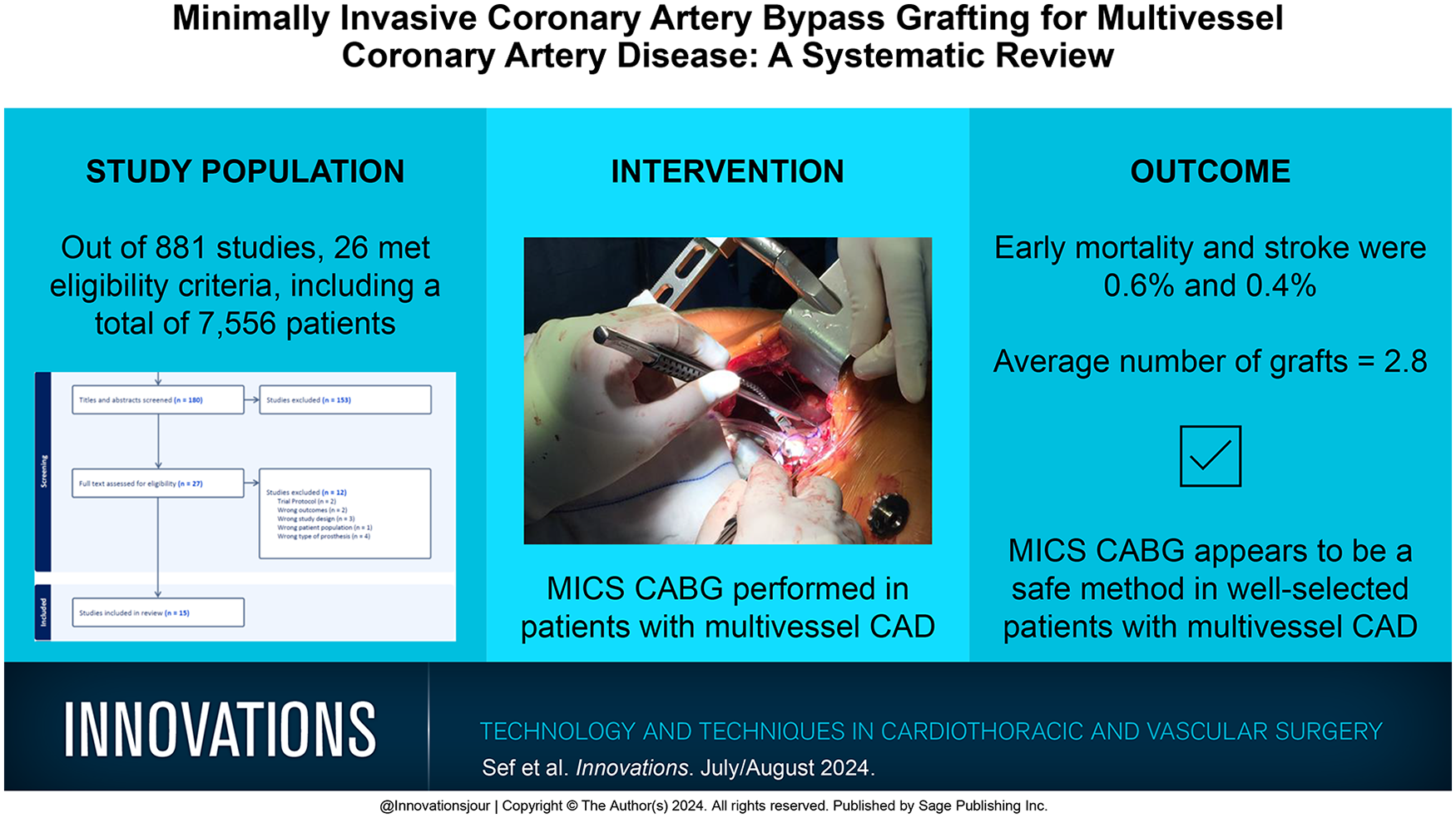
A total of 881 studies were identified, of which 26 studies met the eligibility criteria. The studies included a total of 7,556 patients. The average patient age was 63.3 years (range 49.5 to 69.0 years), male patients were an average of 77.8% (54.0% to 89.8%), and body mass index was 29.8 kg/m2 (24.5 to 30.1 kg/m2). Early mortality and stroke were on average 0.6% (range 0% to 2.0%) and 0.4% (range 0% to 1.3%), respectively. The average number of grafts was 2.8 (range 2.1 to 3.7). The average length of hospital stay was 5.6 days (range 3.1 to 9.3 days).
Conclusions:
MICS CABG appears to be a safe method in well-selected patients with multivessel CAD. This approach is concentrated at dedicated centers, and there is no widespread application, although it has potential to be widely applicable as an alternative for surgical revascularization. However, large randomized controlled studies with longer follow-up are still required to compare the outcomes with conventional CABG and other revascularization strategies.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
