Abstract
In the evolving landscape of cardiac surgery, this article explores the potential of minimally invasive mitral valve replacement procedures as a viable alternative to conventional surgical techniques. Leveraging advancements in automated suturing devices and video endoscopy, our work aims to demonstrate that minimally invasive approaches can be applied across a broad spectrum of surgical scenarios. Herein we highlight preoperative diagnostics and operative techniques, with a focus on infra-axillary anterolateral minithoracotomy as the access point. Our technique utilizes technology from LSI SOLUTIONS® (Victor, NY, USA), including the RAM® Device for automated suturing, which has an ergonomic design and safety features. The device’s capabilities are further enhanced by the SEW-EASY® Device, the RAM® RING, and the COR-KNOT MINI® Device, which streamline suture management and securement. This work outlines how these technological advancements can mitigate concerns about technical complexity and learning curves, thereby encouraging wider adoption of minimally invasive techniques. Clinical benefits may include reduced surgical trauma, quicker recovery, and cost-effectiveness, making it a compelling option in an era of aggressively promoted transcatheter interventions.
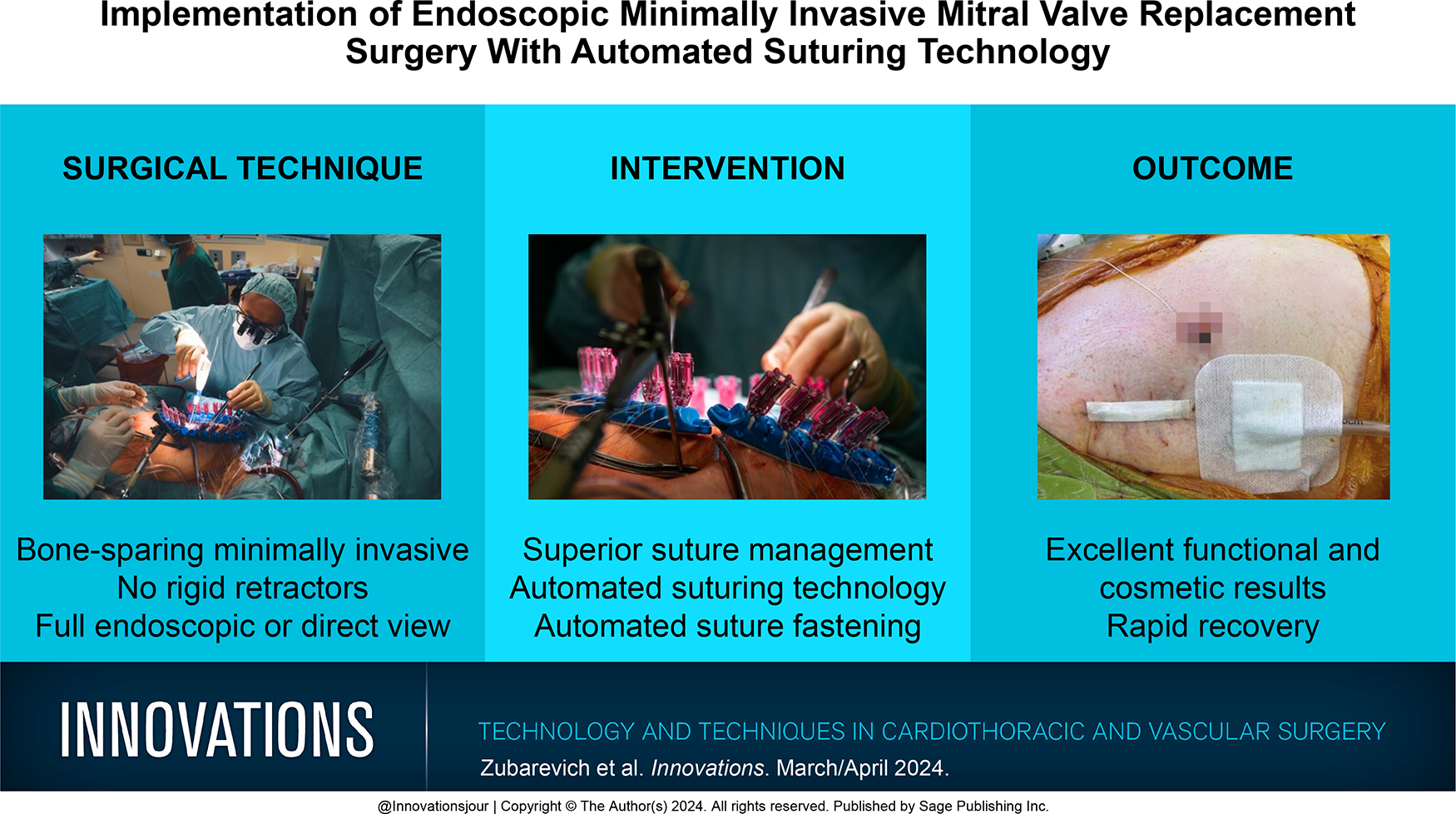
Central Message
Our work aims to demonstrate that minimally invasive approaches can be applied across a broad spectrum of surgical scenarios. Cutting-edge technologies, such as automated suturing devices, offer invaluable support.
Introduction
Over the past decade, the medical field has increasingly embraced less invasive, bone-sparing surgical techniques, offering patients numerous benefits such as reduced surgical trauma, quicker recovery times, and improved cosmetic outcomes. This shift is particularly significant in the context of rapidly evolving transcatheter technologies and the expansion of interventional treatments to moderate-risk and low-risk patient groups. 1 As transcatheter technologies and other interventional treatments continue to advance and expand their indications, traditional surgical disciplines find themselves at a crossroads. The imperative is clear: cardiac surgery should adapt to the technical progress. Adopting less invasive surgical approaches can not only enhance patient care but also reduce hospital and intensive care unit (ICU) stays, thereby lowering overall health care costs. During the past 20 years, several institutions have reported their experience with minimally invasive mitral valve procedures and have demonstrated their noninferiority regarding postoperative survival, morbidity, and safety compared with conventional surgical methods.2 –4
However, the widespread adoption of surgery with much less iatrogenic trauma has been hindered by several challenges, including surgeon apprehensions about the increased technical complexity and the steep learning curve associated with these procedures. In this article, we offer some insight regarding the development of our minimally invasive approach to mitral valve surgery with less surgical trauma due to even smaller incisions with no rigid bone retractors. We demonstrate how the use of automated suturing devices and modern video endoscopy can offer this surgical option to almost every patient throughout the risk spectrum, allowing significant reduction of the operating time and superior stitch precision with no contact to sharp materials.
Surgical Technique
Preoperative Diagnostics
Prior to the operation, all patients undergo transesophageal echocardiography to evaluate the mitral valve pathology. Some surgeons believe that preoperative computed tomography (CT) scans are essential, while others do not believe they are routinely necessary. We consider CT scans to be required for the preoperative planning in all patients undergoing minimally invasive cardiac procedures in order to access the suitability of patient anatomy for minimally invasive access as well as peripheral cannulation. Moreover, in case of severe mitral valve calcification (MAC) or in redo cases, the radiologic assessment of the expansion of the calcification might be crucial for preoperative planning. All patients are discussed in our institutional Heart Team consisting of cardiothoracic surgeons, interventional cardiologists, and anesthesiologists.
Operative Techniques
All patients undergo conventional general anesthesia with a single-lumen endotracheal tube. Transesophageal echocardiography is continuously performed during the procedure in all patients to monitor heart and valve function, guide fluid administration, and evaluate the postoperative results. The mitral valve is preferably accessed through an anterolateral minithoracotomy in the anterior axillary line. A minithoracotomy can be defined as a wound <4 to 6 cm in length, with no rib or cartilage transection, and with the use of only soft-tissue retractors. This highly versatile access can be used for the surgical treatment of all heart valve pathologies.
Automated Suturing Technology
The RAM® Device (LSI SOLUTIONS®, Victor, NY, USA) enables simultaneously passing both ends of a mattress suture through the targeted mitral annular tissue (Fig. 1) with 1 squeeze of the lever (Fig. 2). According to surgeon preference and patient anatomy, the RAM Device is available in 2 tissue width sizes including 3.5 and 5.0 mm (Fig. 3). To reduce the risk of sharps exposure to the surgical team, the distal tip of the RAM Device provides a protective housing that holds the sharp needle tips retracted and unexposed while it is not in use. The RAM Device augments the maneuverability of the human wrist, providing additional articulation (the tip can angle up to 30° in and out relative to the shaft) and rotation (the shaft can rotate 360°) to facilitate a unique degree of ergonomic positioning while placing the annular sutures. After the RAM Device tissue jaw is positioned onto the mitral annulus, a single squeeze and release of the blue lever places the 2 initial bites of the mattress suture. As the device is pulled away from the annulus, the suture pays out of its suture tube (Fig. 1a).
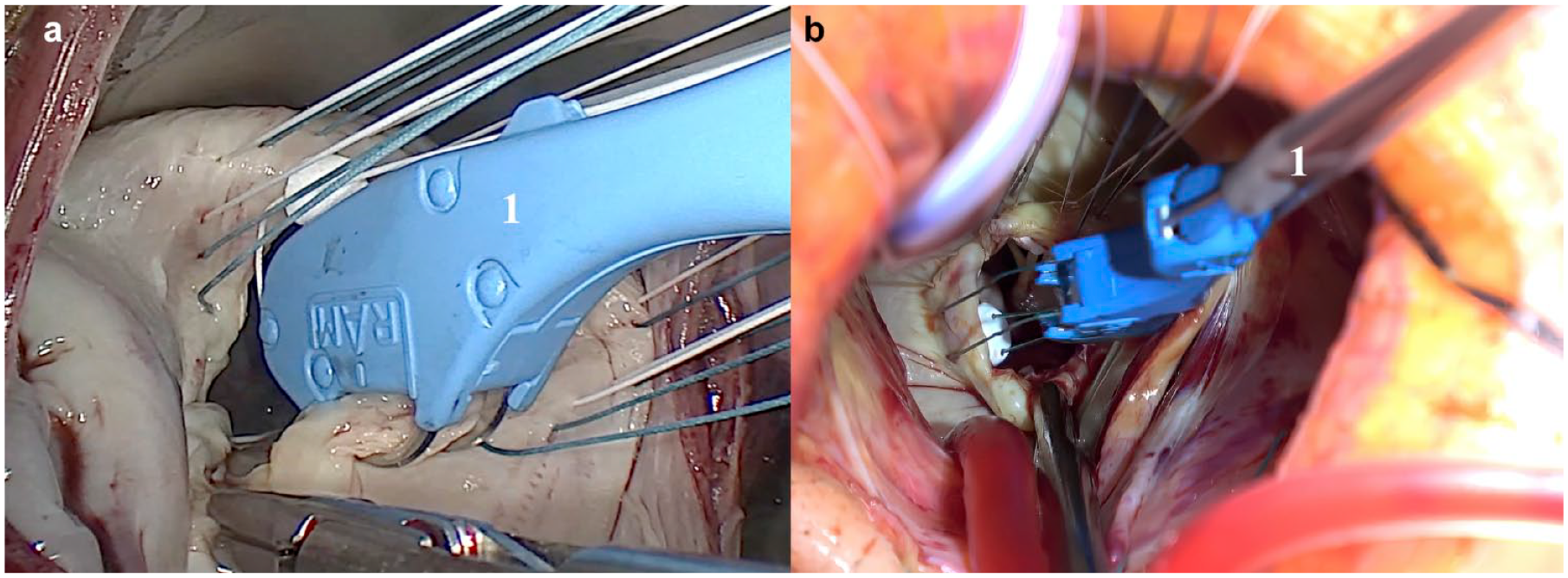
Intraoperative view of the RAM Device (LSI SOLUTIONS, Victor, NY, USA) and video endoscopy. (a) Videoendoscopic image of the RAM Device bite. (b) Direct vision (1 = RAM Device).
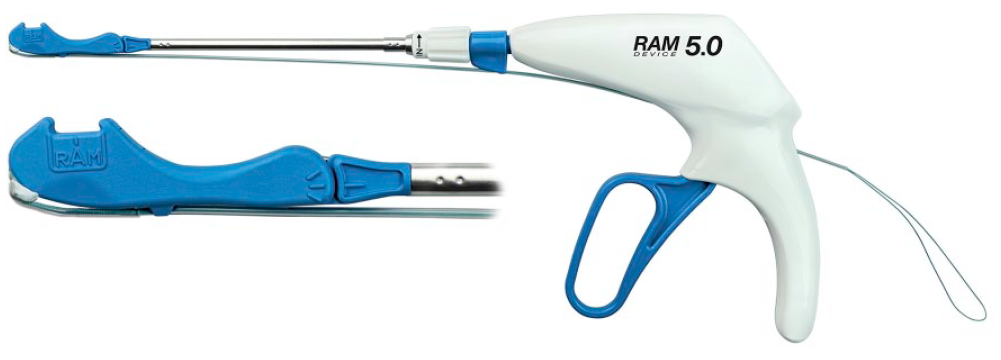
RAM Device (© LSI SOLUTIONS). Reprinted with permission from LSI SOLUTIONS (Victor, NY, USA).

RAM Device size options (© LSI SOLUTIONS). Reprinted with permission from LSI SOLUTIONS (Victor, NY, USA).
Next, an empty SEW-EASY® cassette (LSI SOLUTIONS) is placed into the jaw of the RAM Device. The RAM Device lever is then pulled away from the handle to release the suture from the RAM needle tips and transfer it into the SEW-EASY cassette. Now loaded with suture, the SEW-EASY cassette is placed into a SEW-EASY Device (Fig. 4). The jaw of the cassette is placed on the sewing cuff of the prosthetic valve. Squeezing and releasing the SEW-EASY lever passes both suture ends through the valve prosthesis at the appropriate location (Fig. 5, Fig. 6). The SEW-EASY cassette is then removed from the device and placed into the RAM RACK or the RAM RING (Fig. 7), to provide the additional suture management.
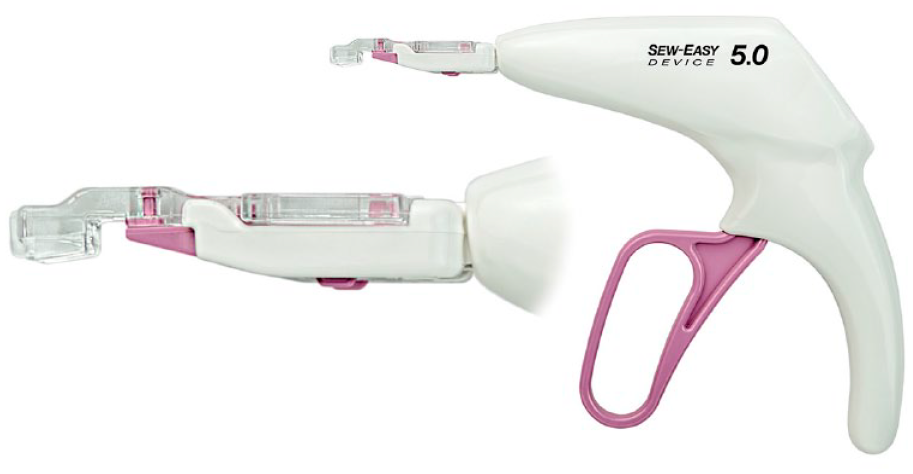
SEW-EASY Device (© LSI SOLUTIONS). Reprinted with permission from LSI SOLUTIONS (Victor, NY, USA).
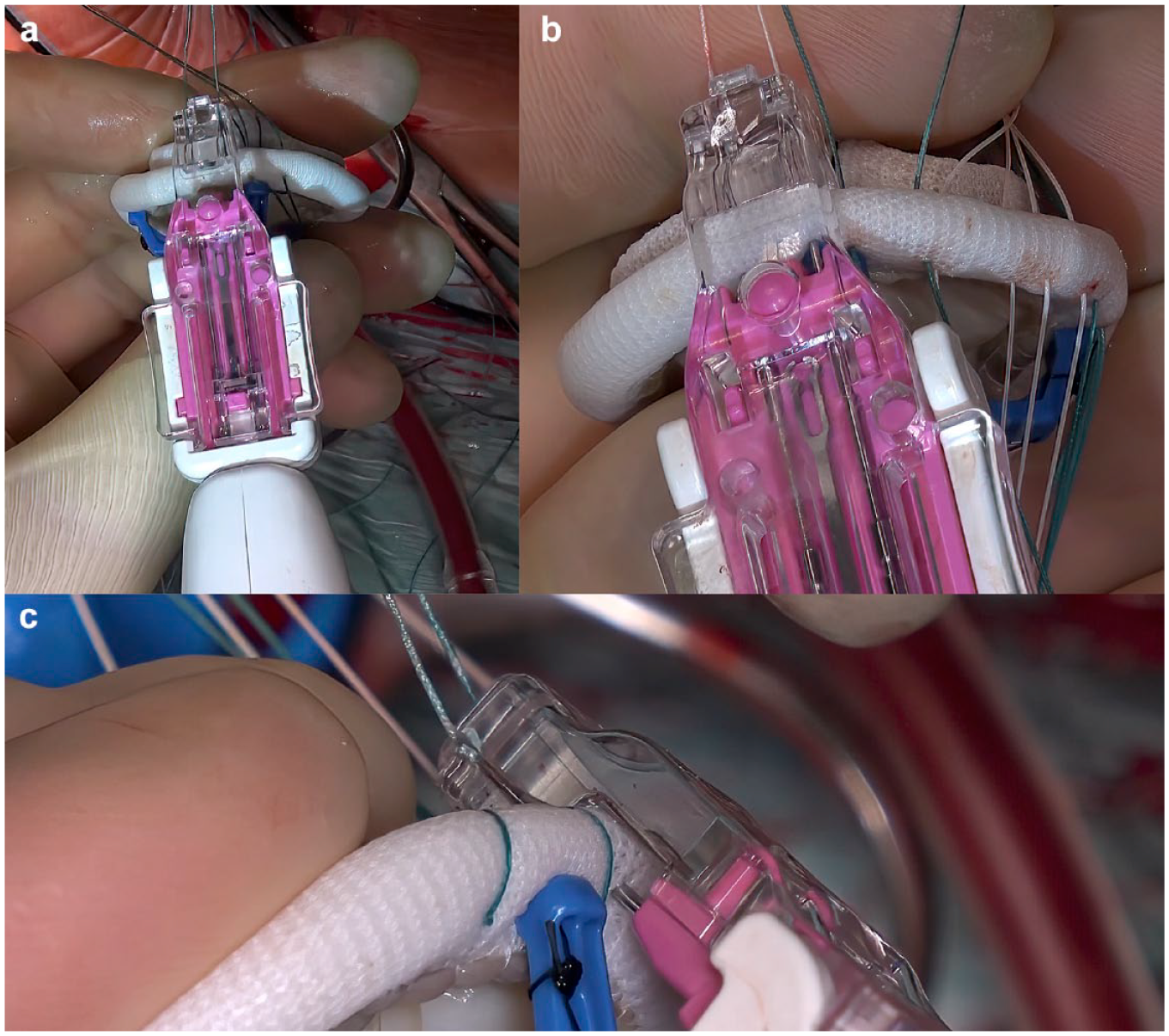
SEW-EASY technology (LSI SOLUTIONS, Victor, NY, USA).
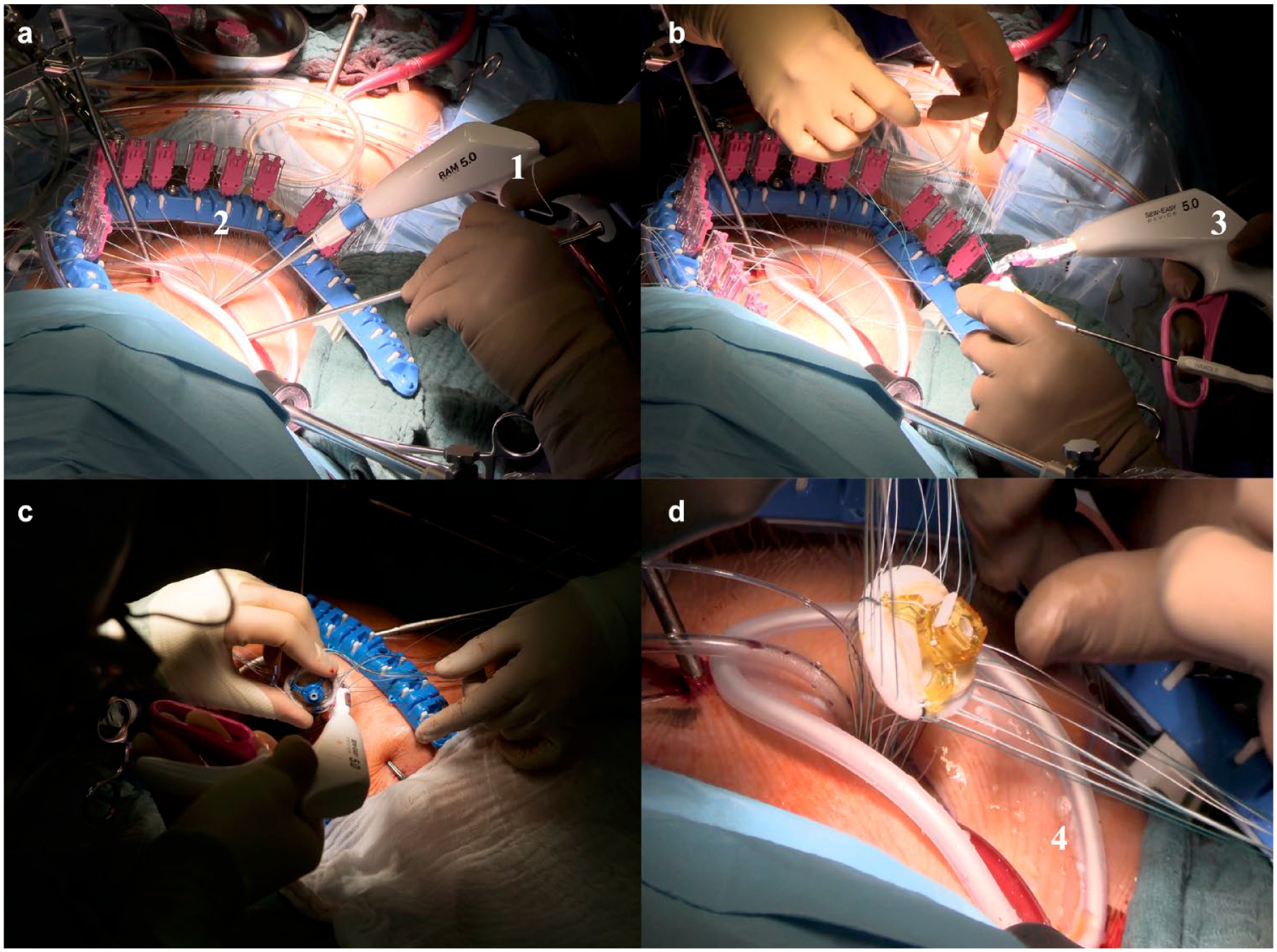
Applying the SEW EASY System (LSI SOLUTIONS, Victor, NY, USA) to the mitral valve prosthesis (1 = SEW EASY System, 2 = RAM RACK, 3 = biological mitral valve prosthesis).
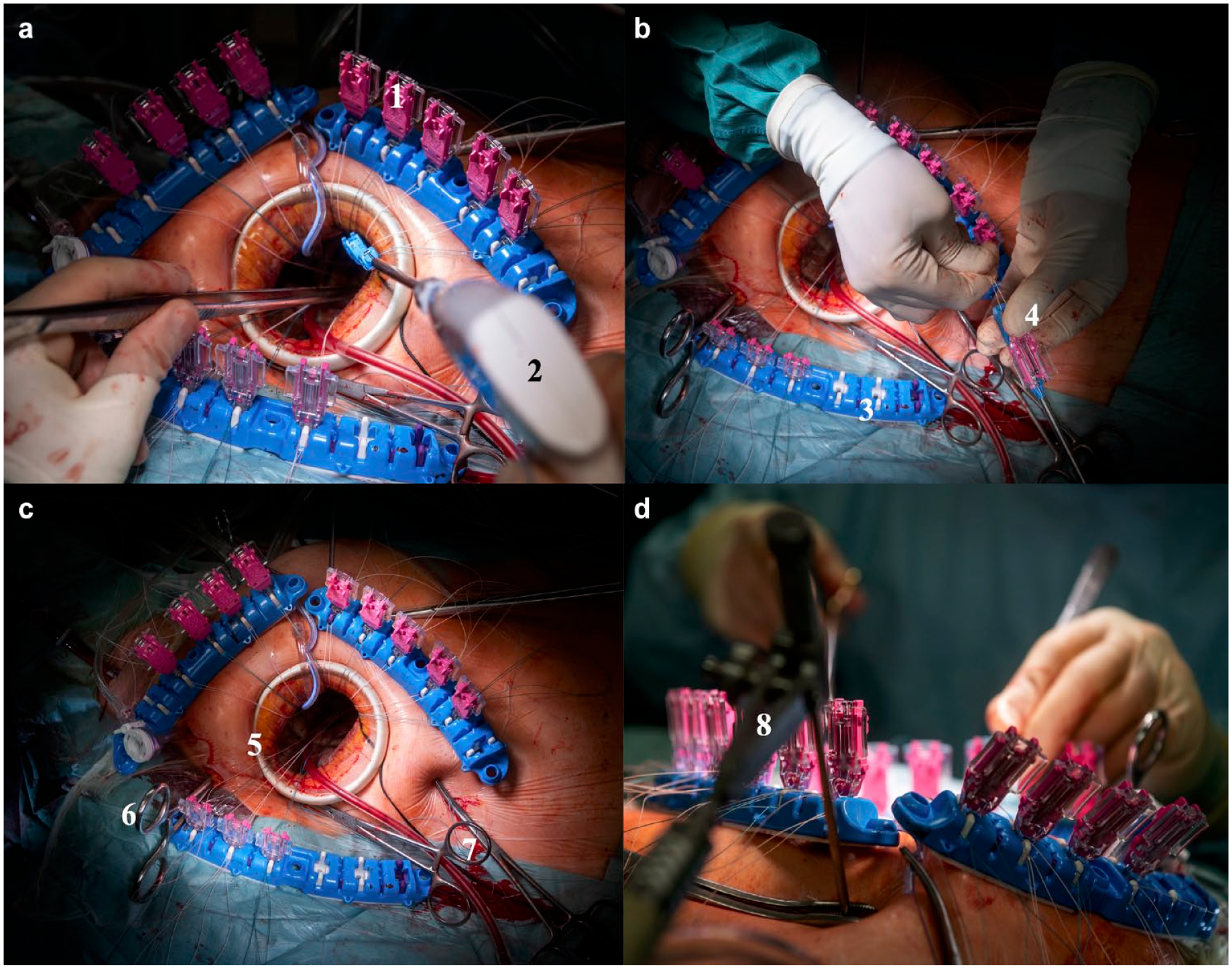
Minimally invasive access and RAM Device (LSI SOLUTIONS, Victor, NY, USA) implementation. (a) 1= RAM cassettes, 2 = RAM Device; (b) 3 = RAM RACK, 4 = loading of the RAM cassette; (c) 5 = soft-tissue retractor, 6 = Chitwood clamp, 7 = fixation of the stay sutures; (d) 8 = positioning of the mitral hook.
With the sewing cuff suture in place, the cassettes are cut off and the sutures are gathered into 3 groups and clamped together. The valve prosthesis is then parachuted down to the level of the annular plane. The sutures are secured with the COR-KNOT MINI Device® (LSI SOLUTIONS; Fig. 8), which features a 4 mm rotating shaft (Supplemental Video).
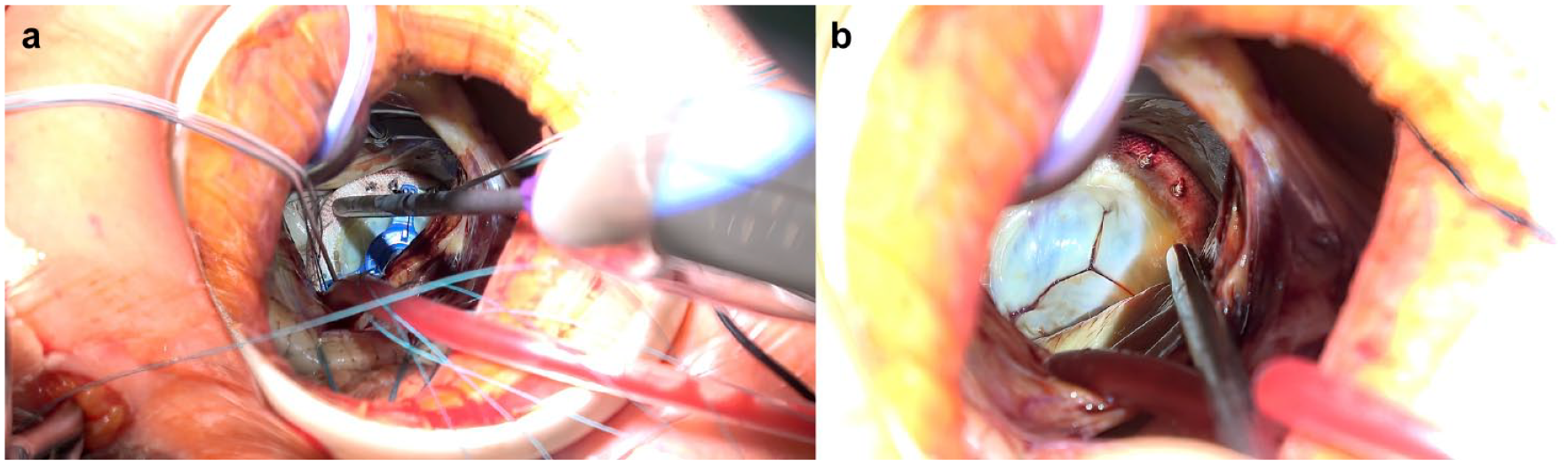
Implementation of the COR-KNOT device (LSI SOLUTIONS, Victor, NY, USA) in minimally invasive mitral valve surgery.
Patient Positioning and Bypass Cannulation
The patient is placed on the operating table in the supine position, with the right hemithorax elevated to 20°. Defibrillator pads are placed across the chest wall. After heparin administration, cannulation for cardiopulmonary bypass (CPB) is achieved via the common groin vessels using the Seldinger technique with the surgical cutdown. In consultation with the perfusionist, the size of each cannula is selected based on the patient’s body surface area and the actual diameter of the groin vessels.
Infra-Axillary Access
A 4 cm vertical skin incision is made in the anterior axillary line. After hemostatic dissection of the muscular tissue, the direction of the internal incision is reoriented horizontally into the fourth intercostal space. The pleural space is opened, and the soft-tissue retractor (ValveGate Soft Tissue Protector; Geister, Tuttlingen, Germany) is inserted. During this procedure, it is not advisable to use the rigid bone retractors to avoid any damaging of the bone structures and to prevent postoperative pain syndrome. The pericardium is opened parallel to the phrenic nerve cranially to the innominate vein and caudally to the inferior vena cava. Then, 4 to 5 stay sutures are attached to the pericardium and pulled under the soft-tissue retractor to enable closer positioning of the heart to the chest opening (Fig. 7c). This allows both the utilization of nonendoscopic instruments and, if necessary, working under the direct vision without video endoscopy.
A 3-dimensional camera port (Aesculap Einstein Vision, B. Braun, Tuttlingen, Germany) is introduced through the small incision in the intercostal space above the thoracotomy incision. A Chitwood aortic clamp (Scanlan International, Inc, St. Paul, MN, USA) is placed through the intercostal space laterally and cranially to the microthoracotomy (Fig. 7c). Carbon dioxide is infused via the camera port throughout the whole procedure to minimize the risk of air embolism.
After cross-clamping the aorta under electrically induced ventricular fibrillation, crystalloid cardioplegic solution (Custodiol®, Dr. Franz Köhler Chemie GmbH, Bensheim, Germany) is then delivered through the needle in the ascending aorta.
The left atrium is accessed through the Waterson groove. The mitral valve is exposed with the aid of a single atrial retractor (Fig. 7d). A ventricular vent catheter is inserted directly through the atriotomy and mitral valve.
Surgical Techniques on the Mitral Valve
One of the major advantages of the minimally invasive access in mitral valve replacement surgery is the applicability of all conventional surgical techniques, such as—in mitral valve repair—leaflet resection, annuloplasty, implantation of the artificial chords, and chords transfer. In cases in which mitral valve replacement is necessary, either stented biological or mechanical valve prostheses can be used, according to the European Society of Cardiology guidelines for valvular heart disease. 5 In all mitral valve replacement procedures via minimally invasive infra-axillary access, automated suture technology is applied to reduce the cross-clamp and CPB times.
After exposing the mitral valve with a single retractor as previously described, the valve is inspected to identify the actual pathology. The annulus is then sized using the standard equipment for a valve prosthesis provided by the manufacturer. The RAM Device is used to place the pledgeted mattress sutures into the mitral annulus. We start at the base of the P2 segment, working our way up clockwise toward the P1 segment. For easier access of the commissures, traction put onto the previous suture can be helpful to expose the annulus. Care should be taken in the left circumflex coronary artery and noncoronary cusp area. With the sequential squeezing and releasing of the RAM Device lever, the device places both initial bites of the horizontal mattress suture through the mitral annulus (Fig. 1a). Each suture end is now attached to the retracted RAM needles. Suture ends are transferred to SEW EASY cassettes, which are placed into the corresponding holding feature in the RAM RACK or RAM RING (Fig. 7b).
When the annular sutures are in place, the SEW EASY Device passes the sutures through the valve prosthesis sewing cuff (Fig. 5). The cassettes are cut away, and the sutures are grouped and clamped in the same manner as in the conventional sewing technique (Fig. 6). After parachuting the valve onto the mitral annulus and confirming its appropriate positioning, the sutures are fastened with the COR-KNOT MINI Device, which crimps the titanium fasteners to automatically secure the sutures and then cuts away the tails (Fig. 8).
After testing the valve competence, the atriotomy is closed with running 4/0 Prolene. The heart is thoroughly deaired, and the aorta is opened. The temporary ventricular pacing wires are placed while the heart is still fully decompressed on the CPB. The cardioplegia needle is removed, and CO2 is discontinued. After securing the surgical hemostasis, protamine is administered at a 1:1 ratio to heparin. Valve function is assessed via transesophageal echocardiography.
Decannulation and Closure
After discontinuing the CPB and decannulation of the groin vessels, the purse string sutures on both femoral vessels are tightened, and hemostasis is confirmed. The wound is closed in anatomic layers. The skin incision is closed with an absorbable 4/0 intracutaneous suture.
Discussion
In recent decades, there has been a transformative shift in the surgical management of patients with valvular heart disease. A variety of minimally invasive techniques have been carefully integrated into routine surgical practice, earning accolades for their superior hemodynamic and cosmetic outcomes. These advancements have not only gained favor among patients but have also been endorsed by referring cardiologists.1,6,7 Multiple clinical studies have substantiated the benefits of less invasive valve surgery, including faster recovery times, reduced postoperative pain, shorter durations of hospital and ICU stays, fewer blood transfusions, and ultimately, lower health care costs.7 –9 Recent clinical trials have further validated the efficacy of less invasive mitral valve surgery, showing it to be on par with, if not superior to, traditional surgical approaches. 4 A landmark effort by Berretta et al. has established an international standard for minimally invasive mitral valve surgery, demonstrating exceptional outcomes across low-risk, intermediate-risk, and high-risk patient groups based on data from the Mini-Mitral International Registry. 10
Despite the compelling advantages and growing body of evidence supporting gentler sternal-sparing surgical techniques, integration into mainstream cardiac surgery has been slow. Initial hesitancy among surgeons stemmed from concerns about a steep learning curve and prolonged CPB and cross-clamp times, which were perceived as additional risks to patients and have been cited as independent predictors of postoperative morbidity and mortality. 11 However, advancements in technology, particularly automated suturing devices, have begun to alleviate some of these concerns. 12 A multicenter study by Malvindi et al. highlighted a notable reduction in CPB and cross-clamp times when employing less invasive mitral valve procedures via infra-axillary access, compared with traditional minimally invasive techniques. 4 Our own experience corroborates these findings; the integration of automated suturing technologies such as the RAM and SEW EASY devices has streamlined the surgical process, enabling rapid and reliable suture placement even in confined remote surgical spaces. These devices have been successfully deployed in more than 600 patients across the world, reinforcing their efficacy and reliability in clinical practice. 13
Malvindi et al. also underscored that the use of less invasive access did not compromise the type of valve prosthesis used or the repair techniques employed on the mitral valve. Infra-axillary access is very versatile and applicable for any type of valve procedure. In addition, the choice of less invasive access had no negative impact on the number of concomitant tricuspid valve procedures.4,14,15
Surgeons with extensive training in minimally invasive aortic and mitral valve procedures may find the learning curve to be relatively flat. As noted by studies from Pitsis et al. and Bakhtiary et al. focusing on minimally invasive aortic valve surgery, surgeons typically adapt to these new techniques within as few as 5 to 10 cases.16 –18 On the other hand, surgeons without regular exposure to minimally invasive methods may require a longer period to become accustomed to the more confined surgical environment. Due to the fact that any conventional valve replacement techniques can be mimicked with the RAM Device, the process of adapting to the novel suturing technology does not take that long. The RAM Device’s enhanced range of motion, coupled with superior suture management via the RAM Rack and customizable stitch widths to suit individual patient anatomy, however, facilitates a quicker mastery of these minimally invasive techniques. Achieving a comfortable level of proficiency in this specialized surgical setting can realistically be accomplished within fewer than 10 cases. The RAM Device can be equally applied on any valve prosthesis, providing an easy and safe implantation.
The definition of minimally invasive mitral valve replacement surgery continues to evolve. In our center, we consider procedures performed through nonsternotomy incisions of less than 6 cm, preferably with the videoendoscopy, to be minimally invasive. The infra-axillary minimally invasive approach significantly reduces chest wall trauma, leaving the patient with no readily apparent postoperative scarring. In some hands, minimally invasive procedures via lateral minithoracotomy are known for causing significant postoperative pain from extensive retraction damaging ribs or compromising of the thoracic nerves by the ports for the endoscopic instruments.19 –21 Since no rigid bone retractors are used during minimally invasive procedures, it is possible to minimize postoperative pain by avoiding bony compression injury. The number of incisions required for endoscopic instruments and imaging can also be reduced. Even with significantly less postoperative pain compared with traditional less invasive mitral valve surgery, we still prefer to offer each patient extra analgesic support with a local anesthetic (e.g., bupivacaine for the first 24 h postoperatively).
It is important to recognize the increased surgical risk in patients presenting with severe MAC. 22 One of the most feared severe intraoperative complications in these patients is annular rupture, especially in patients requiring extensive annular decalcification and debridement. During the placement of the annular sutures, unnecessary suture tension can be reduced by using the RAM automated suture technology. Moreover, the RAM Device, if necessary, provides a deep and stronger bite, as a regular stitch, preventing the needle rotation in the calcified annular plane. Not only can similar tension be simultaneously applied to both of the mattress stitches, avoiding the additional tension on each stitch, but the depth of the bite can also be reduced by holding the RAM Device slightly off the annular plane to create a more superficial bite if desired. One author, with recent success operating on patients with severe MAC, 4 has developed a useful approach to suturing calcific annuli using RAM technology. Publication of this evolving technique and its promising results for patients with MAC is anticipated after additional clinical experience.
The positive impact of minimally invasive procedures on the cost-effectiveness in cardiac surgery is obvious. The addition of the RAM suturing technology, offering even shorter cross-clamp and CPB times, less surgical trauma, shorter ICU stay, fewer deep wound infections, and therefore quicker recovery, contributes to direct cost reduction. Due to the successful implementation of the minimally invasive methods into clinical practice and providing this treatment in high quality and on a regular basis, the hospital reputation among the medical society is increasing. This leads to a significant increase in cardiological referrals not only for endoscopic procedures, which subsequently results in the positive cost balance.
Conclusions
As the field of cardiac surgery continues to evolve, particularly with the rise of transcatheter interventions and the broadening of indications to include lower-risk patient groups, the integration of minimally invasive techniques into standard clinical practice has become imperative for optimal patient care. The health benefits of these minimally invasive approaches are well documented, offering lower postoperative mortality and morbidity rates along with proven procedural safety. Surgeons who provide less traumatic options for patients are offering the most compelling advantages of surgical treatment, allowing them to remain competitive in a rapidly changing medical landscape. Therefore, there should be no reluctance within the surgical community to venture beyond traditional methods and acquire expertise in minimally invasive techniques. Cutting-edge technologies, such as automated suturing devices, coupled with advanced intraoperative suture management systems, soft-tissue retractors, and excellent videoscopic imaging, offer invaluable support in flattening the learning curve. These innovations enable the application of minimally invasive surgical methods across the entire spectrum of surgical risk, further reinforcing the imperative for their adoption.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Farhad Bakhtiary discloses speaker honoraria and consultancy fees from LSI SOLUTIONS. Other authors have no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
At admission, all patients in our institution signed an informed consent on using their anonymized data for research and publication. The approval of the ethical board is not required for this kind of manuscript.
Supplemental Material
Supplemental material for this article is available online.
