Abstract

Introduction
Minimally invasive cardiac surgery (MICS) is increasing in popularity. In this field, endoscopic cardiac surgery (ECS) represents one of the most minimally invasive approaches in the cardiac surgery community, allowing surgeons to conduct cardiac operations under the guidance of an endoscope, used to improve the surgical performance and patient outcomes. 1 In several high-volume centers, ECS is already the standard technique for valvulopathies. However, the ECS approach for multiple valve surgery, due to its complexity, is adopted in few centers,2–5 whereas endoscopic ascending aorta replacement (AAR) has not yet been described in the literature.
At our institution we have adopted the endoscopic approach since 2010. We previously showed its safety and feasibility in isolated aortic and concomitant cases.2,3 In fact, it has become the technique of choice to perform triple valve surgery and, in selected cases, AAR isolated or associated with valve treatment.
We report the case of a 64-year-old woman affected by triple valve disease, permanent atrial fibrillation (AF), and ascending aortic aneurysm (AAAn) who successfully underwent totally endoscopic surgery.
Case Report
A 64-year-old female patient (body mass index, 20 kg/m2; body surface area, 1.6 m2) underwent percutaneous mitral valvuloplasty for a rheumatic stenosis in 1993 and in July 2020 was admitted to our institution with a diagnosis of severe aortic valve regurgitation in tricuspid aortic valve, severe mitral valve stenosis, moderate tricuspid valve regurgitation with annular dilation (annulus size 41 mm), permanent AF on medical treatment with direct oral anticoagulants (DOACs), tubular AAAn of 50 mm, and New York Heart Association (NYHA) functional class III. Echocardiography showed preserved biventricular function, both atria dilation and normal pulmonary pressure. Since the patient refused a standard sternotomy approach, which is the technique of choice in these cases, we opted for a minimally invasive totally endoscopic procedure. A computed tomography scan was done to assess preoperatively the chest anatomy and to exclude the presence of peripheral vascular disease as well as potential contraindications, such as severely calcified aortopathy.
Surgical Technique
Exposition
The patient was placed in a supine position with 30° elevation of the right hemithorax. The operation was started with surgical exposure of the femoral vessels through a small skin incision. Then, a 4 cm right minithoracotomy in the third intercostal space was done at the midclavicular line, representing the working port (Supplemental Fig. 1). A soft-tissue retractor was then placed with no rib spreading and without sacrificing the internal mammary vessels (Supplemental Fig. 1). Three additional 5 mm mini-ports were needed: in the third intercostal space to introduce the vent line; in the second intercostal space to place a 5 mm, 30° thoracoscope; and in the first intercostal space to introduce the aortic cross clamp (Supplemental Fig. 1). A carbon dioxide line was connected to the trocar previously used to place the thoracoscope (Supplemental Fig. 1).
Operation
Cardiopulmonary bypass (CPB) was achieved through femoral vessel cannulation under transesophageal echocardiogram (TEE) guidance. The venous cannula was advanced into the superior vena cava to ensure good venous drainage. Once the patient was under CPB, the pericardium was opened under thoracoscopic vision, with attention paid to avoid phrenic nerve injury. Aortic cross-clamping was achieved using a transthoracic clamp (Supplemental Video 1). Custodiol cardioplegia was delivered directly into the aortic root and then, after the aortotomy, directly into the coronary ostia.
Left atriotomy was made in Sondergaad’s groove, and an atrial retractor was introduced through the right third intercostal space incision. The vent line was placed through the right superior pulmonary vein. The exeresis of the leaflets of a postrheumatic mitral valve was made with preservation of the submitral apparatus (Supplemental Video 2). A 31 mm Epic bioprosthesis (Abbott, Chicago, IL, USA) was implanted, and the left atrial appendage was excluded with double continuous polypropylene suture (Fig. 1a). Then, an oblique aortotomy over the sinotubular junction was made, and the aneurysmatic tract of the ascending aorta was excised. An additional dose of selective cardioplegia was delivered directly into the coronary ostia. After the excision of the aortic valve, a Inspiris Resilia 23 mm bioprosthesis (Edwards Lifesciences, Irvine, CA, USA) was positioned (Fig. 1b, Supplemental Video 3). Then, a Hemashield Platinum 28 mm linear vascular prosthesis (Getinge, Göteborg Sweden) was implanted using 2 continuous polypropylene sutures reinforced with Teflon (Fig. 2a, Supplemental Video 4). The aorta was then declamped after 179 min (Supplemental Video 4). The electromechanical activity of the heart was resumed after 2 direct current shocks at 200 J in sinus rhythm. Finally, the right atriotomy was made, and tricuspid annuloplasty on the beating heart was performed using a 30 mm Cosgrove-Edwards Physio Tricuspid Annuloplasty Ring (Edwards Lifesciences; Fig. 2b, Supplemental Video 5). The de-airing after tricuspid repair was done by filling the heart on pump during the closure of the right atriotomy.
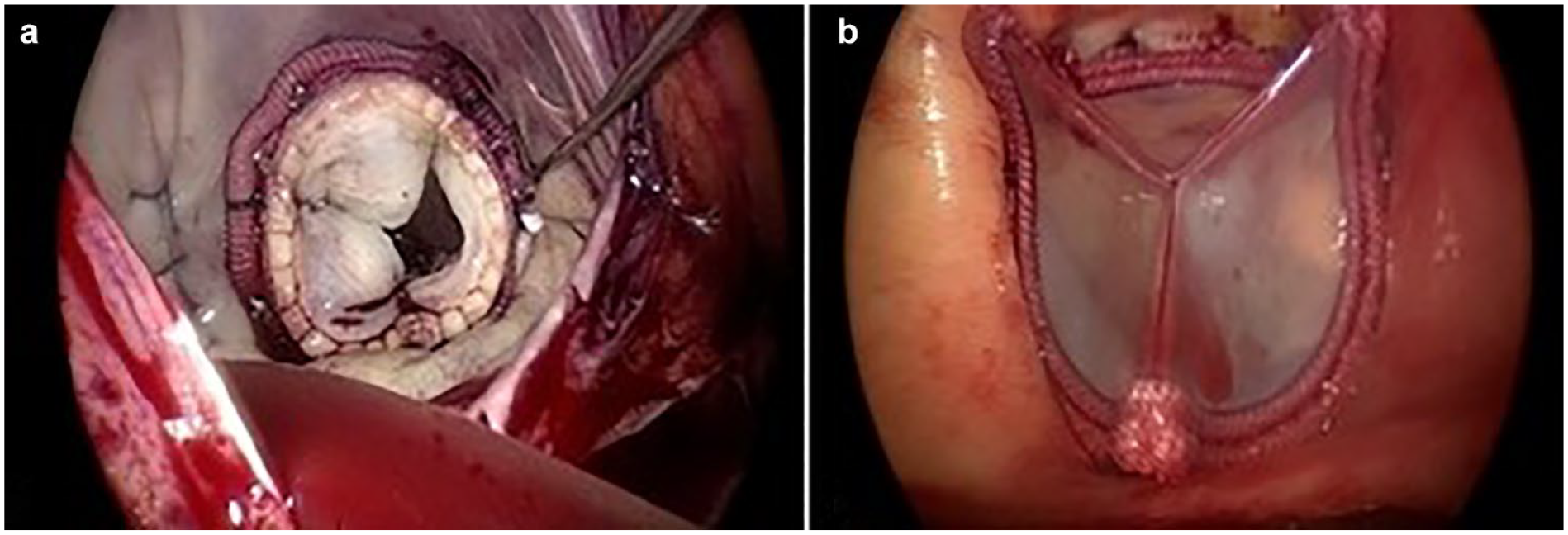
(a) Mitral valve replacement and left atrial appendage exclusion; (b) aortic valve replacement.
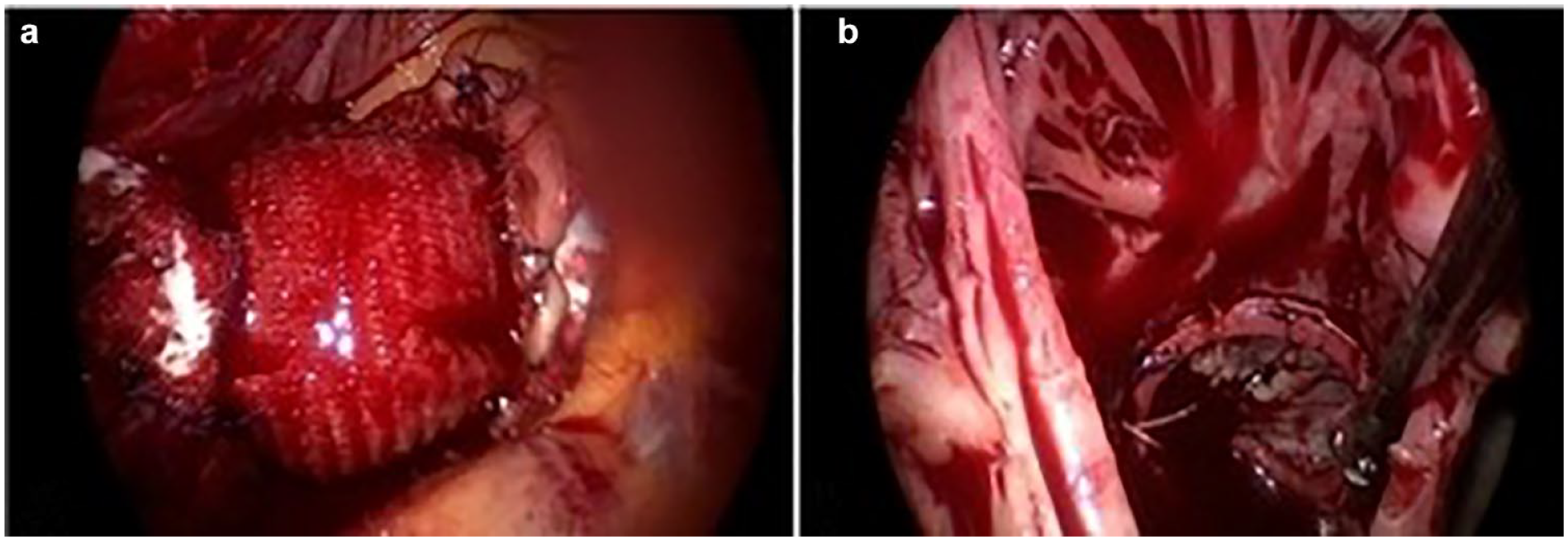
(a) Ascending aorta replacement; (b) beating-heart tricuspid valve annuloplasty.
TEE showed a good mitral and aortic bioprostheses function and no residual tricuspid regurgitation. CPB weaning was easily obtained after 245 min. The operation was completed in the usual fashion.
Postoperative Course and Follow-Up
The patient was transferred to the intensive care unit in stable hemodynamic condition. She was extubated after 12 h and discharged to the rehabilitation center on the eighth postoperative day after a regular course.
At 2-year follow-up, the patient was in NYHA class I. Electrocardiography showed AF rhythm on medical treatment with DOACs. Echocardiography showed good function of the aortic and mitral bioprostheses (medium gradient 12 and 4 mm Hg, respectively), negligible tricuspid regurgitation, normal size, and biventricular function.
Discussion
Patient demand for MICS is growing. Indeed, ECS, compared with sternotomy, is characterized by reduction of postoperative pain, less blood loss, lower risk of infection, and improvement in patient recovery and quality of life. 6
Despite the rapid development of MICS, endoscopic concomitant multiple valve surgery and AAR are still uncommon because of high technical complexity. However, this case report and our institutional experience2,4 demonstrate that this surgery is feasible and could be an alternative to sternotomy in selected patients and in a dedicated center.
We showed that the thoracoscope allows a larger visualization field and is useful for managing concomitant surgical procedures through the same single working port with better exposure and accuracy. 2 Particularly, the thoracoscope vision provides the operator with a direct line of sight into the left ventricle; 7 it allows assessment of the correct position of mitral and aortic prostheses (Supplemental Video 3). Moreover, in concomitant tricuspid valve surgery, the right atrium can be opened on the beating heart 2 to reduce clamping times. Finally, endoscopic AAR is complex but feasible for an expert endoscopic surgeon.
Selected patients should be considered for endoscopic multiple valve surgery and concomitant AAR. To make the patient a good candidate for this technically demanding surgery, we consider inclusion criteria of age less than 80 years, Clinical Frailty Scale point <5, normal body mass index (no obesity, body mass index <30 kg/m2), preserved biventricular function, postjunctional position of the AAAn, and a normal pulmonary pressure. Absolute contraindications are the impossibility of achieving peripheral cannulation, severe pectus excavatum, and the lack of large experience with ECS.
Supplemental Material
sj-pdf-1-inv-10.1177_15569845241231808 – Supplemental material for Pushing the Boundaries in Totally Endoscopic Cardiac Surgery: Combined Triple Valve Surgery and Ascending Aorta Replacement
Supplemental material, sj-pdf-1-inv-10.1177_15569845241231808 for Pushing the Boundaries in Totally Endoscopic Cardiac Surgery: Combined Triple Valve Surgery and Ascending Aorta Replacement by Loris Salvador, Luciana Benvegnù, Daniele Zoni, Tommaso Hinna Danesi, Emad Al Jaber, Olivera Rasovic, Salvatore Poddi and Giovanni Domenico Cresce in Innovations
Footnotes
Acknowledgements
The authors are grateful to Federico Pengo for his technical assistance in video editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
