Abstract

Introduction
Primary cardiac tumors are rare, occurring in about 0.0001% to 0.0003% of autopsies or about 1 in every 500 cardiac surgery cases. 1 About 25% of these are malignant, and of the malignant tumors, about 75% are primary cardiac sarcoma (PCS). 2 PCS tends to involve young patients with no identifiable predisposing factors. 3 The rarity of PCS is highlighted by a recent 43-year review of the Surveillance, Epidemiology, and End Results database, yielding 442 patients with primary cardiac sarcoma. 4 This review showed a median survival of 7 months as well as 1-year, 3-year, and 5-year survival of 40.7%, 15.6%, and 9.8%, respectively. Surgery and chemotherapy were associated with improved survival.
We established a multidisciplinary cardiac tumor team with MD Anderson Cancer Center 20 years ago to manage and study this difficult disease. Our most recent publication from this program had 122 cases of PCS operated on over a 23-year period. 5 The mean age of the patients was 45 years, and 52% were male. The left atrium (40.2%) and right atrium (32.0%) were the most common locations. The most common histology was angiosarcoma (38.5%) and high-grade sarcoma (14.8%). Survival from the initial diagnosis at 1, 3, and 5 years was 88.4%, 43.1%, and 27.8%, respectively. Management of these tumors requires complex decision making and often unique and complex surgical procedures when surgery was appropriate. Currently we have operated on 137 PCS and believe that all PCS should be managed in a center of excellence with a dedicated cardiac tumor team. 6 In this article, we hope to share the 7 pillars of evaluation and treatment for PCS developed over 2 decades of work (Fig. 1).
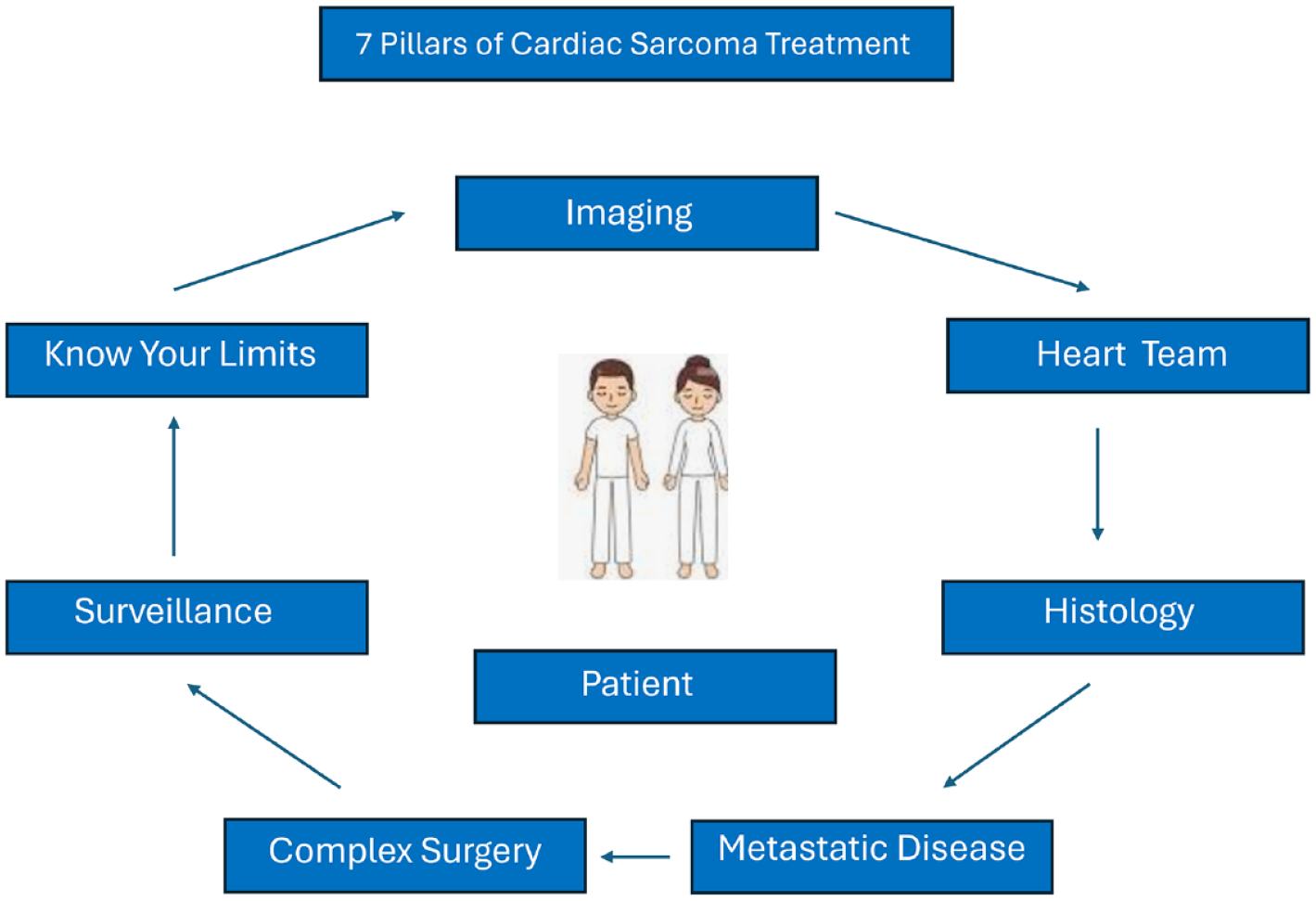
The 7 pillars of cardiac sarcoma treatment.
1. Don’t Be Fooled!
Almost 75% of PCS cases we see were previously operated on elsewhere with a suboptimal resection as the surgeon believed they were dealing with a benign tumor. In fact, all left atrial sarcomas we have seen were initially thought to be myxoma elsewhere and incompletely resected, and our surgery was for residual or recurrent tumor after chemotherapy. Cardiac tumors that are not completely typical for myxoma require additional consideration. Differentiating between benign and malignant tumors of the heart is a key first step.7,8 We studied 130 patients having cardiac magnetic resonance (CMR) imaging for suspected cardiac tumor. Of these, 66 were found to have a definitive tumor, with 39 (59%) being malignant and 27 (41%) benign. Malignant tumor patients were younger (51 vs 65 years) and more often demonstrated tissue invasion (69% vs 0%). 7 Both first-pass perfusion and late gadolinium enhancement were significantly more common in malignant tumors. We have found CMR helpful in differentiating tumor from no mass, thrombus, or pseudotumor. 8
Imaging generally starts with echocardiography as the initial test. Tumors very typical of myxoma or fibroelastoma may not need further imaging other than coronary arteriography if indicated in the patient. 9 For atypical tumors, we move to CMR as the next imaging modality. If malignancy is suspected, a positron emission tomography–computed tomography (PET/CT) scan is done to evaluate for metastatic disease. If PCS remains the primary concern, a CT scan with and without contrast is done to help the surgeon decide if complete resection is possible and to evaluate the needed complexity and risk of the surgery. One tumor that can mimic PCS is cardiac paraganglioma. These tumors can be very large and have intense vascular signal on CMR. Coronary arteriography will show a large parasitic vessel from the coronary circulation to the tumor. 10
2. Meet With Your Multidisciplinary Team
Malignant and large complex cardiac tumors require advanced decision making when planning treatment, and this is best done with a team representing surgery, cardiology, cardio-oncology, oncology, and imaging (Fig. 2). 6 Careful consideration is given to deciding if the tumor is primary or metastatic to the heart. Metastatic tumors are an order of magnitude more common than primary tumors and are only rarely appropriate for surgery. The goals of treatment must be carefully discussed. Surgery can result in 3 potential outcomes. It may make you live longer, make you live better, or hurt you. Incomplete resection of PCS generally results in rapid local recurrence and no improvement in survival. The surgeon must gauge the potential for complete resection and discuss with the oncology team treatment options after resection. For patients who are highly symptomatic from the tumor, palliative resection can be considered. If the patient is not symptomatic and complete resection is not likely, then surgery should be avoided and systemic therapy applied. In these cases, the oncology team members will bring more options to the table in helping the surgeon plan care.
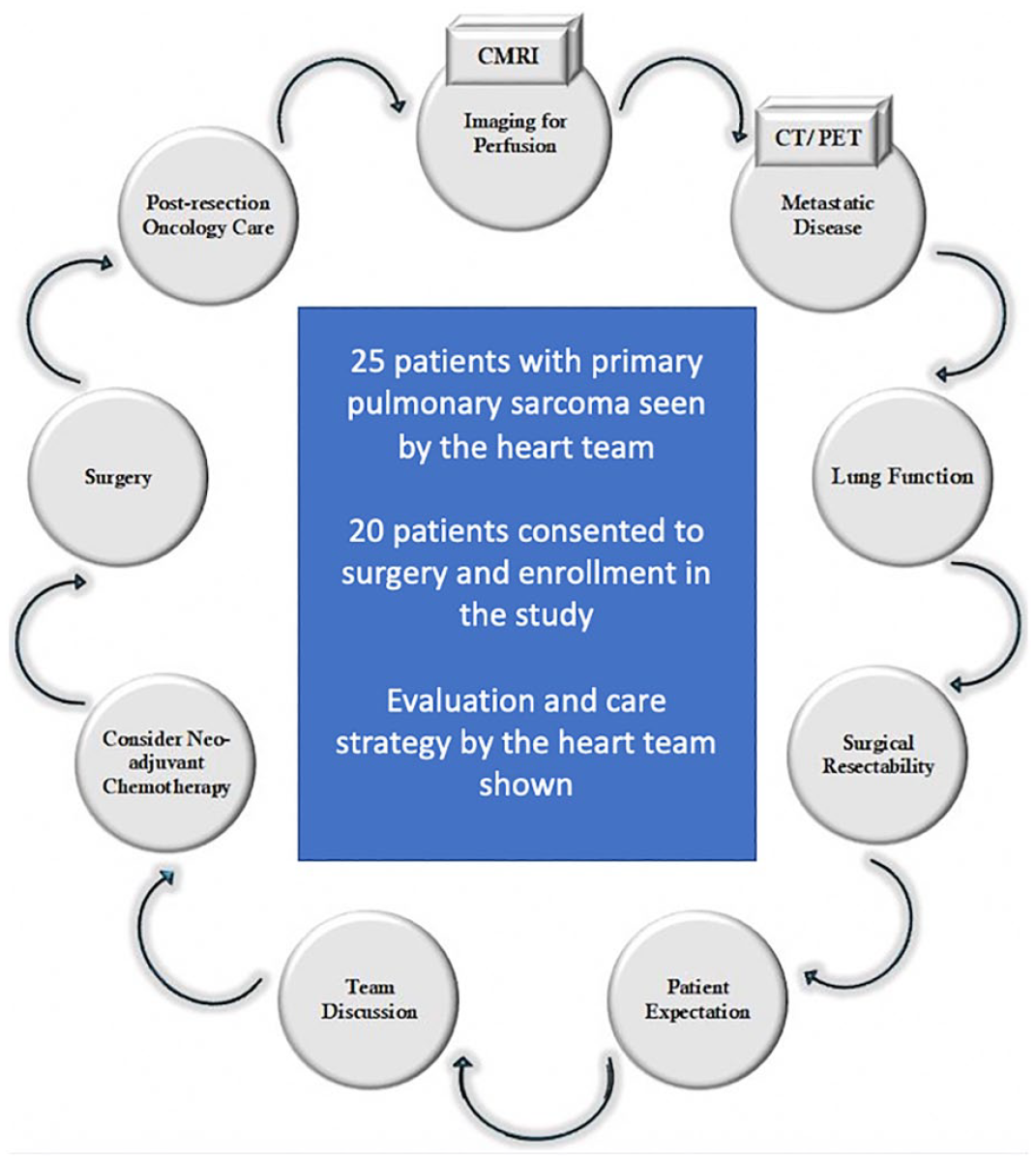
The heart team evaluated 25 patients with primary pulmonary artery sarcoma. Of these, 20 patients consented to surgery and inclusion in the study. The heart team strategy for evaluation and care is shown. CMRI, cardiac magnetic resonance imaging; CT/PET, computed tomography/positron emission tomography. Reprinted from Semin Thorac Cardiovasc Surg, 35(1), Chan et al., Surgical Management of Primary Pulmonary Artery Sarcoma, pp 53–64, Copyright 2023, with permission from Elsevier. 11
3. Get Tissue If Possible
A histologic diagnosis prior to any surgery is preferred if possible. For large right atrial tumors suspected to be PCS, about 10% will turn out on biopsy to be lymphoma, and surgery is not indicated. 12 We have even found a large right atrial plasmacytoma mimicking a malignant tumor, which did not need surgery. 13 We attempt to biopsy and establish a diagnosis in every right-sided tumor. We attempt a transcatheter tumor biopsy as our first approach. If this is not successful, we will move to percutaneous or small anterior thoracotomy needle biopsy. Biopsy of left-sided tumors is more difficult and carries risks of embolization. We have found that neoadjuvant chemotherapy for right atrial PCS will double the negative margin resection rate and patient survival. 14 Over time, we have become more aggressive in pursuing presurgical neoadjuvant chemotherapy. We will currently continue chemotherapy until there is no further tumor response or toxicity levels have been reached. We have not found this to increase the risk of surgery or inhibit recovery. This is usually possible with even large right atrial tumors as they tend to grow externally to the atrium rather than internally causing obstruction. Pulmonary artery sarcoma 11 and left atria sarcoma 15 more often present with obstructive symptoms, making surgery more urgent. If metastatic disease is identified on PET/CT scan, biopsy of this site can be considered.
4. Search for Metastatic Disease
Metastatic disease is common with PCS, especially angiosarcoma. We have found a high incidence of both lung and brain metastatic disease. 16 Metastatic disease that does not respond well to chemotherapy will not do well with surgery and should be treated with systemic therapy. If metastatic disease has a good response to chemotherapy and becomes undetectable, then surgery can be considered for control of the primary tumor.
5. Be Prepared for Complex Surgery
The anatomic extent of PCS is often large (Fig. 3). If surgery is indicated, it is incumbent on the surgeon to understand if complete resection is possible. Unlike most cardiac surgery procedures such as coronary bypass or valve surgery, which are relatively similar case to case, each PCS case is unique and often requires innovate and sometimes complex surgical solutions. Right atrial tumors are often very large and may involve the tricuspid valve and the right coronary artery. 13 When valve replacement is needed, we use a biologic valve despite the patient’s young age, as survival beyond 10 years is unlikely. Similarly, we use reversed saphenous vein to bypass the right coronary rather than internal mammary artery for the same reason. The right ventricular lesion can be resected using a right ventriculotomy, which provides exposure for complete resection and reconstruction and is usually well tolerated. Left atrial PCS is more complex because of the posterior location of the left atrium. When we started our program, we noticed in the literature a very high rate of reoperation for left atrial PCS due to inadequate resections. This led us to develop the procedure of cardiac explantation, ex vivo resection and reconstruction, and reimplantation of the repaired heart–cardiac autotransplantation (Fig. 4, Supplemental Video).15,18,19
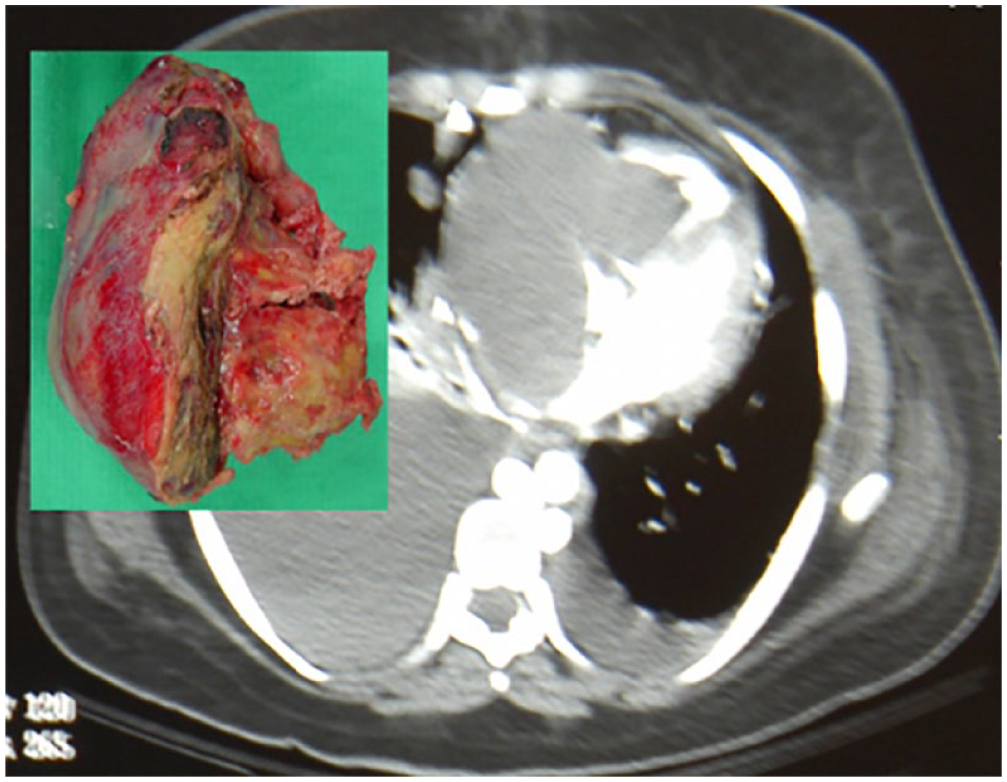
Extensive right atrial and ventricular sarcoma.
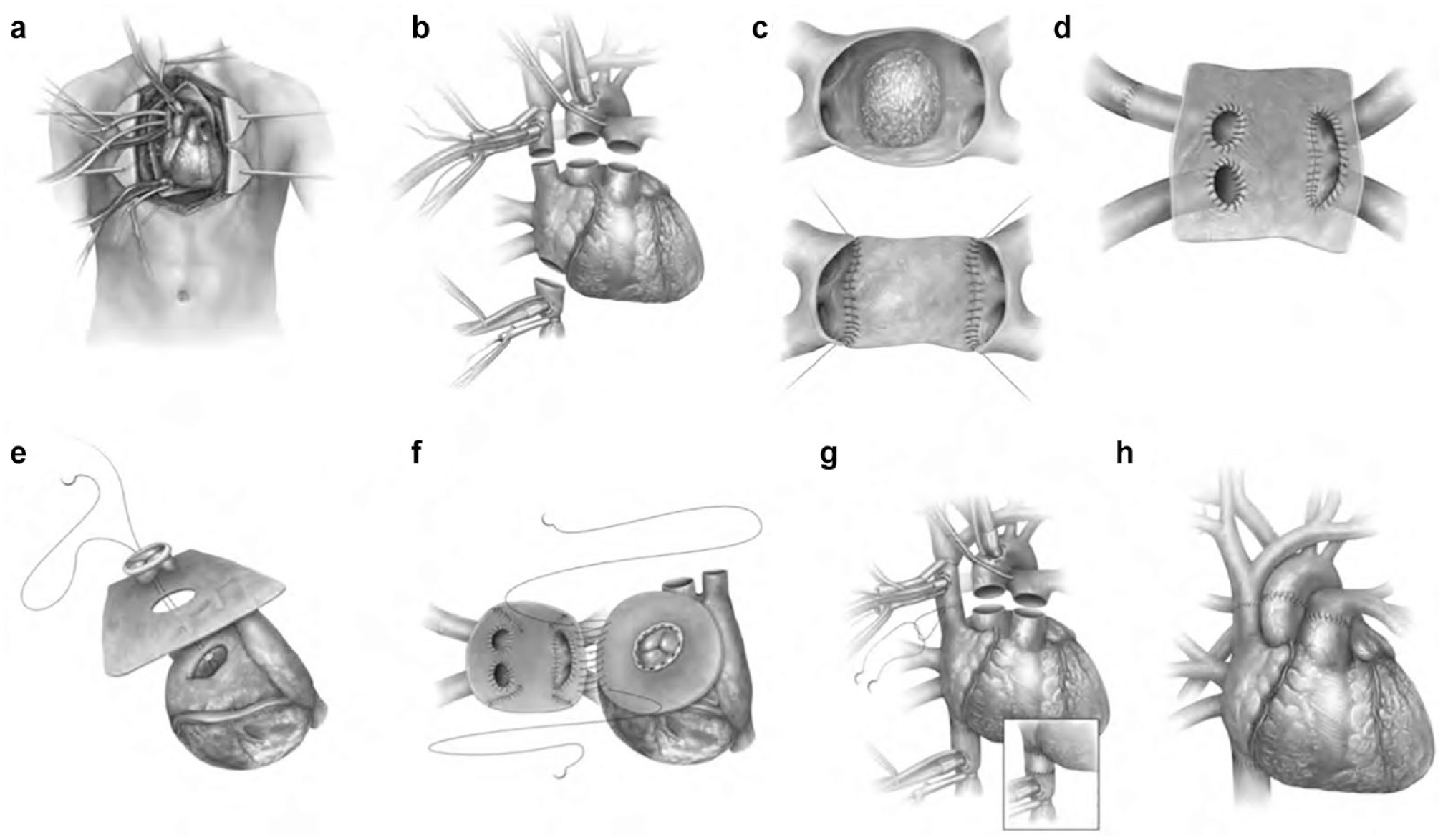
Technique of cardiac autotransplantation. (a) Median sternotomy, cannulation for cardiopulmonary bypass. (b) Explantation of the heart. (c) Ex vivo resection of left atrial tumor, reconstruction of the posterior wall using bovine pericardium. (d) Ex vivo reconstruction of the posterior wall and pulmonary vein using the Gortex graft. (e) Ex vivo reconstruction of the anterior wall using bovine pericardium and mitral valve replacement using bioprosthesis. (f) Orthotopic implantation, left atrial suture line. (g) Caval suture lines. If either cava is too short, an interposition graft might be very useful (insert). (h) After completion of the great vessels anastomoses. Reprinted from Shapira et al., Radical multidisciplinary approach to primary cardiac sarcomas, Eur J Cardiothorac Surg, 2013, 44(2), pp 330–335, by permission of by Oxford University Press on behalf of the European Association for Cardio-Thoracic Surgery. 17
To simplify the technical challenges associated with this procedure, we developed the technique of partial cardiac autotransplantation, leaving the inferior vena cava attached. 20 Left ventricular PCS requires particular consideration concerning resectability as well as approach. We have used an ex vivo approach through the mitral valve with success. 21 For PCS that required an extensive cardiac resection as well as pneumonectomy for complete resection, the mortality was high when done as a single procedure due to bleeding into the post-pneumonectomy space resulting from coagulopathy associated with the long pump time. We have subsequently separated these operations into 2 steps, the Texas Two Step procedure, doing the cardiac resection and delaying the pulmonary resection as a separate procedure several days later. 22 We have since done 10 Texas Two Step procedures with no deaths. The surgeon taking on these cases should plan carefully and understand the complexity they will encounter.
6. Plan for Lifelong Surveillance
Local recurrence has been low with complete resection, but distant disease remains the prominent cause of death. We recommend postsurgery chemotherapy to all patients. Follow-up imaging and visits are conducted every 3 months to evaluate for both local and distant disease occurrence. Most physicians will not have treated many or often any PCS, so close contact with the primary cardiac tumor team is imperative. We have created an institutional review board–approved database and follow all patients indefinitely. Teams willing to take on these rare and complex cases should also strive for complete and permanent follow-up.
7. Don’t Let Your Ego Cause Harm
PCS are rare and challenging. Cardiac surgeons generally like to rise to challenges, but surgeons should be cognizant of the complexity and risk these cases bring and the potential harm to patients if approached inappropriately. We have established a web-based monthly cardiac tumor conference not only to discuss our own cases but also to review cases with other institutions to help provide a team-based approach. Literature exists to help orient the surgeon and heart tumor teams to the medical and surgical approach to these tumors.1,5,11,14,23 Finally, surgeons and heart tumor teams should take to heart the complexity of primary cardiac sarcoma and understand that it is a systemic disease requiring a systemic approach.24,25
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
