Abstract
Objective:
The aim of this study was to investigate the outcomes of robotic mitral valve surgery with and without concomitant tricuspid valve surgery.
Methods:
Patients who underwent robotic mitral surgery between March 2010 and September 2022 were included. Patients were grouped according to the presence of concomitant tricuspid interventions. The groups were compared for baseline factors, operative parameters, and early postoperative outcomes. Age- and gender-matched groups were also compared for outcomes.
Results:
The study included 285 robotic mitral surgery patients. There were 59 patients who underwent concomitant tricuspid interventions. In the concomitant tricuspid surgery group, cardiopulmonary bypass time (150.1 vs 128.4 min, P < 0.001) and cross-clamp time (99.2 vs 82.4 min, P < 0.001) were longer. Prolonged intubation was more frequent in the concomitant tricuspid intervention group (5.2% vs 0.5%, P = 0.029). The groups did not differ in terms of mortality, permanent pacemaker (PPM) requirement, or other morbidities. Perioperative outcomes were similar after matched group analysis.
Conclusions:
Operative mortality and early adverse outcomes did not increase with the addition of tricuspid intervention in our cohort of robotic mitral surgery patients. The robotic approach for mitral disease and coexisting tricuspid disease may offer safe results without an increased risk of postoperative PPM requirement.
Central Message
The results from this cohort of robotic mitral surgery patients found that concomitant tricuspid intervention was not associated with a greater incidence of operative mortality or early adverse outcomes, including permanent pacemaker requirement.
Introduction
Robotic mitral surgery has been applied safely with excellent results for more than 2 decades. With the increase in surgical experience and developments in robotic apparatus, additional interventions during mitral surgery, including correction of the atrial septal defect, surgical cryoablation, and tricuspid valve surgery, are performed easily.1–3
Tricuspid valve disorders often accompany mitral valve disease, with a reported coincidence of up to 64%.4–6 Correction of tricuspid valve pathology has proven to have long-term benefits in terms of survival and quality of life. The presence of pulmonary hypertension or signs of right ventricular failure may require the tricuspid valve pathology to be addressed during mitral surgery. 4 The addition of right-sided procedures can be safely performed during conventional mitral valve surgery.6–9 Minimally invasive approaches in mitral surgery offer several additional benefits to mitral surgery and allow concomitant intervention to the tricuspid valve. There are concerns regarding the higher risk of permanent pacemaker (PPM) requirement when tricuspid valve repair or replacement is added to mitral valve operations with conventional or minimally invasive approaches. However, there is a lack of literature regarding robotic mitral surgery with additional right-sided procedures. 10 In this study, we report our early operative results with robotic mitral surgery, with and without additional tricuspid intervention.
Methods
Patients who underwent robotic mitral surgery between March 2010 and September 2022 at a single institution were included. Patients with underlying hypertrophic cardiomyopathy or congenital disorders were excluded from the study. Approval from the Acibadem Maslak Hospital Institutional Review Board was obtained before beginning the study. Informed consent was waived due to the retrospective nature of the study. The study was conducted in accordance with the Declaration of Helsinki.
The patients were grouped according to the presence of concomitant tricuspid interventions. The indications for concomitant tricuspid procedures included moderate or severe tricuspid regurgitation. All severe tricuspid regurgitations were intervened. Patients with infective endocarditis were excluded from the study. Moderate tricuspid regurgitation at preoperative echocardiography was assessed with operative transesophageal echocardiography (TEE), and a decision was made on a per-patient basis. The presence of organic tricuspid valve disease, right ventricular disfunction, annular size greater than 40 mm, and clinical signs of heart failure were factors that influenced our decision to intervene on patients with moderate tricuspid regurgitation. The final decision was made by the heart team considering all of the above factors. Clinical, procedural, and postoperative data were prospectively recorded in an institutional database and retrieved for this retrospective review. The groups were compared for baseline, operative, and early postoperative results. The groups were then matched for age and gender and compared for perioperative outcomes. Prolonged intubation was defined as invasive mechanical ventilation longer than 48 h.
Surgical Technique
All procedures were completed by a single surgical team using the robotic da Vinci Xi or Si systems (Intuitive Surgical, Sunnyvale, CA, USA). Patients of advanced age with concomitant coronary artery disease or peripheral arterial disease underwent routine preoperative computed tomographic angiography. Peripheral cannulation was performed under ultrasound and echocardiography guidance. 11 In cases of extensive atherosclerotic femoral arteries or plaque/thrombus formation at the level of the abdominal or thoracic aorta, the axillary artery was cannulated with surgical exposure. In both patient groups, undergoing either isolated mitral valve surgery or combined mitral and tricuspid valve surgery, the cannulation consisted of a percutaneously placed superior vena cava cannula through the internal jugular vein and an inferior vena cava cannula placed through the femoral vein, under TEE guidance. If additional tricuspid intervention was planned, the bulldog clamps were then placed on the superior and inferior vena cava before right atriotomy. In patients with severe calcifications at the level of the ascending aorta, the operation was performed under hypothermic fibrillation without cross-clamping.
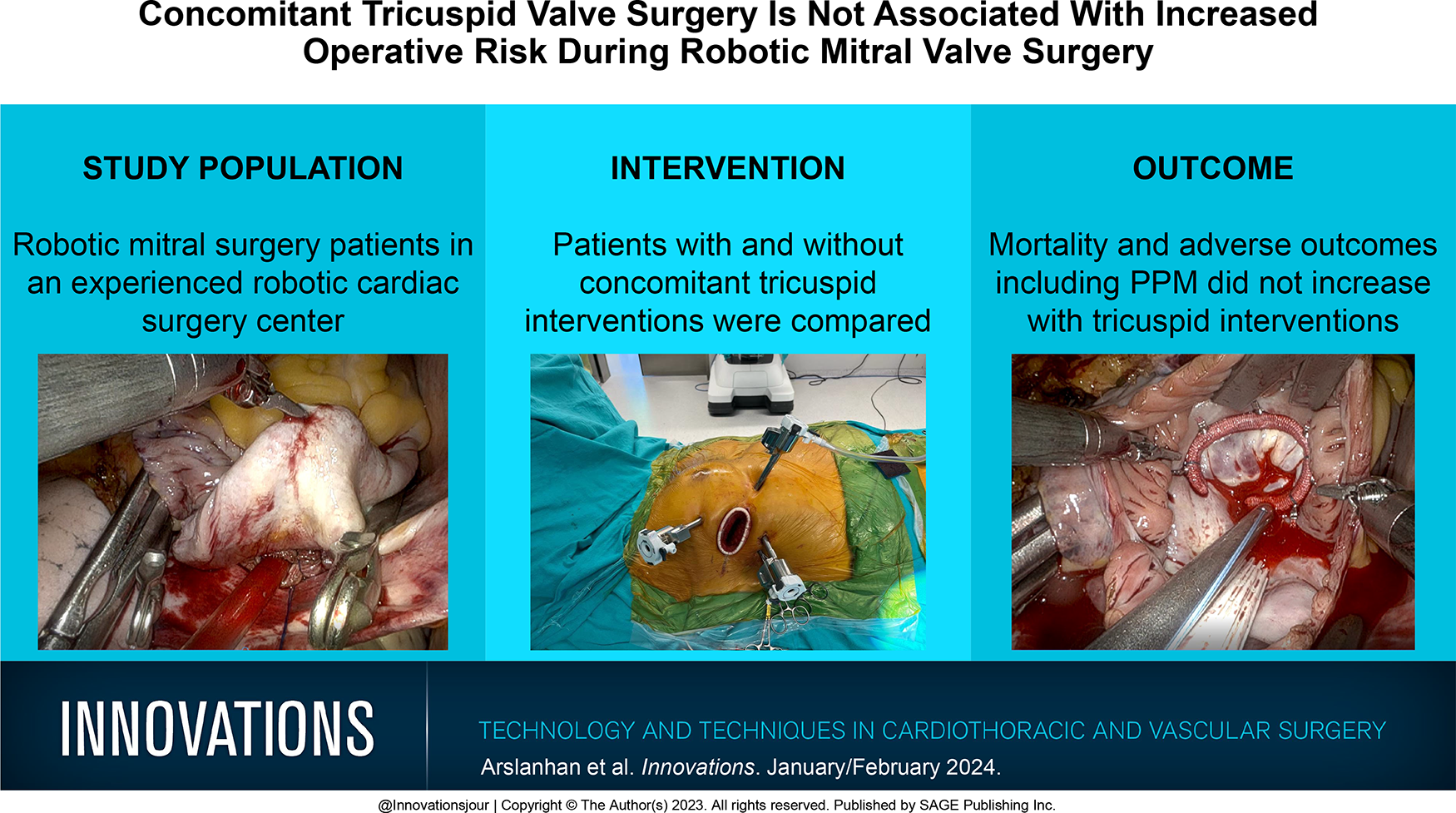
A minithoracotomy, usually 4 cm, was performed at the right fourth intercostal space. The third and fifth intercostal spaces were used for the insertion of left and right working ports, respectively (Fig. 1). The XS sized working port was more comfortably used during valve replacements; an XXS sized port does not easily allow the introduction of the new valve. In mitral repair cases, an XXS port was sufficient. The Chitwood clamp and a suction vent were placed through a stab incision at the second intercostal space in the anterior axillary line. A soft tissue retractor was placed after the port implantation. The camera was placed through the soft tissue retractor, and cardiopulmonary bypass (CPB) was initiated.
Operative setup for the robotic approach to mitral and tricuspid valves.
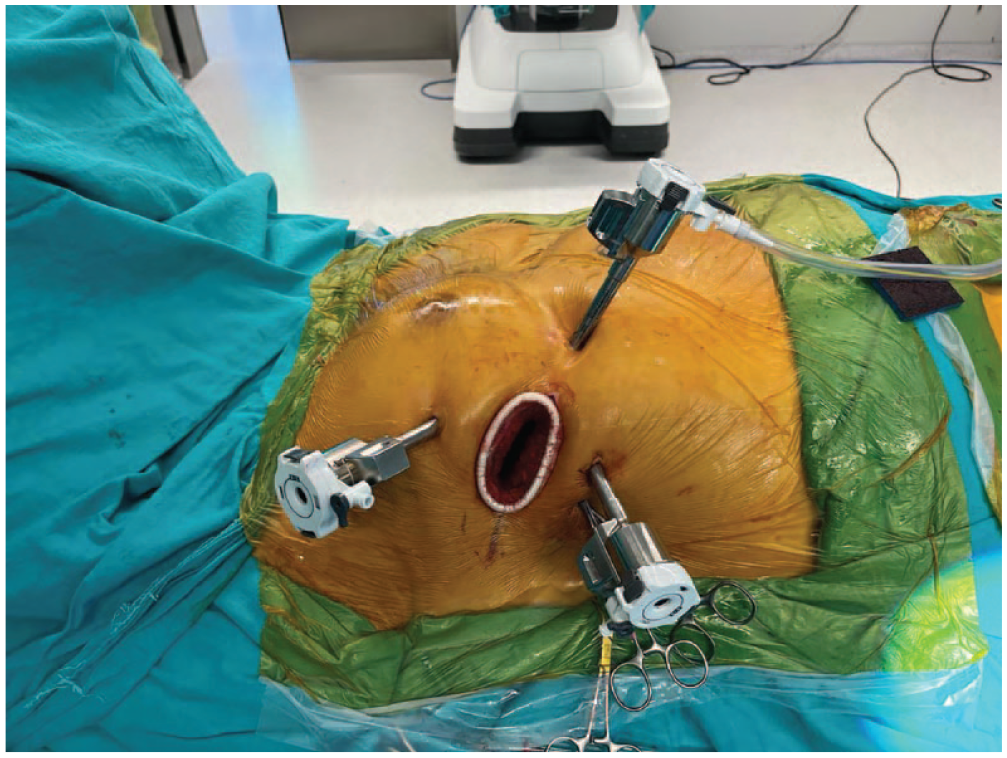
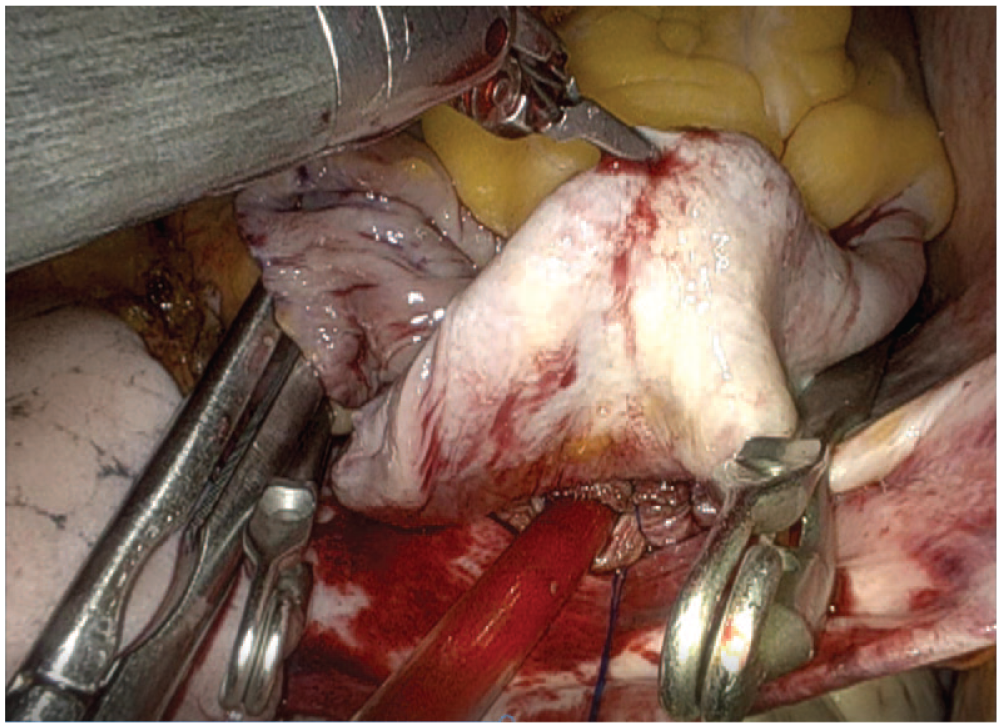
The commencement of CPB was followed by the opening of the pericardium, classically with an incision of 2 cm anterior and parallel to the phrenic nerve and further fixation externally. A Chitwood aortic clamp was inserted in the direction of the transverse sinus. Cardiac arrest was established with a cross-clamp and antegrade cardioplegia delivered through a temporary needle in the ascending aorta through the soft-tissue retractor. The preferred cardioplegia solution was Custodiol HTK Solution, which was switched to modified del Nido Solution for all robotic mitral cases after 2020. The interatrial groove was dissected, left atriotomy was performed, and an atrial retractor was placed into the left atrium (LA). The LA appendage was ligated in all patients with atrial fibrillation (AF) using a double-layered running Prolene 3.0 suture from inside the LA. In patients with AF, the cryoablation probe (Cardioblate, CryoFlex; Medtronic, Dublin, Ireland) was delivered through the working port. The probe was fit to the shape of the atrial wall, creating cryolesions of 1 to 10 cm in length. The LA was closed following the completion of mitral valve repair or replacement. In the case of tricuspid valve pathology, the inferior and superior vena cavae were occluded with bulldog clamps (Fig. 2), followed by right atriotomy and placement of an atrial retractor into the right atrium. The tricuspid valve was examined, the annulus was sized for prosthesis, and sutures were implanted. A semi-rigid ring was used for all tricuspid repair cases. Pledgeted sutures were used for tricuspid valve replacement cases, and nonpledgeted sutures were used for tricuspid repair cases. Apart from this, pledgeted sutures were used as necessary in parts of the annulus in repair cases if the atrial tissue was observed to be fragile. Once all the sutures were completed, they were passed through the annulus of the valve or ring prosthesis outside the thoracic cavity. The valve or ring was removed from its holder and deployed through the soft-tissue retractor. The pledgeted sutures were secured using an automatic mechanical knot fastening system (COR-KNOT; LSI SOLUTIONS, Victor, NY, USA; Fig. 3) The right atriotomy was closed using a premade loop suture. Both bulldog clamps in the vena cavae were released. After deairing, the aortic cross-clamp was removed. Subsequently, TEE was performed to check for any abnormal findings or air traps. The patient was then weaned off CPB.
Bulldog clamps used to clamp the vena cavae.
Placement of the tricuspid ring using the automated suture-fastening system.
Evaluation of PPM Requirement
Patients were monitored with 12-lead electrocardiogram daily after the operation until discharge. Patients with Mobitz type 2 or third-degree atrioventricular (AV) block were observed with telemetry for 7 days on a temporary pacemaker prior to the decision for a PPM implantation. Patients with persistent AV block were monitored with 24 h continuous rhythm Holter to confirm the decision, and a PPM was implanted if Mobitz type 2 or third-degree AV block was observed after 7 days.
Statistical Analysis
IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. Continuous parameters are given as mean and standard deviation, while categorical parameters are given as numbers and percentages. The chi-squared test was applied to compare categorical variables. Student’s t test was used for comparisons between 2 groups of normally distributed continuous variables, and Mann–Whitney U test was used for comparisons between 2 groups of non-normally distributed continuous variables. The groups with and without tricuspid interventions were matched for age and gender to compare outcomes.
Results
During the study period, 451 patients underwent robotic cardiac surgeries. A total of 301 patients underwent surgery for mitral valve disease, and 16 patients were excluded from this study. Of the 285 patients included, 59 received concomitant tricuspid interventions. The mean age of the included patients was 56.5 ± 15.1 years, 171 patients (60.0%) were female, 29 patients had a history of prior cardiac surgery, and 233 patients (81.8%) had New York Heart Association class 2 or more severe symptoms. The demographic and operative characteristics of the patients are summarized in Table 1. The main indication for mitral surgery was mitral insufficiency (n = 207, 72.6%), and the main mitral and valve pathology was degenerative disease (n = 146, 52.1%). In mitral valve repair due to degenerative valve disease, ring annuloplasty was done routinely, and chorda implantation to anterior and posterior leaflets was performed when necessary. Rarely, leaflet resection (triangular/quadrangular P2 resection) was also performed. In rheumatic disease, mitral valve replacement was more frequently performed. Mitral valve repair was performed in 150 patients (52.6%), and 52 of 59 patients (88.1%) with tricuspid intervention received tricuspid annuloplasty. The remaining patients who received tricuspid valve replacement had organic tricuspid disease. The distribution of procedures performed is summarized in Table 2.
Baseline Characteristics of Study Patients.
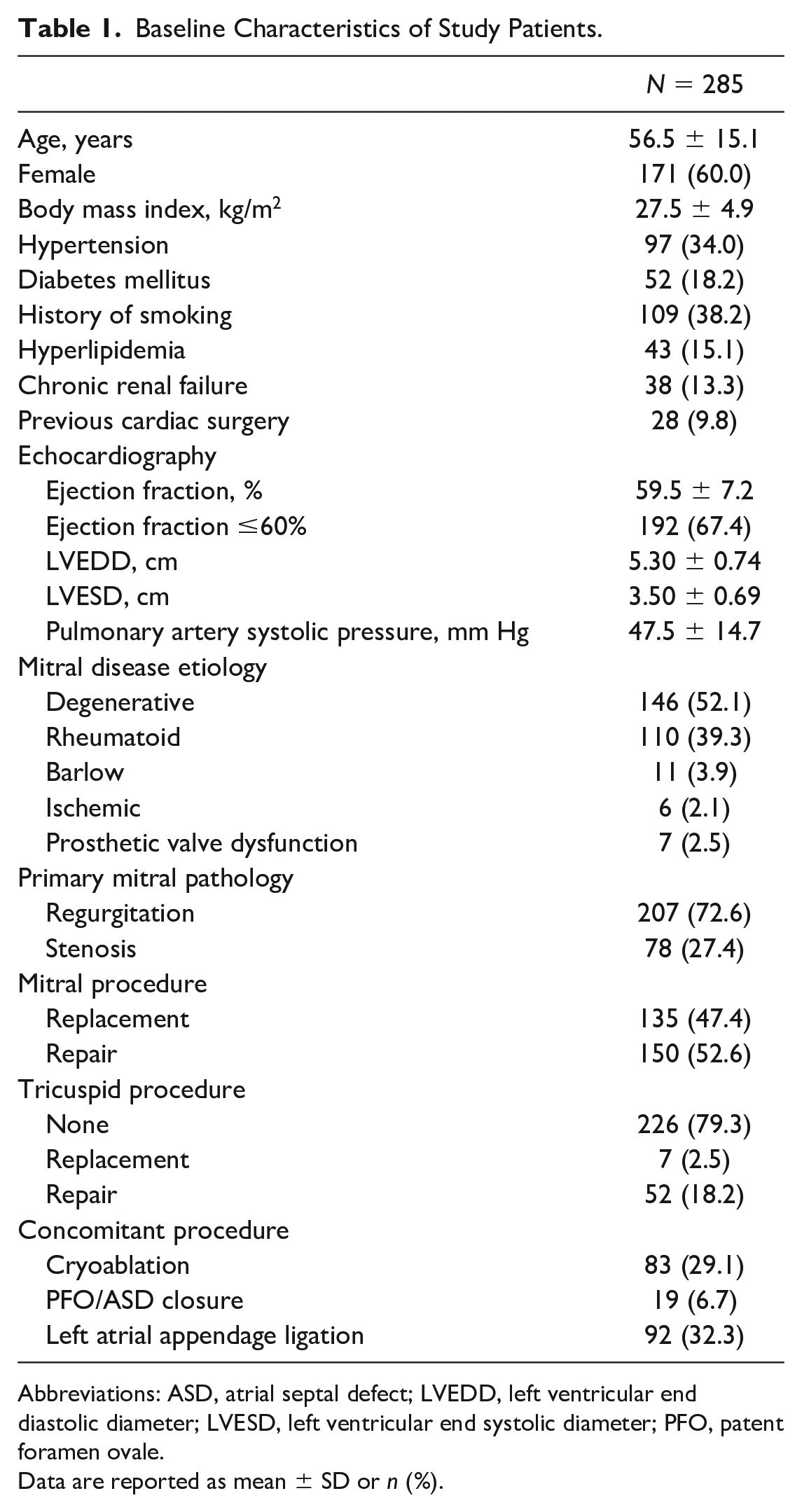
Abbreviations: ASD, atrial septal defect; LVEDD, left ventricular end diastolic diameter; LVESD, left ventricular end systolic diameter; PFO, patent foramen ovale.
Data are reported as mean ± SD or n (%).
Distribution of Operations Performed.
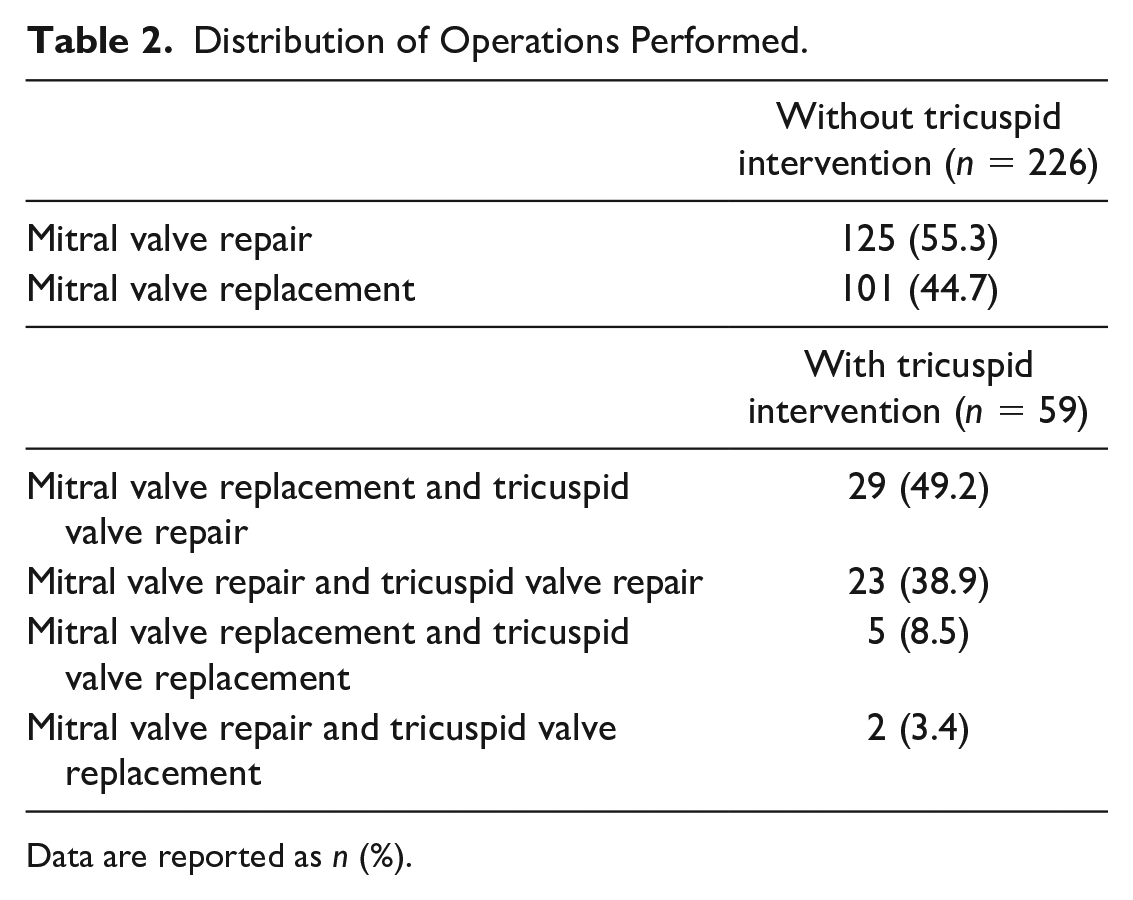
Data are reported as n (%).
The demographic and operative characteristics of the groups were compared (Table 3). Patients who required concomitant tricuspid intervention were older (66.3 vs 53.9 years, P < 0.001), were more frequently female (86.4% vs 53.1%, P < 0.001), and more frequently had diabetes (30.5% vs 15.0%, P = 0.006), chronic renal disease (20.3% vs 11.5%, P = 0.015), AF (69.5% vs 19.9%, P < 0.001), and a history of previous cardiac surgery (69.5% vs 19.9%, P < 0.001). Based on echocardiography, patients who required tricuspid intervention had lower ejection fractions (58.3% vs 59.9%, P = 0.009) and higher pulmonary arterial pressure (53.8 vs 45.6 mm Hg, P < 0.001). Additional cryoablation and LA appendage ligation procedures were performed more frequently in patients undergoing tricuspid intervention.
Comparison of Baseline Characteristics Among Groups.
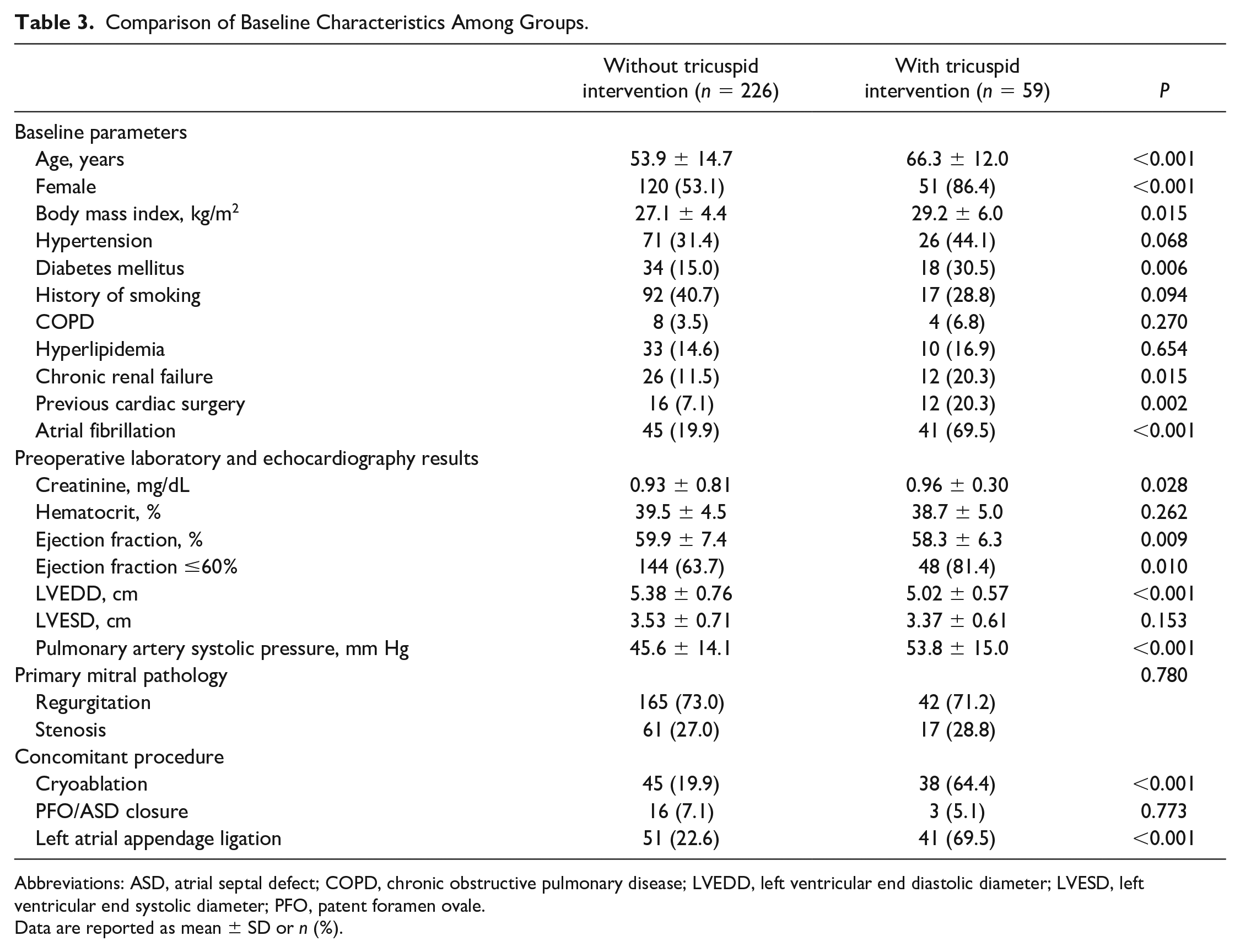
Abbreviations: ASD, atrial septal defect; COPD, chronic obstructive pulmonary disease; LVEDD, left ventricular end diastolic diameter; LVESD, left ventricular end systolic diameter; PFO, patent foramen ovale.
Data are reported as mean ± SD or n (%).
In patients who underwent concomitant tricuspid intervention, the CBP (150.1 vs 128.4 min, P < 0.001) and cross-clamp times (99.2 vs 82.4 min, P < 0.001) were longer. Prolonged intubation was more frequent in the concomitant tricuspid intervention group (5.2% vs 0.5%, P = 0.029). The groups did not differ in terms of acquired AF and PPM requirements. Three patients in the isolated mitral surgery group and 1 patient in the concomitant tricuspid surgery group had third-degree AV block for 7 days postoperatively. These 4 patients were implanted with PPMs. Mortality and other early postoperative outcomes were similar between the groups (Table 4). A comparison was made with age- and gender-matched groups for perioperative results (Table 5). Mortality and morbidity, including PPM requirement, were similar between the groups with and without tricuspid intervention after matching.
Comparison of Groups for Postoperative Outcomes.
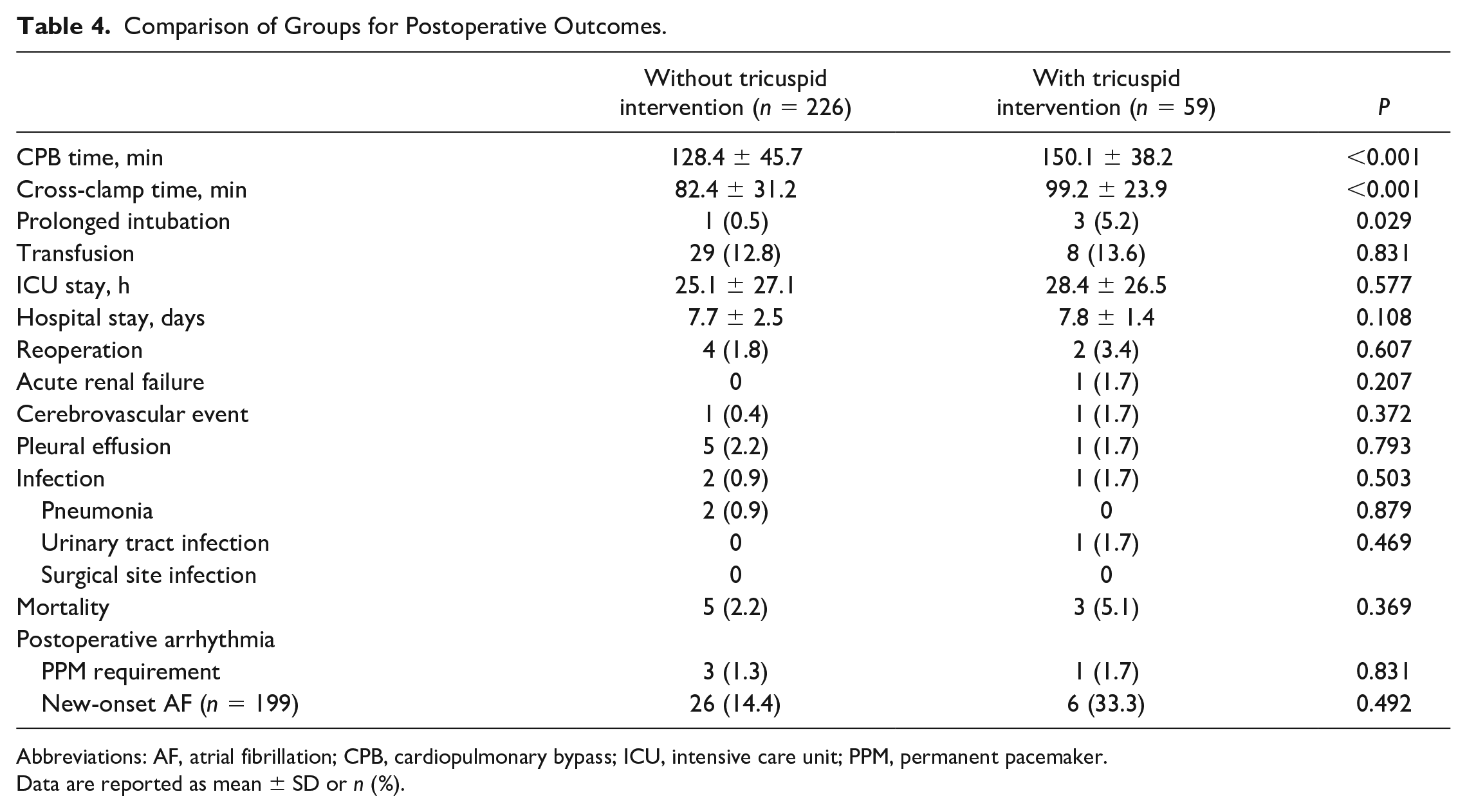
Abbreviations: AF, atrial fibrillation; CPB, cardiopulmonary bypass; ICU, intensive care unit; PPM, permanent pacemaker.
Data are reported as mean ± SD or n (%).
Comparison of Age- and Gender-Matched Groups.
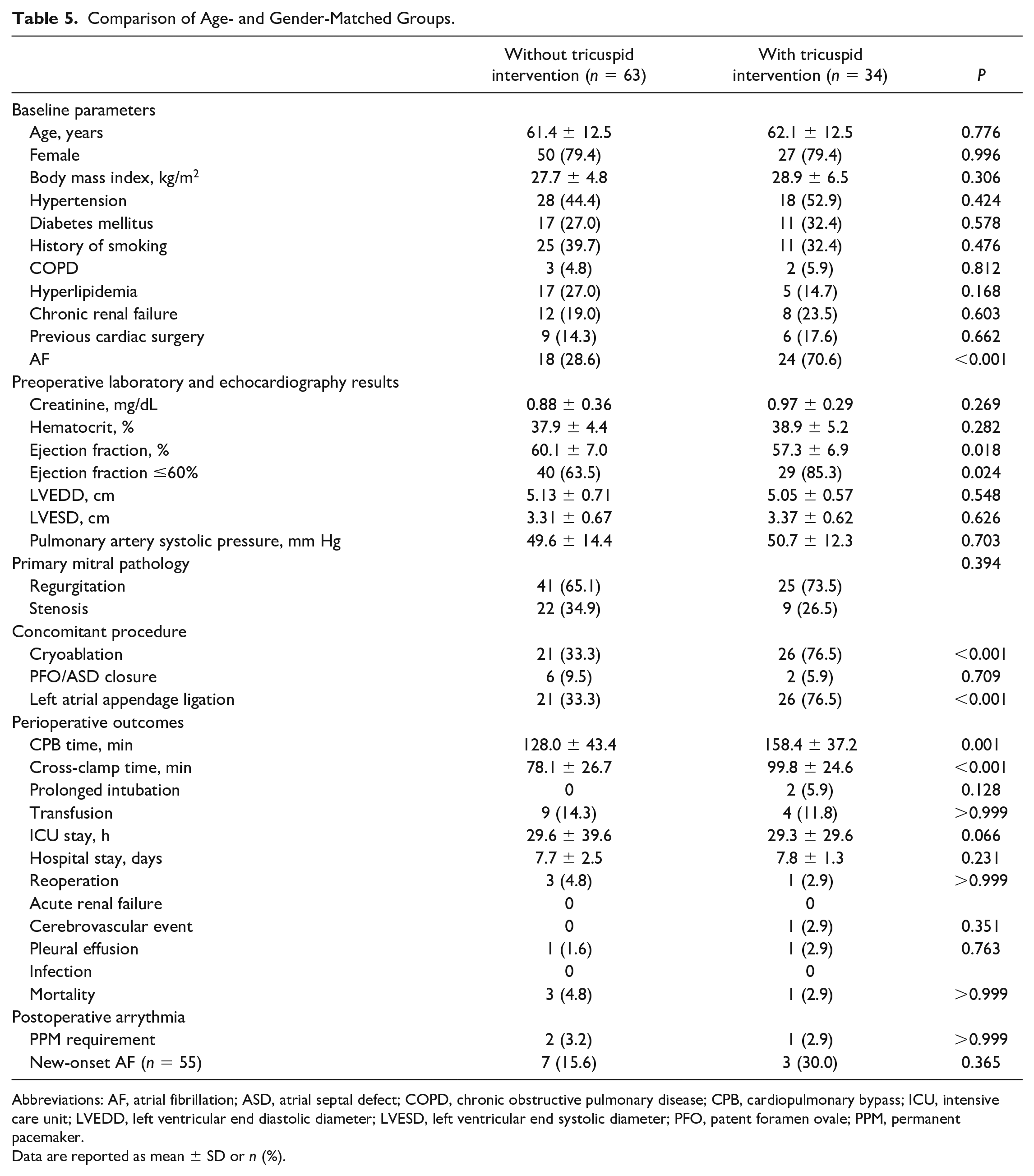
Abbreviations: AF, atrial fibrillation; ASD, atrial septal defect; COPD, chronic obstructive pulmonary disease; CPB, cardiopulmonary bypass; ICU, intensive care unit; LVEDD, left ventricular end diastolic diameter; LVESD, left ventricular end systolic diameter; PFO, patent foramen ovale; PPM, permanent pacemaker.
Data are reported as mean ± SD or n (%).
Discussion
In our study, we analyzed 285 patients who underwent robotic mitral surgery and compared patients with and without concomitant tricuspid surgery. Despite increased cross-clamp and CPB durations, operative mortality and early adverse outcomes did not increase in our cohort with the addition of a tricuspid intervention to robotic mitral surgery. The outcomes were also similar between the age- and gender-matched groups.
There is a concern for worse outcomes with the addition of a tricuspid intervention to mitral valve surgery. One multicenter study found an increased adjusted mortality risk with concomitant tricuspid valve surgery. 12 However, the reported mortality in patients with tricuspid interventions is generally not increased in other single-center studies. In a series of patients operated via median sternotomy and in cohorts of minimally invasive mitral repair patients, perioperative mortality was similar with and without an additional tricuspid procedure.7,9,13 An analysis of the Society of Thoracic Surgeons national database supports this view that additional tricuspid valve surgery was not associated with an increase in adjusted mortality. 14 Although the group that underwent additional tricuspid surgery in our cohort was older and had more comorbidities, we did not find an increased risk of mortality.
The risk of other early postoperative adverse outcomes was similar with and without additional tricuspid intervention in our series of robotic mitral surgery. Cerebrovascular events, acute renal failure, pneumonia, other infections, and transfusion requirements are not more common in concomitant tricuspid valve surgery patients in reports from single-center studies.9,15,16 On the other hand, prolonged intubation was more frequent in our cohort in the group with additional tricuspid surgery. Longer ventilation times have been reported from a single-center matched cohort of mitral surgery patients with and without tricuspid valve repair. 7 However, this increase in ventilation times did not translate to higher rates of infection or intensive care unit stay in our cohort or in the literature.
The addition of tricuspid surgery to mitral surgery was safe in terms of mortality and morbidity in our patients, despite the additional cross-clamp and CPB durations with right-sided interventions. In our series, the addition of tricuspid surgery resulted in a mean increase of 17 min in cross-clamp time and 22 min in CPB time. Similar increases in cross-clamp and CPB times have been reported with both conventional and minimally invasive approaches in mitral surgery. 17 To avoid excessive cross-clamp times in robotic surgery, we utilize certain techniques such as the Leyla loop for atriotomy closure and endoscopic bulldog clamping for bicaval occlusion.18,19 Considering the added complexity of a minimally invasive approach in mitral and tricuspid surgery, centers may consider performing isolated mitral surgeries during the learning curve period of their robotic surgery programs and including patients with right-sided pathologies later on to avoid long cross-clamp and CPB times to ensure good outcomes with combined mitral and tricuspid procedures.
A significant risk with additional tricuspid intervention in mitral surgery is the increase in PPM requirement.6,8,9,17,20 The conduction tissues in close proximity to the mitral and tricuspid annulus are at risk of trauma from sutures or prosthetic devices. In isolated mitral surgery, the need for PPM has been reported at a range of 2.5% to 8.8%.20–22 Our rate in isolated mitral cases was lower at 1.3%. With additional right-sided interventions, the PPM requirement was reported at a range of 14.1% to 21.0% in other series.9,17,20,22–24 The timing of PPM implantation in valve surgery patients is not well-established with a paucity of recommendations for the optimal time in the early postoperative course. In a study of more than 15,000 cardiac surgery patients, PPM implantation on the seventh postoperative day was safe and prevented unnecessary implantations. 25 Longer cross-clamp times have been implicated in the increased PPM need. 26 Cross-clamp times are generally longer in minimally invasive methods compared with surgery via sternotomy, and patients who require tricuspid intervention are more frequently comorbid. However, our PPM rates with concomitant tricuspid surgery were not increased despite these factors. In cases that required both a tricuspid valve intervention and cryoablation, we performed the cryoablation procedure only in the LA and took care to place our annular sutures with as low traction as possible for both the mitral and tricuspid valves. These technical points may explain the low PPM rate despite a greater rate of cryoablation in the tricuspid intervention group.
Robotic surgery is generally adopted very slowly in mitral valve surgery, due to increased costs, the need for dedicated nursing care, and, more importantly, a lack of proper training. Although the mitral repair technique is similar whether performed via sternotomy or robotic assistance, the setup and challenges of these 2 approaches are highly different. The learning curve of complex cases may require a long time period. It is highly important to start a robotic program with suitable, low-risk cases with simple pathologies. Experience in conventional mitral repair can improve the outcomes during the initial phases of a robotic program. We experienced a substantial decrease in cross-clamp and CPB times as we became familiar with and experienced in the robotic technique. Moreover, the learning curve concerns not only the surgeon but also the whole surgical team including anesthesiologists, scrub nurses, and perfusionists. Members of the robotic team were exchanged very rarely, as we believe that working with a stable, consistent team improves all outcomes.
The robotic approach for coexisting tricuspid disease during mitral surgery may offer a safe approach without an increased risk of pacemaker requirement. The robotic approach may provide benefits in protection against PPM requirement. These benefits may arise from the 3-dimensional vision camera and enhanced exposure by the dynamic atrial retractor that allow easier visualization and correction of the mitral pathologies that assist in lowering cross-clamp times. Improved suture precision with the tactile feedback of the robotic arms may also be important in avoiding damage to the surrounding conduction tissues.
Limitations
The study was performed at a single center with a single surgical team. Surgical outcomes may be influenced by varying surgeon experience and different patient cohorts in different centers. Another limitation is the low sample size of included patients. Our study did not include a control group of sternotomy patients, which could provide additional information on the safety of the robotic approach.
Conclusions
Operative mortality and early adverse outcomes did not increase with the addition of tricuspid intervention in our cohort of robotic mitral surgery patients. The robotic approach for mitral disease and coexisting tricuspid disease may offer a safe approach without an increased risk of postoperative PPM requirement.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
