Abstract
Objective:
The optimal approach and choice of initial aortic valve replacement (AVR) is evolving in the growing era of transcatheter AVR. Further survival and hemodynamic data are needed to compare the emerging role of rapid deployment (rdAVR) versus stented (sAVR) valve options for AVR.
Methods:
The Northern New England Cardiovascular Database was queried for patients undergoing either isolated AVR or AVR + coronary artery bypass grafting (CABG) with rdAVR or sAVR aortic valves between 2015 and 2021. Exclusion criteria included endocarditis, mechanical valves, dissection, emergency case status, and prior sternotomy. This resulted in a cohort including 1,616 sAVR and 538 rdAVR cases. After propensity weighting, procedural characteristics, hemodynamic variables, and survival outcomes were examined.
Results:
The breakdown of the overall cohort (2,154) included 1,164 isolated AVR (222 rdAVR, 942 sAVR) and 990 AVR + CABG (316 rdAVR, 674 sAVR). After inverse propensity weighting, cohorts were well matched, notable only for more patients <50 years in the sAVR group (4.0% vs 1.9%, standardized mean difference [SMD] = −0.12). Cross-clamp (89 vs 64 min, SMD = −0.71) and cardiopulmonary bypass (121 vs 91 min, SMD = −0.68) times were considerably longer for sAVR versus rdAVR. Immediate postreplacement aortic gradient decreased with larger valve size but did not differ significantly between comparable sAVR and rdAVR valve sizes or overall (6.5 vs 6.7 mm Hg, SMD = 0.09). Implanted rdAVR tended to be larger with 51% either size L or XL versus 37.4% of sAVR ≥25 mm. Despite a temporal decrease in pacemaker rate within the rdAVR cohort, the overall pacemaker frequency was less in sAVR versus rdAVR (4.4% vs 7.4%, SMD = 0.12), and significantly higher rates were seen in size L (10.3% vs 3.7%, P < 0.002) and XL (15% vs 5.6%, P < 0.004) rdAVR versus sAVR. No significant difference in major adverse cardiac events (4.6% vs 4.6%, SMD = 0.01), 30-day survival (1.5% vs 2.6%, SMD = 0.08), or long-term survival out to 4 years were seen between sAVR and rdAVR.
Conclusions:
Rapid deployment valves offer a safe alternative to sAVR with significantly decreased cross-clamp and cardiopulmonary bypass times. Despite larger implantation sizes, we did not appreciate a comparative difference in immediate postoperative gradients, and although pacemaker rates are improving, they remain higher in rdAVR compared with sAVR. Longer-term hemodynamic and survival follow-up are needed.
Central Message
In this analysis of a multicenter database, rapid deployment aortic valves were found to be safe with similar short-term and long-term survival compared with stented AVR. Rapid deployment valves have significantly faster operative times and are associated with larger valve implants. Although pacemaker rates are higher overall, they are steadily declining and do not differ by type of rapid deployment valve implanted.
Introduction
The development of rapid deployment aortic valve replacement (rdAVR) technology has arisen as an alternative to standard surgical AVR (sAVR). Compared with sAVR, rdAVR offers faster implantation and decreased cardiopulmonary bypass (CPB) and cross-clamp times.1–5 Compared with transcatheter AVR, rdAVR offers the benefits of a self-expanding bioprosthesis with proper surgical debridement of the annulus contributing to more favorable hemodynamic profiles and low gradients.6–9 The rdAVR has excelled in the promotion of minimally invasive platforms to approach AVR as well as a useful adjunct in complex cardiac procedures,10–12 and it has been shown in some studies to have improved perioperative outcomes such as length of stay, atrial fibrillation rates, and transfusion rates.1,2,13 In addition, short-term survival, hemodynamic, and durability data are comparable with those of sAVR.14 –16 However, rdAVR has been shown to have an increased permanent pacemaker (PPM) rate compared with sAVR, which in some studies has been greater than 10%.16–18 The morbidity and long-term mortality impacts of this associated PPM rate remain undetermined. There is a growing use of rdAVR and a need for further data to define its role within the realm of AVR. As such, we seek to analyze outcomes of rdAVR and sAVR within a large multicenter regional cardiovascular database specifically as it relates to survival and important periprocedural outcomes previously mentioned, such as rates of PPM and hemodynamic performance.
Methods
Data Source
The Northern New England Cardiovascular Disease Study Group (NNECDSG) is a consortium of hospitals in Vermont, New Hampshire, and Maine. The multicenter NNECDSG collects data on coronary artery bypass grafting (CABG), valve replacement surgery, percutaneous coronary interventions, and structural heart procedures. The registries are validated with administrative data and linked to the Social Security Death Index (through 2018) such that analysis of long-term survival beyond hospital discharge could be analyzed. Information on patient and procedural characteristics and outcomes is collected on a standard data collection form with associated data definitions. The institutional review boards have designated the NNECDSG registry as a quality improvement registry, and therefore patient consent was not required. The analysis dataset is deidentified and is considered not human subjects research and does not require institutional review board approval.
Study Cohort
Between the years of 2015 and 2021 within 3 contributing centers in the NNECDSG, adult patients aged >18 years undergoing a primary sternotomy for single (aortic) valve replacement, with or without concomitant CABG, were included. Exclusion criteria were endocarditis, aortic dissection, redo sternotomy, emergency case status, and mechanical valve replacement. When comparing isolated aortic valve procedures, patients with cardiovascular disease (left main stenosis >50% or 3-vessel disease) were excluded. The rapid deployment aortic valves included either Edwards Intuity (Edwards Lifesciences, Irvine, CA, USA) or Corcym Perceval (Corcym, London, UK). This resulted in overall sAVR (n = 1,616) and rdAVR (n = 538) cohorts that were then further stratified into isolated AVR and AVG + CABG cohorts. Within the overall cohort, 407 (76%) of the rdAVR valves were Perceval and 132 (24%) were Intuity.
Study Outcomes
The primary endpoints were short (30-day) and long-term (4-year) survival. Secondary endpoints included procedural characteristics such as operative times, size of implant, and postoperative outcomes including length of stay, stroke, bleeding, renal failure, atrial fibrillation, readmission, myocardial infarction, and PPM rates. In addition, hemodynamic data such as postoperative gradient and paravalvular leak (PVL) rates were recorded. As published effective orifice areas for rapid deployment aortic valves are not available, comparable sizing analysis was constructed between rdAVR and sAVR in the following construct: small rdAVR comparable with 19 mm sAVR, medium rdAVR comparable with 21 to 23 mm sAVR, large rdAVR comparable with 25 mm sAVR, and extra-large rdAVR comparable with 27 mm sAVR. These results, including survival outcomes, were propensity matched and stratified by overall, AVR only, and AVR + CABG cohorts. Further subgroup analyses of PPM rates by valve type, center, and size were completed.
Statistical Analysis
Univariate comparisons of patient characteristics by valve type stratified by a single AVR or by single AVR with a CABG procedure were summarized using percentages for categorical variables and means or medians for continuous variables, with the χ² test and Student’s t test (or Wilcoxon rank-sum test for nonparametric data). All P values were two-sided, and P < 0.05 was considered statistically different.
To balance differences in patient and disease characteristics between the 2 groups, we used inverse probability weighting. The nonparsimonious logistic regression propensity model included the following variables: age, gender, body surface area, peripheral vascular disease, diabetes, chronic obstructive pulmonary disease, congestive heart failure, prior dialysis or creatinine ≥2, ejection fraction, New York Heart Association Class IV, atrial fibrillation, new coronary artery disease, white blood cell >12,000, concomitant CABG, recent myocardial infarction, priority at surgery, and hospital at which the index procedure was done. We verified the performance of the weighted model by comparing the distribution of covariates and propensity scores between groups before and after inverse probability weighting. We further used propensity score trimming to remove an extreme outlier. Assessment of covariate balance showed improvement after trimming. To assess the balance between groups, we compared the standardized mean differences (SMD). Difference in values <0.1 are generally considered to indicate a negligible difference in the mean between the 2 groups.
Crude survival rates were calculated using Kaplan–Meier estimates, with differences in survival by valve type assessed with log-rank tests. We used Cox proportional hazards models to estimate hazard ratios and their 95% confidence intervals for each outcome. A weighted log-rank test was used to compare group differences in survival function. Stata Statistical Software: Release 17 (StataCorp LLC, College Station, TX, USA) was used for all analyses.
Results
Patient Characteristics
The overall cohort consisted of 1,616 sAVR and 538 rdAVR patients. These were further subdivided into isolated AVR (942 sAVR and 222 rdAVR) and AVR + CABG (674 sAVR and 316 rdAVR) groups. Inverse propensity weighting was conducted, resulting in a well-balanced overall cohort with a slightly greater percentage of younger patients under 50 years old noted in the sAVR group (4% vs 1.9%, SMD = −0.12). Otherwise, baseline characteristics such as sex, body surface area, comorbidities, and ejection fraction were evenly weighted within the overall cohort. Other significant differences in patient characteristics existed within isolated AVR and AVR + CABG subgroups such as age, body mass index, peripheral vascular disease, prior cerebral vascular accident, and 3-vessel disease, but these did not carry significance in the overall cohort (Table 1).
Patient and Disease Characteristics After Propensity Score Weighting.
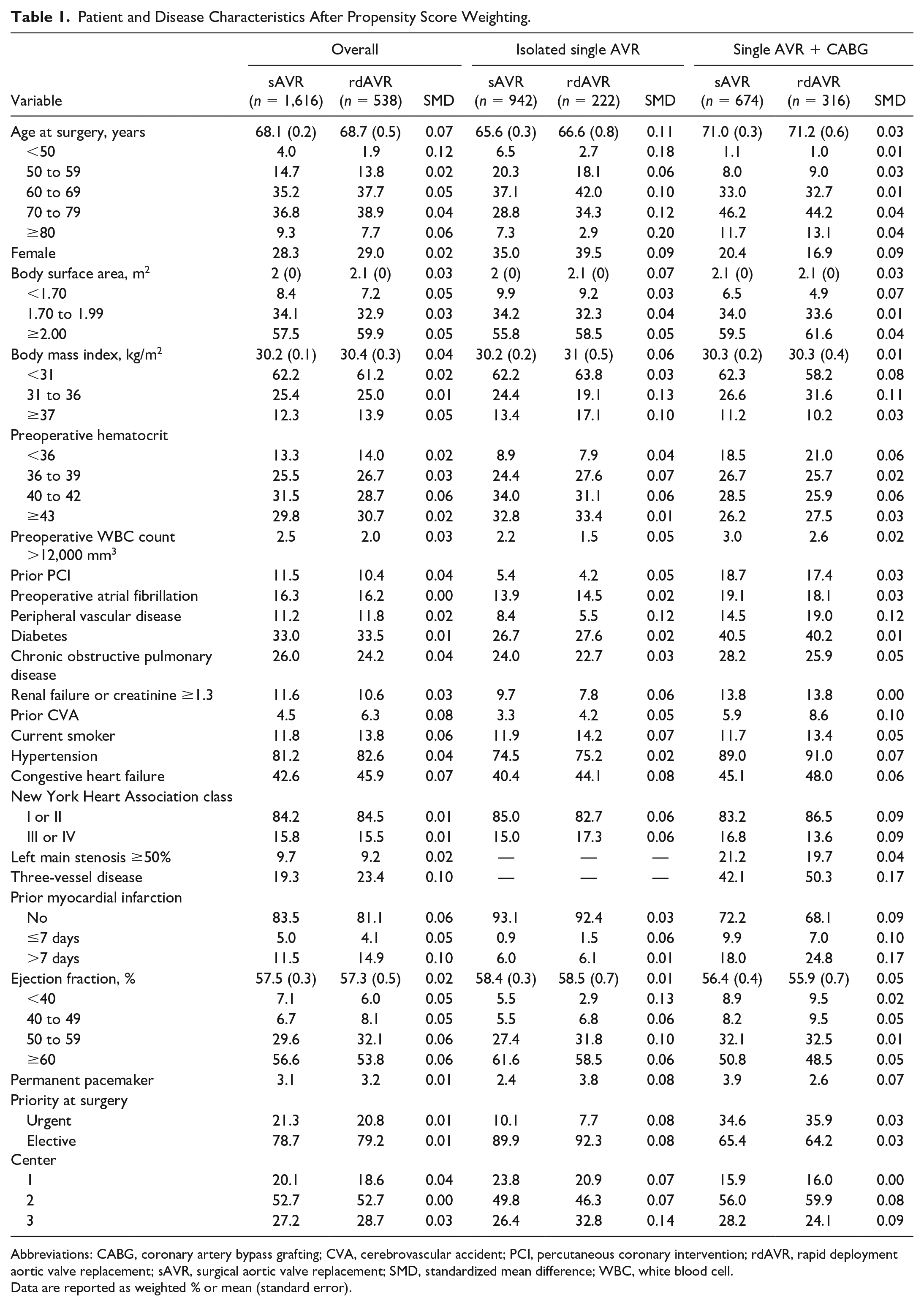
Abbreviations: CABG, coronary artery bypass grafting; CVA, cerebrovascular accident; PCI, percutaneous coronary intervention; rdAVR, rapid deployment aortic valve replacement; sAVR, surgical aortic valve replacement; SMD, standardized mean difference; WBC, white blood cell.
Data are reported as weighted % or mean (standard error).
Procedural Characteristics
Operative duration as it relates to both CPB (121 vs 90 min, SMD = −0.68) and cross-clamp times (89 vs 65 min, SMD = −0.71) were 30 and 24 min less, respectively, for the overall rdAVR group compared with the sAVR group. Both of these results persisted with significance within each of the isolated AVR and AVR + CABG groups (Table 2).
Intraoperative Characteristics by Valve Type.
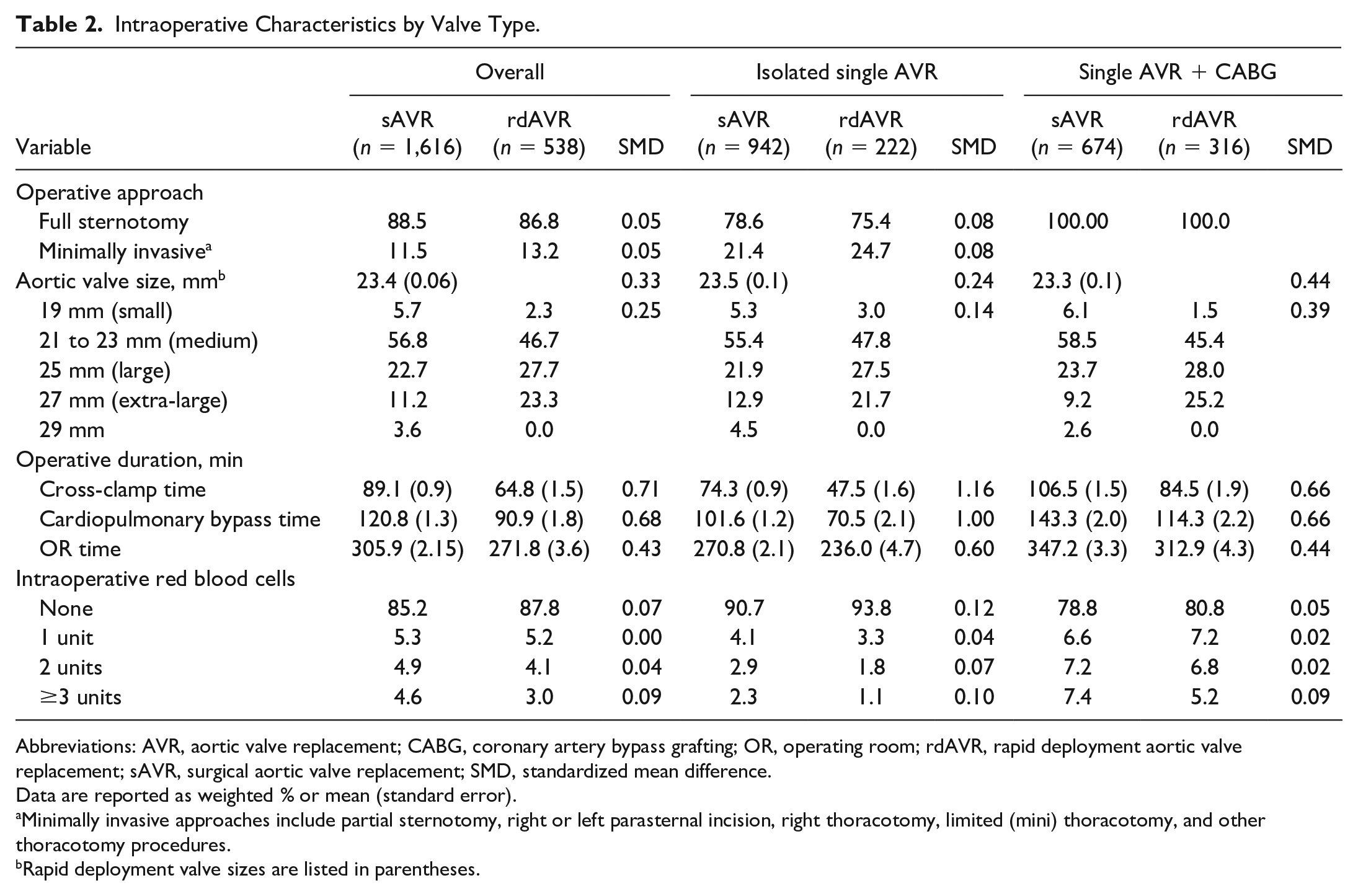
Abbreviations: AVR, aortic valve replacement; CABG, coronary artery bypass grafting; OR, operating room; rdAVR, rapid deployment aortic valve replacement; sAVR, surgical aortic valve replacement; SMD, standardized mean difference.
Data are reported as weighted % or mean (standard error).
Minimally invasive approaches include partial sternotomy, right or left parasternal incision, right thoracotomy, limited (mini) thoracotomy, and other thoracotomy procedures.
Rapid deployment valve sizes are listed in parentheses.
The most common rdAVR size implanted was size medium (47%), and 51% of rdAVRs were either size large or extra-large. The 21 and 23 mm valve sizes were the most commonly implanted sAVR valves (57%), with 33% being either size 25 or 27 mm. With regard to smaller prostheses, 5.7% of sAVR valves were 19 mm and only 2.3% of rdAVR were size small (Table 2). Isolated rdAVR had a higher rate of zero intraoperative blood transfusions compared with sAVR (94% vs 91%, SMD = 0.12), and when the operative approach was examined by center, 56% of isolated rdAVR were done with a minimally invasive approach.
Short-Term Outcomes
Postoperative characteristics are reported in Table 3. There were no differences in rates of red blood cell transfusion, bleeding, stroke, thrombocytopenia, length of stay, atrial fibrillation, ventilation times, freedom from major adverse cardiac events (MACE), PVL, or readmission rates. Rates of acute kidney injury and infection were slightly higher within rdAVR. Mean postoperative gradients did not differ significantly overall or between comparable sAVR and rdAVR valve sizes or valve type. Lower gradients were associated with larger valve prostheses (Fig. 1, Fig. 2). Overall PPM rates were significantly lower in sAVR compared with rdAVR (4.4% vs 7.4%, SMD = 0.12; Table 3). PPM rates were significantly associated with larger rdAVR valve sizes (10.9% large rdAVR and 16.3% extra-large rdAVR; Fig. 3) and differed regionally throughout the centers (Fig. 4). Temporal trends of PPM rates showed a steady decrease in PPM frequency in the rdAVR group over time beginning at 25% overall in 2015 and declining to 6.3% in 2020 (Fig. 5).
Postoperative Outcomes by Valve Type.
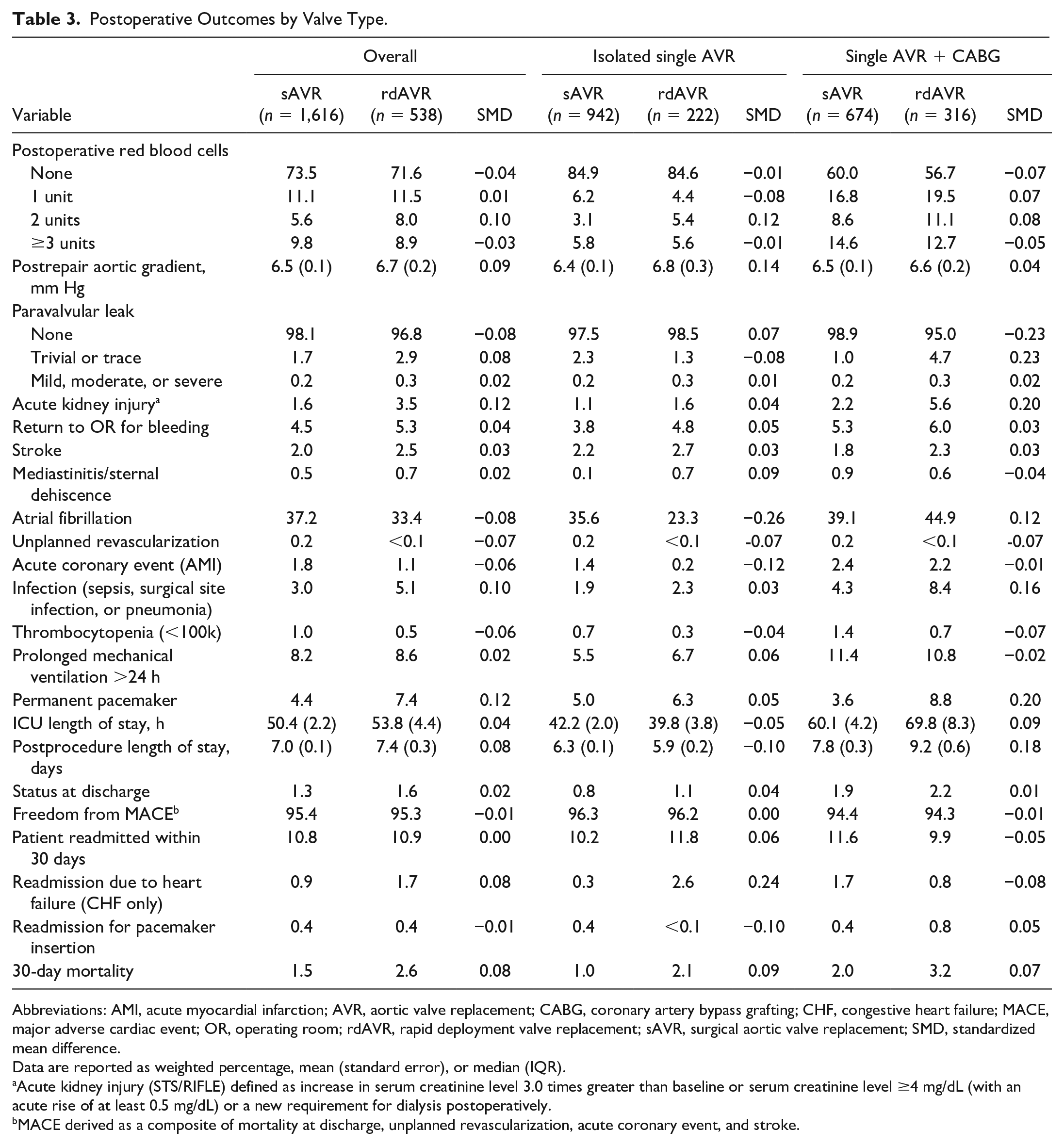
Abbreviations: AMI, acute myocardial infarction; AVR, aortic valve replacement; CABG, coronary artery bypass grafting; CHF, congestive heart failure; MACE, major adverse cardiac event; OR, operating room; rdAVR, rapid deployment valve replacement; sAVR, surgical aortic valve replacement; SMD, standardized mean difference.
Data are reported as weighted percentage, mean (standard error), or median (IQR).
Acute kidney injury (STS/RIFLE) defined as increase in serum creatinine level 3.0 times greater than baseline or serum creatinine level ≥4 mg/dL (with an acute rise of at least 0.5 mg/dL) or a new requirement for dialysis postoperatively.
MACE derived as a composite of mortality at discharge, unplanned revascularization, acute coronary event, and stroke.
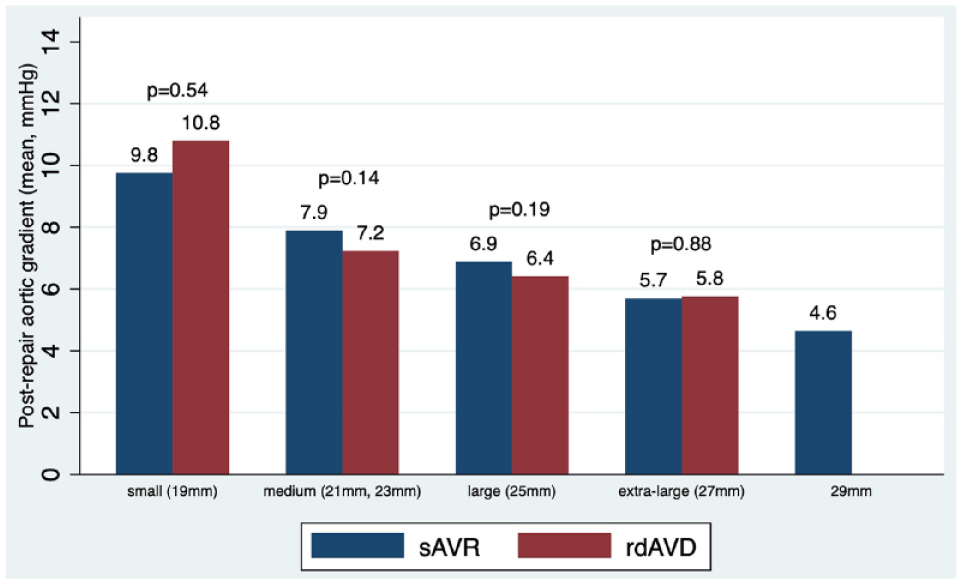
Mean postrepair gradient by valve size. rdAVR, rapid deployment valve replacement; sAVR, surgical aortic valve replacement.
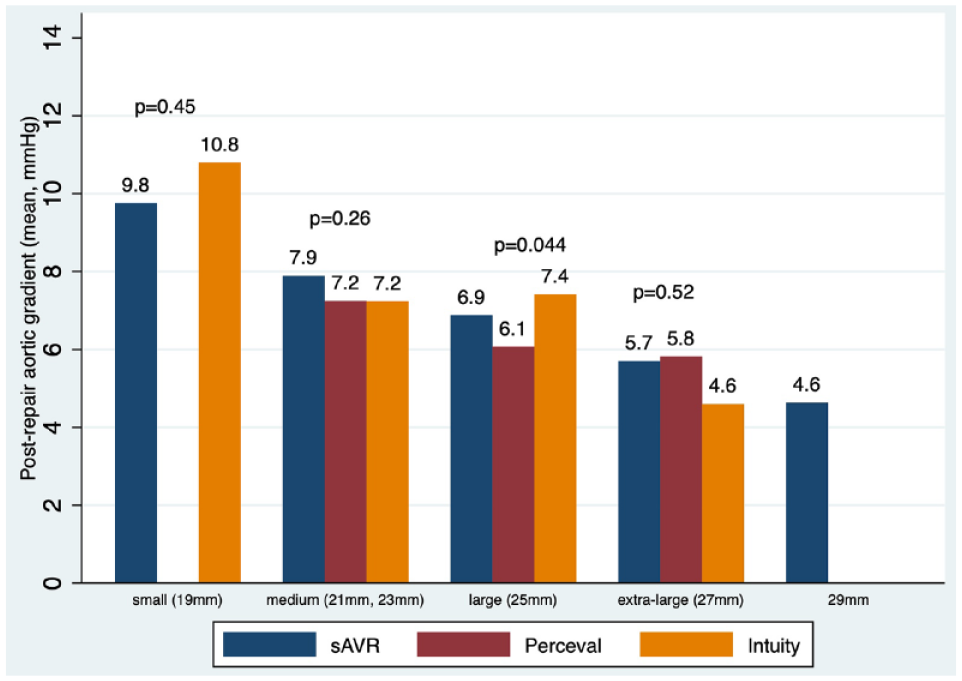
Postoperative aortic gradient by valve size and valve type. sAVR, surgical aortic valve replacement.
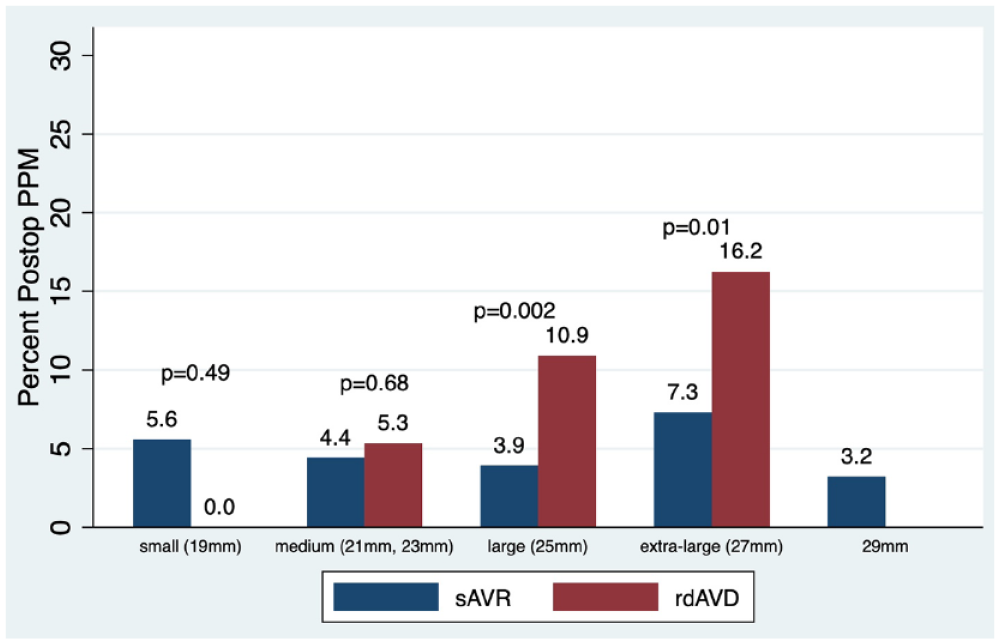
Postoperative pacemaker by valve size. PPM, permanent pacemaker implantation; rdAVR, rapid deployment valve replacement; sAVR, surgical aortic valve replacement.
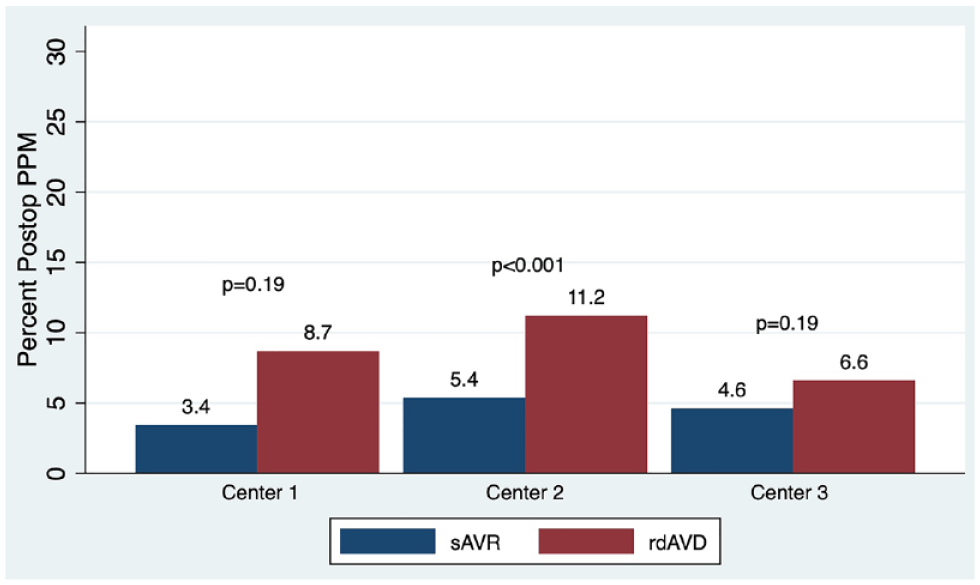
Postoperative pacemaker by center. PPM, permanent pacemaker implantation; rdAVR, rapid deployment valve replacement; sAVR, surgical aortic valve replacement.
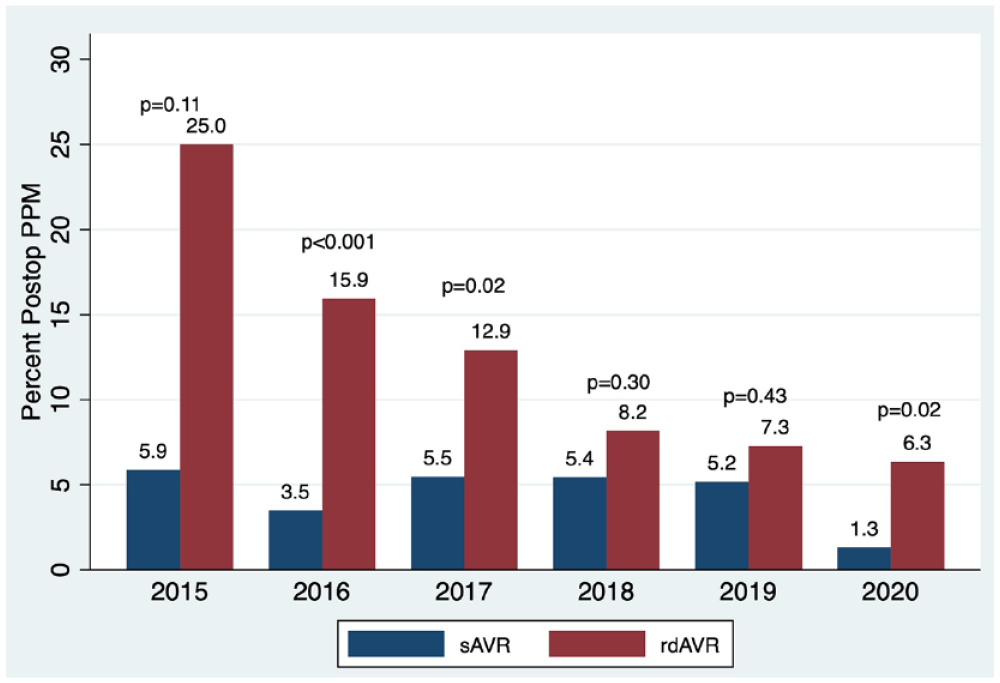
Postoperative pacemaker by year. PPM, permanent pacemaker implantation; rdAVR, rapid deployment valve replacement; sAVR, surgical aortic valve replacement.
Survival
Short-term 30-day mortality was not significantly different between sAVR and rdAVR (1.5% vs 2.6%, SMD = 0.08). Adjusted long-term survival was not significantly different between sAVR and rdAVR out to 4 years within overall, isolated AVR, or AVR + CABG cohorts (Fig. 6). Temporal mortality distribution was examined and after 30-day adjusted survival curves showed a possible trend of ongoing late deaths in the sAVR group, but this did not achieve significance (Fig. 7).
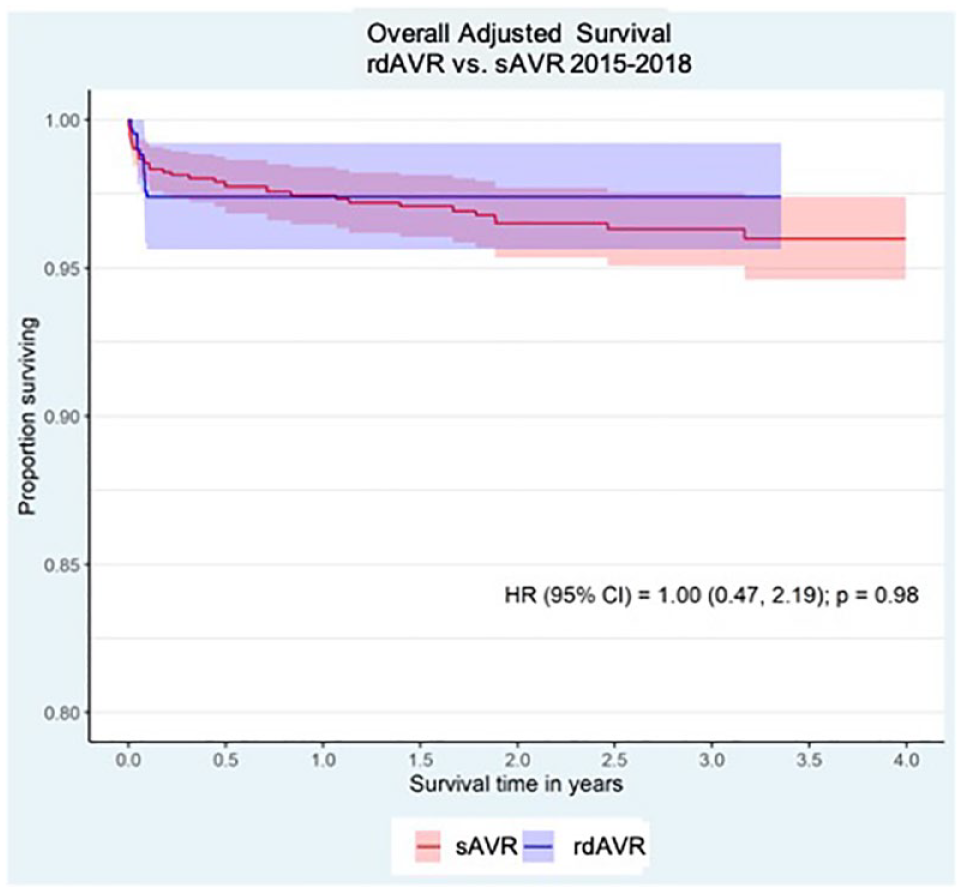
Overall adjusted survival. CI, confidence interval; HR, hazard ratio; rdAVR, rapid deployment valve replacement; sAVR, surgical aortic valve replacement.
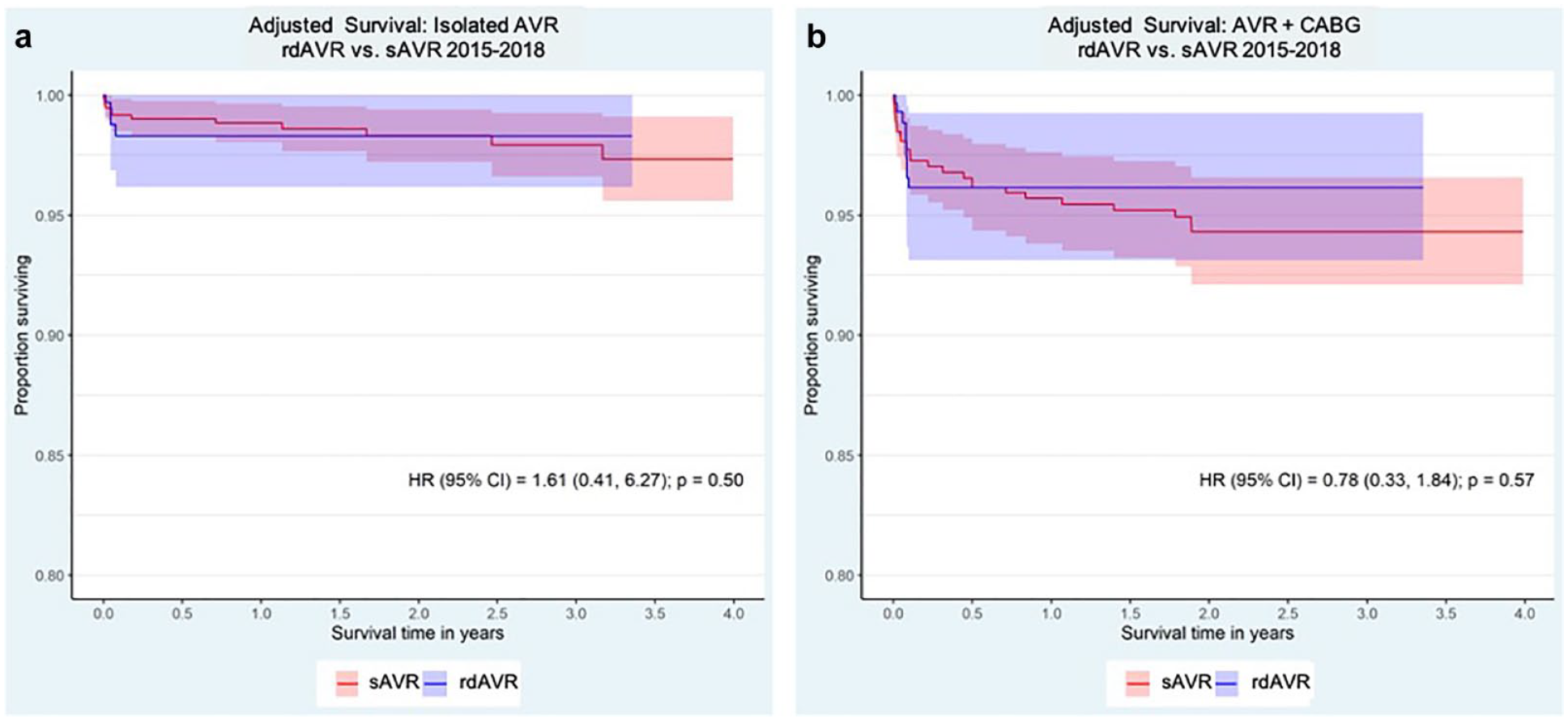
Adjusted survival by (a) isolated AVR and (b) AVR + CABG. AVR, aortic valve replacement; CABG, coronary artery bypass grafting; CI, confidence interval; HR, hazard ratio; rdAVR, rapid deployment valve replacement; sAVR, surgical aortic valve replacement.
Discussion
This large regional analysis of sAVR versus rdAVR has 3 main findings. First, hemodynamic and survival indices were no different between sAVR and rdAVR. Second, our study has shown a notable decrease in CPB and cross-clamp times with rdAVR compared with sAVR. Third, although the PPM rate for rdAVR remains higher than sAVR, this complication varies by center and has declined steadily over time.
Our finding of a similar safety profile insofar as morbidity and mortality of rdAVR is consistent with that previously reported in the literature. We found no difference in short-term 30-day mortality of rdAVR compared with sAVR, which has been previously reported.16,18,19 In addition, our adjusted long-term survival out to 4 years showed no significant difference between rdAVR and sAVR (Fig. 6) in any of the overall or isolated AVR and AVR + CABG cohorts that were examined. Fischlein and the PERSIST investigators in a randomized fashion showed noninferiority in survival between the Perceval rdAVR and conventional bioprosthesis at 1 year, 20 and Haverich et al. showed good performance and outcomes of the Intuity valve out to 3 years. 21 A recent large national analysis by Erfe et al. using the Society of Thoracic Surgeons database also showed no difference in survival of rdAVRs compared with standard stented sAVR outcomes at 2-year follow-up. 16 Our large regional analyses of short-term and long-term follow-up out to 4 years are consistent with these findings and further support the safety and durability of rdAVR.
Hemodynamic data with regard to rates of PVL of rdAVR have been varied. Some studies have shown greater rates of any PVL in rdAVR but no significant differences in significant PVL (moderate or greater),16,22 whereas others have shown no difference. 15 Our analysis showed excellent rates (<1%) of mild/moderate/severe PVL across all rdAVR cohorts and a very low rate of any PVL (3.2%), which was lower than rates previously reported by Lazkani et al. (4.2%). 22 Only in the rdAVR + CABG group did we see slightly higher rates of trivial or trace PVL (4.7% vs 1.0%, SMD = 0.23), which is of questionable hemodynamic importance. Contrary to published reports of lower gradients being associated with rdAVRs,17,23 overall postoperative hemodynamic gradients in our study did not differ by rdAVR versus sAVR valve type. Not surprisingly, we found an association with decreased valve gradients with increased valve size (Fig. 1) and no significant difference in gradients by rdAVR type (Fig. 2).
While it has been well established that increased CPB and cross-clamp times are associated with poorer outcomes,1,4,24 these benefits were hard to capture in our data. Our study showed a reduction of 30 and 24 min in CPB and cross-clamp times, respectively, in the overall rdAVR group compared with the sAVR group. Procedural times have consistently been found to be faster in rdAVR than sAVR, but these differences in CPB and cross-clamp times are greater than previously reported, which are typically in the 15 to 20 min range.10,16,17,25 Unlike other groups, we did not see these faster operative times translate into improvements in length of stay, rates of atrial fibrillation, transfusion, mechanical ventilation times, or freedom from MACE.10,25 –30 Instead, we noticed increased rates of acute kidney injury (3.5% vs 1.6%, SMD = 0.12) and infection (5.0% vs 3.0%, SMD = 0.1) in the rdAVR group. Although difficult to explain, this could in part be related to higher rates of transfusion of multiple units of packed red blood cells in the isolated AVR group or a reflection of case complexity as rdAVR + CABG had higher rates of 3-vessel disease and a slightly increased number of bypass grafts (2.3 vs 2.1, P < 0.001) compared with sAVR + CABG. However, our study is not the first to associate an increase in acute kidney injury with rdAVR, as Erfe et al. found renal failure of 3.9% in their rdAVR group versus 3.0% in sAVR. 16 As suggested by others, the benefits of shorter CPB and cross-clamp times may not be linear, 24 and certain high-risk subgroups who may benefit the most from faster operating room times deserve further analysis. Although not statistically significant, we did see a trend toward improved long-term survival in the rdAVR + CABG cohort (Fig. 7). As these patients are typically higher risk, this may be a group that sees long-term benefits of faster operative times.
Although early single-center reports suggested similar PPM rates,10,28 more recent larger studies have consistently shown significantly increased rates of PPM with rdAVR compared with sAVR.16,22,31–33 Our data show an overall PPM rate of 7.4% versus 4.4% associated for rdAVR compared with sAVR. This is on the lower end of previously reported PPM rates for rdAVR, which have previously been shown in the 7.9% to 13% range.16,17,22,34 We found a steady decline in our PPM rate over the study time period (Fig. 5), which was as high as 25% in 2015 and 6.3% in 2020. These findings are consistent with those reported by Berretta et al. in 2020, summarizing trends from the Sutureless and Rapid Deployment International Registry in which PPM rates declined from 12.8% to 5.9% over their 11-year study period. 34 As we saw a higher frequency of extra-large rdAVR implants in the earlier years of our study, these improvements in PPM rates may be attributable to surgeon learning curve and better understanding of implant technique.
Overall, rdAVR tended to be associated with larger prostheses and promoted minimally invasive techniques. We found 51% of rapid deployment implantations to be size large or extra-large compared with 37.4% of sAVR implants greater than 23 mm. Therefore, the majority (62.5%) of sAVR valves were 19, 21, or 23 mm. In the rdAVR cohort, rdAVR sizes large and extra-large had significantly higher PPM rates of 10.9% and 16.3%, respectively. Larger rdAVR implantation size is a known risk factor for PPM, 32 and the higher rate of larger rdAVR implants may have contributed to our PPM rate. Minimally invasive techniques were used more frequently with rdAVR. Within the largest center in our study, the rate of minimally invasive sternotomy in the AVR only cohort was notably higher for rdAVR versus sAVR (55.9% vs 18%, P < 0.001).
Several limitations of our study exist beyond those typically associated with data collection of a large regional database. First, long-term follow-up beyond 30 days was available for survival only out to 4 years. Longer-term survival and durability data continue to be critical in consideration for adoption of new valve technology. Hemodynamic variables insofar as valve performance such as transvalvular gradient, structural degeneration, PVL, and overall ejection fraction are important in the overall assessment of valve choice and were not available from the database. Current efforts are focused on obtaining midterm and long-term echocardiography follow-up to specifically address these durability questions. In addition, further subgroup analysis as it relates to high-risk groups (elderly, comorbid conditions, low ejection fraction) and survival implications of those receiving PPM deserve further investigation. Overall case volumes and PPM rates differed by center, and although our propensity model corrected for center size, some degree of institutional bias may persist.
Conclusions
In this large regional multicenter analysis, we found that short-term and long-term adjusted survival out to 4 years were no different between sAVR and rdAVR. The rdAVR group was associated with notable decreases in CPB and cross-clamp times and promoted minimally invasive surgical approaches for isolated AVR. PPM rates continue to be higher in rdAVR compared with sAVR but are improving. The benefits of faster operative times as well as implications of PPM rate and valve durability deserve further long-term follow-up and subgroup analysis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The member centers of the Northern New England Cardiovascular Disease Group.
