Abstract
Objective:
Psoas muscle size is a reliable marker of sarcopenia and frailty that correlates with adverse outcomes after cardiac surgery. However, its use in mitral and minimally invasive cardiac surgery is lacking. We sought to determine whether frailty, as measured by psoas muscle index, increases surgical risk for minimally invasive mitral valve surgery.
Methods:
Patients undergoing isolated minimally invasive mitral surgery via right minithoracotomy were identified. Patients who underwent maze, tricuspid intervention, and those who were emergent were excluded. Total psoas muscle area was calculated using the average cross-sectional area at the L3 vertebra on computed tomography scan and indexed to body surface area. Sarcopenia was defined as <25th gender-specific percentile. Patients were stratified by sarcopenia status and outcomes compared.
Results:
Of 287 total patients, 192 patients met inclusion criteria. Sarcopenic patients were 6 years older (66 vs 60 years, P = 0.01), had lower preoperative albumin levels (4.0 vs 4.3 g/dL, P < 0.001), and had higher Society of Thoracic Surgeons risk of morbidity/mortality (13.1% vs 9.0%, P = 0.003). Operative major morbidity or mortality was 6.4% versus 5.5% (P = 0.824), while the 1-year mortality rate was 2.1% versus 0% (P = 0.08). After risk adjustment, psoas index did not predict operative morbidity or mortality. However, sarcopenia was associated with higher odds of readmission (odds ratio = 0.74, P = 0.02).
Conclusions:
Contrary to other cardiac operations, for patients undergoing isolated minimally invasive mitral valve surgery, sarcopenia was not associated with increased perioperative risk except for higher readmission rates. Minimally invasive surgical approaches should be strongly considered as the approach of choice in frail patients.
Central Message
Frailty, as measured by sarcopenia, was not found to be a predictor of major morbidity or mortality in this sample of patients undergoing isolated minimally invasive mitral valve surgery. Minimally invasive approaches may benefit the frail population compared with sternotomy, and further studies are warranted.
Introduction
The number of elderly patients undergoing cardiac surgery continues to increase; among those undergoing mitral valve surgery, the median age is 75 years.1,2 These patients possess a greater number of comorbidities, making preoperative risk assessment of utmost importance. 3 Frailty is defined as a vulnerability to stressors due to loss of physiologic reserve.4,5 Although older age does not necessarily translate to frailty, the two are linked, and as the cardiac surgery population ages, they will be more prone to a frail phenotype. Frailty has been previously demonstrated to be a risk factor for adverse outcomes in patients undergoing conventional cardiac surgery via sternotomy, resulting in prolonged length of stay, increased rate of discharge to intermediate care facilities, higher readmissions, and higher risk of operative and midterm mortality.4,6,7 In cardiac surgery, the gold standard of risk assessment remains the Society of Thoracic Surgeons (STS) risk scores, although these underestimate perioperative risk in frail patients.7,8 With recognition of the importance of frailty, the STS added preoperative 5-meter gait speed as a variable within the Adult Cardiac Surgery Database (ACSD), which was validated by Afilalo and colleagues as a mortality predictor. 9 Despite this, there are still many proxies used for frailty, with more than 20 different validated objective assessment tools. Many of these tools are cumbersome to apply with no consensus as to which assessments should be applied in practice. 10
Skeletal muscle mass is an excellent objective predictor of the frail phenotype that is free from acute changes in clinical status or patient effort–related measurements. 11 One validated method of quantifying sarcopenia is via measurement of psoas muscle area, a muscle heavily involved from a functional standpoint in recovery after major surgery. 12 Psoas muscle size has been shown to correlate heavily with other measures of frailty, including short physical performance battery, grip strength, and VO2 max.13,14 Although there is no consensus on exact psoas muscle size cutoffs for measuring sarcopenia, some studies have defined sarcopenia as a psoas muscle area falling within the lowest gender-specific quartile of a study cohort or by indexing psoas muscle area to body surface area with use as a continuous variable.14–16 Studies within cardiac surgery utilizing psoas muscle size to quantify sarcopenia have demonstrated psoas muscle size as an excellent predictor of adverse postoperative outcomes and demonstrated worse morbidity and mortality in patients undergoing median sternotomy who possess lower total psoas indices.13,16–20
In the current realm of mitral valve disease, increasing patient frailty is pushing heart teams toward percutaneous approaches due to the perceived high risk for surgery. In a study by Sorajja et al. analyzing the Transcatheter Valve Therapy registry, frailty was the most commonly cited indication for transcatheter edge-to-edge mitral repair. 21 Although certainly an option in higher-risk patients, this modality can be suboptimal to durable, surgical mitral valve repair or replacement.
Minimally invasive cardiac surgery (MICS) is increasing in utilization based on patient preference and potential benefits of MICS including avoidance of deep sternal wound infection and sternal instability, fewer blood transfusions, and shortened intensive care unit (ICU) and hospital length of stay with comparable short-term morbidity and mortality.22–24 In addition, sternotomies may limit rehabilitation in the early postoperative period due to strict sternal precautions and greater dependence on leg strength, which a minithoracotomy may avoid. However, the benefit of MICS in frail patients is not yet established. Prior studies looking at percutaneous edge-to-edge mitral valve repair have demonstrated frailty as an independent predictor of worse morbidity and mortality, although these patients tended to be of prohibitive surgical risk.25,26 We hypothesized that a minimally invasive approach to mitral surgery in a frail population, as defined by sarcopenia, would not have worse postoperative outcomes.
Methods
This study was approved by the University of Virginia’s Institutional Review Board (#23305) and the University of Michigan’s Institutional Review Board (#00148119). Using the STS ACSD, patients from 2 centers undergoing minimally invasive mitral valve surgery were identified from 2013 to 2021. Only those undergoing MICS through a right anterior minithoracotomy were included. Surgical approach to the mitral valve was via direct vision with thorascopic assistance, without utilization of the robotic approach in any of the participating patients. Patients with endocarditis, emergent indications, or concomitant procedures including tricuspid valve intervention and atrial fibrillation ablation procedures were excluded from analysis. Those without a preoperative computed tomography (CT) scan within 6 months of their index operation were also excluded.
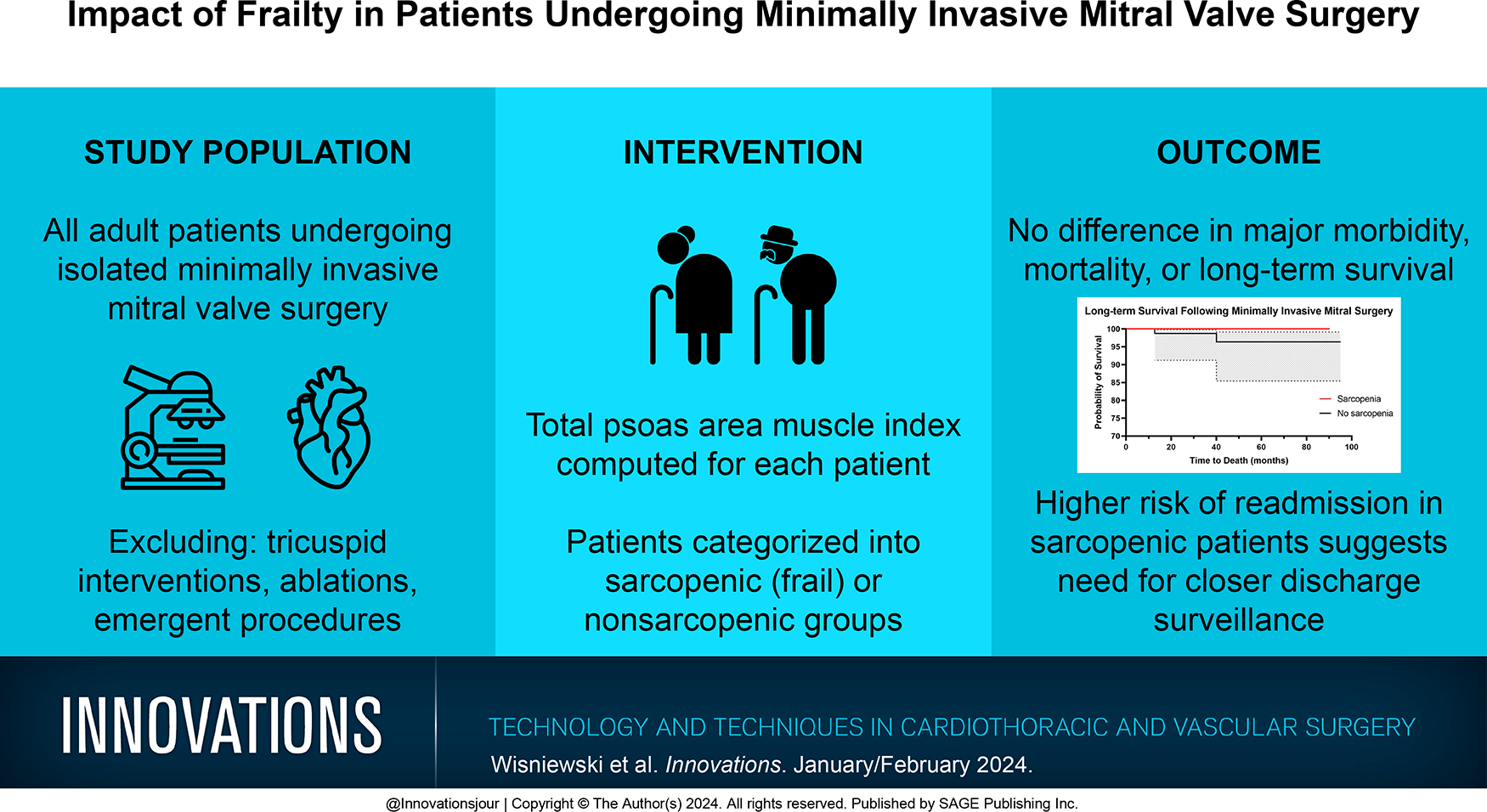
Total psoas muscle area (TPA) was calculated using the average cross-sectional area at the caudal level of the L3 vertebra of both the left and right psoas muscle on CT scan, as suggested by previous studies. 27 Multiplanar reconstruction was utilized to take psoas muscle measurements at the appropriate cross-sectional area perpendicular to the muscle body length (Fig. 1). Only preoperative CT scans within 6 months of each patient’s index operation were utilized. If patients had multiple preoperative CT scans, the scan closest in proximity to the index operation was used for measurements. Two independent observers from each institution measured total psoas muscle area in triplicate. The mean of these values was then indexed to body surface area to generate the total psoas area index (TPAI). Patients were categorized as sarcopenic if falling below the 25th gender-specific percentile for psoas index.
Multiplanar computed tomography reconstruction for psoas muscle area measurement with representative images of a sarcopenic and nonsarcopenic patient.
Standard STS definitions were used for all variables. Interobserver variability for psoas muscle measurement was assessed by Pearson correlation. Categorical values were analyzed via χ2 test and continuous variables via independent t tests. Multivariable logistic regression was used to evaluate psoas index as a predictive measure for categorical outcomes. Risk adjustment was performed with logistic regression using STS predicted risk scores for the relevant outcome. If no risk score was applicable to the relevant outcome, then the analysis was performed utilizing predicted risk of morbidity or mortality (PROMM) for adjustment. Kaplan–Meier analysis was done to determine predicted long-term survival based on most recent status at follow-up. Patient follow-up from hospital discharge was determined utilizing electronic medical records for appointment visits, hospital readmissions, telephone calls, medication refills, or evidence of death. All statistical analyses were carried out using SAS version 9.4 (SAS Institute, Cary, NC, USA), with a P value less than 0.05 determining significance.
Results
Preoperative Risks in Sarcopenic Patients
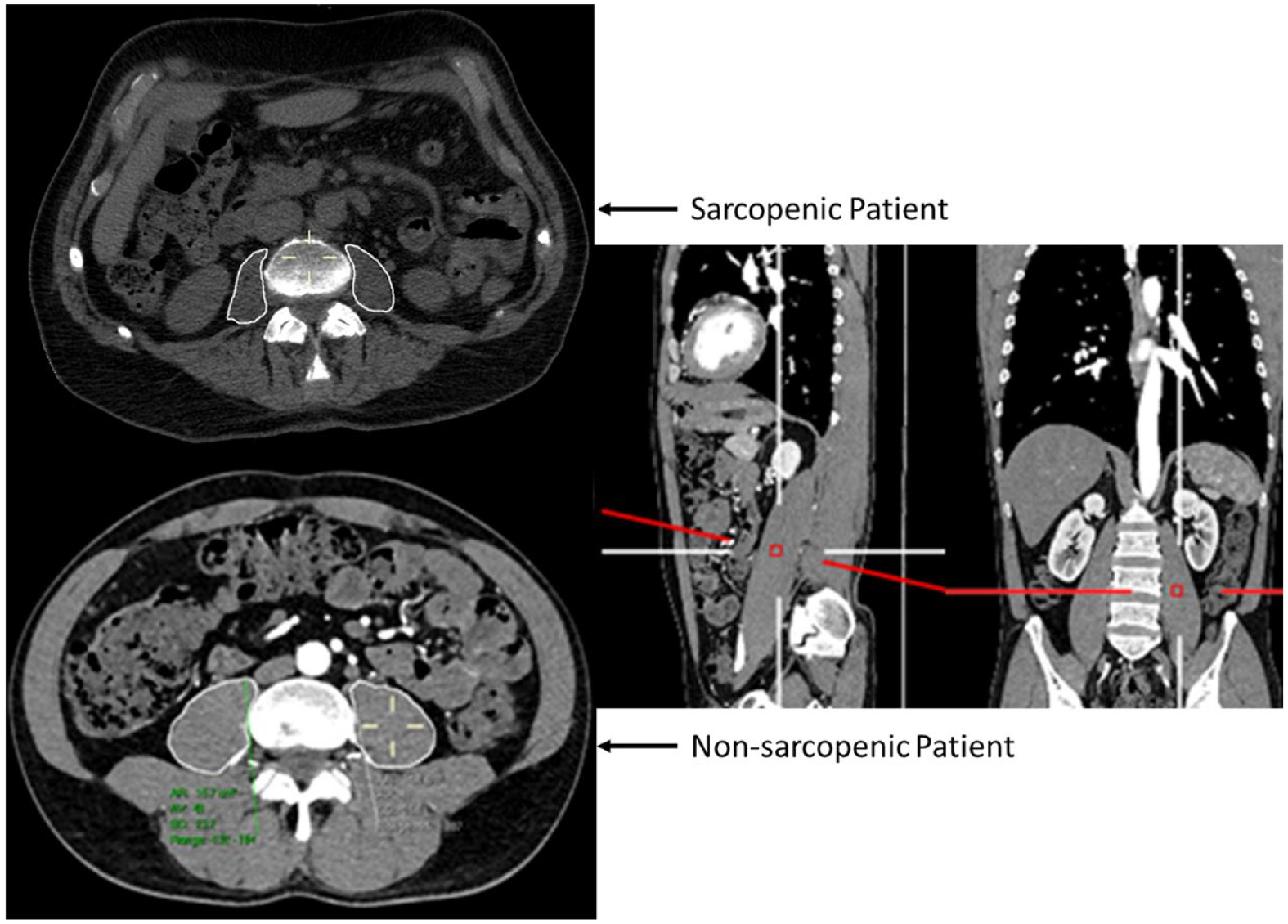
From 2013 to 2021, a total of 446 patients underwent minimally invasive mitral valve surgery. Following exclusions, a total of 192 patients underwent pure, isolated minimally invasive mitral valve surgery (Fig. 2). The mean predicted risk of mortality (PROM) was 1.25%, the mean age was 61 years, and 50% of patients were female. Interobserver variability of total psoas muscle measurements was minimal between observers, with a Pearson correlation coefficient of 0.91 (Fig. 3). The median TPA was 18.2 (5.9 to 39.8) cm2, with men having higher TPA compared with women (25.3 vs 14.8 cm2, P < 0.001; Fig. 4a). When adjusting for body surface area, the median psoas index was 9.8 cm2/m2. Men had a higher psoas index as compared with women (12.2 ± 2.4 vs 8.4 ± 2.1 cm2/m2, P < 0.001; Fig. 4b). The 25th percentile gender-specific cutoff for defining sarcopenia was 10.32 cm2/m2 for male patients and 7.22 cm2/m2 for female patients, resulting in 47 sarcopenic patients and 145 nonsarcopenic patients.
Consort diagram of patient cohort.
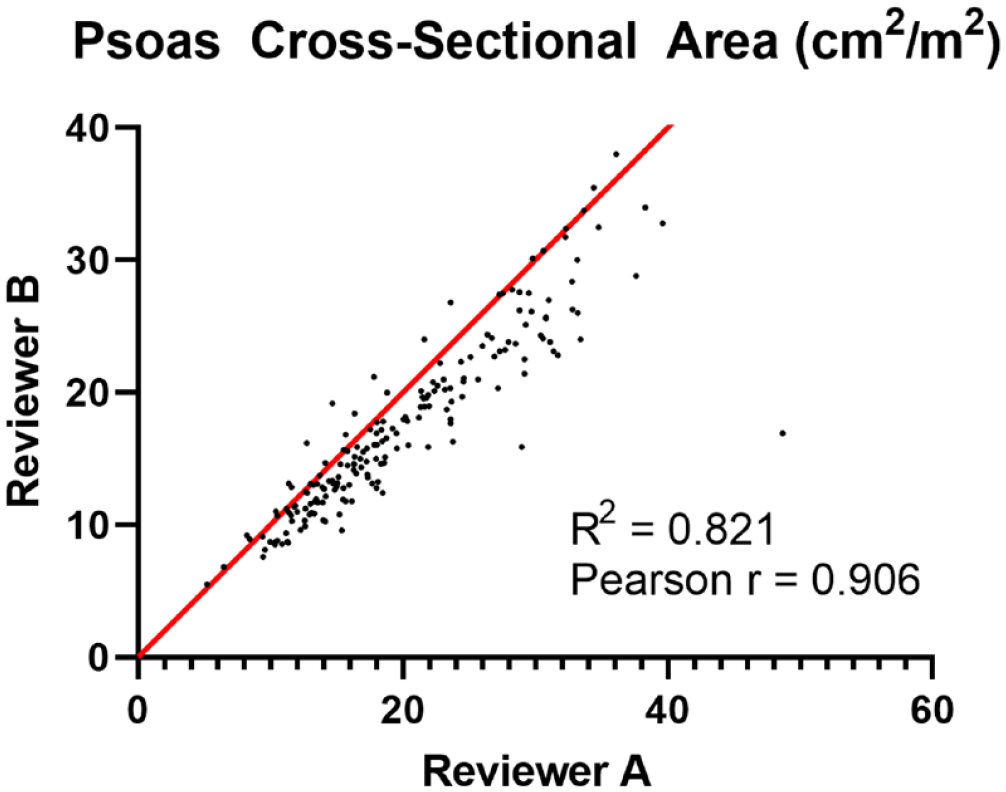
Total psoas cross-sectional area plot of measurements by independent reviewers with Pearson correlation coefficient and linear regression.
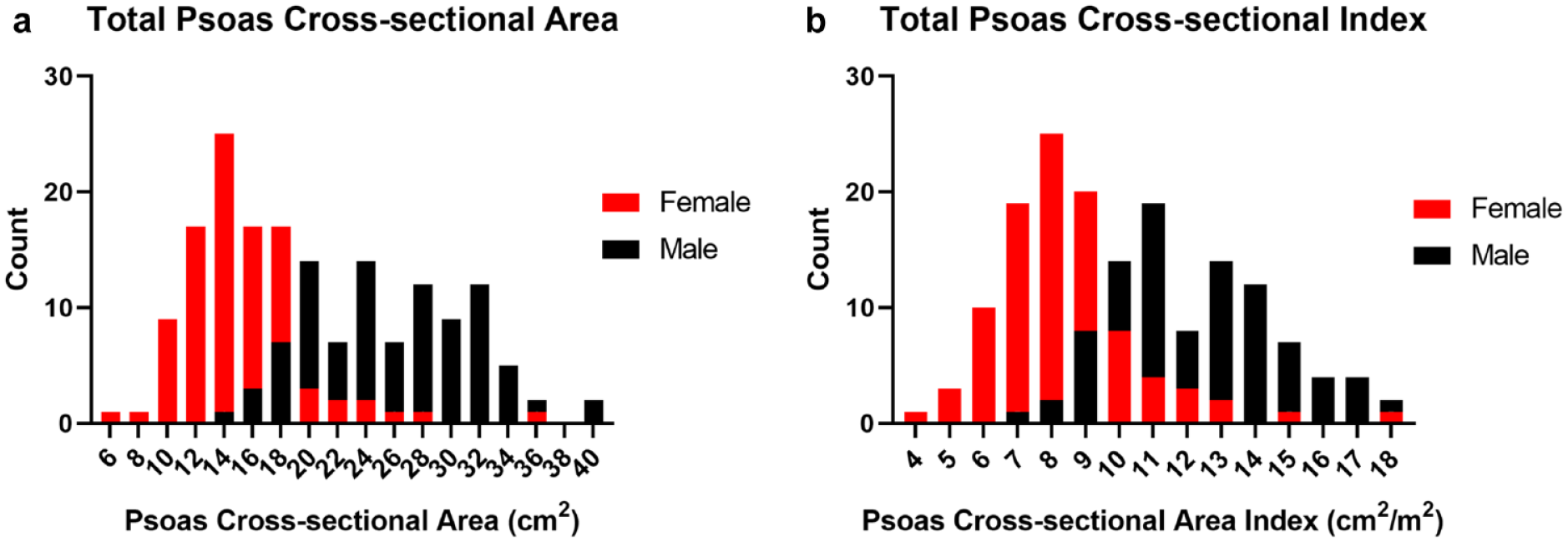
Histograms of (a) total psoas cross-sectional area and (b) total psoas cross-sectional area index by sex.
Sarcopenic patients were older (60 vs 66 years, P = 0.01), had lower preoperative albumin levels (4.0 vs 4.3 g/dL, P < 0.001), were less likely to undergo elective operations (91.5% vs 98.6%, P = 0.01), and had higher STS PROM (2.0% vs 1.0%, P = 0.01) and STS PROMM (13.1% vs 9.0%, P = 0.003). In addition, sarcopenic patients had significantly higher predicted risk of other STS major morbidities including renal failure (2.7% ± 3.3% vs 1.4% ± 1.4%, P = 0.01), permanent stroke (1.5% ± 0.9% vs 1.1% ± 0.7%, P = 0.003), reoperation (5.3% ± 3.0% vs 4.1% ± 2.0%, P = 0.01), and prolonged ventilation (7.2% ± 5.6% vs 5.4% ± 4.5%, P = 0.048). Other baseline characteristics and comorbidities were similar between the 2 groups (Table 1).
Baseline Characteristics by Sarcopenia.
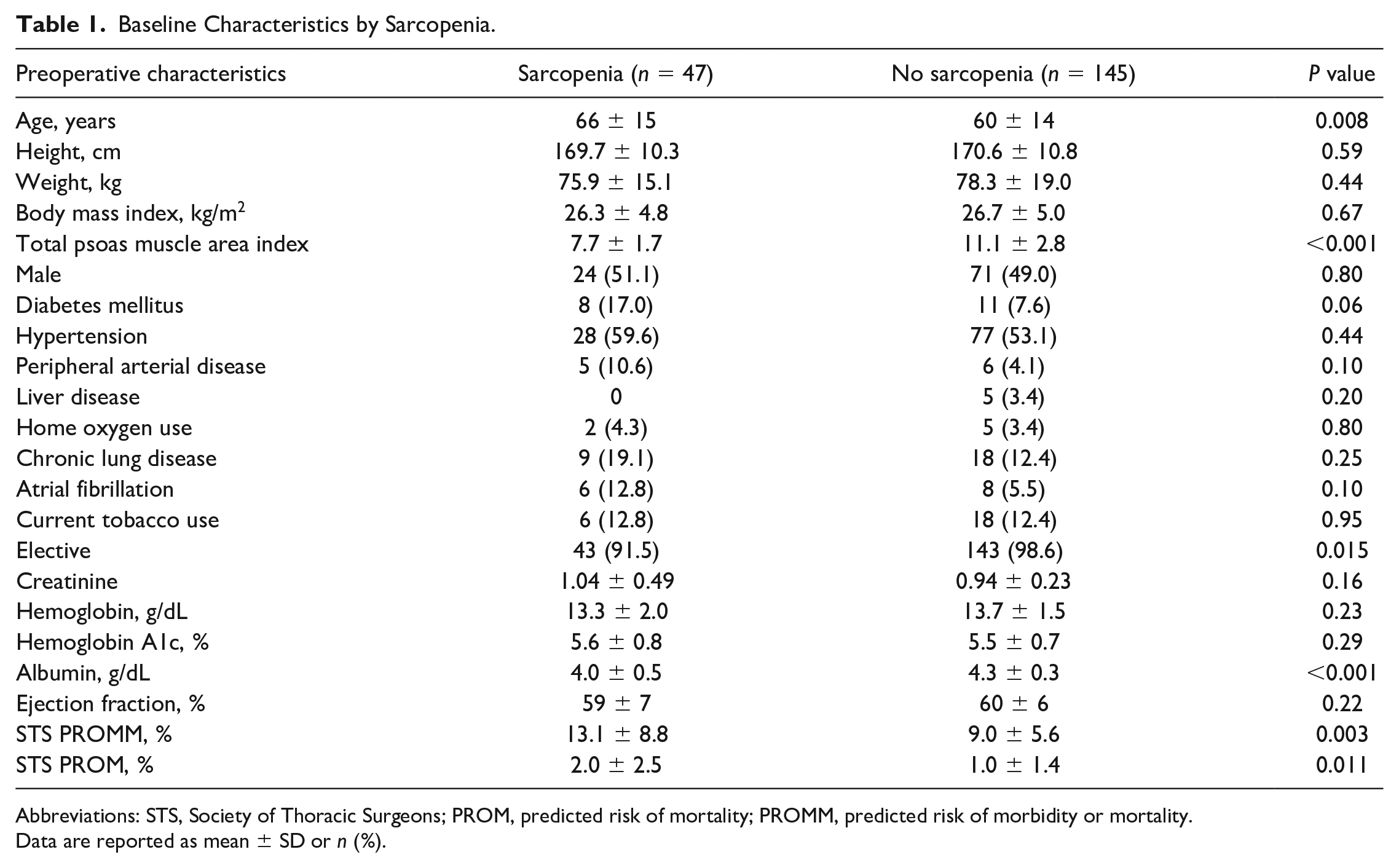
Abbreviations: STS, Society of Thoracic Surgeons; PROM, predicted risk of mortality; PROMM, predicted risk of morbidity or mortality.
Data are reported as mean ± SD or n (%).
Unadjusted and Risk-Adjusted Outcomes
There was no difference in operative mortality, with 1 death in the sarcopenic group (2.1%) and none in the nonsarcopenic group (0%, P = 0.092). Rates of other complications including stroke (2.1% vs 0.7%, P = 0.40), pneumonia (0% vs 1.4%, P = 0.42), and renal failure (0% vs 0%) were similar compared with nonsarcopenic patients (Table 2). There was no difference in postoperative ventilator hours (3 ± 3 vs 5 ± 17 h, P = 0.18), ICU hours (57 ± 46 vs 54 ± 55 h, P = 0.79), or hospital length of stay (7 ± 4 vs 6 ± 3 days, P = 0.26) between the sarcopenic and nonsarcopenic patients. Sarcopenic patients had similar rates of discharge to a facility as compared with nonsarcopenic patients (17.0% vs 20.0%, P = 0.65). At 1 year, there was no difference in mortality, with a single death in the sarcopenic group and no deaths in the nonsarcopenic group (2.1% vs 0%, P = 0.078).
Postoperative Outcomes by Sarcopenia.
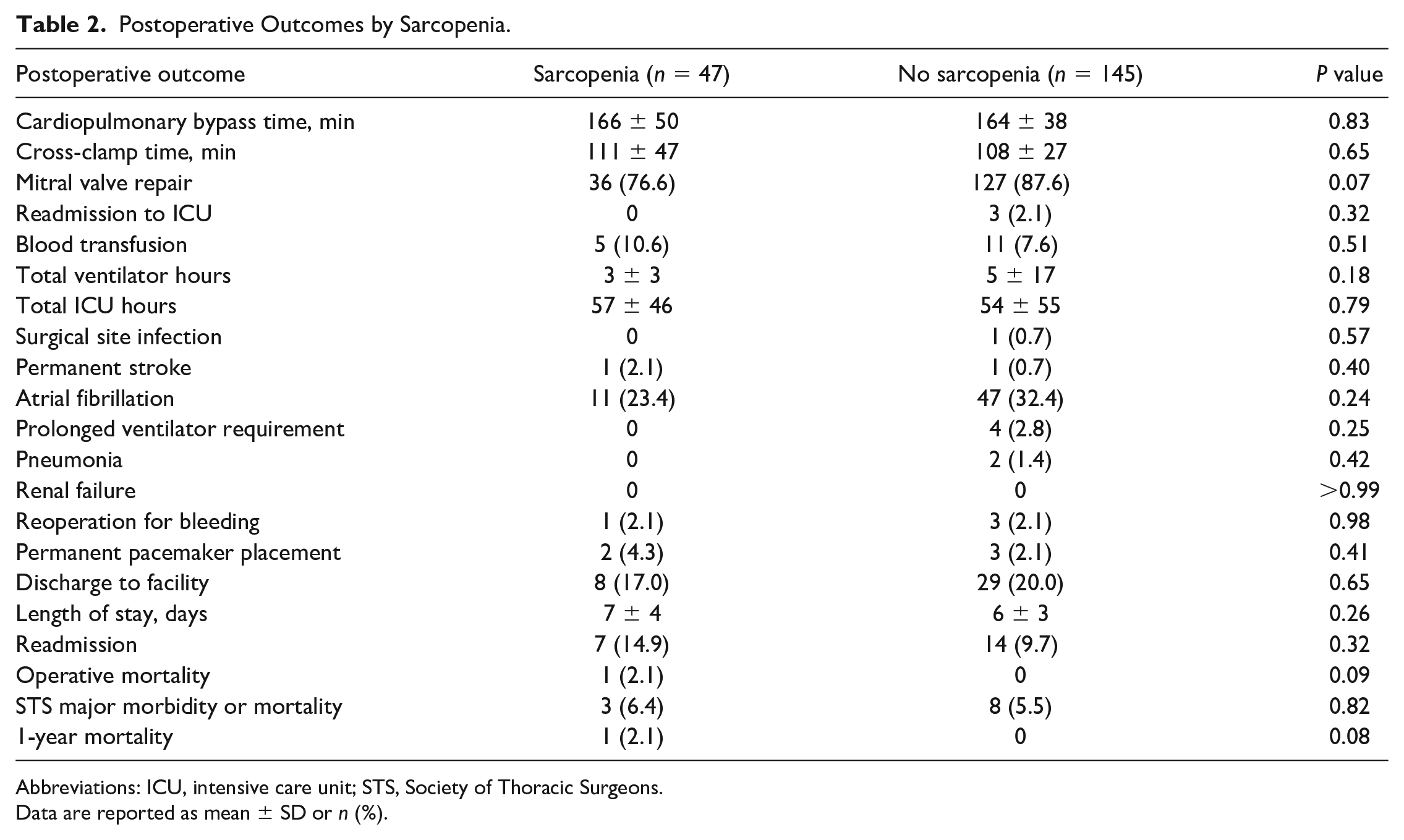
Abbreviations: ICU, intensive care unit; STS, Society of Thoracic Surgeons.
Data are reported as mean ± SD or n (%).
When using the psoas index, logistic regression for mortality and major morbidity between the 2 groups was similar (Table 3). Regression analysis demonstrated lower TPAI as a risk factor for readmission (OR = 0.776, P = 0.01), longer ICU stay (estimate = −2.59, P = 0.046), and increased hospital length of stay (estimate = −0.24, P = 0.002). Following risk adjustment, lower TPAI was significantly associated only with risk of readmission (OR = 0.74, P = 0.02) and no other STS major morbidities or mortality (Table 4). Reasons for readmission among the sarcopenic patients varied but included groin infection (n = 1), lymphocele (n = 1), lower extremity deep vein thrombosis (n = 1), cholecystitis (n = 1), palpitations (n = 1), and altered mental status in setting of subtherapeutic international normalized ratio (n = 1). Lastly, Kaplan–Meier analysis demonstrated no difference in long-term survival between sarcopenic and nonsarcopenic patients (log rank, P = 0.631; Fig. 5).
Unadjusted Outcomes for Psoas Index.
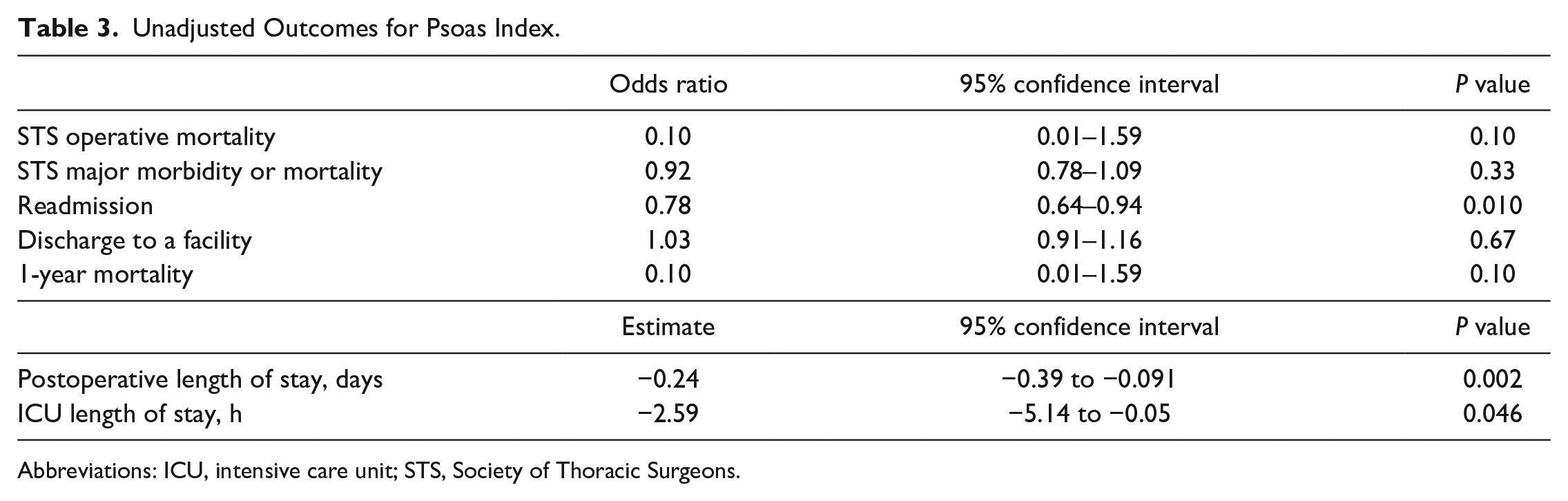
Abbreviations: ICU, intensive care unit; STS, Society of Thoracic Surgeons.
Risk-Adjusted Outcomes for Psoas Index.
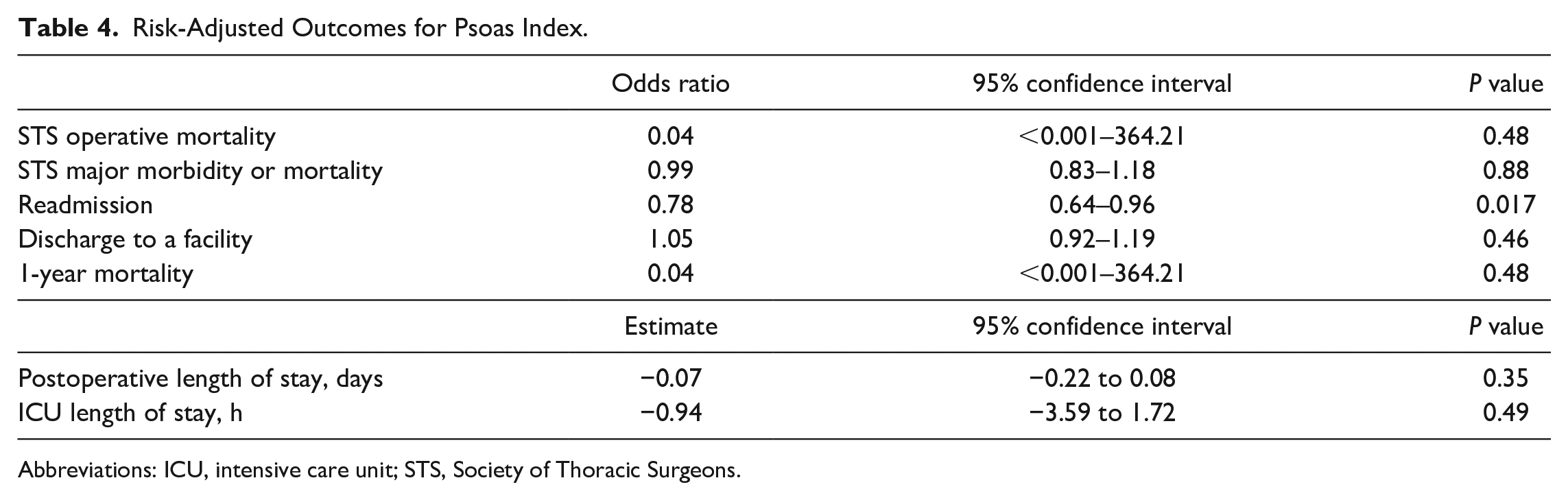
Abbreviations: ICU, intensive care unit; STS, Society of Thoracic Surgeons.
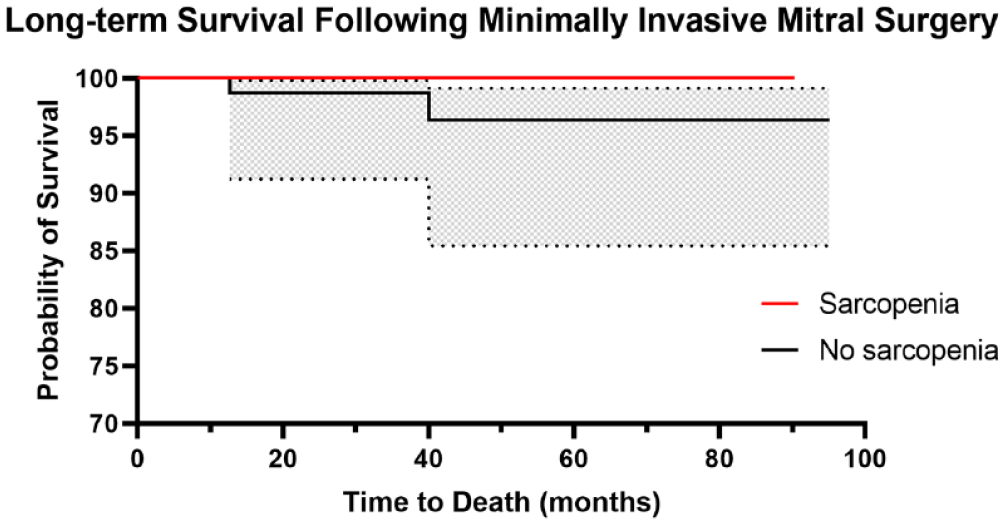
Kaplan–Meier curve of long-term survival of sarcopenic versus nonsarcopenic patients (log rank, P = 0.63).
Discussion
There are a multitude of instruments proposed for frailty measurement, although most rely on patient effort and are subject to change based on a patient’s current health status. Therefore, objective measures of frailty are increasingly important. Sarcopenia is a reliable and reproducible measure that is routinely available in MICS mitral patients, as these patients nowadays undergo preoperative CT imaging of the abdomen and pelvis to assess femoral vessels for cannulation. 28 The psoas muscle area has been extensively shown to correlate with the frail phenotype and has been validated in other studies of cardiac surgery patients. In this study involving 2 institutions with experience in MICS, psoas index and sarcopenia correlated with a frailty phenotype, such that these patients were also older and had lower preoperative albumin levels compared with nonsarcopenic patients. In addition, sarcopenic patients at baseline had higher STS PROMM, which is prevalent in the frail phenotype. Unlike in previous studies of frail patients undergoing sternotomy, sarcopenia was not associated with increased risk of morbidity or mortality in isolated mitral valve surgery. Long-term survival analysis also did not suggest worse survival odds in sarcopenic patients following isolated mitral valve surgery. After risk adjustment, psoas index was also not associated with postoperative complications, although a larger psoas index was associated with a lower risk of readmission.
Frailty is a well-established risk factor for adverse outcomes following cardiac surgery, and it is becoming increasingly utilized for risk assessment preoperatively in multiple cardiac surgical procedures.12,13,16,18,19,29 In patients undergoing surgical aortic valve replacement through sternotomy, sarcopenic patients had higher mortality at 1 year. Following risk adjustment, these patients still had a high risk of major morbidity, 1-year mortality, and long-term mortality with increased resource utilization from extended hospital stay. 18 In another study looking at patients undergoing mitral valve replacement through sternotomy, frail patients had significantly higher rates of postoperative complications, higher resource utilization, and higher in-hospital mortality, although this study utilized a different measure of frailty as opposed to sarcopenia. 29 Okamura and colleagues evaluated the impact of sarcopenia in patients undergoing valve surgery via median sternotomy, including aortic and tricuspid valve interventions. 19 They found sarcopenia, as measured by total psoas muscle area, to be a predictor of adverse long-term outcomes, including decreased long-term survival and increased risk of major adverse cardiac and cerebrovascular events, although they did not look specifically at their isolated mitral valve patients. Interestingly, their study did not find an increased risk of in-hospital mortality, which is congruent with our study and previous studies. 16
Previous studies looking at STS risk predictions in the frail population have demonstrated that this risk model may underestimate patient risk of complications, at least following surgical aortic valve replacement.8,30 We did not find this to be true, as our patients had no differences in STS major morbidity and mortality despite having higher preoperative risk scores. When looking at long-term survival, our sarcopenic patients did not have worse survival, unlike other studies of sarcopenia in cardiac surgery. Following risk adjustment, sarcopenic patients fared just as well as nonsarcopenic patients, aside from risk of readmission, demonstrating that perhaps minimally invasive approaches to the mitral valve may mitigate this added risk of frailty. Reasons for hospital readmission after surgery varied among the sarcopenic patients, and some, including deep vein thrombosis and altered mental status, had an internal relationship to the patients’ frailty status. Given the higher rate of readmission in frail patients, extended surveillance may be warranted upon discharge to home for prevention of this complication.
The application of psoas muscle measurements in patients undergoing MICS is easy to incorporate into practice and has been validated as a modality for stand-alone frailty measurement with high correlation to previously validated tests such as grip strength, short performance physical battery scores, and 5-meter walk test.12–14 At our institutions, all MICS patients routinely undergo preoperative CT scanning to delineate anatomy for peripheral cannulation strategies and also anterior minithoracotomy incision location. These scans are also performed prior to surgery, allowing for an accurate representation of their skeletal mass at the date of their operation.
Transcatheter options for mitral valve disease are continuing to increase in utilization, especially for higher-risk patients with prohibitive risk or those considered too frail for conventional surgery.23,24 These options may also extend to patients without optimal anatomy for a transcatheter device given their limited surgical options from a risk standpoint. Minimally invasive approaches, as opposed to sternotomy, may offer an alternative for this patient group and allow for more durable repair or replacement of their mitral valve.
With the development of newer, less invasive techniques and increased patient awareness of surgical modalities, patients moving forward are likely to opt for avoidance of sternotomy when possible. Thus, with an aging population and likely increasing frequency of frailty, proper risk stratification in minimally invasive approaches will become necessary. Through simple and reproducible psoas muscle measurement as a proxy of sarcopenia, this objective frailty characteristic can be added to traditional risk calculations that lack appropriate measures of frailty.
This article adds to the growing body of literature supporting the importance of frailty in preoperative risk assessment and is the first to evaluate its impact in MICS and isolated mitral valve surgery. This study is the first to characterize sarcopenia and its risk in isolated mitral valve surgery and in MICS. Perhaps there is added benefit to minimally invasive approaches in the frail patient. Utilization of the psoas muscle index continues to be an excellent measurement of sarcopenia and therefore of the frail phenotype, although it remains prudent to combine this index with other measures of frailty for comprehensive evaluation.
Our study has limitations that should be noted. Surgeries were performed largely by 2 surgeons at high-volume centers, which may limit the generalizability of the results. The number of patients included in the study was also low due to the inclusion of only pure, isolated mitral valve operations; therefore, differences may be present that were not detected due to the study power. Psoas muscle index, although an excellent objective measure, is not comprehensive in evaluating frailty, and other components of frailty were either not available or had high rates of missingness. A direct comparison with a sternotomy approach was not possible, as CT imaging of the pelvis is not routinely available for these mitral patients. Because of the low event rate, this study is likely underpowered to detect differences in individual complications. Lastly, as the predicted risk of patients undergoing isolated mitral valve surgery was generally low, further investigation is needed to determine if these results extend to higher-risk mitral patients.
Conclusions
Frailty as measured by the psoas index was routinely available, highly reproducible, and associated with other measures of a frailty phenotype. Although sarcopenia has been associated with an increased risk of complications after a sternotomy approach for cardiac surgery, in patients undergoing minimally invasive mitral surgery, sarcopenia and psoas index were not associated with morbidity or mortality. The higher readmission rate for sarcopenic patients as measured by a smaller psoas index suggests postdischarge care coordination may be a target for quality improvement, specifically in high-risk frail patients. Minimally invasive approaches may prove to be of great benefit in the frail population compared with sternotomy, and further comparative studies between these groups are warranted.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was in part funded by NIH/NHLBI T32 training grant (No. T32HL007849).
