Abstract
Heart transplantation utilizing deceased after circulatory death (DCD) donors has expanded the donor pool through the use of ex vivo normothermic perfusion. Compared with brain death donation, the conventional method of performing DCD heart transplantation includes an additional period of warm and cold ischemia. We have developed a beating heart implantation technique that obliviates the need for a second cardioplegic arrest and the associated reperfusion injury. We hypothesize this reproducible method may improve short-term and long-term outcomes to mirror results seen in brain death donors and provide details on how to perform beating heart transplantation.
Central Message
Beating heart transplantation avoids an additional ischemic arrest typically involved in DCD heart transplantation. We provide a detailed discussion of how to perform a beating heart transplant.
Introduction
The use of normothermic ex vivo organ perfusion devices has enabled the increased utilization of organs from deceased after circulatory death (DCD) donors and made it possible for more aggressive organ selection and expansion of the donor pool.1,2 Platforms such as the Organ Care System (OCS™; TransMedics, Andover, MA, USA) have enabled better assessment and resuscitation of the heart allograft prior to transplantation. 3 However, current practice necessitates 2 cardioplegic arrests, exposing the heart to multiple ischemia reperfusion injuries as well as necessitating long reperfusion times after removal of the cross-clamp.2,4 We have developed a method of performing DCD heart transplantation with continuous, uninterrupted, warm blood perfusion after placement on OCS, removing the second cardioplegic arrest, and implanting the heart while beating. 5
Surgical Technique
Inclusion and Exclusion Criteria
Patients are considered for DCD heart transplantation with a beating heart implantation method when a suitable donor who meets Maastricht Category III criteria is matched. Extended criteria donors, which includes those older than 50 years, size mismatch determined by a greater than 10% difference in predicted heart mass, presence of left ventricular (LV) hypertrophy, ejection fraction less than 50%, and history of diabetes, are also considered and discussed by a multidisciplinary team of transplant cardiologists and cardiac surgeons. Exclusion criteria include the need for any concomitant cardiac surgical procedure such as total arch replacement at the time of heart transplant or multiorgan transplantation.
Procurement
Procurement of the heart proceeds as normal in DCD heart transplantation. Typically, this entails withdrawal of life support in the donor, a period of observation prior to the demise of the donor, and declaration of circulatory death. Once circulatory death is confirmed, the chest is rapidly opened, and ice is placed in the chest. First, a venous cannula is placed in the right atrium to drain typically greater than 1 L of donor blood to prime the perfusion circuit. Heparin is added to the collection bags.
Next, the donor root is cannulated with an aortic root cannula, a cross-clamp applied in the ascending aorta, and 1 L of cold del Nido cardioplegia is delivered. The heart is vented either by transecting the left lower pulmonary vein or the left atrial appendage. The aorta is then transected as far distal into the arch as possible, to maximize the length of aorta available for intraclamp sewing in the recipient. Then the main pulmonary artery is transected, followed by the superior vena cava at its confluence with the innominate vein and the inferior vena cava. Depending on whether the lungs are procured or not, the pulmonary veins versus the left atrium are transected.
The heart is then inspected, prepared, and attached to the normothermic perfusion system via cannulas in the aorta and the pulmonary artery. At this time, warm blood perfusion begins, and the heart is reanimated. Flow is maintained at a rate of 650 to 900 mL/min into the root. An LV vent is placed via the left atrium to ensure that the left ventricle is decompressed. The prior right atrial cannulation site for donor blood priming is closed. The heart is ready for transport back to the recipient. Serial lactates are measured throughout the period of normothermic perfusion. A target aortic pressure of 65 to 90 mm Hg is maintained, flow is increased to 1.5 L/min as tolerated, and a heart rate of 65 to 100 beats per min is maintained.
Positioning and Preparation
It is imperative to minimize the distance between the organ perfusion platform and the recipient, as this will minimize the length of tubing, pacing wires, potential obstacles, and amount of time that the heart will be draining into a bucket instead of the machine or the recipient chest. The perfusion circuit is prepped and draped just adjacent to the recipient operating room table. Prior to arrival of the organ, an additional antegrade perfusion line is connected to the arterial circuit of the recipient cardiopulmonary bypass (CPB) circuit. It is cut to the appropriate length and primed with recipient blood. Additional cardiotomy suckers are prepared, and adequate length to reach the organ perfusion platform is ensured. A root cannula is prepared as well as pacing wires and a pacing box.
The operating room staff, surgical team, and anesthesia team discuss the overall order of events prior to arrival of the organ:
Rotation of the heart 180° to anatomic position
Aortic root cannulation
Placement of bipolar epicardial pacing wires
Initiation of flow from the CPB circuit and decrease of flow on the organ perfusion device
Placement of a cross-clamp
Separation from the organ perfusion device
Transfer to the basin
Beating heart implantation
Prior to arrival of the donor organ, the recipient undergoes median sternotomy and is placed on CPB. We recommend preparing the pericardial well in such a fashion that drainage is directed into an angled chest tube with a cardiotomy sucker placed within. This is especially important in patients with an LV assist device in place, as upon removal of the diseased heart, there will be a large potential space that can hide drainage returning from the perfused donor heart. Typically, explant of the recipient’s diseased heart is performed immediately prior to or upon arrival of the donor organ to the recipient operating room.
Preparation on Organ Perfusion Device
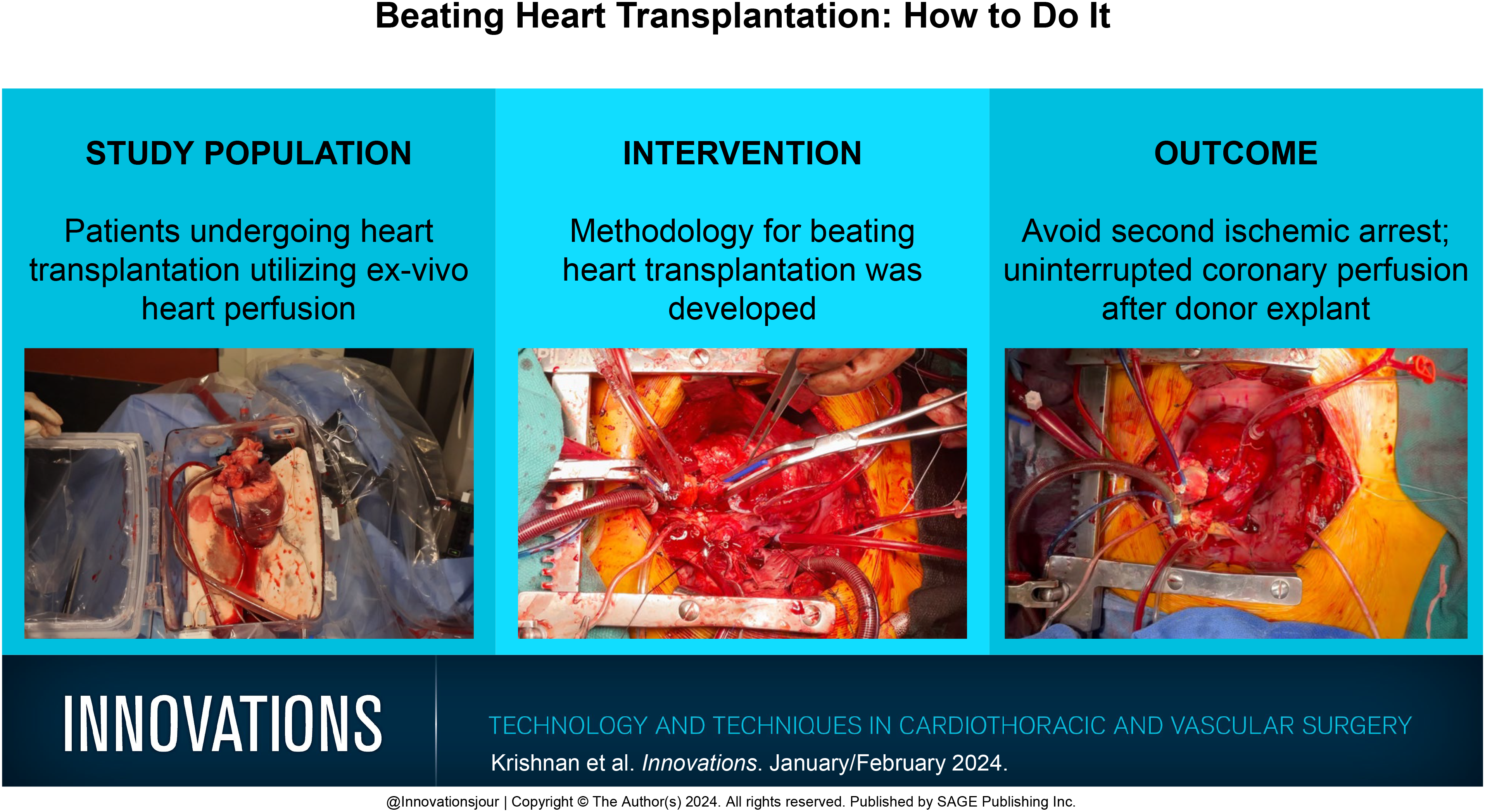
Once the heart arrives and the perfusion platform is prepped and draped adjacent to the recipient, the aortic cannula attaching the donor heart to the organ perfusion circuit is loosened. Then, the surgeon rotates the heart 180°, so the heart appears in anatomic position (Supplemental Video). This rotation facilitates placement of a root cannula on the anterior surface of the aorta. In addition, this will allow for low cross-clamp placement, which will add ease to the aortic anastomosis that takes place between 2 cross-clamps in this procedure. The surgeon must pay attention to the LV vent drainage at this stage, as the rotation may compromise adequate drainage, and LV distention must be avoided. After ensuring that the heart continues to drain well, the cannula is tightened once again.
Next, the root is cannulated with a standard aortic root cannula. The root cannula is then connected via a wet-to-wet connection with the previously prepared antegrade perfusion limb originating from the recipient CPB circuit. Flow from the recipient bypass circuit is not directed to the donor heart yet, and flow to the heart is still exclusively from the organ perfusion circuit.
Bipolar pacing wires are then placed in the epicardium and attached to the pacing box. The heart is paced at 90 beats per min during this time. A basin is brought to the heart, with previously prepared cardiotomy suction cannulas in place to facilitate transfer to the recipient while receiving the drainage from the donor heart and returning it to the CPB circuit.
Now, the organ perfusion circuit flow is progressively decreased to less than 1 L per min. Simultaneously, flow is initiated from the CPB circuit to the heart via the root cannula. Once a target flow of 600 to 800 cc/min is achieved from the CPB circuit, the organ perfusion circuit flow is further reduced to less than 500 cc/min. Next a cross-clamp is placed low on the donor aorta above the root vent with the handles facing the heart, ensuring that all flow is coming from the CPB circuit, functionally isolating the heart from the organ perfusion device. Perfusion to the heart remains uninterrupted during this process. It is imperative to quickly separate the heart from the organ perfusion device at this point by removing it from the aortic and pulmonary cannulas, as blood that drains from the heart into the organ perfusion reservoir is not draining into the CPB circuit but rather the organ perfusion circuit. Once separated from the organ perfusion device, the heart is transferred to the basin, and the drainage is returned to cardiotomy suction, allowing it to be scavenged into the CPB circuit. We recommend that this step from placement of a cross-clamp to transfer of the heart into the basin take no more than 10 s to reduce the risk of blood loss. The basin should be moved to the operative field, and further preparations can be made prior to sewing.
Preparation for Implantation
While the heart is beating in the basin and drainage is adequately captured via the cardiotomy suction, the surgeons can use this opportunity to inspect for a patent foramen ovale and close if necessary. This is an opportunity to prepare sewing cuffs from each of the anastomotic sites. It is important to preserve length on the aorta as this will facilitate sewing between the donor cross-clamp and the recipient cross-clamp. The previously placed LV vent stitch should be tied down or removed at this time. The pulmonary artery cannulation stitch should also be removed. The coronary sinus should be identified clearly at this point as well. We have previously attempted placement of a coronary sinus catheter attached to cardiotomy suction but have found that maintaining position within the sinus is cumbersome with the heart beating and may interfere with sewing. Furthermore, the negative pressure often causes collapse of the coronary sinus and poor drainage thereafter.
At this time, a new LV vent should be prepared for the donor organ as well. We favor placing a vent in the left superior pulmonary vein on the recipient side and then placing it into the left ventricle when the heart is in the chest.
Implantation
Left atrium
Implantation begins with the left atrial anastomosis. We decrease the pacemaker to 45 to 60 beats per min to facilitate sewing. We recommend that the experienced surgeon holds the beating heart in position, ensuring that (1) the heart continues to drain adequately and that the cardiotomy suckers are adequately capturing the drainage, (2) that the aorta does not twist and the root remains pressurized, and (3) that exposure is adequate for sewing. We start with a parachute technique with the first few bites taken between the heart in the basin and the recipient left atrial cuff. After 4 to 5 bites, the heart is moved to the chest. It is imperative that the root remains pressurized at this time and that drainage is continued to be captured to avoid blood loss. Once the heart is in place, the experienced assistant continues to maintain exposure, simultaneously managing the position of the cross-clamp on the donor aorta and ensuring that the drainage cannulas adequately drain the heart. The back wall is completed first, followed by the front wall. Bites are easiest when the heart is relaxed; thus, the operating surgeon will typically take multiple bites while the heart is in diastole. Prior to completion of the anastomosis, the LV vent is ensured across the mitral valve, and anastomosis is then completed. Throughout this time, the assistant and perfusionist maintain communication to ensure adequate root pressurization. The perfusionist targets a root pressure of 150 mm Hg to overcome the length of the tubing, and the assistant constantly monitors the root pressure manually and communicates changes to the perfusionist.
Aorta
The aorta is next in the sequence. There should be ample length of the donor aorta and the recipient aorta, and further trimming can be performed now if the donor clamp and the recipient clamp have been positioned appropriately to maximize aorta available to sew. The donor clamp should be directed caudal, and the recipient cross-clamp should be directed cranial. A standard end-to-end anastomosis is then performed between the clamps. Special attention should be paid to the back wall, as this region will be very hard to access after the anastomosis is complete. Upon completion of the anastomosis, the donor cross-clamp is removed first followed by the recipient cross-clamp. Now flow to the coronaries is provided by both the root cannula and the aortic cannula in the recipient aorta. The root cannula can now be repurposed into a deairing needle when appropriate. The remainder of the operation can proceed as a standard heart transplant. 6
Pulmonary artery, inferior vena cava, and superior vena cava
Pulmonary artery anastomosis is next, with a standard end-to-end approach, with the backwall proceeding first. The inferior vena cava anastomosis is then performed, and the implantation is complete with the superior vena cava anastomosis. These three anastomoses are identical to nonbeating heart methods.
Reperfusion and separation from CPB
Given that there has been uninterrupted coronary perfusion with warm blood since the initial arrest at the time of organ harvesting, and the heart has remained beating, we find that the function of the heart looks excellent with minimal reperfusion. Nevertheless, the function of the heart is assessed thoroughly via transesophageal echocardiogram as we begin to wean the CPB circuit. Standard measures including favorable arterial blood gas, appropriate rhythm, adequate function with a loaded heart, and labs within a normal range are ensured prior to separation from CPB. Cannulas and vents are removed in appropriate sequence, and chest closure commences after satisfaction with the function of the heart and ensuring hemostasis.
Discussion
The use of DCD donors for heart transplantation has marked a pivotal step forward in expanding the donor pool. Currently, it is standard practice to use normothermic organ perfusion to resuscitate the organ in the setting of circulatory death to enable assessment and normothermic perfusion during transportation. The use of normothermic organ perfusion platforms typically requires 2 cardioplegic arrests: one during procurement and the other just prior to implantation. We hypothesized that rearresting the heart exposes it to another bout of ischemia reperfusion injury.7,8 Additionally, we sought to make DCD heart transplantation comparable with brain death donor heart transplantation, which uses only 1 cardioplegic arrest.
In our method, there is only 1 period of ischemia, beginning with warm ischemia while the donor’s life support is withdrawn and then cold ischemia while the organ is being placed on the organ perfusion platform. Once on the device, there is uninterrupted, warm blood perfusion to the coronaries, effectively providing continuous perfusion to the organ. In standard DCD heart transplantation, there is a period of warm ischemia during withdrawal of life support, then cold ischemia during placement on the device, reperfusion, then another period of cold ischemia as the heart is rearrested, and then a final period of warm ischemia when the heart is implanted. In the gold standard of heart transplantation from brain death donors, there is an extended period of cold ischemia during procurement and transport and a period of warm ischemia during implantation of the heart. In removing an entire cardioplegic arrest from DCD heart transplantation, we hypothesize our method of beating heart implantation may lead to better preservation of the allograft and, ultimately, improved short-term results (i.e., decreased intensive care unit length of stay and decreased use of mechanical circulatory support) and long-term performance and survival of the organ.
Notably, the other prevailing methodology of performing DCD heart transplantation is through normothermic regional perfusion (NRP), which does not routinely use normothermic ex vivo organ perfusion. As such, NRP curtails a period of cold ischemia, resulting in 2 warm ischemic periods (one in which the donor is agonal and the other while sewing the organ in the recipient) and 1 prolonged cold ischemic period during cold storage. Our described method of beating heart DCD transplantation involves 1 fewer warm ischemic periods and may be the preferred approach in centers that favor the use of normothermic ex vivo organ perfusion as a method of DCD heart transplantation.
Conclusions
We describe a method of performing DCD heart transplantation with the heart beating, removing the additional period of warm and cold ischemia typically associated with DCD heart transplantation.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
