Abstract
Objective:
The goal of minimally invasive surgery is to reduce trauma to patients and improve their postoperative outcomes. In this context, the utilization of robot-assisted thoracic surgery (RATS) in the treatment of lung cancer has increased worldwide. The feasibility of single-incision major pulmonary resections by RATS was recently reported, with the objective of minimizing the surgical trauma of the traditional multiportal RATS approach. However, both techniques require intercostal incisions, potentially causing immediate and chronic pain resulting from intercostal nerve injury. To reduce postoperative pain resulting from intercostal approaches, we developed a nonintercostal, outside the thoracic cage (OTC) approach for RATS lobectomy, avoiding intercostal instrumentation. This report aims to describe the results of the first reported series of OTC subcostal RATS lobectomies.
Methods:
Retrospective analysis of a series of the first consecutive patients operated on using the novel OTC subcostal RATS lobectomy technique.
Results:
Between August and December 2022, a total of 10 consecutive cases were analyzed. The median age was 63 (55 to 84) years, the mean body mass index was 29 (24 to 45) kg/m2, and the median American Society of Anesthesiologists score was III (II to IV). No serious adverse events were observed, and there was no conversion of the surgical technique. The mean operative time was 132.6 (98 to 223) min. The median length of stay was 2 days. No pain-related complications, readmissions, or 30-day mortality were observed.
Conclusions:
This series demonstrates that OTC RATS lobectomy is feasible and safe. A phase I clinical trial is currently underway to prospectively assess the safety of the technique as well as its clinical relevance.
Central Message
Current robotic approaches for major pulmonary resections require intercostal incisions potentially causing intercostal nerve injury. Avoiding intercostal instrumentation could have a positive impact on postoperative recovery. Our novel nonintercostal OTC RATS approach was feasible and safe.
Introduction
The purpose of minimally invasive surgery (MIS) is to reduce trauma to patients and improve their postoperative and long-term outcomes.1 –7 Recent advances in MIS and robotic platforms have democratized the use of robot-assisted thoracic surgery (RATS) for the treatment of lung cancer. The current standard RATS technique for pulmonary resections relies on the use of 4 intercostal ports.8,9 With the objective of further minimizing surgical trauma, recent reports have suggested that these interventions are feasible through a single incision (i.e., uniportal RATS). 10 However, both approaches involve intercostal incisions that may harm the intercostal nerve bundles, causing immediate as well as chronic pain and the resulting consequences. To spare the intercostal nerves and reduce postoperative pain, subxiphoid and subcostal approaches have been suggested for video-assisted thoracic surgery (VATS) major pulmonary resections.11,12
Using these reports as a premise, we developed a nonintercostal, outside the thoracic cage (OTC) approach for RATS lobectomy, avoiding intercostal instrumentation and potentially improving postoperative course for patients.
Methods
We retrospectively analyzed data from a prospectively maintained surgical database for thoracic surgery including the first patients who underwent OTC RATS lobectomies for the treatment of lung cancer between August 1 and December 1, 2022, at the Centre Hospitalier de l’Université de Montréal (CHUM), an academic center in Montréal, QC, Canada (IRB 2023-11100). Data were collected in the preoperative, intraoperative, and postoperative setting to assess the feasibility and safety of this novel approach. All procedures were performed on the DaVinci Xi platform (Intuitive, Sunnyvale, CA, USA). Statistical analysis was performed using IBM SPSS Statistics, Version 29.0 (IBM Corp, Armonk, NY, USA). Descriptive characteristics are presented as mean, median, and interquartile range for continuous parameters and frequency distributions (number and proportion) for categorical parameters for all patient demographics and outcomes.
Technical Aspects: Right OTC RATS Lobectomy
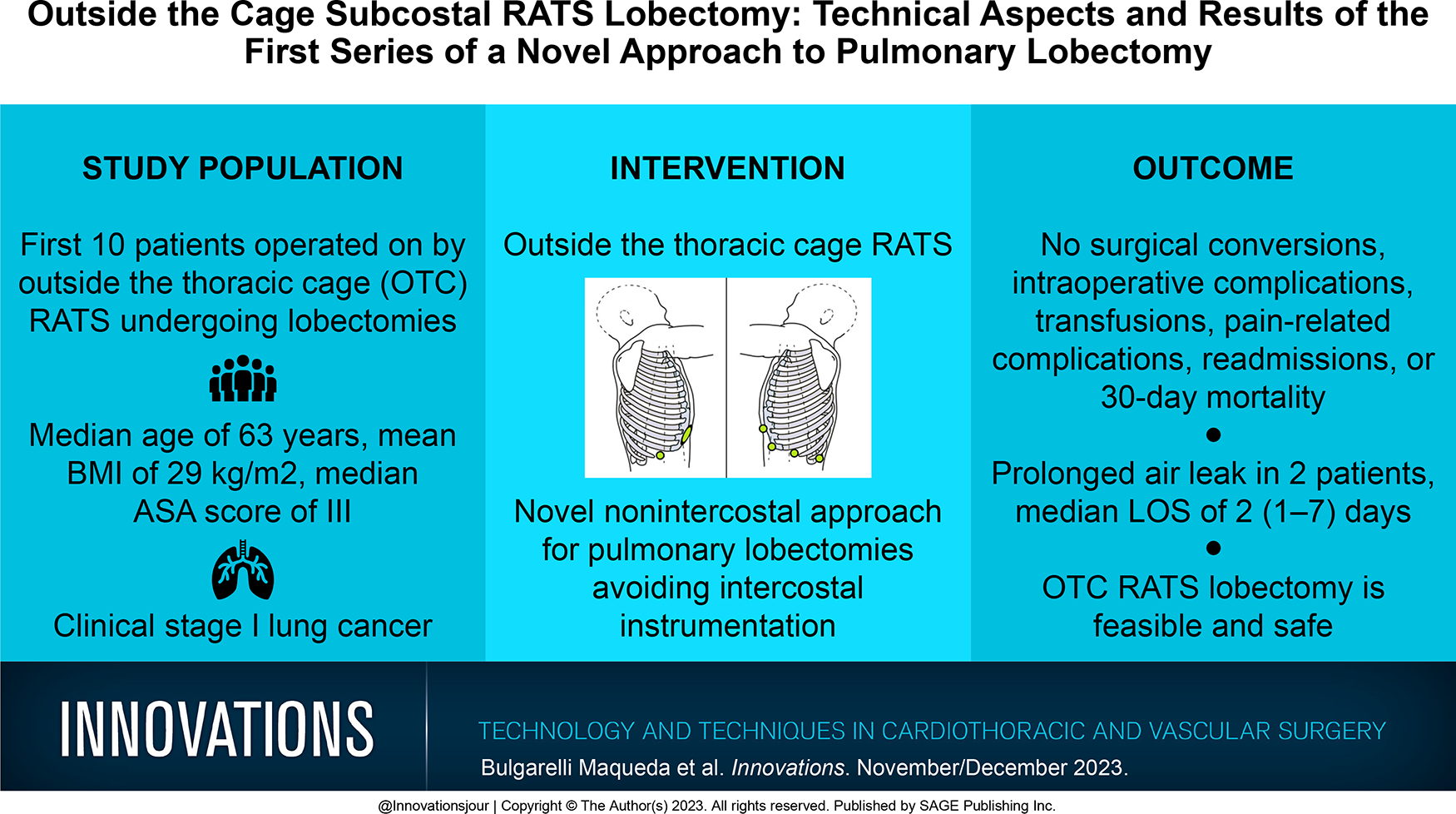
Under general anesthesia and single-lung ventilation, the patient was positioned in a lateral decubitus position with dorsal supports allowing the table to be tilted approximately 15° posteriorly. A 4 cm anterior subcostal incision was performed lateral to the xiphoid process and parallel to the costal margin. The abdominal muscles were partially divided using cautery until the transverse abdominis fascia was reached. Blunt dissection was then used to create a subcostal tunnel to enter the pleural cavity under endoscopic vision (Supplemental Video 1). A wound protector was placed to keep the working port open and dry for the duration of the procedure. Next, a second subcostal incision was performed posteriorly below the 10th rib and medial to the 11th rib, where a 12 mm trocar was inserted under direct vision using the camera through the anterior port (Fig. 1). Alternative port placements included adding additional subcostal ports and replacing the anterior working incision with two 8 mm or 12 mm ports allowing for the use of CO2 if needed.
Subcostal port positioning for right hemithorax outside the thoracic cage robot-assisted thoracic surgery.
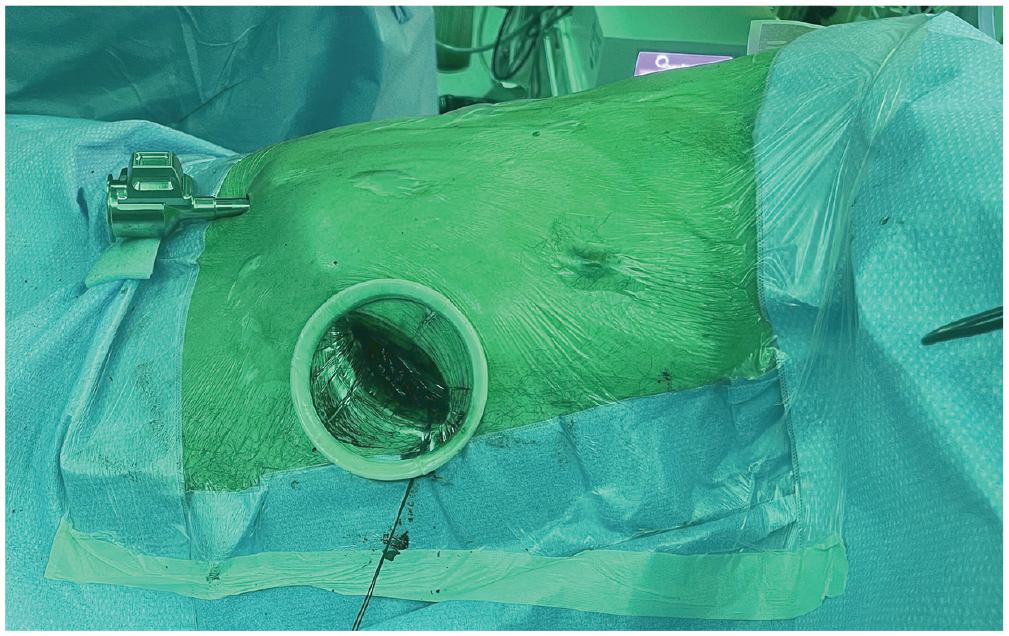
The surgical cart of the robotic system was then positioned at the front of the patient. The first robotic arm was extended caudally and posteriorly, whereas arms 2, 3, and 4 were extended caudally and anteriorly toward the anterior subcostal incision (Supplemental Video 1). First, the camera was docked in arm 2 with a 30° downward setting at the posterior angle of the working incision. Next, a robotic tip-up fenestrated forceps was inserted in arm 1 through the posterior port under direct vision. Arm 3 was then docked in the anterior subcostal wound medial to the camera with a robotic Maryland bipolar forceps, and arm 4 was docked in the medial angle of the wound with a robotic hook (Fig. 2). One of the advantages of robotic systems such as the one used for this series lies in the ability to easily interchange the attachments between arms when needed.
Robotic arm positioning in the anterior subcostal working port for right-side outside the thoracic cage robot-assisted thoracic surgery.
In this setting, the assistant could use a curved suction inserted through the anterior port to facilitate dissection and keep the field free of cautery smoke. The lobectomies were performed using a standard anterior fissure-less approach; in this case, a right upper lobectomy is shown (Supplemental Video 1). During the procedure, hilar lymph nodes (LN) were individualized and resected while dissecting the hilum structures. Subcarinal and paratracheal LN were dissected out after lobectomy was completed, and the specimen was moved caudally in the thoracic cavity (Supplemental Video 1). Once complete, the robotic arms and ports were removed from the chest, a drain was inserted through the posterior incision, and “figure of 8” stitches with absorbable sutures were placed to close the diaphragmatic openings of the port sites. A stay suture was placed around the drain using a trocar site closure device that was inserted through the skin and then tied when removing the drain.
Technical Aspects: Left OTC RATS Lobectomy
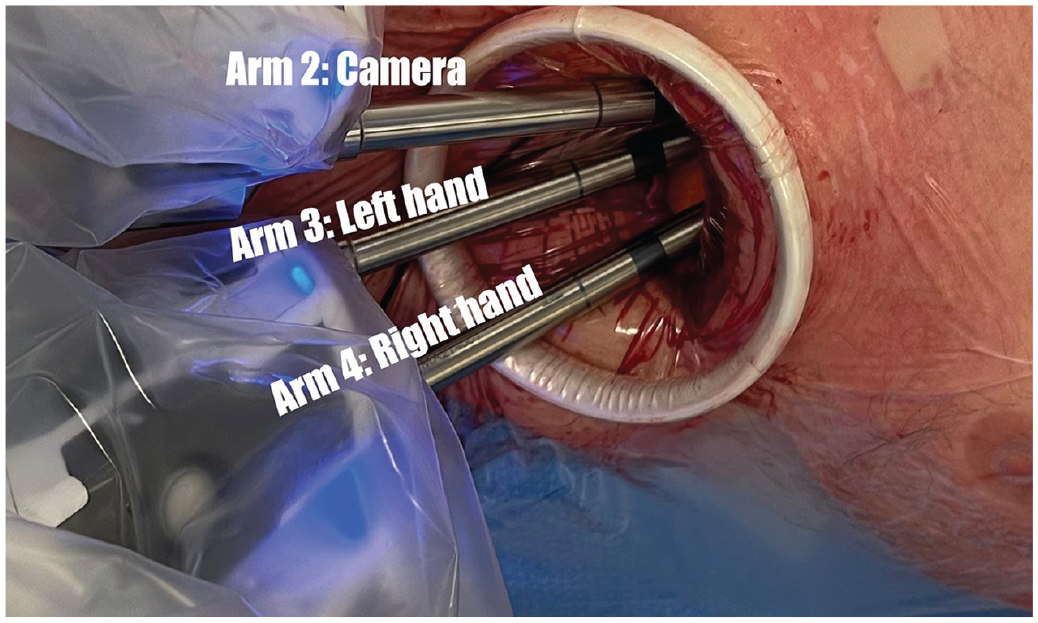
Using the same anesthesia technique, patient position, and robot setup, a subcostal port was placed in the midaxillary line below the 10th rib using an 8 mm robotic trocar where the camera was inserted. CO2 insufflation was used at 8 mm Hg to descend the diaphragm and cause a mild contralateral mediastinal shift, to allow the surgeon to safely insert the remaining subcostal ports in proximity to the insertion point of the diaphragm on the rib cage under thoracoscopic vision. A second subcostal 8 mm port for the camera was placed 4 cm lateral to the xiphoid process, avoiding mediastinal and phrenic impairment. A third subcostal 12 mm robotic trocar was then placed at the level of the posterior axillary line below the 10th rib and medial to the 11th rib. Lastly, an 8 mm subcostal port was placed along the anterior axillary line (Fig. 3). The first robotic arm was docked in the most anterior port, arm 2 in the anterior axillary line port, arm 3 in the midaxillary line port, and arm 4 in the most posterior 12 mm port. A Maryland bipolar forceps was placed in arm 1, the camera in arm 2, and a hook in arm 3. Throughout the procedure, arm 4 was used for the tip-up fenestrated forceps as well as the robotic staplers and suction. As mentioned, all attachments may have been interchanged if needed.
Subcostal port positioning for left hemithorax outside the thoracic cage robot-assisted thoracic surgery.
The left-sided lobectomies were also performed in a standard anterior fissure-less fashion; in this case, a left upper lobectomy is displayed (Supplemental Video 2). LN dissection was carried out in the same fashion as detailed for the contralateral technique. Once complete, the robotic arms and ports were removed, and a drain was inserted through the midaxillary line port and secured accordingly. The specimen was retrieved using a specimen retrieval pouch through the most anterior port that was extended by 2 cm. The anterior incision was sutured in layers, whereas the ports were closed using absorbable sutures, as previously described.
Of note, for this series we maintained the same strategy applied to all our VATS and intercostal RATS procedures, which is based on a paravertebral block performed at the end of every procedure (Supplemental Video 2), and standard doses of regular acetaminophen and hydromorphone as needed.
Results
A total of 10 consecutive cases were completed in this study. These were the first 10 patients operated on using the novel OTC RATS technique. Demographic data described in Table 1 show an equal sex distribution, a median age of 63 (55 to 84) years, a mean body mass index (BMI) of 29 (24 to 45) kg/m2, and a median American Society of Anesthesiologists (ASA) score of III (II to IV). All patients presented with preoperative clinical stage I (≤cT2N0M0) disease according to the International Association for the Study of Lung Cancer non-small cell lung cancer tumor, node, and metastasis classification, 8th edition. Table 1 describes the patient characteristics and comorbidities. Right-sided lesions in the right upper lobe were predominant (n = 8, 80%). Surgical procedures were uneventful with no conversions of the surgical technique to open or intercostal RATS or VATS, no intraoperative complications, and no 30-day mortality. Surgical outcomes are displayed in Table 2.
Baseline Characteristics.

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; HTN, hypertension; LLL, left lower lobe; LUL, left upper lobe; RLL, right lower lobe; RML, right middle lobe; RUL, right upper lobe.
Data are reported as mean, median (IQR), or n (%).
Surgical Outcomes.
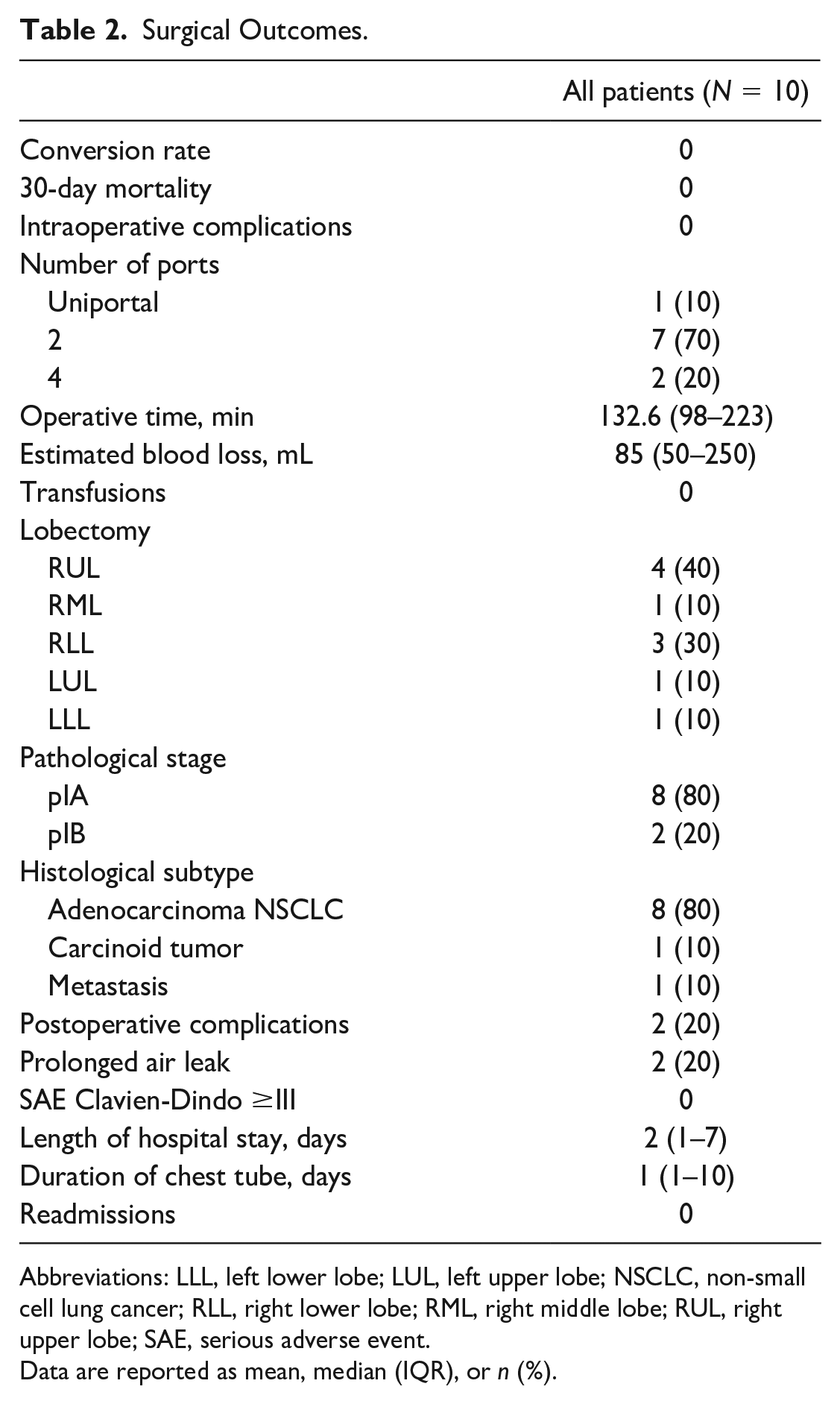
Abbreviations: LLL, left lower lobe; LUL, left upper lobe; NSCLC, non-small cell lung cancer; RLL, right lower lobe; RML, right middle lobe; RUL, right upper lobe; SAE, serious adverse event.
Data are reported as mean, median (IQR), or n (%).
Most procedures were performed in the right hemithorax (n = 8, 80%) and used only 2 ports (n = 7, 70%). A single case was completed with a uniportal OTC RATS approach, and it was for a cT1cN0 pure ground-glass lesion located in the middle lobe. The mean operative time was 132.6 (98 to 223) min, with an estimated blood loss of 85 (50 to 250) mL. No patient required a blood transfusion. All patients had pathological stage I disease, and the most common histological diagnosis was lung adenocarcinoma (n = 8, 80%). The median hospital length of stay (LOS) was 2 (1 to 7) days, and the median chest tube duration was 1 (1 to 10) day. The postoperative complication rate was 20% (n = 2), namely, prolonged air leaks. There were no significant adverse events and no Clavien-Dindo classification complications of III or above. There were no pain-related complications or readmissions observed in this series.
Discussion
Since the early 1990s, evidence in favor of reducing surgical trauma has prompted the development of MIS techniques, including VATS, resulting in smaller incisions. Surgical approaches in thoracic surgery have continued to evolve in favor of minimizing the size and quantity of ports, 5 as demonstrated by recent publications on the feasibility of uniportal RATS for major anatomical resections. 10 Regardless of size or quantity of access ports, all require intercostal incisions that put patients at risk of developing acute or chronic neuropathic pain resulting from intercostal nerve injury, which is the most common complication after thoracic surgery affecting nearly 30% to 40% of patients. 13
In an effort to reduce chronic pain by avoiding intercostal nerve instrumentation, less traumatic approaches have been described for major pulmonary resections by VATS. These strategies involve entering the thorax through subxiphoid or subcostal incisions, which studies suggest results in significantly lower postoperative pain in comparison with the standard intercostal approach. 12 On one hand, although the subxiphoid approach is less traumatic and avoids intercostal instrumentation, it often requires resection of the xiphoid process. In addition, its medial position, although beneficial for certain surgeries such as thymic resections, could lead to mediastinal or cardiac impairment during lung resection, especially in the left hemithorax. On the other hand, subcostal incisions 5 may be less traumatic, as they do not require resection of the xiphoid process and present less risk of mediastinal compression.
With the rise in the use of multiportal and uniportal robotic platforms in thoracic surgery,14,15 it is becoming increasingly pressing to develop less traumatic techniques that are well adapted to these technologies. Following the rationale described for VATS, we developed a subcostal approach for RATS lobectomy using the most readily available robotic platform. This approach avoids intercostal instrumentation, potentially providing short-term and long-term benefits to patients while giving full control of the instruments to the surgeon, including the robotic stapler.
The demographic characteristics in this series of cases suggest that although the sample size is small, it is still an accurate representation of a “real world” sample. The selection of patients was primarily based on their clinical stage to limit surgical complexity until feasibility could be properly assessed. It is of note that the median ASA score was III (II to IV), BMI ranged from 24 to 45, age ranged from 55 to 84 years, most patients were smokers (n = 8, 80%), and most presented with multiple comorbidities. The heterogeneity of the patients is representative of the target population.
Most cases were completed in the right hemithorax (n = 8, 80%) due to selection bias of the surgical team, who felt more comfortable developing the technique on the right side to avoid a possible impairment of the mediastinum. With experience and confidence in the technique, an increasing volume of left-sided lobectomies are currently being performed. Regarding surgical outcomes, the absence of conversion, intraoperative complications, transfusions, pain-related complications, readmissions, and 30-day mortality are all indicative of the feasibility and safety of this approach for lobar resections.
Prolonged operative time in MIS pulmonary resections has been reported to be associated with increased postoperative complications.16 –18 In this series, the mean operative time was 132.6 (98 to 223) min, which is lower than the threshold proposed by the cited authors at which complications are more frequently observed. In addition, a 25% decrease (mean 99.4 min) was observed in the second half of cases. This trend could imply that increasing the surgical volume and improving mastery of the technique could further decrease operative time and therefore potentially improve the safety of the technique.
Pathological staging was stage I in all cases, which is consistent with the preoperative clinical stage, with most histological diagnosis being lung adenocarcinoma (n = 8). Postoperative complications, specifically prolonged air leak, were present in 20% of the cases (n = 2), which resulted in prolonged LOS and discharge with Heimlich valves. Both patients had severe emphysema. There were no significant adverse events with a Clavien-Dindo score of III or greater. No patient required readmission. The median LOS was 2 (1 to 7) days and the median chest tube duration was 1 (1 to 10) day, both of which are consistent with the average for other patients undergoing MIS pulmonary resections by our team.
We observe obvious limitations given the retrospective design of this study, its small sample size, the selection bias toward clinical stage I lung cancer patients, and the fact that all cases were performed by a single surgeon. Nonetheless, the feasibility and safety of the technique were demonstrated based on observed surgical outcomes, which gave rise to a phase I clinical trial (NCT05832112). Long-term studies will be necessary to evaluate chronic pain rates as well as diaphragmatic hernia rates using the OTC technique.
Conclusions
OTC RATS lobectomies were feasible and safe in this first series serving as our benchmark for procedural development. The observed results have prompted the continuation of the study in an ongoing human phase 1 clinical trial to further assess the large-scale feasibility and safety of this novel technique as well as its long-term clinical relevance. Multicenter studies will be required to assess the generalizability of the technique.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
