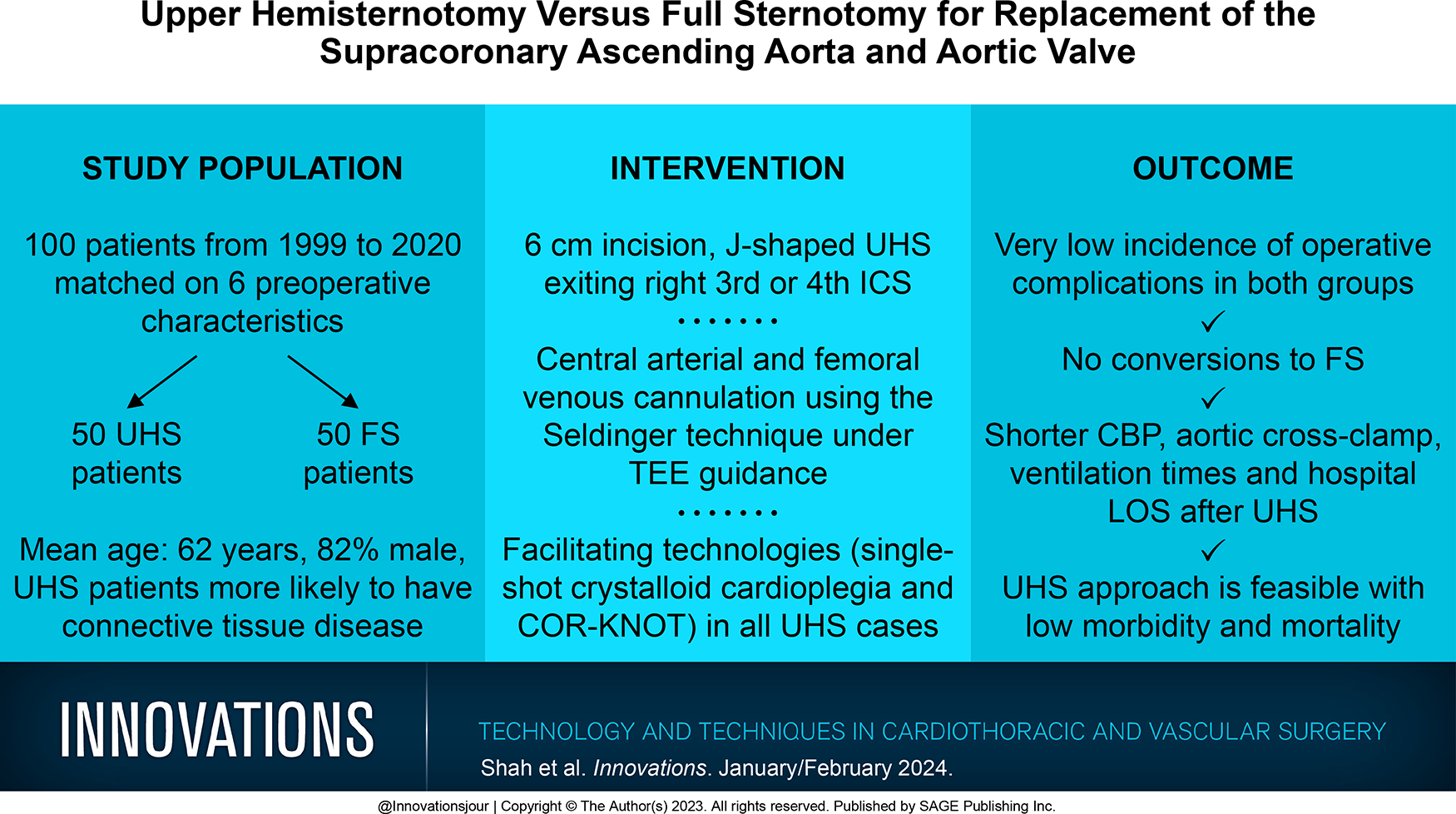
Abstract
Objective:
Upper hemisternotomy (UHS) for supracoronary ascending aorta replacement (scAAR) with concomitant aortic valve replacement (AVR) results in less trauma and potentially faster convalescence compared with full sternotomy (FS). Direct head-to-head studies are lacking. We compared a group of UHS patients with a matched group of FS patients undergoing scAAR and AVR.
Methods:
There were 198 patients who underwent scAAR and AVR procedures by a single surgeon between 1999 and 2020. After matching 6 preoperative characteristics, there were 50 UHS and 50 FS patients. Patients who required acute type A aortic dissection repair, reoperations, concomitant procedures, or hypothermic circulatory arrest were excluded.
Results:
In the matched sample, the hospital mortality rate was 1% (1 of 100). The median cardiopulmonary bypass time was 150 (interquartile range [IQR], 131 to 172) min and 164.5 (IQR, 138 to 190) min, respectively, for the UHS and FS groups (P = 0.08). The median aortic cross-clamp time was 121 (IQR, 107 to 139) min during UHS and 131 (IQR, 115 to 159) min during FS (P = 0.05). The median ventilation time was 7 (IQR, 3 to 14) h versus 17 (IQR, 10 to 24) h, respectively, after UHS and FS (P = 0.005). The median hospital length of stay was 7 (IQR, 6 to 9) days after UHS and 8 (IQR, 7 to 11) days after FS (P = 0.05).
Conclusions:
The low morbidity and mortality support the wider use of UHS for scAAR and AVR in appropriately selected patients. Larger studies are needed to confirm these initial findings.
Keywords
Central Message
The UHS approach is a safe and reproducible technique for supracoronary ascending aorta and aortic valve replacement. The low morbidity and mortality further support its use in select patients and experienced hands.
Introduction
Supracoronary ascending aorta replacement (scAAR) with aortic valve replacement (AVR), chronicled by Groves et al. in 1964, is the operation of choice in patients with concomitant ascending aorta aneurysm and aortic valve disease. 1 This operation is conventionally performed with a full sternotomy (FS) to provide abundant exposure to the ascending aorta and aortic valve. Over the past 2 decades, upper hemisternotomy (UHS) has been increasingly adopted for isolated AVR and now has spread to proximal aortic surgery.2–5 Our recent studies on the UHS David and Bentall (i.e., mini-Bentall) procedures demonstrated equivalent or superior outcomes compared with the FS counterparts.6–8 Except for a single multisurgeon European study, a direct comparison of UHS with FS for scAAR and AVR is lacking presumably due to the steep learning curve involved with utilization of UHS for complex proximal aortic surgery and concerns about adequate exposure using UHS with a dilated aorta.9,10 Consequently, the aim of this study was to compare perioperative results between UHS and FS after scAAR and AVR in a case-matched patient population over a 20-year single-surgeon experience.
Methods
Patient Selection
All patients who underwent scAAR with concomitant AVR between 1999 and 2020 by a single surgeon across several institutions were included (Fig. 1). Patients were captured from a prospectively compiled Aortic Wellness Database containing preoperative, intraoperative, and postoperative data, and their data were analyzed retrospectively. Patients who had an acute type A aortic dissection, root abscess, infectious endocarditis or aortitis, reoperation, concomitant cardiac procedure (i.e., mitral or tricuspid surgery, coronary artery bypass grafting [CABG]), or open distal anastomosis, hemiarch, or total aortic arch replacement using hypothermic circulatory arrest were excluded from the study. A total of 198 consecutive patients underwent an elective or urgent scAAR and AVR procedure performed by a single surgeon (K.A.P.). From October 1999 to October 2014, 104 patients underwent elective or urgent FS scAAR and AVR procedures. From December 2014 to April 2020, 94 patients underwent elective or urgent UHS scAAR and AVR procedures. Since December 2014, all elective or urgent scAAR and AVR procedures were performed using UHS, with no patients undergoing FS. Institutional review board approval for use of the deidentified database for analysis (No. 20D.802) and the study (iRISID-2023-2417) was obtained.
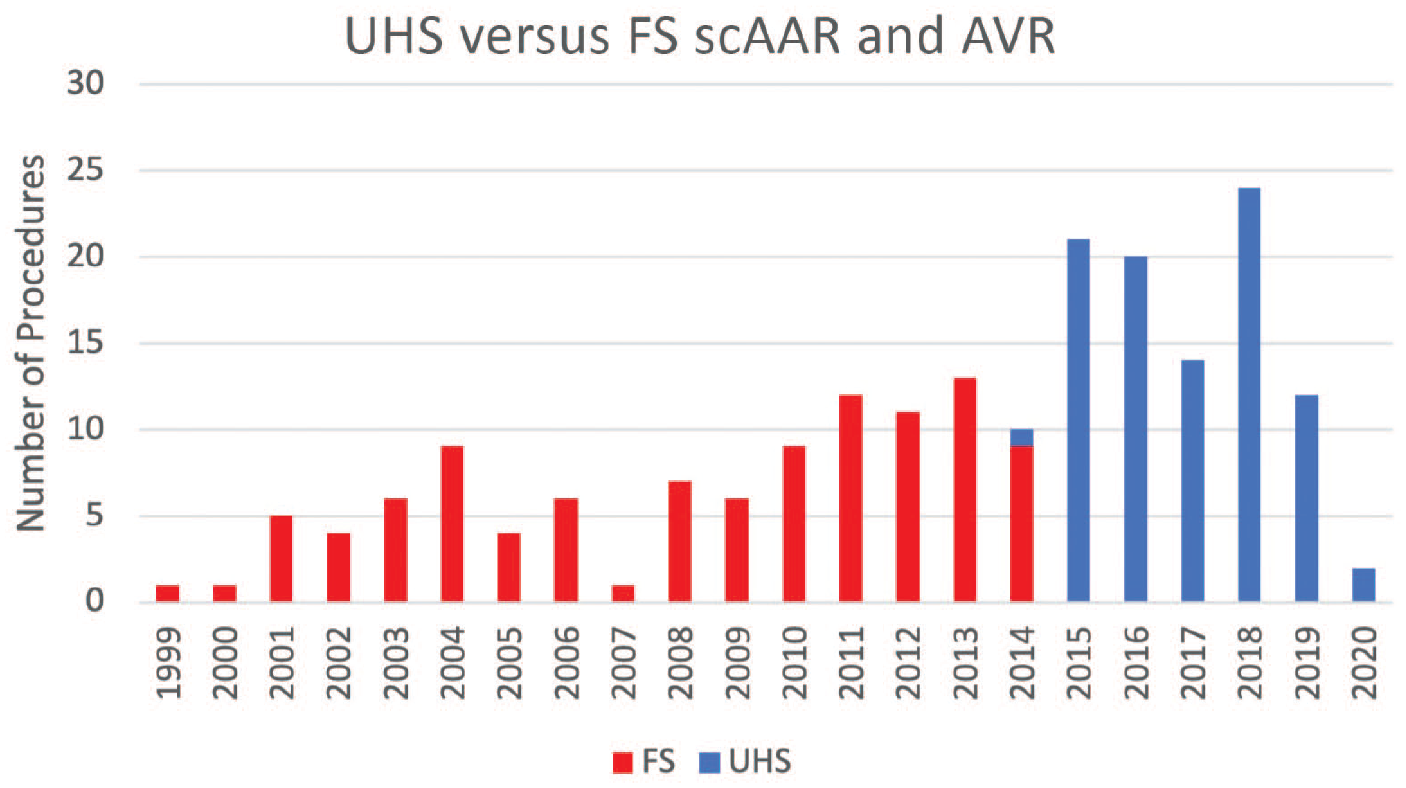
Upper hemisternotomy versus full sternotomy procedures between 1999 and 2020.
Endpoints
The primary endpoint was 30-day mortality. Secondary endpoints were cardiopulmonary bypass (CPB), aortic cross-clamp (ACC), and mechanical ventilation times; occurrence of stroke, transient ischemic attack, renal insufficiency, wound infection, prolonged ventilatory support >24 h, reoperation for bleeding, and blood product transfusions; and intensive care unit (ICU) and hospital length of stay (LOS). The definitions adopted for the endpoints were in accordance with the Society of Thoracic Surgeons Adult Cardiac Surgery Database specifications.
Surgical Technique
Our UHS approach to proximal aortic surgery has been previously published.6 –8 A J-shaped UHS was performed 2 cm inferior to the sternal notch to the midpoint of the third or fourth intercostal space and exiting the right lateral third or fourth intercostal space, carefully avoiding injury to the right internal mammary artery and vein. Adjunctive techniques to help maximize visualization during UHS were strategic placement of pericardial traction sutures to both anteriorize the ascending aorta toward the incisional surface and position the aorta to the midline in addition to cephalad traction sutures around the ACC and through the pericardium. Also, in patients with deep mediastinal anatomy, pericardial sutures were placed and tagged, the sternal retractor was removed, upward traction was then placed on the pericardial sutures, and the sternal retractor was reinserted.
The proximal aortic arch was directly cannulated using the Seldinger technique under transesophageal echocardiographic guidance. The right femoral vein was directly cannulated, and a 25F long femoral venous cannula (Bio-Medicus; Medtronic, Dublin, Ireland) was advanced into the superior vena cava using the Seldinger technique under transesophageal echocardiographic guidance. The femoral venous cannula was “Y-ed” to a smaller right atrial cannula in the event of inadequate venous drainage. In all UHS cases, 2 L of single-dose, long-acting crystalloid antegrade cardioplegia, either Custodiol histidine-tryptophan-ketoglutarate (HTK; Essential Pharmaceuticals LLC, Durham, NC, USA) or del Nido (Nephron Pharmaceutical Corporation, West Columbia, SC, USA) solution, was administered into the aortic root after ACC. In cases of severe aortic insufficiency, an initial 1 L dose of antegrade cardioplegia was delivered, and the remaining 1 L solution was infused into the coronary ostia after aortotomy. During FS, the right atrium was cannulated directly, and cold blood potassium-based cardioplegia was injected in most cases, with repeated doses every 15 to 20 min. A direct main pulmonary arterial or, less commonly, a right superior pulmonary vein vent was inserted. The field was flooded with carbon dioxide in both groups.
Subsequently, AVR was performed in a standard fashion with either a stented mechanical or biological valve based on age and patient preference. Noncoronary sinus resection with reapproximation was performed for moderate aortic root ectasia. The COR-KNOT automated suture fastener (LSI SOLUTIONS, Victor, NY, USA) was used in all UHS cases, while manually hand-tied knots were used in most FS cases. An appropriately sized graft was anastomosed to the sinotubular junction with Teflon felt-reinforced 4-0 polypropylene in a continuous fashion. The ascending aorta was divided 1 cm below the ACC. The distal anastomosis was performed with a Teflon felt-reinforced continuous 4-0 polypropylene suture with reinforcing sutures applied to the inside of the posterior suture line. In UHS cases, the chest was closed with 3 or 4 stainless steel wires placed around the hemisternum.
Statistical Analysis
The UHS patients were matched with the FS patients based on age, sex, hypertension, diabetes, Marfan syndrome status, and diameter of the ascending aorta. It was impossible to select patients from the 2 groups who were exactly matched using the 6 preoperative characteristics. A computer program was written to find the best possible matched pairs. The statistical technique of matching by propensity score was not employed; adopting this method to match patients would have resulted in extremely small sample sizes, precluding a meaningful data analysis. The 2 groups of patients were then compared using the two-sample t test for continuous outcomes (Mann–Whitney U test when the normality assumption was not tenable) and the chi-square test (Fisher’s exact test when expected frequencies were small) for categorical outcomes. Statistical methods appropriate for independent samples in lieu of paired samples were employed because matching patients was not meant to induce dependence within pairs. Matching was simply a tool to reduce the imbalance between the 2 surgical groups with respect to the 6 preoperative characteristics. All tests of hypotheses were two-sided and used a 0.05 significance level. SAS 9.4 (SAS Institute, Cary, NC, USA) was used for all data analyses.
Results
Before matching, there were 94 UHS and 104 FS patients. After matching, there were 50 UHS and 50 FS patients. Baseline patient characteristics in the matched and unmatched groups are presented in Table 1 and the Supplemental Table, respectively. Table 1 shows the success of the matching based on the 6 preoperative characteristics. There was a preoperative imbalance with respect to some of the characteristics that were not used in matching patients.
Matched Preoperative Characteristics.
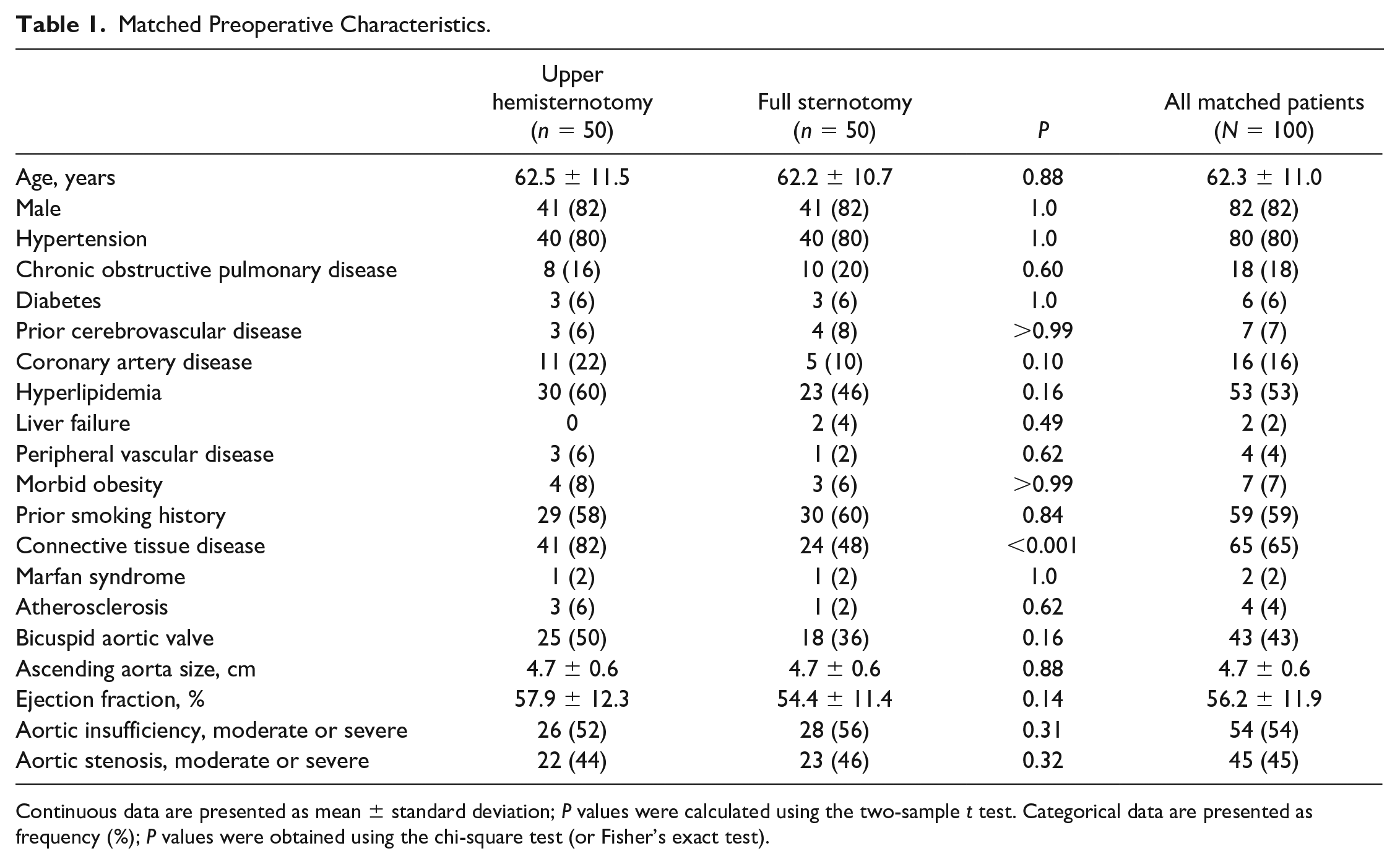
Continuous data are presented as mean ± standard deviation; P values were calculated using the two-sample t test. Categorical data are presented as frequency (%); P values were obtained using the chi-square test (or Fisher’s exact test).
There was a male predominance in both groups (82% vs 82%, P = 1.0), and the mean patient age was 62.5 ± 11.5 years in the UHS group and 62.2 ± 10.7 years in the FS group (P = 0.88). The mean diameter of the ascending aorta was 4.7 ± 0.6 cm for UHS patients and 4.7 ± 0.6 cm for FS patients (P = 0.88). The usual comorbid conditions were hypertension (80% vs 80%, P = 1.0), hyperlipidemia (60% vs 46%, P = 0.16), prior smoking history (58% vs 60%, P = 0.84), and bicuspid aortic valve (50% vs 36%, P = 0.16) in the UHS and FS cohorts, respectively. The UHS group had a significantly greater prevalence of connective tissue disease (82% vs 48%, P < 0.001). There was a similar distribution of patients with diabetes (6% vs 6%, P = 1.0), chronic obstructive pulmonary disease (16% vs 20%, P = 0.60), and morbid obesity (8% vs 6%, P > 0.99). The mean preoperative ejection fraction was 57.9% ± 12.3% in the UHS group and 54.4% ± 11.4% in the FS group (P = 0.14). Also, there was no difference in the prevalence of moderate or severe aortic insufficiency (52% vs 56%, P = 0.52) and moderate or severe aortic stenosis (44% vs 46%, P = 0.32) between the groups.
Operative results are shown in Table 2. A small number of patients in both groups required urgent procedures (6% vs 8%, P > 0.99). The median CPB time was 150 (interquartile range [IQR], 131 to 172) min and 164.5 (IQR, 138 to 190) min, respectively, for the UHS and FS groups (P = 0.08). The median ACC time was 121 (IQR, 107 to 139) min for UHS patients and 131 (IQR, 115 to 159) min for FS patients (P = 0.05). The total median blood product use for packed red blood cell (0 [IQR, 0 to 1] U, P = 0.11), fresh frozen plasma (0 [IQR, 0 to 2] U, P = 0.05), cryoprecipitate (0 [IQR, 0 to 1] U, P = 0.10), and platelet (0 [IQR, 0 to 2] U, P = 0.75) transfusions was very low. A biological valve was more likely to be implanted in UHS (94%) and FS (92%) patients (P > 0.99). The median prosthesis size implanted was 25 mm for UHS (IQR, 25 to 27 mm) and FS (IQR, 23 to 27 mm) patients (P = 0.88). Starting in January 2013, the simultaneous implementation of 2 facilitating technologies commenced in all cases, which included COR-KNOT and single-dose cardioplegia (i.e., Custodiol-HTK cardioplegia from January 2013 to January 2020, and del Nido cardioplegia from January 2020 to present). COR-KNOT was used in all UHS cases and 19 FS cases (38%), whereas manually hand-tied knots were used in 31 FS cases (62%, P < 0.001). Custodiol-HTK and del Nido cardioplegia were administered in 49 (98%) and 1 (2%) UHS cases, respectively, while Custodiol-HTK and cold blood potassium-based cardioplegia were injected in 19 (38%) versus 31 (62%) FS cases, respectively (P < 0.001).
Matched Intraoperative Results.
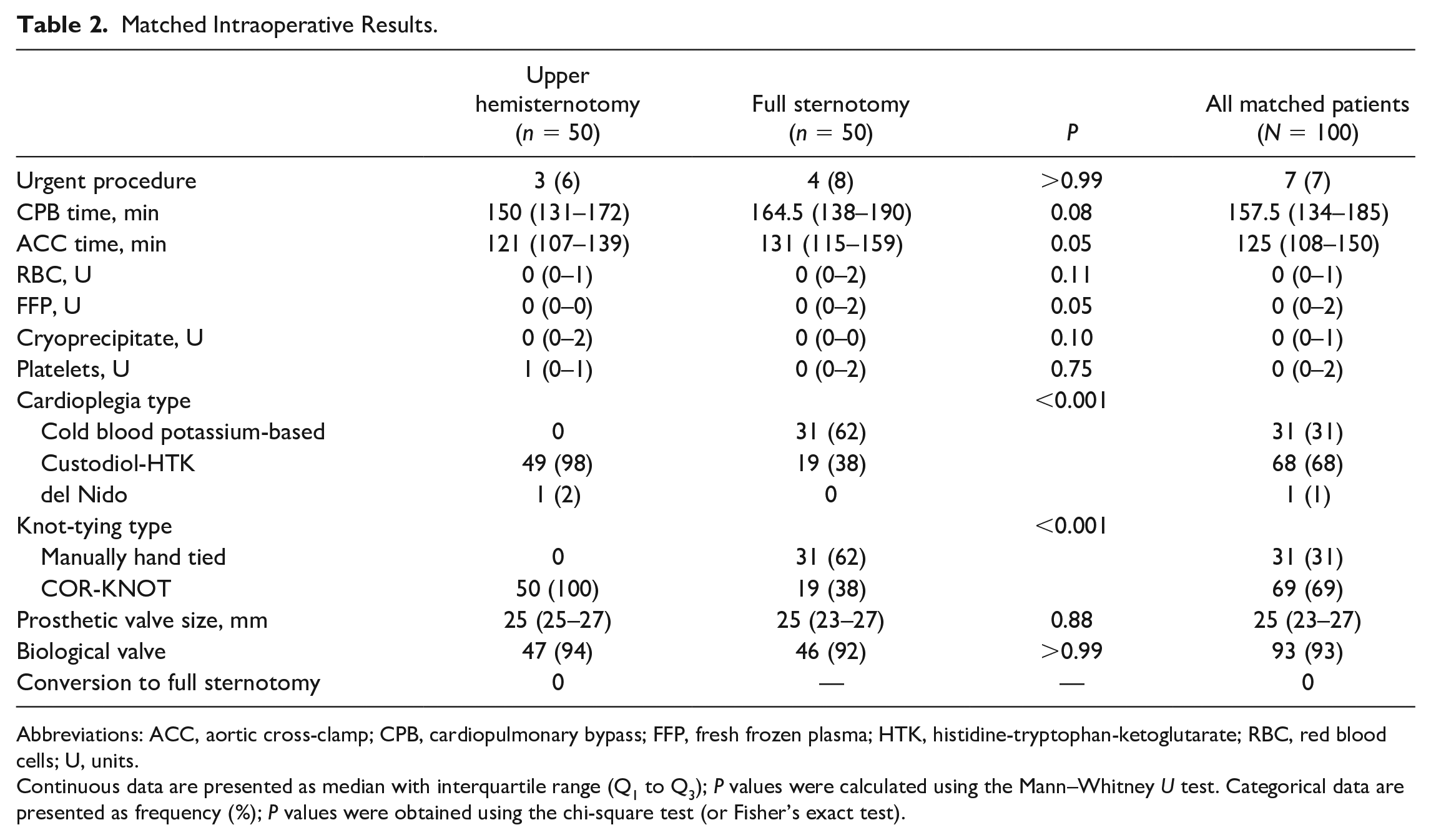
Abbreviations: ACC, aortic cross-clamp; CPB, cardiopulmonary bypass; FFP, fresh frozen plasma; HTK, histidine-tryptophan-ketoglutarate; RBC, red blood cells; U, units.
Continuous data are presented as median with interquartile range (Q1 to Q3); P values were calculated using the Mann–Whitney U test. Categorical data are presented as frequency (%); P values were obtained using the chi-square test (or Fisher’s exact test).
Thirty-day mortality was 1% (1 of 100), specifically a UHS patient who died in the hospital after ventricular fibrillation and cardiac arrest on postoperative day 7. Other postoperative results are shown in Table 3. The occurrence of stroke (2% vs 2%, P = 1.0), transient ischemic attack (2% vs 0%, P > 0.99), and prolonged ventilatory support (6% vs 6%, P = 1.0) were similar between the UHS and FS groups, respectively. There were no patients with renal insufficiency, wound infections, or reintubations in either cohort. The UHS group had a shorter median ventilation time compared with the FS group (7 [IQR, 3 to 14] h vs 17 [IQR, 10 to 24] h, P = 0.005). There were no conversions to FS during UHS. One UHS patient (2%) and 2 FS patients (4%) underwent reoperation for bleeding (P > 0.99). The median ICU stay was 3 days for the UHS (IQR, 2 to 5 days) and FS (IQR, 2 to 4 days) groups (P = 0.51). The median LOS was 7 (IQR, 6 to 9) days for the UHS group and 8 (IQR, 7 to 11) days for the FS group (P = 0.05).
Matched Postoperative Results.
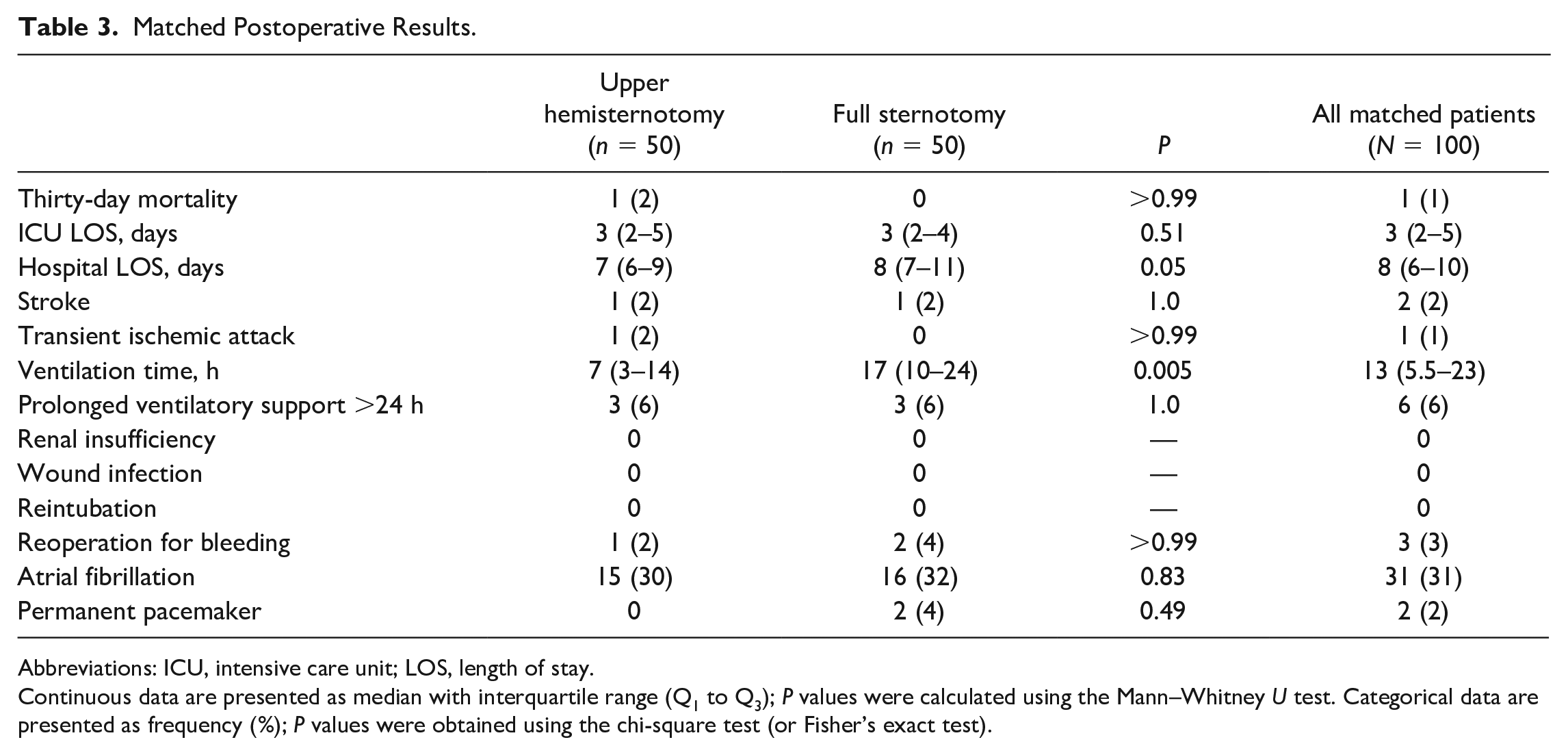
Abbreviations: ICU, intensive care unit; LOS, length of stay.
Continuous data are presented as median with interquartile range (Q1 to Q3); P values were calculated using the Mann–Whitney U test. Categorical data are presented as frequency (%); P values were obtained using the chi-square test (or Fisher’s exact test).
Discussion
A recent comprehensive meta-analysis comparing minimally invasive aortic surgery with FS reported reduced rates of reoperation for bleeding, renal insufficiency, ICU and hospital LOS, and shorter CPB time with no difference in mortality. 11 After exploring outcomes of a J-shaped UHS for proximal aortic operations of which 209 of 483 patients (43.2%) underwent AAR and AVR, Levack and Svensson demonstrated reduced ICU and hospital LOS and hospital cost besides better cosmesis. 4 Moreover, Staromlyński and Suwalski revealed that a V-shaped UHS is feasible in supracoronary AAR and AVR in 44 patients. 5 In a 9-year experience using a J-shaped UHS for aortic procedures including concomitant AVR with scAAR in 65 patients, Kaneko et al. published a 30-day mortality rate of 2.7%, very low incidence of operative complications with no conversions to FS, ventilation time of 5.9 h, and ICU LOS of 27 h. 12 In a subgroup of 9 patients undergoing UHS scAAR and AVR, Byrne et al. found no deaths and 1 patient requiring reoperation for bleeding. 13 Our prior studies also demonstrate that UHS provides safe and equal access for complex proximal aortic operations with other advantages.6–8 We found less ventilation time and fewer reoperations for bleeding after the mini-Bentall procedure in a contemporary cohort of 48 patients. 7 Moreover, we found no postoperative differences in a select cohort of 43 patients undergoing isolated elective UHS versus FS David procedures. 6 In a head-to-head comparison between 117 UHS and 234 FS scAAR and AVR propensity-matched patients at a multisurgeon European center, Haunschild et al. reported equally low morbidity and mortality without significant differences between groups. 9 The principal findings in this study also include low rates of complications and mortality in a total of 100 scAAR and AVR matched patients.
Thirty-day mortality was 1% (1 of 100) in this study, compared with 0.8% (3 of 357) reported by Haunschild and colleagues and 0% in 44 UHS patients reported by Staromlyński and colleagues after AAR and AVR.5,9 After UHS, 1 patient (2%) had a stroke, and 1 patient (2%) had a transient ischemic attack. There were no occurrences of renal insufficiency, wound infection, or reintubations in either cohort. Staromlyński et al. published an occurrence of 5% (2 of 44) neurologic complications, 7% (3 of 44) acute kidney failure, and 5% (2 of 44) wound infections after UHS. 5 Following UHS, Haunschild et al. observed that revision was required in 5.1% (6 of 117) for sternal instability or wound-healing disorders, reintubation occurred in 2.6% (3 of 117) and postoperative stroke was found in 1 patient (0.9%). 9 We had no conversions to FS, in keeping with the 0% to 0.8% reported conversion rate in 2 studies, affirming that excellent exposure of the ascending aorta and aortic valve can be achieved without compromising safety, while also avoiding superfluous visualization of the right ventricle.5,9 Moreover, UHS is easily converted to FS if the need arises. One UHS patient (2%) had reoperation for bleeding, whereas 2 FS patients (4%) underwent reoperation for bleeding (P > 0.99). The Haunschild group published a 5.9% (7 of 117) versus 16.2% (19 of 117) revision rate for bleeding and pericardial effusion after UHS and FS, respectively, while the Staromlyński group demonstrated a 7% (3 of 44) incidence for cardiac tamponade and/or rethoracotomy for bleeding after UHS.5,9 Techniques we used to avoid bleeding in both groups included liberal use of Teflon felt for construction of the anastomoses and pledgeted sutures to buttress the posterior wall of the distal anastomosis. Although UHS confers less trauma and possible bleeding, Levack and others found no particular differences in the administration of perioperative blood transfusions between UHS and FS. 4 The Staromlyński group found a mean intraoperative transfusion rate of 1.03 U for red blood cells, 1.92 U for fresh frozen plasma, and 0.60 U for platelets after UHS. 5 In comparison, our total median blood product use was 0 U for all blood products.
The CBP (median 150 min) and ACC (median 121 min) times were nearly consistent with that of Staromlyński et al. (mean CBP 161.4 min; mean ACC 108.3 min) and longer than that of Haunschild et al. (median CBP 89 min; median ACC 65 min) in UHS patients.5,9 During UHS, our CBP and ACC times were shorter by 14.5 min (P = 0.08) and 10 min (P = 0.05), respectively, compared with FS. These differences could be due to tackling the learning curve with the cumulative experience of performing >200 FS scAAR and AVR and isolated UHS AVR procedures before embarking on the first UHS scAAR and AVR and the simultaneous application of 2 facilitating technologies in all UHS cases. Specifically, COR-KNOT may save CBP and ACC times, and Custodiol-HTK and del Nido solutions offer up to 90 min of safe myocardial ischemic arrest with a single dose.2,14 Cold blood potassium-based cardioplegia must be repeated every 15 to 20 min, and manually hand-tied knots are cumbersome in a limited working space, which interrupts the surgical workflow and fluidity.2,14 Moreover, single-dose antegrade cardioplegia obviates the need for a retrograde coronary sinus catheter, which is more challenging to place in the deep and narrow space of a UHS. The UHS patients spent less time on the ventilator (median 7 h vs 17 h, P = 0.005), possibly related to preserved respiratory mechanics. Comparably, median ventilation time was 8.2 h after UHS in the Haunschild et al. study. 9 Our UHS cohort also had a 1-day shorter median hospital LOS (P = 0.05), perhaps from faster convalescence. These time savings encourage the development of a perioperative fast-track pathway after UHS proximal aortic surgery akin to that established after minimally invasive CABG and AVR.15,16 Haunschild et al. instituted a fast-track protocol after elective Bentall procedures and documented shorter ventilation times and ICU LOS without an increase in perioperative complications. 17
The ideal UHS candidate is an otherwise healthy young or middle-aged patient with no major concomitant cardiac disease and few comorbidities. These patients take maximum benefit from less trauma and sternal spreading, especially those with connective tissue disease, decreased dissection and subsequent adhesion formation if reoperation is needed, faster convalescence, and superior cosmesis. For surgeons interested in performing less invasive proximal aortic surgery, they should first perform a high-volume of FS aortic operations and isolated UHS AVRs.2,7,8 By using a stepwise UHS progression from AVR to scAAR to scAAR and AVR, surgeons can achieve proficiency in a reproducible manner. 18 Only after gaining sufficient experience should surgeons seek to perform more complex aortic operations. Notwithstanding surgeon experience, patients with acute type A aortic dissection, those requiring concomitant CABG or total arch replacement with hypothermic circulatory arrest, or those with prior CABG and patent bypass grafts crossing the midline are not UHS candidates considering the need for greater operative exposure and technicality as well as meticulous inspection of the coronary ostia in acute type A aortic dissection. Patients with infective endocarditis complicated by an aortic root abscess or who require reoperations, concomitant mitral valve surgery, or open distal hemiarch anastomosis may be UHS candidates for experienced surgeons.
Strengths and Limitations
The strengths of this study are that it is derived from a large, prospectively collected database of a single surgeon whose inclusion of patients, surgical technique, and postoperative management were the same for each procedure and stable over time. This eliminates inter-operator bias and minimizes possible surgeon-driven confounders.
This study had some limitations. We matched patients on 6 preoperative characteristics; propensity score matching was not employed because this would have resulted in extremely small sample sizes. All cases were performed by a surgeon with >25 years of experience in aortic surgery and >10 years of experience with UHS across several institutions, which limits the generalizability of findings. Inevitable advances in surgical techniques and perioperative care over the study period may have influenced outcomes in the contemporary group of patients. Moreover, because surgical procedure was not randomized, unmeasured confounding cannot be ruled out. Late outcomes (i.e., freedom from reoperation and valve-related complications, follow-up echocardiographic data, and survival) were not reported due to incomplete follow-up. That said, this study was not designed to assess late outcomes. Because of a relatively small sample size, these hypothesis-generating results require further confirmation in a large, randomized controlled trial, which would provide increased power to detect potential differences between groups.
Conclusions
The low morbidity and mortality support the application of UHS for scAAR and AVR in select patients and experienced hands.
Supplemental Material
sj-pdf-1-inv-10.1177_15569845231213074 – Supplemental material for Upper Hemisternotomy Versus Full Sternotomy for Replacement of the Supracoronary Ascending Aorta and Aortic Valve
Supplemental material, sj-pdf-1-inv-10.1177_15569845231213074 for Upper Hemisternotomy Versus Full Sternotomy for Replacement of the Supracoronary Ascending Aorta and Aortic Valve by Vishal N. Shah, Jose Binongo, Jane Wei, Brian M. Till, Colin King, Jacqueline McGee and Konstadinos A. Plestis in Innovations
Footnotes
*Presented as a poster (presentation on demand) at the AATS aortic symposium, Boston, MA, USA, May 13, 2022
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
