Abstract
Objective:
Robot-assisted minimally invasive coronary bypass surgery is one of the least invasive approaches that offers multivessel revascularization and accelerated recovery. We investigated the benefits of computed tomography angiography (CTA) guidance in robotic coronary bypass (RCAB) by analyzing perioperative outcomes.
Methods:
Between April 2022 and April 2023, 60 consecutive patients who underwent RCAB under preoperative CTA guidance were included. The intercostal space of the minithoractomy incision was determined based on the distance from the thoracotomy site to the midsection of the left anterior descending artery (LAD) on preoperative CTA. Peripheral vascular findings on preoperative CTA guided the decision for the cannulation site. Perioperative parameters and early outcomes were evaluated.
Results:
The mean age of the patients was 62.3 ± 10.5 years, and 51 patients were male (85.0%). The mean number of revascularized vessels was 2.9 ± 1.1. Left thoracotomy guided by CTA measurements was performed in the fourth intercostal space in 37 patients (61.7%) and in the third intercostal space in the remaining patients. Axillary cannulation was performed in 28 (46.7%) patients because of prohibitive findings in the iliac vessels and aorta. All target coronary arteries with an indication for bypass were revascularized with CTA-guided RCAB. The left internal mammary artery (LIMA) was anastomosed to the LAD in all patients, and the LIMA was anastomosed sequentially to the diagonal artery in 17 patients (28.3%). No operative mortality or cerebrovascular event was observed. One patient underwent reoperation due to bleeding.
Conclusions:
Robot-assisted minimally invasive multiple-vessel coronary bypass under preoperative CTA guidance is safe and can be performed with excellent results.
Keywords
Central Message
Robot-assisted minimally invasive multivessel coronary bypass under preoperative CTA guidance is safe and can be performed with excellent results despite anatomic variability among patients. Cannulation and thoracotomy sites as well as a more feasible use of the LIMA graft are influenced by CTA guidance and robotic assistance.
Introduction
With the advancement of surgical technologies, minimally invasive cardiac surgery has seen a steep rise in acceptance in recent years, as it presents many benefits to the patient with the avoidance of sternotomy, less tissue traumatization, less pain, less bleeding, and timely recovery. 1 Coronary bypass surgery techniques were quickly adapted to minimally invasive approaches and opted for by an increasing number of clinics worldwide. Ensuring access to the heart from the most favorable intercostal space (ICS), harvesting the longest possible segment of the left internal mammary artery (LIMA), and the decision to use alternative grafts are important points of consideration in minimally invasive coronary bypass. Robotic assistance adds many advantages to the minimally invasive technique. A longer portion of the LIMA can be harvested, such that the graft is more suitable for sequential anastomosis and a “T” graft configuration becomes more feasible. 2 During the harvest of the LIMA, no retractor placement or thoracic wall retraction is necessary, avoiding potential nerve or muscle injury, injury to the ribs, and pain resulting from the distension of the chest. Computed tomography (CT) guidance is a useful tool for the surgical team in preoperative planning and achieving optimal surgical outcomes. We investigated the safety and benefits of CT angiography (CTA)–guided robotic coronary bypass (RCAB) by analyzing perioperative outcomes.
Methods
Study Population
The 60 consecutive patients who underwent RCAB with preoperative CTA guidance in our clinic between April 2022 and April 2023 were included in the study. No RCAB cases were excluded. Emergent cases, patients with severe comorbidity, left pleural adhesions, or ascending aortic disease prohibiting robotic surgery did not undergo RCAB during the study period. Nine female (15.0%) and 51 male (85.0%) patients with a mean age of 62.3 ± 10.5 years made up the study cohort. The mean EuroSCORE II of the patients was 2.3 ± 1.9. The patient demographics are summarized in Table 1.
Patient Demographics.
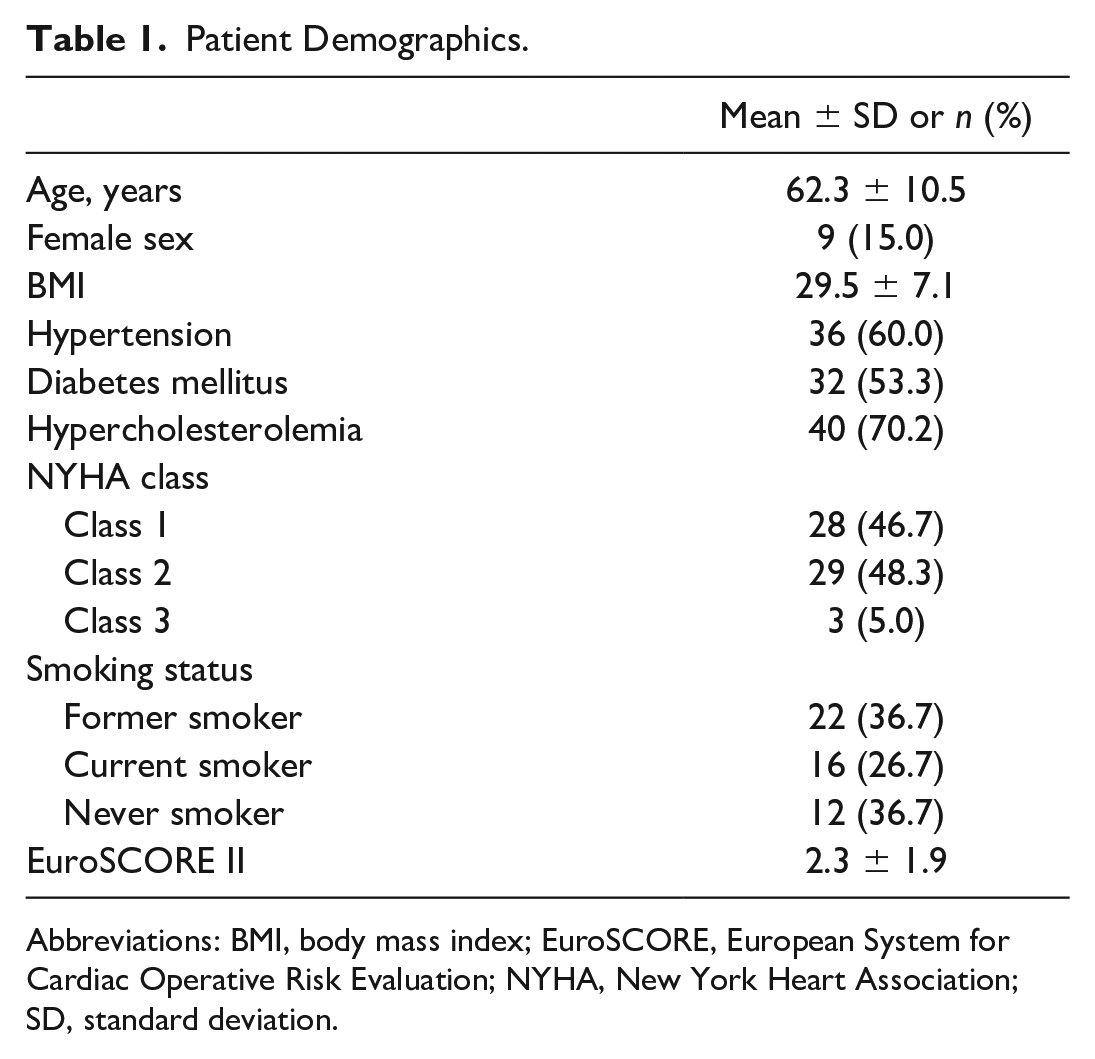
Abbreviations: BMI, body mass index; EuroSCORE, European System for Cardiac Operative Risk Evaluation; NYHA, New York Heart Association; SD, standard deviation.
CT Guidance
CTA from the neck to the thigh was obtained 5 days before the operation. Evaluations of the LIMA anatomy, angulations of the target diagonal artery from the left anterior descending artery (LAD; Fig. 1), the aorta, access site for minithoracotomy, and aortoiliac disease affecting the choice cannulation site were using these preoperative CTA scans. The preoperative CTA evaluation was made with a collaborative effort by the radiologist and the surgeon. After agreement on the findings of the preoperative CTA, the final decision on how to carry out the surgery was made by the surgeon.
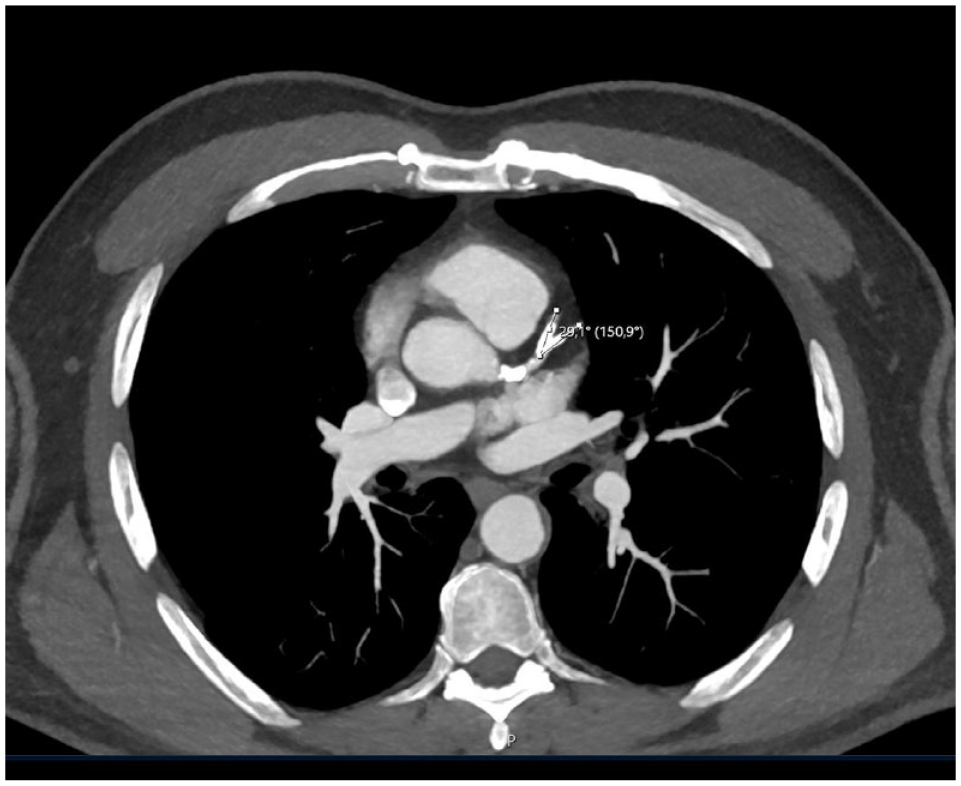
Evaluations of the angle between the target diagonal artery and the left anterior descending artery with preoperative computed tomography.
Thoracic Cavity and LIMA Measurements
A comprehensive evaluation of the entire arterial system, from the neck to the thigh, was conducted using a third-generation dual-source CT scanner (Siemens Somatom Force; Siemens Healthcare AG, Erlangen, Germany). The CTA employed a collimation of 2 × 192 × 0.6 mm, a temporal resolution of 66 ms, a spatial resolution of 0.24 mm, a generator power of 240 kW (2 × 120 kW), a maximum scan speed of 737 mm/s, and a rotation time of 0.25 s. Sublingual nitroglycerin spray was used to induce vasodilation and improve image quality, except for those patients presenting with severe aortic stenosis or hypotension. Medications to control heart rate were not introduced except for those patients who were already under a beta-blocker regimen.
The CTA was ECG gated and performed in a retrospective spiral mode. This comprehensive and detailed imaging protocol allowed for an in-depth examination of the arterial system. The images were evaluated by a senior radiologist experienced in cardiovascular imaging, using Sectra IDS7 version 23.2 software (Sectra AB, Linköping, Sweden). The decision for the cannulation site was made taking into consideration any occlusions, thrombi, or calcifications in the iliofemoral vessels or the aorta (Fig. 2). The atherosclerosis of the ascending aorta was evaluated to decide on the proximal aortic anastomosis strategy and the choice of grafts.
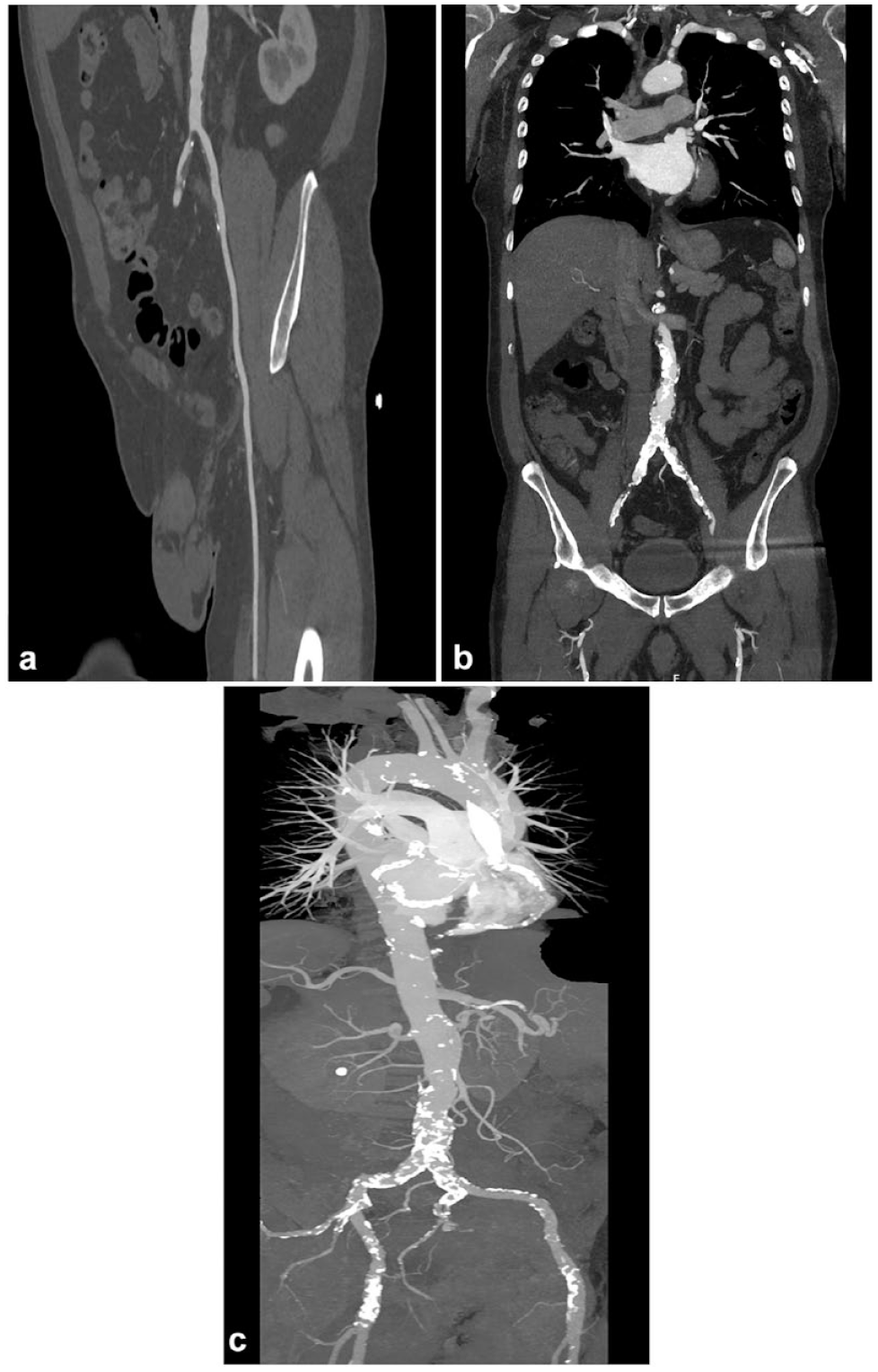
Evaluation of the aorta and the iliofemoral vessels on preoperative computed tomography. (a) Thrombi, (b) occlusive disease, or (c) severe calcifications prohibit femoral cannulation and axillary cannulation is opted for these patients.
In 3-dimensional reconstructions, measurements of the LIMA length were made from its origin to the fourth ICS and to the level of its bifurcation using the straightened view of the artery (Fig. 3). The distances of the sinotubular junction and the midportion of the LAD to the third and fourth ICSs were measured for each patient (Fig. 4). The upper margin of the fourth rib was used for the distance from the third ICS, and the upper margin of the fifth rib was used for the distance from the fourth ICS. The angle between the target diagonal artery and the LAD was also measured.
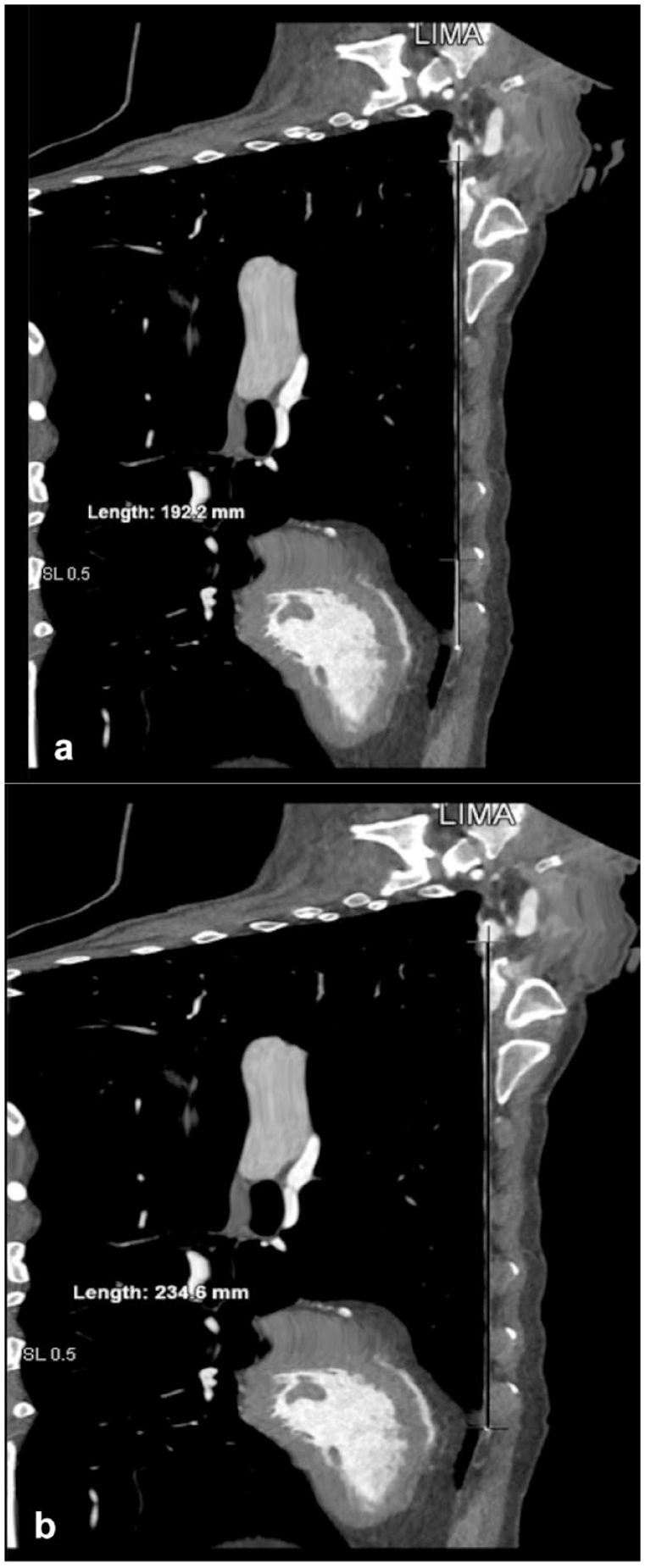
Measurements of the LIMA length (a) from its origin to the fourth intercostal space and (b) to the level of its bifurcation using the straightened view of the artery in 3-dimensional reconstructions. LIMA, left internal mammary artery.
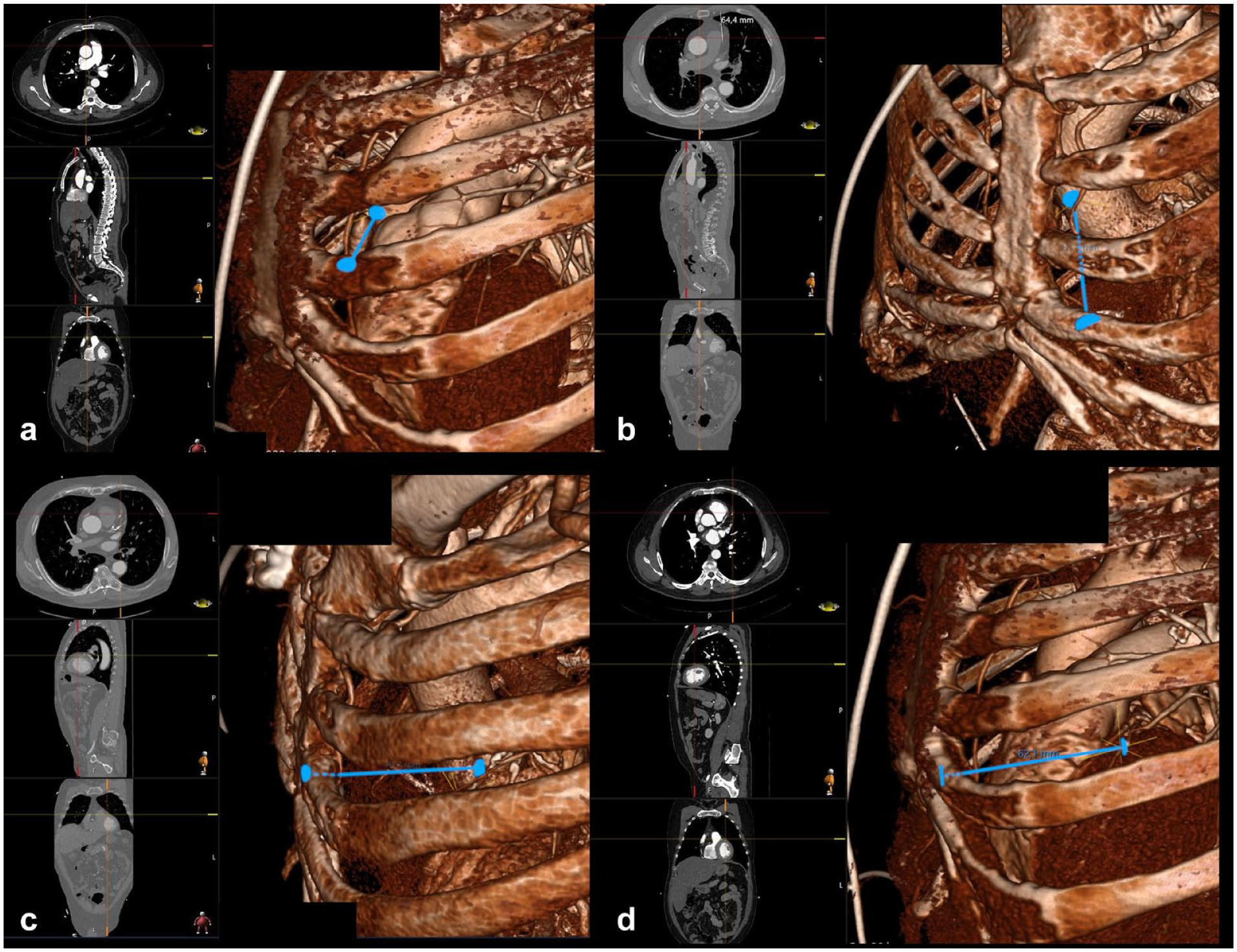
The distances of (a) the sinotubular junction to the third intercostal space, (b) the sinotubular junction to the fourth intercostal space, (c) the midportion of the LAD to the third intercostal space, and (d) the midportion of the LAD to the fourth intercostal space measured in 3-dimensional reconstructions of computed tomography images shown with blue lines. LAD, left anterior descending artery.
Surgical Technique
After induction of general anesthesia, the patient was positioned for robotic setup. A transesophageal echocardiography (TEE) probe was placed, and a double-lumen intubation was performed in all patients. The right internal jugular vein was cannulated with activated clotting time targeted (ACT) at 200. Robotic trocar docking sites and thoracotomy sites were marked on the patient’s skin as planned during the preoperative evaluation with CTA (Fig. 5). After surgical draping, the robotic trocars were positioned. The right robotic trocar was placed through the second ICS, which was later used for the insertion of the Chitwood clamp; the left robotic trocar was placed from the sixth or seventh ICS; and the camera was placed through the fourth ICS lateral to the nipple line. After placement of the trocars, robotic docking was completed. According to the LIMA topography visualized in the preoperative CTA, the LIMA was harvested with careful attention to specific variations in its course (Fig. 6). The endothoracic fascia was dissected anterior and posterior to the LIMA until the level of LIMA bifurcation was reached. Additional heparin was administrated with a target ACT above 250 after dissection of the entire length of the LIMA was completed. The LIMA was clipped and transected distally to the bifurcation site (Fig. 7). After completion of LIMA harvesting, bleeding control of the thoracic wall was performed with robotic assistance, and the robotic trocars are undocked subsequently. At this stage, peripheral cannulation, either axillary or femoral as planned with preoperative CTA findings, was performed under TEE guidance. Simultaneously with peripheral cannulation, the other surgeon performed the thoracotomy incision. The Babliak minithoracotomy retractor was placed. Cardiopulmonary bypass (CPB) was initiated, and the heart was decompressed before opening the pericardium. The pericardium was incised, the pulmonary artery and the aorta were separated from each other, and a Dacron tape was placed around the aorta, using the Babliak technique previously described. 3 The root cannula for cardioplegia was placed, and the Chitwood clamp was inserted through the second ICS, the previous site of the right robotic trocar. The heart was arrested with the delivery of cardioplegia. The left pulmonary veins and the inferior vena cava were encircled with tape to achieve distal exposure to the right side. Distal anastomoses were then performed. The LIMA graft was used for the LAD in all patients, and a sequential anastomosis to the diagonal artery was performed if the angle between the LIMA and diagonal artery was smaller than 40° in the preoperative CTA evaluation. When possible, the radial artery—and the saphenous vein otherwise—were used as additional grafts if needed. All of these additional grafts were harvested endoscopically. The cross clamp was removed after completion of the distal anastomoses, and a side-biting clamp was placed on the aorta for the proximal anastomoses (Fig. 8). Alternatively, the proximal anastomoses were done onto the LIMA. In 2-vessel bypass cases in which the LIMA graft was used sequentially to the LAD and the diagonal artery, no proximal anastomoses were required. Transit-time flow measurements were carried out after the completion of all anastomoses to ensure adequacy and quality of revascularization. The patient was weaned off CPB when optimal conditions were achieved. A chest tube was placed through the access site of the left robotic trocar, and the other incisions were sutured. A temporary pacemaker lead was implanted only when necessary.
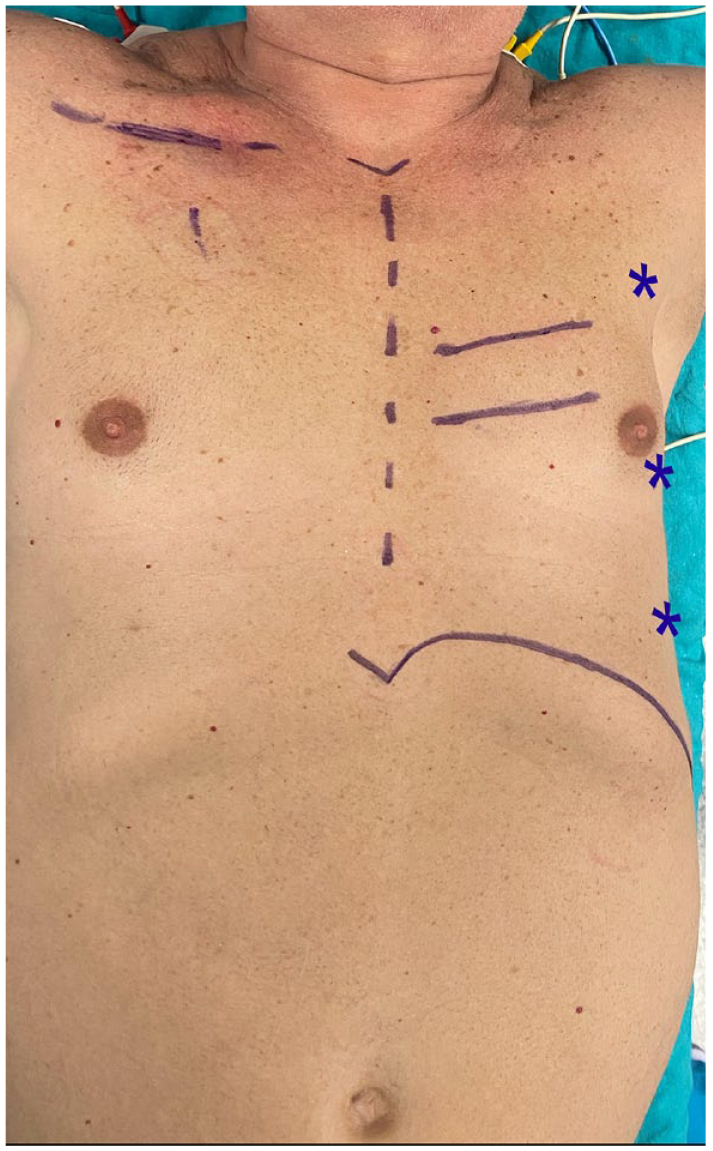
Robotic trocar docking sites (marked with stars) and thoracotomy sites (marked with lines) are marked on the patient’s skin as planned during the preoperative evaluation.
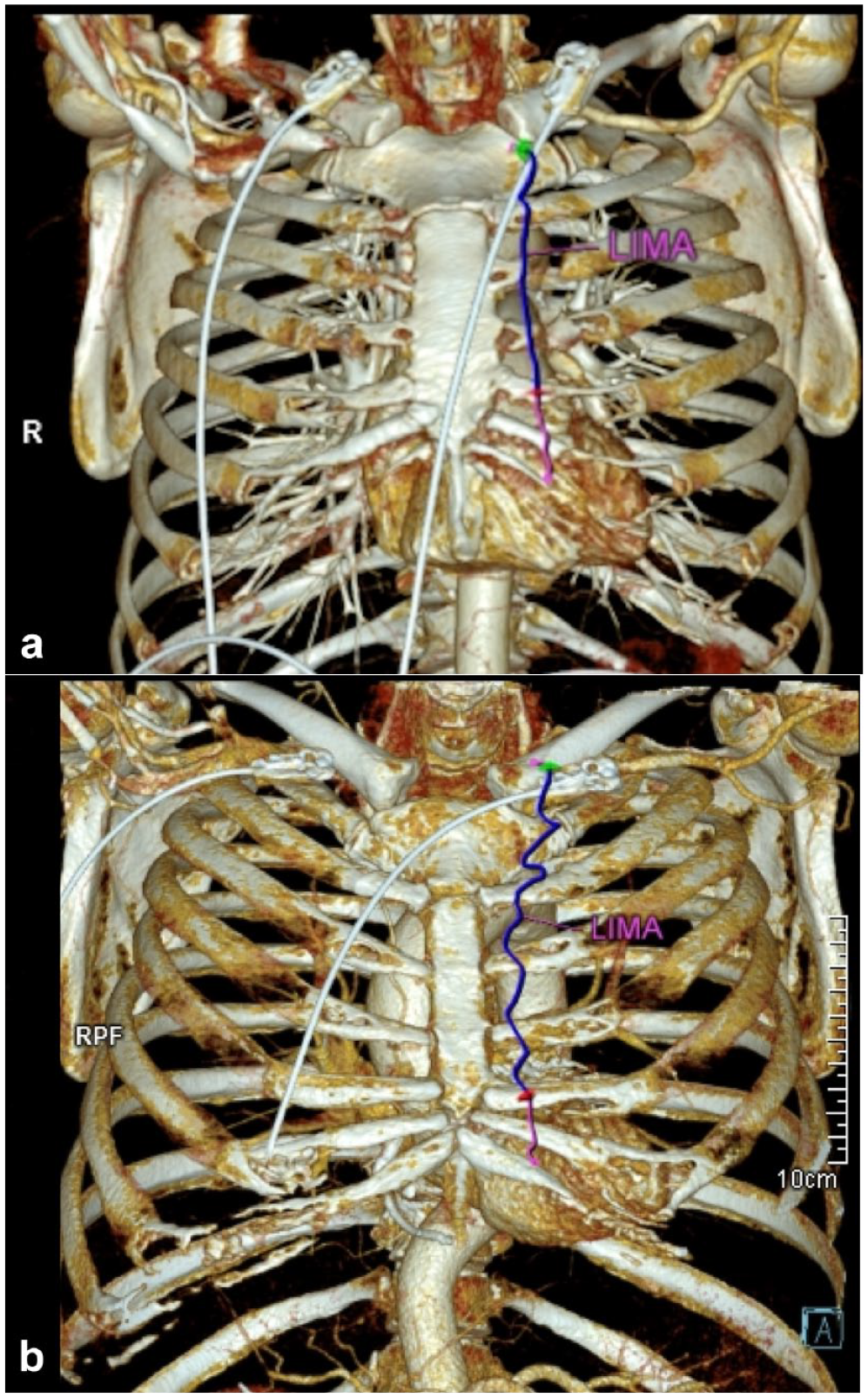
The surgical team can be aware of a (a) linear or (b) tortuous course of the LIMA when the course of the artery is delineated on computed tomography images. The lengths of the arteries harvestable by direct vision are shown in blue, and additional lengths harvestable with robotic assistance are shown in magenta. LIMA, left internal mammary artery.
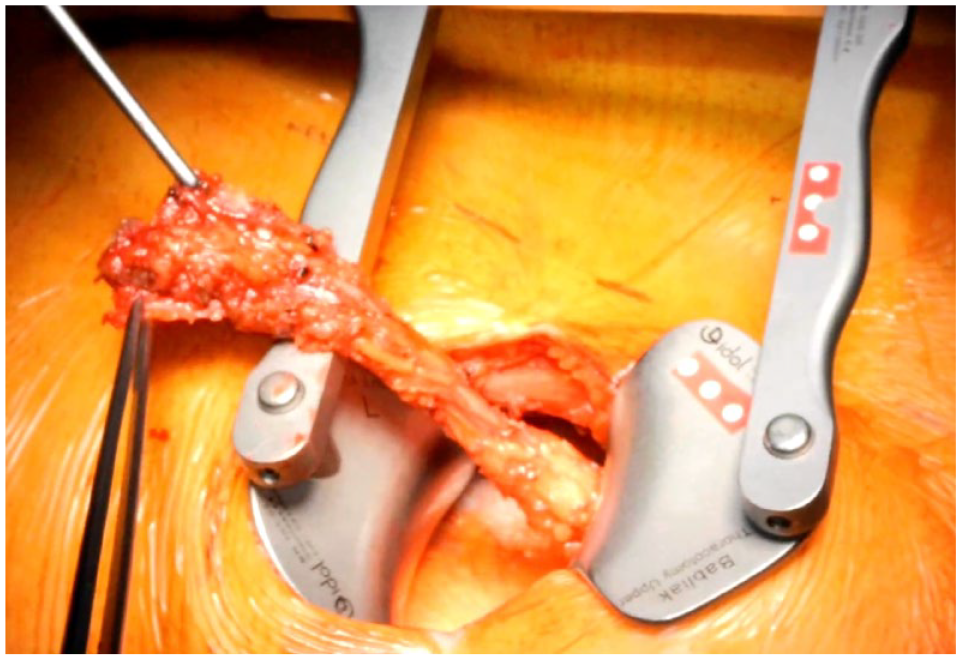
Operative view of the left internal mammary artery harvested to its bifurcation, beyond the intercostal space of the thoracotomy incision.
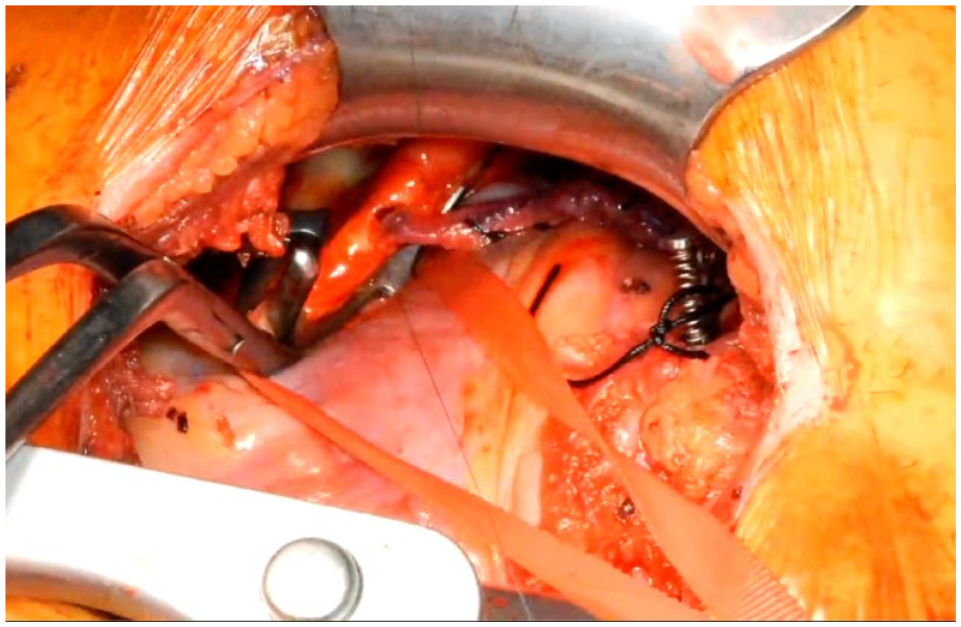
If proximal anastomoses need to be made onto the aorta, then a side-biting clamp can be placed through the thoracotomy incision.
Ethical Statement
The study was conducted in accordance with the Declaration of Helsinki. Patient data for this study were retrieved from the institutional database retrospectively. Approval from Acıbadem Maslak Hospital Institutional Review Board was obtained before establishing the study.
Statistical Analysis
IBM SPSS Statistics, Version 25.0 (IBM Corp., Armonk, NY, USA) and RStudio (Posit, Boston, MA, USA) with ggplot package were used for statistical analysis. Continuous parameters are given as mean and standard deviation, while categorical parameters are given as numbers and percentages. The Shapiro–Wilk test was used to assess the normal distribution of parameters. The Student’s t test was used for comparisons between 2 groups of normally distributed quantitative variables, and the Mann–Whitney U test was used for comparisons between 2 groups of non-normally distributed quantitative variables. Receiver operating characteristic (ROC) analysis was carried out to determine a cutoff for the angulature between the target diagonal artery and the LAD artery in decision making for sequential anastomosis. Density plots were charted using the R ggplot package.
Results
The median CPB and cross-clamp times were 152.7 ± 44.0 min and 74.2 ± 21.4 min, respectively. In the CTA evaluation of the aorta for peripheral cannulation, 28 patients (46.7%) presented with an aortic pathology, and the cannulation site of choice was axillary in these patients. Femoral cannulation was performed in the remaining 32 patients (53.3%). Of the patients with aortic pathologies, 57% had severe calcification, 13% had thrombi, and 30% had both. Based on CTA measurements, the left thoracotomy was performed in the fourth ICS in 37 patients (61.7 %) and in the third ICS for the remaining patients. All of the target coronary arteries with an indication for bypass were revascularized with CTA-guided RCAB. The mean number of revascularized vessels was 2.9 ± 1.1. The LIMA was anastomosed to the LAD in all patients. As a second graft for the non-LIMA grafts, the radial artery and saphenous vein were preferred. In young patients and left-sided vessel anastomoses, we preferred radial artery when possible, and for the rest of the anastomosis and when radial artery use was not feasible, saphenous vein grafts were used. The diagonal artery was revascularized in 28 patients (46.7%), and the LIMA was anastomosed sequentially to the diagonal artery in 17 of these patients (28.3%). The ROC curve for the LAD-diagonal artery angle and sequential use of the LIMA graft showed that the LIMA was less likely to be used for sequential anastomosis to the diagonal artery when the angle was greater than 40°, and a separate graft was more frequently harvested in these cases (Fig. 9). A radial artery graft was used in 15 patients (25.0%). Proximal anastomoses were done onto the aorta in 39 patients (65.0%) and onto the LIMA in 11 patients (18.3%). For the remaining patients, no proximal anastomoses were necessary. The perioperative data are presented in Table 2.
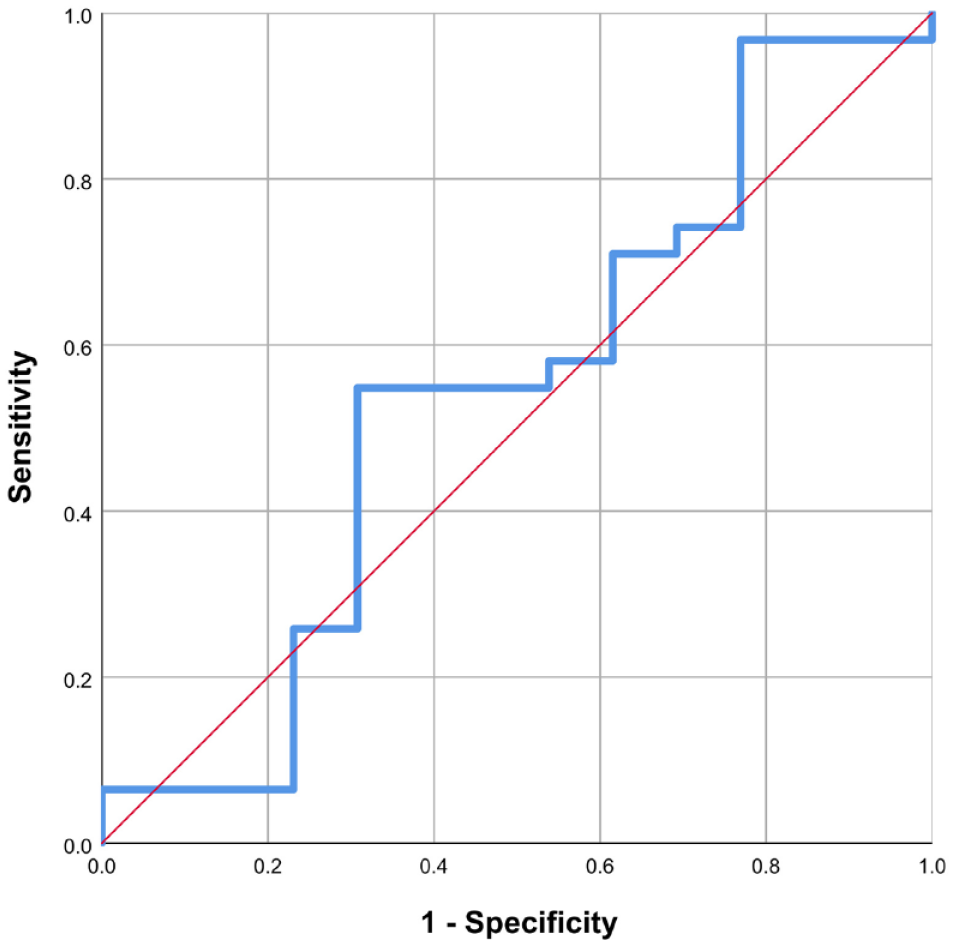
The ROC curve constructed for the angle between the target diagonal artery and the left anterior descending artery. The left internal mammary artery was less likely to be used as a sequential graft when the angle was greater than 40° with a sensitivity of 51.6% and a specificity of 69.2%.
Operative Data of RCAB Patients.

Abbreviations: CPB, cardiopulmonary bypass; ICS, intercostal space; LIMA, left internal mammary artery; RCAB, robotic coronary bypass; SD, standard deviation.
According to the CTA measurements, the mean LIMA length that could be harvested with robotic assistance was longer (22 ± 2.5 cm vs 19 ± 2 cm, P = 0.001) than the mean length possible with direct vision through the fourth ICS (Fig. 10a). The mean distance from the third ICS to the LAD middle portion was 5.2 ± 1.2 cm, and the mean distance from the fourth ICS to the LAD middle portion was 5.6 ± 1.1 cm (P = 0.079; Fig. 10b). The mean distance from the third ICS to the aortic sinotubular junction was shorter (7.1 ± 1.1 cm vs 8.1 ± 1.3 cm, P < 0.001) than the mean distance from the fourth ICS to the aortic sinotubular junction (Fig. 10c). The mean intubation time was 4.7 ± 2.9 h, the mean intensive care unit (ICU) stay was 20.8 ± 9.6 h, and the mean hospital stay was 7.8 ± 2.6 days. The mean chest tube output in the ICU was 538.5 ± 504.6 mL, and 1 patient underwent reoperation due to bleeding. Eight patients (13.3%) received packed red blood cell transfusions. No operative mortality or cerebrovascular event was observed among the study patients (Table 3).
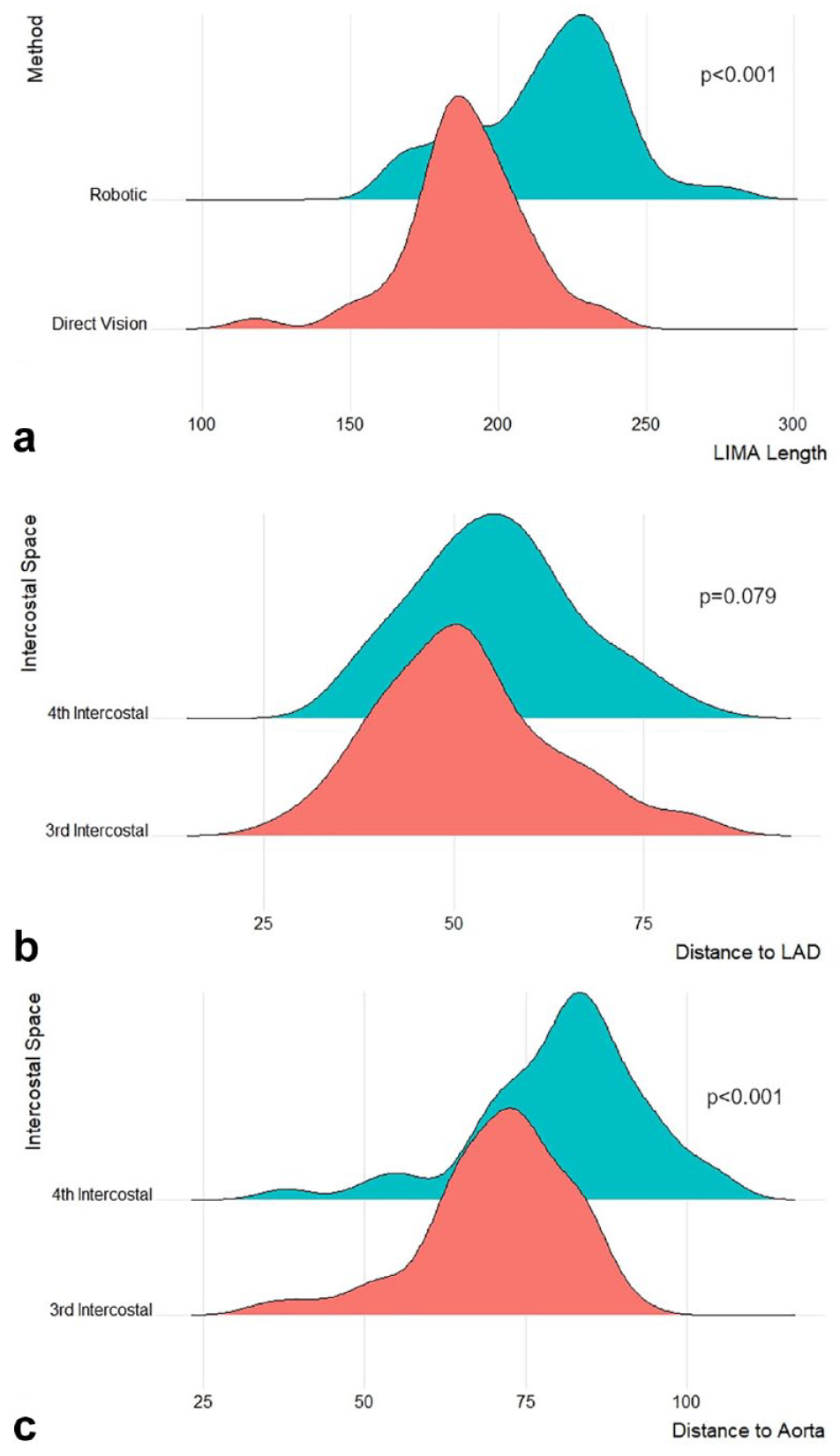
Density plots for (a) estimated length of the LIMA available for harvest with robotic assistance and estimated length of the artery available for harvest by direct vision based on computed tomography measurements, (b) distance to the midportion of the LAD from the third and fourth intercostal spaces, and (c) distance to the sinotubular junction of the aorta from the third and fourth intercostal spaces. LAD, left anterior descending artery; LIMA, left internal mammary artery.
Clinical Outcomes.
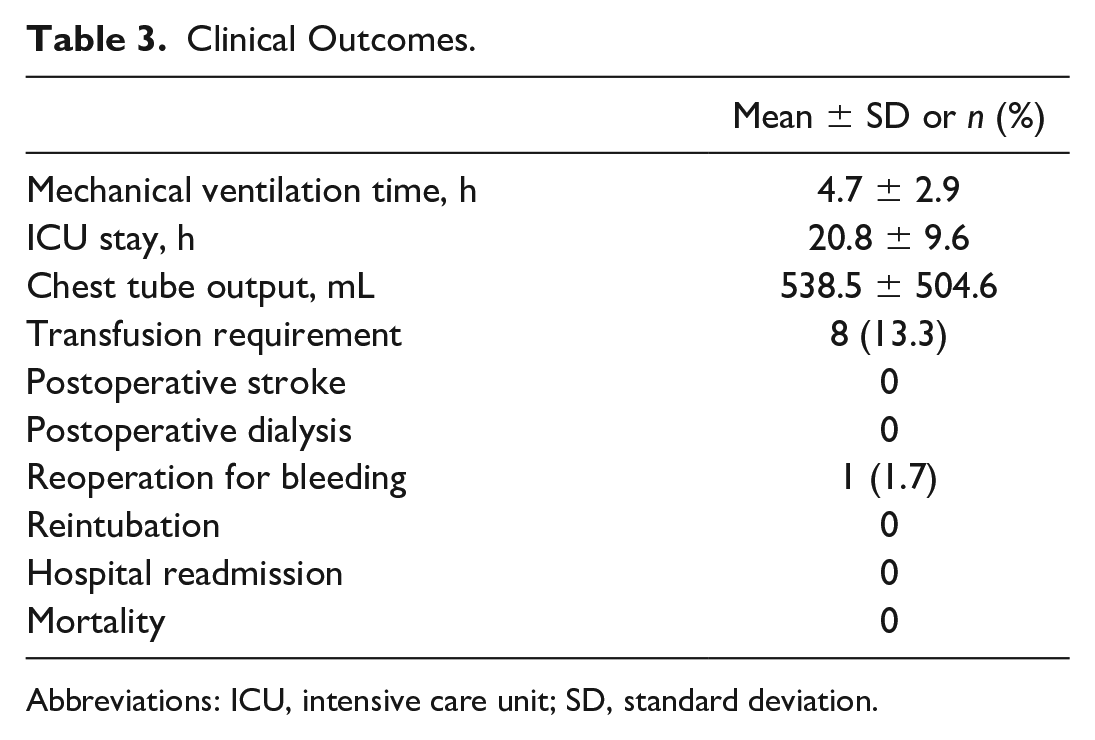
Abbreviations: ICU, intensive care unit; SD, standard deviation.
Discussion
Minimally Invasive and Robot-Assisted Coronary Bypass
With the introduction of minimally invasive techniques to cardiac surgery, better operative outcomes in terms of bleeding, postoperative pain, patient mobility, and better cosmetic results with minimal scars have been reported. 4 Robotic assistance in cardiac surgery has further advantages over other minimally invasive approaches. In the case of multivessel coronary bypass, robot-assisted LIMA harvest results in less tissue traumatization without the need for thoracic wall retraction, the harvest of a longer LIMA graft, and a higher potential for sequential LIMA use. 5
Our short-term postoperative results support the use of robotic assistance in minimally invasive cardiac surgery, with no mortality, infection, postoperative stroke, need for dialysis, reintubation, or hospital readmission observed in our cohort. Only 1 patient was reoperated due to intercostal venous bleeding. In addition to the advantages of RCAB, preoperative CTA planning can help achieve excellent results with detailed knowledge of the patient’s anatomic topography of the heart, the LIMA, and the optimal thoracotomy access points.
LIMA Harvest and Decision on Thoracotomy Access Site
In a study by Itagaki et al. in which the options for LIMA harvest in minimal access coronary surgery such as minimally invasive direct coronary artery bypass (MIDCAB), thoracoscopic, and robotic harvest were compared, the robotic option was shown to be superior in harvested LIMA length, providing a less difficult harvesting process for the surgeon, and less pain and better cosmetic results in the postoperative period for the patient. 6 Robotic harvest of the LIMA was shown to be effective and advantageous in 2 large case series in studies by Srivastava et al. 7 and Balkhy et al. 8 In minimally invasive coronary bypass surgery, access is usually gained from the fourth ICS. 9 In another study by Rams et al., in which the LIMA topography in MIDCAB patients was retrospectively analyzed by CTA, the fourth ICS was favored for minimally invasive bypass surgery. 10 As the chest wall distal to the incision cannot be visualized during MIDCAB, it is generally preferred to make the incision from a lower ICS for the ability to harvest a longer LIMA. While this is a simple solution, it also brings about the problem of a long distance between the access site and the proximal aorta whilst performing proximal anastomoses, cannulation, or clamping. With robotic LIMA harvesting, the LIMA can be harvested to its whole length, independent of which ICS the access is achieved from. The decision of the ICS to perform the minithoracotomy was made based on the distance to the midsection of the LAD from the skin using preoperative CTA. The third ICS would be a potentially feasible and practical access site in this condition as aortic manipulation, proximal anastomoses, and placement of the cardioplegia cannula and the cross-clamps are among the potentially troublesome aspects of the minimally invasive technique. Among the 60 cases that were operated on, sequential LIMA-diagonal artery bypass was performed for 17 patients. This was possible due to the robotic harvest of the LIMA to its full length. Robotic LIMA harvesting enables the surgeon to use the artery more often sequentially for multiple-vessel revascularization. Many robotic minimally invasive cardiac surgeons tend to choose the thoracotomy access site according to the robotic camera port site, which is the fourth ICS. But preoperative planning with CTA and the decision regarding the thoracotomy site accordingly present many advantages. In the preoperative CTA, the closest ICS to the site of the proximal anastomosis can be determined. The distal anastomosis can be easily made from both the third and fourth ICSs. The distance of the proximal anastomosis site to the thoracotomy access site is the main factor in our decision for which ICS to employ, with the aim of performing both the distal and proximal anastomoses in a safe and comfortable manner. The final decision is made by the surgeon, taking all parameters into consideration. In our practice, we started to opt more commonly for the third ICS as an access site due to the previously mentioned reasons. In total, 23 patients (38.3%) in this study were operated through the third ICS, and most of the patients operated through the third ICS were our more recent patients, as our familiarity with robotic LIMA harvest increased and we experienced easier exposure of the aorta through the third ICS. However, we refrain from recommending the routine use of the third ICS for RCAB. The most practical rule that we established for the thoracotomy site is to choose the ICS that is closest to the right ventricle outflow tract, thereby allowing easier manipulation of the aorta for proximal anastomosis and root cannulation.
Topographic Anatomy of the LIMA
The topographic anatomy and the course of the LIMA are visualized preoperatively with CTA imaging. The LIMA course in the thoracic wall varies anatomically between patients, and when an abnormal course is observed in the CTA during preoperative mapping, this is taken into consideration during the planning stage. The LIMA is then harvested more cautiously, which decreases the risk of injury to the vessel as it is not always visible to the naked eye due to a dense fatty tissue while opening the fascia before harvesting. 11 The use of CTA may be safer than other methods for evaluating the course of the LIMA, as reported by Gasparovic et al., who reported that the preoperative CTA was superior in providing a more accurate assessment of the mediastinal structures compared with chest X-ray or conventional angiography, evidenced by reduced rates of complications such as injury to the LIMA during the harvest. 12
Measurement of the Angle Between the LAD and Diagonal Artery
The angle between the LAD and the diagonal artery was found to be an important factor in the decision to perform a sequential anastomosis with the LIMA. An ROC curve was constructed to investigate the relationship between the LAD-diagonal angle and the sequential use of the LIMA graft. The ROC curve analysis revealed a cutoff of 40° for the likelihood of using a separate graft for the diagonal artery. At angles greater than 40°, the LIMA graft was less often used for sequential anastomosis, and an additional radial artery or saphenous vein graft was more frequently needed. With greater angulature, the sequential use of the LIMA graft is less feasible, and preoperative understanding of the LAD-diagonal angle allows the surgical team to make the decision to use additional grafts such as the radial artery or the saphenous vein beforehand, so that the preparation can be completed accordingly and the cross-clamp times are not increased during this decision process.
Decision of the Cannulation Strategy
With the preoperative evaluation of CTA images, we not only decided on the thoracotomy site but also obtained valuable information that guided the cannulation strategy. We assess the complete thoracic and abdominal aorta and the iliofemoral arteries for peripheral arterial disease, calcifications, or thrombotic lesions. If any extensive atherosclerotic plaques or thrombi at any level of the aorta or the iliofemoral vessels were observed, axillary artery cannulation via surgical exposure was preferred. This CTA guidance has contributed to the minimalization of cerebrovascular or peripheral vascular complications during RCAB.
Conclusions
Robot-assisted minimally invasive multiple-vessel coronary bypass under preoperative CTA guidance is safe and can be performed with excellent results. CTA guidance enables the surgeon to evaluate the LIMA topography and the position of the patient’s heart and influences the choice of cannulation site and the preparation of additional grafts. Despite anatomic variability among patients, safe outcomes and ideal surgical exposure can be achieved with CTA-guided RCAB.
Footnotes
*This study was presented at the 2023 ISMICS Annual Scientific Meeting in Boston, MA, USA, as an oral presentation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
