Abstract
Objective:
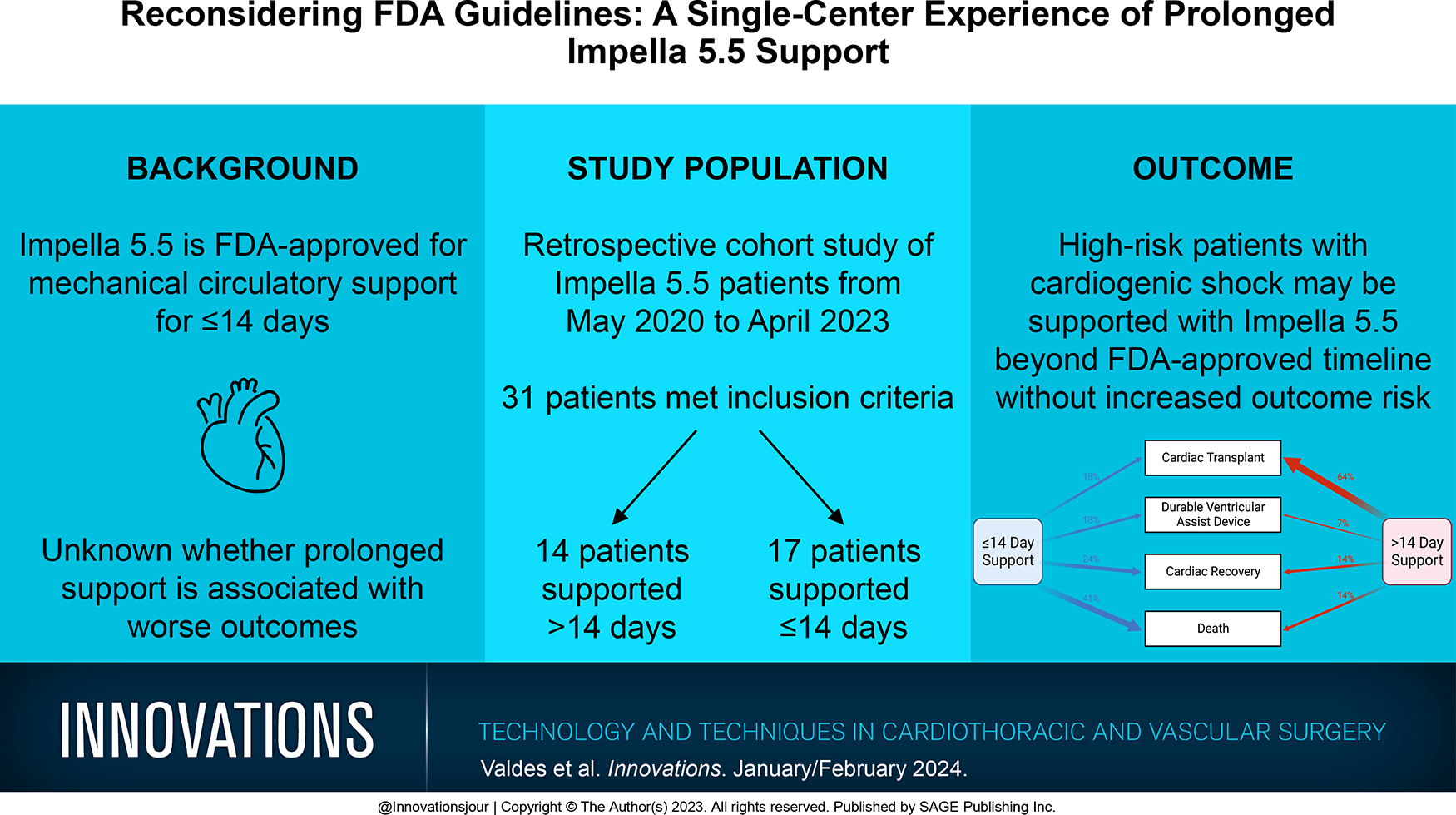
Impella 5.5 (Abiomed, Danvers, MA, USA) is approved by the US Food and Drug Administration (FDA) for mechanical circulatory support for ≤14 days. It is unknown whether prolonged support is associated with worse outcomes. We sought to review our single-center experience with Impella 5.5 and compare outcomes based on support duration.
Methods:
We retrospectively reviewed adult patients (≥18 years old) supported with Impella 5.5 at our institution (May 2020 to April 2023). Patients on prolonged support (>14 days) were compared with those supported for ≤14 days.
Results:
There were 31 patients supported with Impella 5.5 including 14 (45.2%) supported >14 days. Median support duration for those on prolonged support was 43.5 (interquartile range [IQR] 25 to 63.5) days versus 8 (IQR 6, 13) days for those who were not (P < 0.001). Overall, the device-related complication rate was 9.7% and did not differ between groups (P = 0.08). Overall, 30-day postimplant survival was 71% and did not differ by support duration (P = 0.2). In-hospital mortality was 32% and did not differ between cohorts (P > 0.99). Among those surviving to explant (n = 22), long-term strategy included bridge to durable ventricular assist device (18%, n = 4), cardiac transplant (55%, n = 12), and cardiac recovery (27%, n = 6).
Conclusions:
High-risk patients with cardiogenic shock may be supported with Impella 5.5 beyond the FDA-approved duration without increased risk of complications or mortality.
Keywords
Central Message
The use of Impella 5.5 beyond the 14-day FDA-approved timeline was not associated with increased risk of complications or mortality in high-risk patients with cardiogenic shock. Future studies with larger and more representative samples are needed.
Introduction
Temporary mechanical circulatory support (tMCS) devices are used in patients with refractory cardiogenic shock to serve as a bridge to implantation of long-term MCS devices, heart transplantation, heart recovery, or to provide circulatory support in the setting of a high-risk surgical or procedural intervention.1–3 The Impella 5.5 (Abiomed, Danvers, MA, USA) is one form of tMCS that functions to unload the left ventricle as blood is ejected into the ascending aorta via a catheter-mounted microaxial impeller lying across the aortic valve.2,4 Minimally invasive devices, such as Impella, show an advantageous safety profile compared with conventional, open tMCS approaches. 5 Minimally invasive tMCS is associated with easier implantation, lower risks of bleeding, lower perioperative mortality, elimination of sternotomy, and preservation of the pericardium.1,5,6
Although Impella offers a rapid and minimally invasive option for temporary MCS, the maximum duration of US Food and Drug Administration (FDA)–approved support with Impella 5.0, LD, and 5.5 is 14 days. There is concern that with longer support, there is an increased risk for device malfunction, thromboembolic events, hemolysis, and thrombocytopenia.7,8 In a study comparing short-term (<10 days) versus long-term (>10 days) support of Impella CP, 5.0, and RP, Zeschky and colleagues found no significant difference in major complications or survival, suggesting a potential role for prolonged support. 9 Rock and colleagues reported similar findings and reduced evidence of end-organ damage in a subgroup of patients with prolonged Impella 5.5 support greater than 14 days when compared with the entire cohort of patients supported with Impella 5.5. 10
However, there is a paucity of data surrounding prolonged Impella 5.5 use beyond 14 days.7,8,11,12 The purpose of this study is to examine Impella support among patients supported at our institution to evaluate the safety and efficacy of prolonged support >14 days compared with support ≤14 days.
Methods
Study Design and Population
Institutional review board approval was obtained for this single-center study evaluating prolonged Impella support beyond 14 days in adult patients (≥18 years of age) and the association between duration of support and outcomes (protocol No. IRB202202335). Inclusion criteria included patients supported with and separated from the Impella 5.5 device implanted by a single surgeon between May 1, 2020, and April 25, 2023. Exclusion criteria included patients <18 years old, those not supported with an Impella 5.5, and those still on Impella 5.5 support at the end of the study period.
Data Collection
Data were collected retrospectively from the electronic medical record. Patients were categorized according to Impella support duration—those supported for ≤14 days constituted the “short-term” cohort and those supported for >14 days constituted the “prolonged support” cohort. Demographic and baseline data collected included age, sex, body mass index (BMI), medical comorbidities, preoperative heart function, type of heart disease, and inotropic therapy dependence.
Risk of in-hospital mortality was estimated using the European System for Cardiac Operative Risk Evaluation (EuroSCORE) II. 13 EuroSCORE II uses age, sex, preexisting conditions, cardiac-specific parameters, and procedural factors to calculate a score associated with an estimated risk of in-hospital mortality after major cardiac surgery. We also used the Model for End-Stage Liver Disease (MELD) Score, which takes into account dialysis use and laboratory factors (creatinine, bilirubin, international normalized ratio [INR], and sodium) to stratify risk of transplant. 14
Impella 5.5 support details included indication for placement, surgical technique (axillary vs central placement), support duration, ambulation status while on Impella, use of concomitant support, and type of anticoagulation. Hemodynamic and laboratory values were also collected preoperatively and at the time of Impella explant, as well as on postimplant day (POD) 1 and POD5. Hemodynamic values included central venous pressure (CVP), systolic pulmonary artery pressure, diastolic pulmonary artery pressure (dPAP), pulmonary capillary wedge pressure (PCWP), cardiac index, and left ventricular ejection fraction. Laboratory values included creatinine, hemoglobin, platelets, INR, and albumin.
Primary and Secondary Outcomes
The primary outcomes were in-hospital and 30-day survival. Secondary outcomes included complications, hospital length of stay, disposition after Impella support, need for device repositioning, and hemodynamic changes. Complications were defined as any event that required significant medical or surgical intervention.
Statistical Analysis
Patient demographics and comorbidities were summarized and compared by duration of Impella 5.5 support. Continuous variables were summarized using mean and standard deviation or median (interquartile range [IQR]). Categorical variables were summarized using frequency and percentage. Unpaired t tests and Fisher’s exact tests were used to compare continuous and categorical variables, respectively. All statistical analyses were performed in R software, version 4.2.0 (R Core Team, Vienna, Austria). A P value <0.05 was considered statistically significant.
Results
Study Population
During the study period, 31 patients were supported with and separated from Impella 5.5 at the study institution. Of these 31 patients, 14 (45.2%) were on prolonged support (>14 days). Summary demographics and baseline characteristics are displayed in Table 1. Mean support duration for those on prolonged support was 51.7 ± 36.4 days compared with 8.5 ± 4.4 days for those on ≤14-day support (P < 0.001). The overall cohort was 80% male (n = 25). Prolonged support patients were younger compared with patients on ≤14-day support (48.9 ± 13.9 years vs 61.7 ± 9.3 years, P = 0.008). The mean BMI of the cohort was 28.08 ± 4.61 kg/m2. Comorbidity burden was similar between groups, with similar prevalence of diabetes, hypertension, peripheral vascular disease, and hyperlipidemia.
Demographics and Baseline Characteristics.
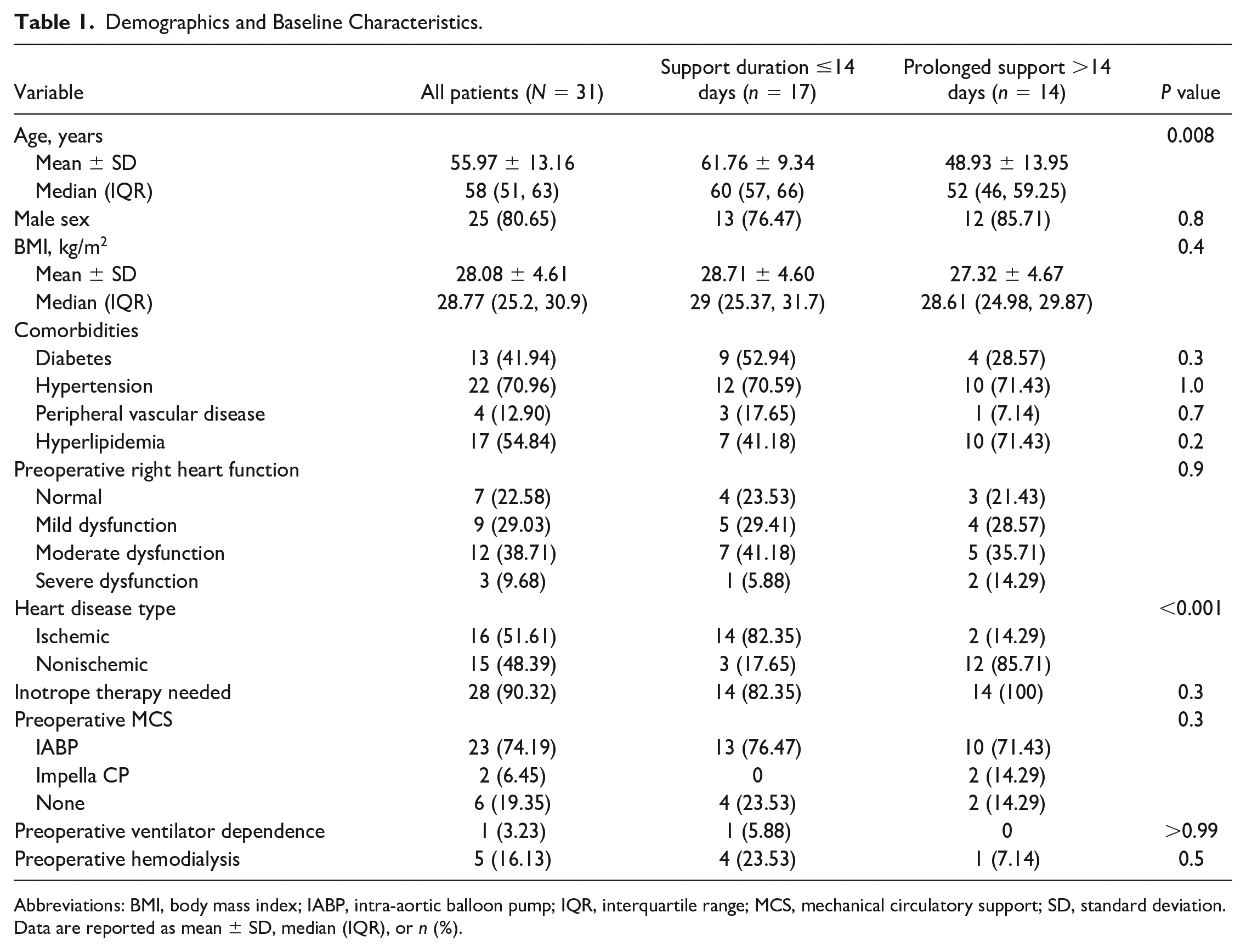
Abbreviations: BMI, body mass index; IABP, intra-aortic balloon pump; IQR, interquartile range; MCS, mechanical circulatory support; SD, standard deviation.
Data are reported as mean ± SD, median (IQR), or n (%).
The groups did not differ in preoperative right heart function, inotropic support requirement, ventilator support requirement, hemodialysis requirement, intra-aortic balloon pump (IABP) usage, Impella CP usage, or concomitant IABP and extracorporeal membrane oxygenation (ECMO) support. However, those on prolonged support were more likely to have nonischemic rather than ischemic cardiomyopathy compared with the ≤14-day cohort (85.7% [n = 12] vs 17.6% [n = 3], P < 0.001).
Preimplant and Explant Risk Stratification
Risk stratification prior to Impella implant and at explant are displayed in Table 2. There was no difference in either preimplant (17.1 ± 5.6 vs 14.3 ± 6.4, P = 0.2) or explant (15.4 ± 6.5 vs 16.6 ± 7.1, P = 0.6) MELD scores between prolonged Impella support and support ≤14 days. A similar result was found for the preimplant (22.8 ± 13.1 vs 24.9 ± 16.3, P = 0.7) and explant (20.5 ± 11.8 vs 23.2 ± 12.9, P = 0.6) EuroSCORE II values.
Risk Stratification.
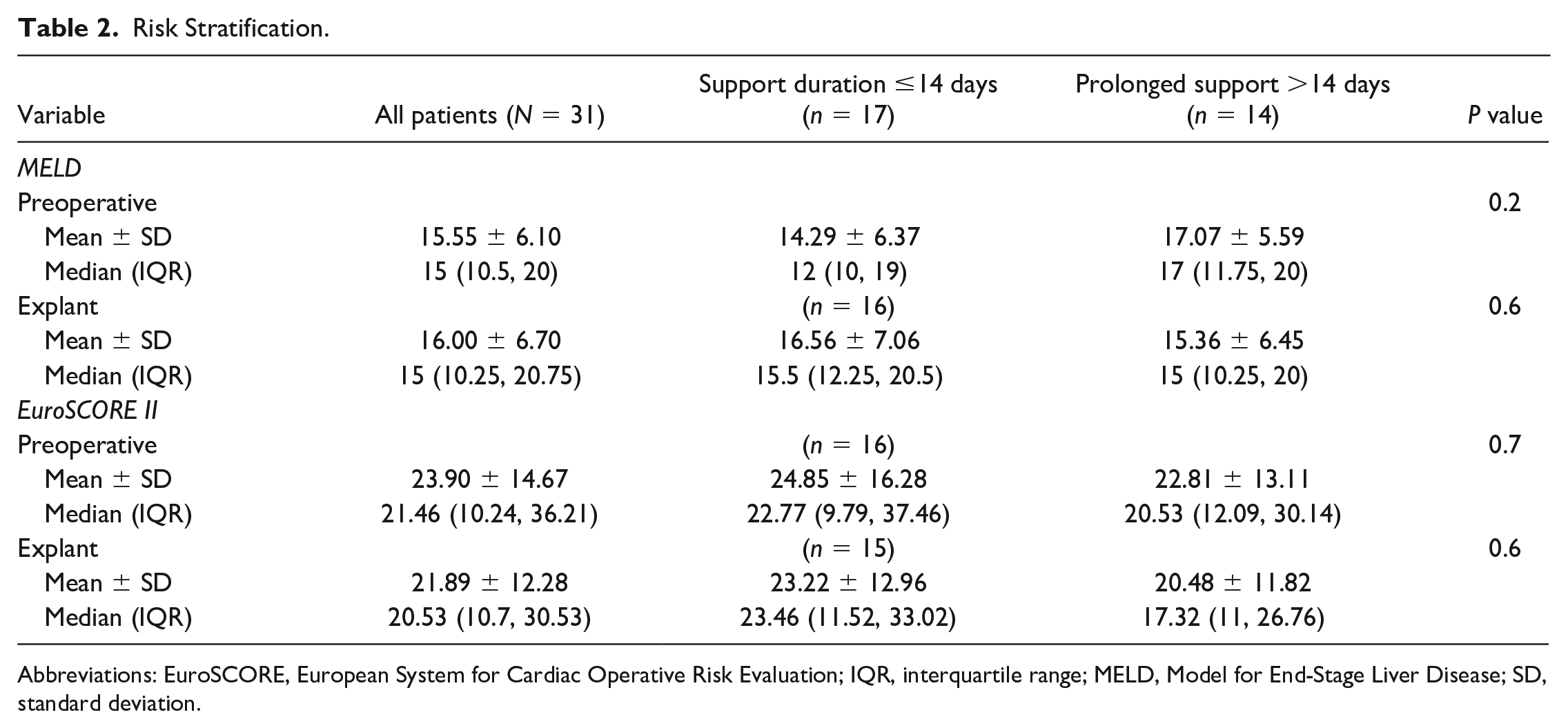
Abbreviations: EuroSCORE, European System for Cardiac Operative Risk Evaluation; IQR, interquartile range; MELD, Model for End-Stage Liver Disease; SD, standard deviation.
Impella Device Details
Impella 5.5 device details are displayed in Table 3. Impella indications were similar between the groups, with similar proportions (P = 0.2) of patients with decompensated heart failure (71.4% [n = 10] vs 52.9% [n = 9]), ventricular arrhythmia (21.4% [n = 3] vs 11.76% [n = 2]), and myocardial infarction (7.1% [n = 1] vs 35.3% [n = 6]). While most patients (77.4%, n = 24) underwent Impella placement via right axillary cutdown with a 10 mm gelweave graft conduit, 7 of the patients on ≤14-day support underwent placement via central cannulation. The bicarbonate protocol was followed for all patients implanted with Impella 5.5. This consisted of using a bicarbonate-based purge solution (sodium bicarbonate 8.4% 25 mEq in 1L D5W) to neutralize dextrose’s acidic pH, increase protein stability, and reduce hematologic protein denaturation, allowing for stable purge flow resistance without contributing to the overall systemic heparin exposure.
Impella Details.
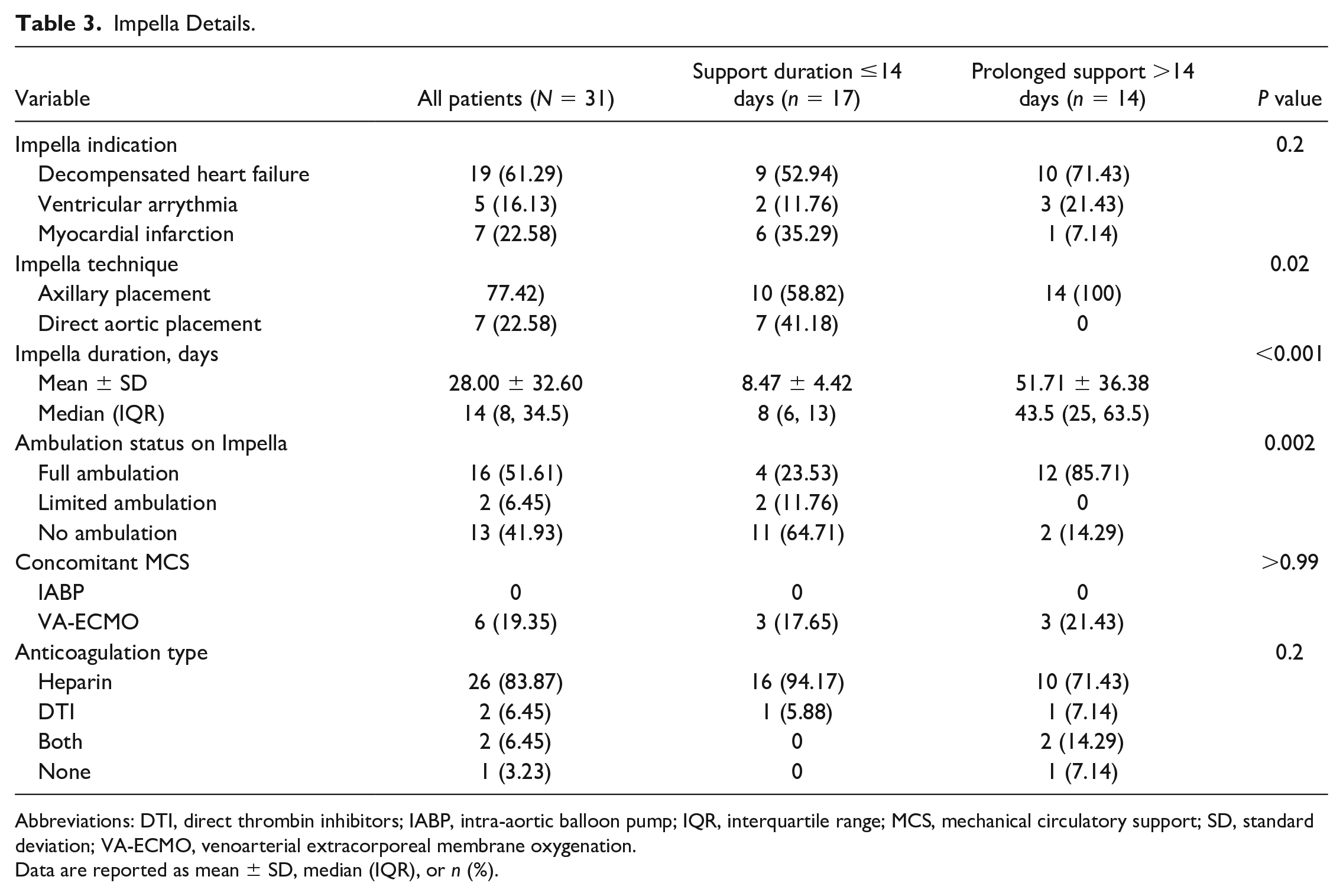
Abbreviations: DTI, direct thrombin inhibitors; IABP, intra-aortic balloon pump; IQR, interquartile range; MCS, mechanical circulatory support; SD, standard deviation; VA-ECMO, venoarterial extracorporeal membrane oxygenation.
Data are reported as mean ± SD, median (IQR), or n (%).
Concomitant MCS use was similar between groups, with no patients on concomitant IABP and 6 patients on venoarterial (VA) ECMO, 3 from each cohort. In the short-term cohort, indications for VA-ECMO consisted of right ventricular dysfunction in 2 patients and inability to sustain cardiac output with only Impella in 1 patient. In the prolonged cohort, 1 patient experienced malignant arrhythmias and VA-ECMO was placed as salvage therapy and bridge to potential heart transplant (although the patient ultimately expired), and in the last 2 patients, 1 was upgraded to VA-ECMO due to worsening ventricular function with only Impella and the other from concerns of sepsis attributed to prolonged support. Of the 3 patients on VA-ECMO in the short-term support group, only 1 was decannulated simultaneously with explant Impella. For the rest, ECMO was discontinued 3 and 5 days prior to Impella explant, respectively. Of the 3 patients on VA-ECMO in the prolonged support group, 1 was supported with ECMO for the last 16 days of Impella, another was supported concomitantly with ECMO and Impella for 4 days prior to transplant, and the last patient was supported for 4 days prior to Impella explant. Anticoagulation type did not differ between groups, with similar proportions of patients on heparin, direct thrombin inhibitors (DTI), and both heparin and a DTI (P = 0.2). Most patients on prolonged support were fully ambulatory (85.71%, n = 12), while most patients on support ≤14 days were either immobile or had limited ambulation (64.71% [n = 11] and 11.76% [n = 2], respectively, P = 0.002).
Outcomes
Primary and secondary outcome comparisons are shown in Table 4. Short-term and long-term mortality were similar between groups (Fig. 1). The overall mortality rate while supported with Impella was 19.4% (n = 6), at 30 days postimplant was 29.0% (n = 9), before hospital discharge was 32.3% (n = 10), and at follow-up was 35.5% (n = 11). The median follow-up time for the cohort was 106 (IQR 26 to 276) days.
Comparison of Outcomes.
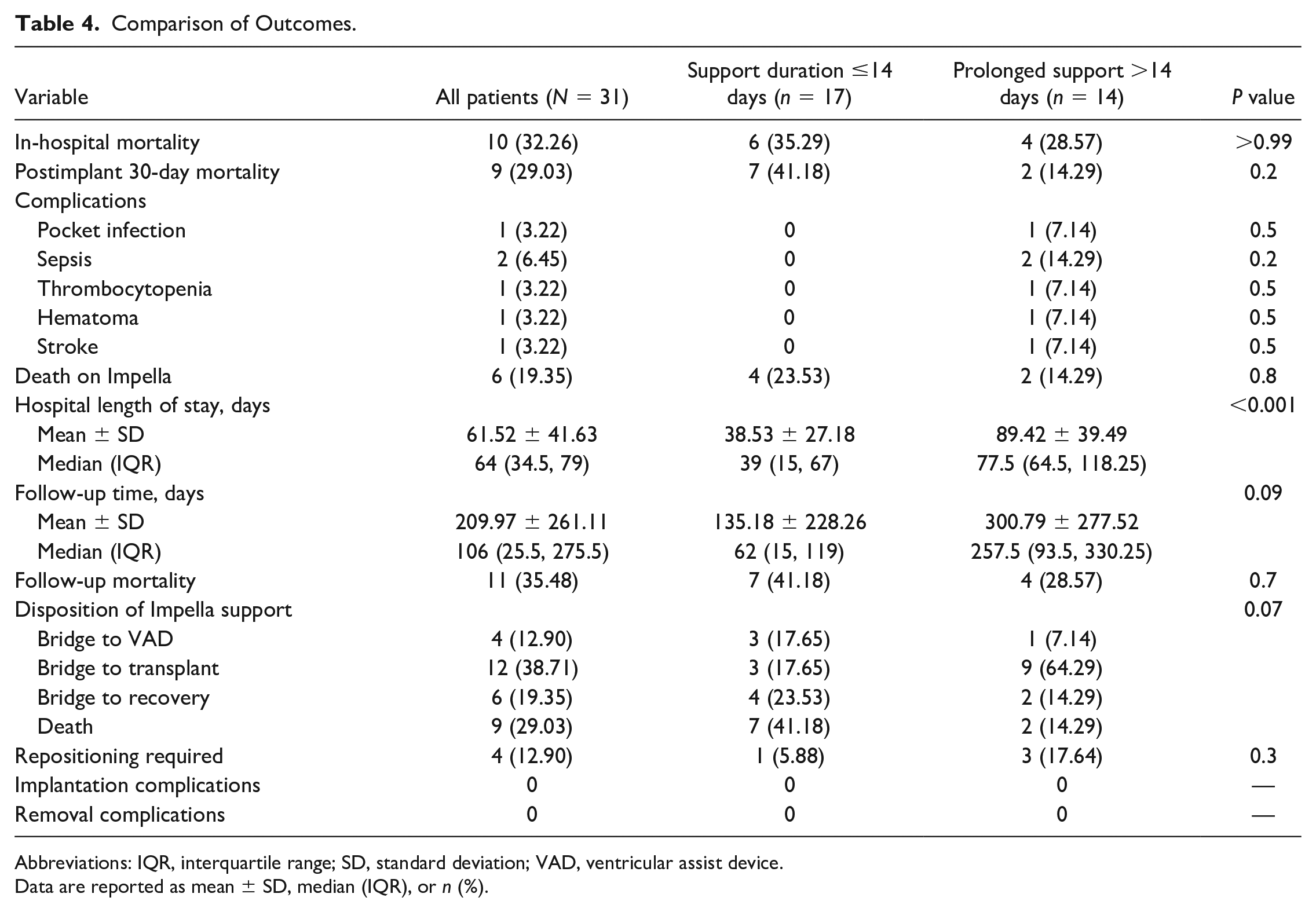
Abbreviations: IQR, interquartile range; SD, standard deviation; VAD, ventricular assist device.
Data are reported as mean ± SD, median (IQR), or n (%).
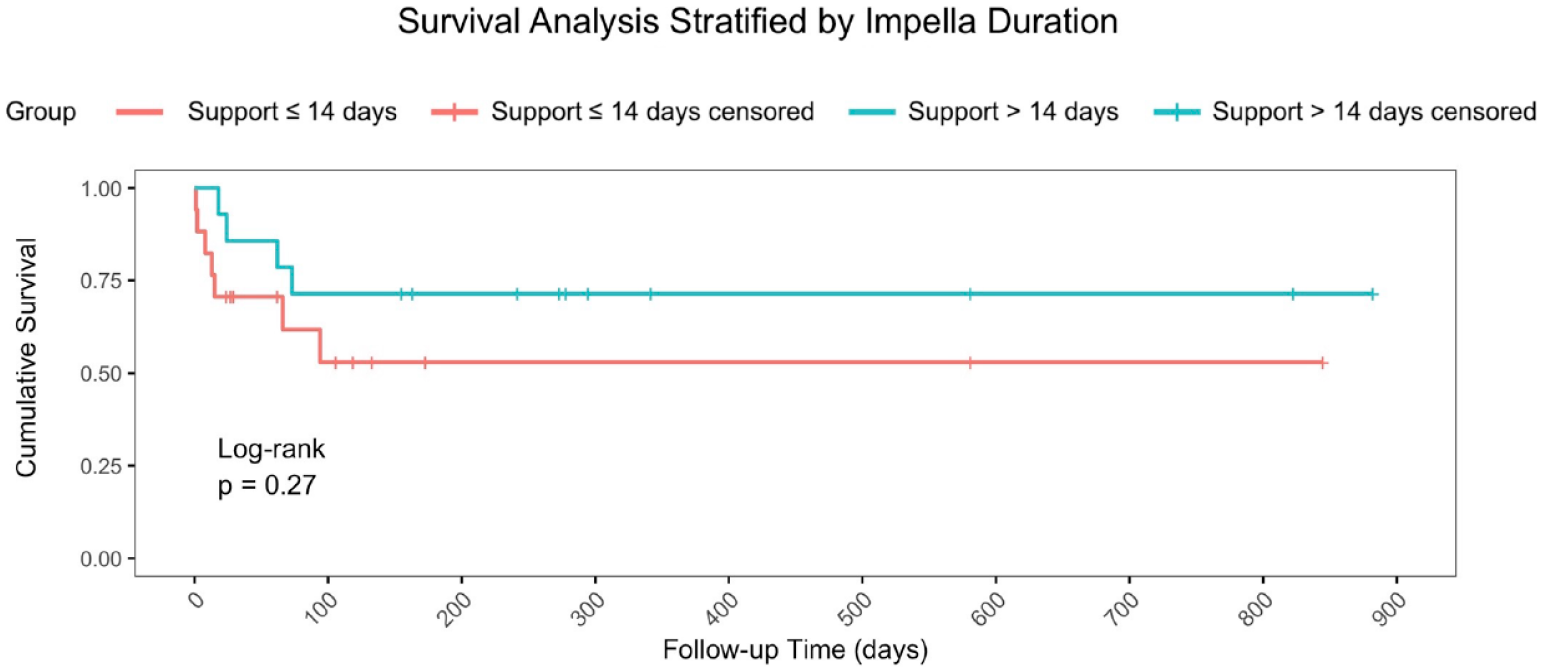
Longitudinal Kaplan–Meier survival after Impella 5.5 (Abiomed, Danvers, MA, USA) insertion for all 31 patients, stratified by support duration. This reveals similar survival after Impella 5.5 insertion (log-rank P = 0.27). There were no significant differences between the short-term and prolonged support groups on in-hospital, 30-day, and follow-up mortality.
In addition, 3 patients experienced 5 complications, including pocket infection (3.2%, n = 1), sepsis (6.5%, n = 2), thrombocytopenia (3.2%, n = 1), and stroke (3.2%, n = 1). The overall complication rate and each individual complication rate did not differ between groups. Four patients (12.9%) required device repositioning, and no patients experienced Impella implant or explant complications.
Bridge strategy differed between groups although this did not reach statistical significance (P = 0.07; Fig. 2). Most patients on prolonged support received a transplant at explantation of Impella (64.3%, n = 9), compared with 3 patients (17.7%) on short-term support. Three of the patients on short-term support (17.7%) received a durable ventricular assist device (VAD), compared with 1 patient on prolonged support (7.14%). In total, 6 patients were bridged to myocardial recovery, 2 in the prolonged group (14.3%) and 4 in short-term support group (19.4%).
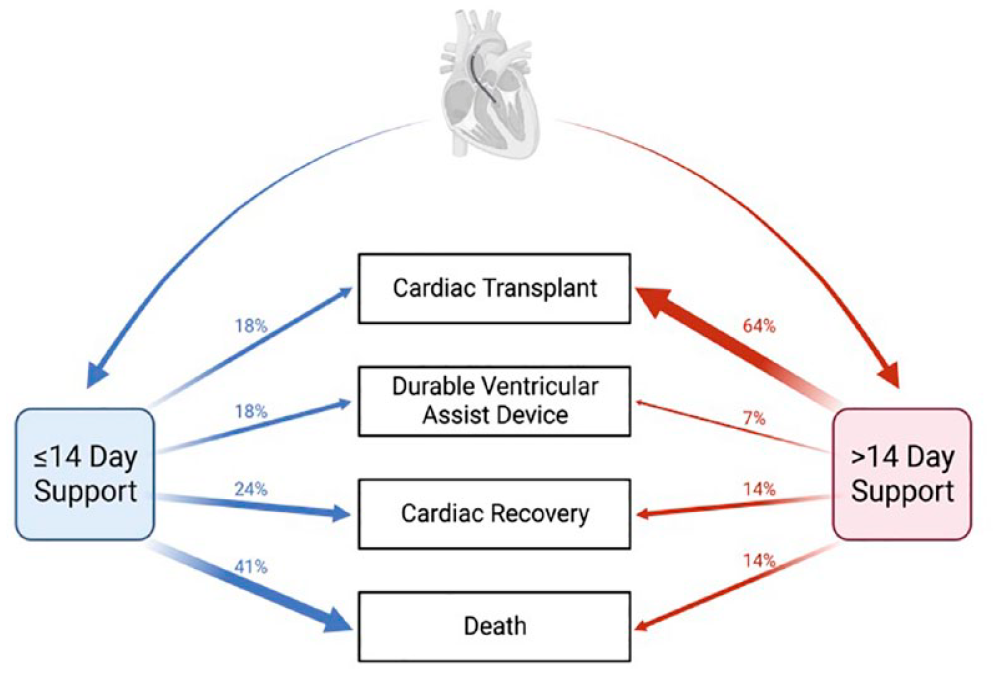
Disposition after Impella 5.5 support stratified by cohort. Created with BioRender.com.
Hemodynamic and Lab Value Changes
Hemodynamic and laboratory values were collected before Impella implantation, on POD1, on POD5, and just prior to Impella removal. These values are displayed in Supplemental Table 1 and Supplemental Table 2, respectively. Four patients supported <5 days did not have POD5 hemodynamic or laboratory data. Patients supported ≤14 days did not have PCWP data on POD1 or at Impella explant. In terms of hemodynamic and laboratory data, the prolonged cohort had higher explant albumin compared with the short-term cohort (3.6 ± 0.4 vs 2.9 ± 0.6, P = 0.004). There were no other differences between the groups in preimplant, peri-implant, or postimplant hemodynamic, echocardiographic, or laboratory variables.
Discussion
Impella 5.5 is an effective tMCS device used for the management of cardiogenic shock. Impella has become a popular tMCS option in part due to the possibility of minimally invasive insertion. Although FDA approved for up to 14 days of support, prolonged Impella 5.5 support is sometimes indicated for complex cases and for patients awaiting cardiac transplantation. This single-center experience evaluated the safety and efficacy of Impella 5.5 support beyond 14 days compared with 14 days or less.
In the present study, the prolonged cohort was supported for an average of 52 days, while the short-term cohort was supported for an average of 8 days. The support duration cohorts differed minimally in their baseline characteristics, comorbidities, and severity of preimplant disease. However, the prolonged support patients had a higher rate of nonischemic heart disease. Risk stratification calculations (MELD and EuroSCORE II), Impella device indication, concomitant MCS, and anticoagulation strategy did not differ between cohorts. Ambulation status differed between the groups, with more prolonged support patients who were fully ambulatory. Compared with the short-term cohort, more patients in the prolonged support cohort received a heart transplant. Yet, complication rates, need for device repositioning, and short-term and long-term mortality were similar between cohorts. Hemodynamic, echocardiographic, and laboratory values did not differ between groups, aside from the prolonged support group having higher explant albumin levels.
Evaluation of the safety and efficacy of prolonged Impella support is important when advocating for the adoption of new guidelines. Although there are few studies evaluating outcomes of prolonged Impella support, the studies available have yielded promising results. In a study by Rock and colleagues, 24 patients were implanted with 26 Impella 5.5 devices. 10 Of these 24 patients, 10 had a support duration greater than 14 days. These patients were managed for an average of 27 days on prolonged support and experienced a 64% survival rate to hospital discharge. After Impella support, 50% of patients experienced cardiac recovery, 33% had heart transplants, and 17% had a durable VAD implanted. In another study by Bernhardt and colleagues, 46 patients underwent Impella 5.5 implantation, with mean support time of 15 days, at different German centers. 11 Overall, 30-day and 90-day survival estimates were 73.9% (95% confidence interval [CI]: 63.3% to 88.9%) and 71.7% (95% CI: 60.7% to 87.1%), respectively. In their cohort, the reported disposition after Impella was composed of durable VAD (41%), cardiac recovery (35%), and cardiac transplantation (2.2%). In the present study, the prolonged support cohort experienced longer support duration and improved outcomes compared with patients in the studies by Rock et al. and Bernhardt et al. Our prolonged cohort was supported for an average of 52 days and experienced an overall postimplant 30-day survival rate of 85.7% and a 71.4% survival rate to hospital discharge. In addition, 64.3% were successfully bridged to heart transplantation compared with 33% and 2.2% in the previously mentioned studies.10,11 However, the rates of disposition after Impella vary in the literature, as these are dependent on institutional protocols and individual patient factors, such as comorbidities and therapeutic management.
When considering complications, Rock and colleagues found an overall complication rate of 73% (8 of 11) in their prolonged group, with axillary hematoma, device malfunction, and gastrointestinal bleeding responsible for 18% each. 11 The study also noted single instances of axillary infection, stroke, heparin-induced thrombocytopenia, and new-onset renal failure, each responsible for 9% of the total complication rate. Bernhardt and colleagues found that 34.8% of patients (16 of 46) experienced bleeding that required transfusions. 11 Further, 15.2% of the study cohort experienced pump thrombosis. Bernhardt and colleagues attributed this to suboptimal placement of the inflow orifice as 10 patients were found to have inflow cannulas in the aortic root. There was a low but considerable rate of thromboembolism (2.2%) with 1 occurrence of stroke. Others have found that Impella 5.5 is associated with a lower incidence of stroke compared with previous models and to other tMCS devices. 15 This may be attributed to the lack of pigtail catheter with Impella 5.5 as this may have been a potential source of thromboembolic events. 16 In the present study, 21.4% of patients (3 of 14) on prolonged support experienced complications. This is an improvement from the 73% complication rate reported in the study by Rock and colleagues. 10 Further, unlike these other studies, the patients in our study did not experience significant bleeding complications. This may be due to the fact that Impella 5.5 does not require heparin in the purge solution. One patient from our study experienced 2 complications on different days of support—a pocket infection on day 4 and a right middle cerebral artery stroke on day 29. In the other 2 patients, 1 had separate episodes of thrombocytopenia and sepsis on days 30 and 78, respectively, and the other experienced sepsis on day 57. Overall, the present study shows that patients on prolonged Impella 5.5 support do not have an increased risk of complications or mortality compared with those supported for ≤14 days. In addition, it appears that outcomes with the Impella 5.5 are improving with time and experience.
Impella 5.5 is used for hemodynamic support in patients with cardiogenic shock. In a recent study evaluating the impact of Impella 5.5 hemodynamics on patients with moderate to severe functional mitral regurgitation, Kataria and colleagues found that Impella 5.5 improved hemodynamic response 24 h postimplant with increased cardiac index and decreased CVP, dPAP, and systemic vascular resistance. 17 There was also a significant decrease in serum lactate, and although nonsignificant, all other laboratory values (serum creatinine, alanine aminotransferase, aspartate aminotransferase, and bilirubin) showed improvement after 24 h on Impella 5.5. In the present study, hemodynamic and laboratory data were similar between the prolonged and short-term cohorts on POD1 and POD5, and at the time of explant, and there was a trend toward improved hemodynamics and laboratory values in the prolonged cohort. Our findings corroborate with the existing literature and suggest that patients on prolonged Impella 5.5 support will benefit from improved hemodynamics until durable therapy is implemented.
As a single-center, retrospective review, this study has several intrinsic limitations. First, the collection of variables for our study through a comprehensive retrospective chart review introduces the possibility of bias. The inconsistencies and differences in electronic health record documentation may have led to missing variables from our sample population. Further, our sample consisted of disproportionately more male than female patients directly affecting the generalizability of our results. Also affecting the generalizability of our results is our limited sample size, as small samples can affect the internal and external validity. Although the patients in our study differed minimally in their baseline and preoperative characteristics, the complexity of each individual case also makes comparisons between patients difficult.
Conclusions
Impella 5.5 is a safe and effective tMCS device used for the management of cardiogenic shock. It is not known if extended use of the Impella 5.5 past the 14-day FDA-approved support time is associated with increased mortality and complication rates. The current study suggests that the Impella 5.5 can be used safely past the 14-day time point without an increase in adverse outcomes. This makes the Impella 5.5 an attractive choice for patients whose clinical status may require a long support time before durable therapy can be established. Future studies should include a larger sample size that is more representative of the general population.
Supplemental Material
sj-pdf-1-inv-10.1177_15569845231212157 – Supplemental material for Reconsidering FDA Guidelines: A Single-Center Experience of Prolonged Impella 5.5 Support
Supplemental material, sj-pdf-1-inv-10.1177_15569845231212157 for Reconsidering FDA Guidelines: A Single-Center Experience of Prolonged Impella 5.5 Support by Carlos Alberto Valdes, Griffin Stinson, Omar M. Sharaf, Fabian Jimenez Contreras, Ahmet Bilgili, Mustafa M. Ahmed, Juan Vilaro, Alex M. Parker, Mohammad A. Z. Al-Ani, Daniel Demos, Juan Aranda, Mark Bleiweis, Thomas M. Beaver and Eric I. Jeng in Innovations
Footnotes
*Abstract was accepted and presented as an oral presentation at the 2023 ISMICS Annual Scientific Meeting in Boston, MA, USA.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
