Abstract
Objective:
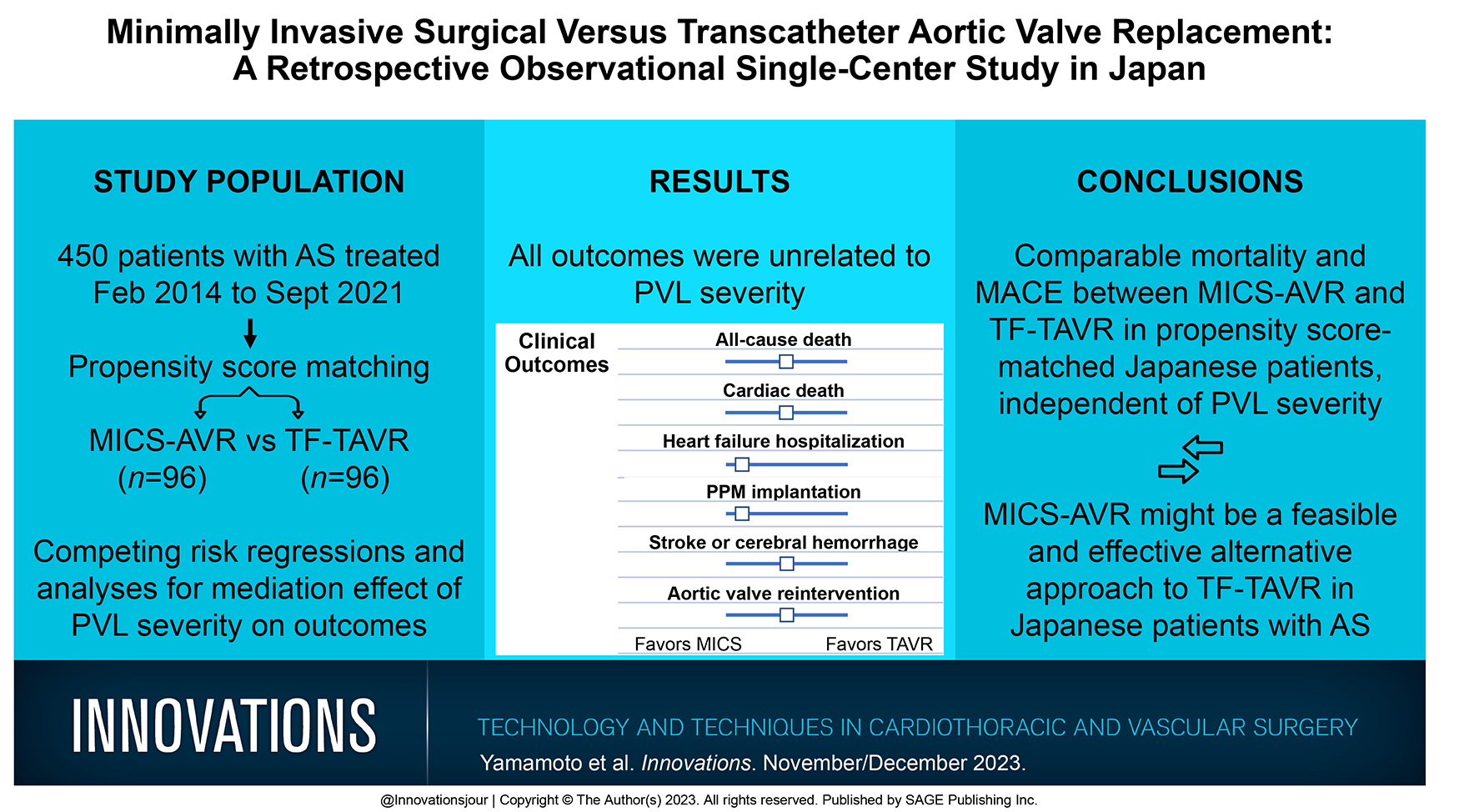
This study aimed to compare the outcomes of minimally invasive aortic valve replacement (MICS-AVR) versus transfemoral transcatheter aortic valve replacement (TF-TAVR) in Asian patients.
Methods:
We conducted a retrospective, observational, single-center study in Japan, including cases of MICS-AVR (n = 202) and TF-TAVR (n = 248) between 2014 and 2021. In a total of 450 cases, propensity score matching was performed at a ratio of 1:1, resulting in 96 pairs. Furthermore, we performed competing-risk regression and mediation analyses to determine the treatment effect on outcomes of interests, considering death as a competing risk, and to evaluate the mediation effect of paravalvular leak (PVL) severity.
Results:
There were similar incidences of all-cause death, cardiac death, stroke and cerebral hemorrhage, and aortic valve reintervention between the 2 groups. However, the TF-TAVR cohort had a longer hospital length of stay and higher rates of significant PVL compared with the MICS-AVR cohort. Multivariable-adjusted Cox regression analyses revealed that heart failure hospitalization (hazard ratio [HR] = 0.129, 95% confidence interval [CI]: 0.038 to 0.445, p = 0.001) and permanent pacemaker implantation (HR = 0.050, 95% CI: 0.006 to 0.409, p = 0.005) favored MICS-AVR. Competing-risk regression analyses confirmed similar findings. All outcomes were unrelated to PVL severity.
Conclusions:
To our knowledge, this is the first comparative study of clinical outcomes in Asian patients undergoing MICS-AVR versus TF-TAVR, revealing that MICS-AVR could be a feasible and efficient alternative to TF-TAVR. Future larger-scale randomized controlled trials are needed to validate the present results.
Keywords
Central Message
There were comparable mortality and major adverse cerebrovascular event rates between minimally invasive aortic valve replacement and transfemoral transcatheter aortic valve replacement in propensity score–matched Japanese patients with aortic stenosis, independent of paravalvular leak severity.
Introduction
Degenerative severe aortic stenosis (AS) is a potentially life-threatening disorder, and its incidence continues to steadily increase in aging societies. Although surgical aortic valve replacement (SAVR) was the standard of care in patients with severe AS, supportive care including diuretic administration and balloon valvuloplasty was the only palliative treatment in those considered inoperable or at high risk. The rise in transcatheter aortic valve replacement (TAVR) use resulted in a paradigm shift in severe AS management. TAVR, initially introduced as an alternative to SAVR in inoperable or high-risk patients with AS, proved to be feasible. 1 Since recent randomized controlled trials (RCTs) proved the superiority of TAVR over SAVR even in intermediate-risk to low-risk patients with AS,2,3 TAVR is widely accepted in patients with AS at any level of risk.
Minimally invasive aortic valve replacement (MICS-AVR) has recently evolved with technological improvements. Compared with the conventional SAVR, MICS-AVR, which avoids full sternotomy, has improved the outcomes of severe AS due to the potential benefits from avoiding sternal wound complications, bleeding, systemic inflammation, and pulmonary dysfunction. 4 Previous comparative studies of MICS-AVR versus TAVR, mainly in Europe and the United States, have reported that MICS-AVR might be a feasible alternative to TAVR.5,6 However, to date, no similar studies in Asian countries have been conducted.
In addition, since patients with AS undergoing MICS-AVR or TAVR were relatively very old, death could be considered a censoring event in the evaluation of outcomes other than death. Therefore, outcomes of interests may have not been correctly evaluated in previous studies; death should have been considered a competing risk in the assessment of outcomes other than death. Moreover, since the contribution of postprocedural paravalvular leaks (PVLs) to each outcome remains uncertain, the treatment effect through PVL severity on each outcome should be determined between the 2 approaches. Hence, we performed a propensity score matching (PSM) analysis of 450 consecutive patients with symptomatic severe AS who underwent MICS-AVR or transfemoral TAVR (TF-TAVR) at our single center in Japan. Moreover, competing-risk regression and mediation analyses were performed to determine treatment effects on outcomes of interests, considering death a competing risk, and to evaluate the mediation effect of PVL severity.
Methods
Patient Selection
The treatment strategy of MICS-AVR or TF-TAVR was determined based on a multidisciplinary consensus of our heart team. A total of 450 consecutive patients with severe AS who underwent MICS-AVR or TF-TAVR at our hospital from February 2014 to September 2021 were included. Data collection and analysis were performed after obtaining approval by the Institutional Review Board. Written informed consent was obtained from all patients for the publication of the study data.
Preoperative Patient Characteristics
Preoperative variables including the following 28 factors were recorded for each patient. These variables comprised the EuroSCORE II components including age, female sex, chronic obstructive pulmonary disease, extracardiac arteriopathy, poor mobility, previous cardiac surgery, active endocarditis, critical preoperative state, baseline creatinine clearance, and diabetes managed by insulin. They also included angina, left ventricular ejection fraction (LVEF), recent myocardial infarction, pulmonary hypertension, New York Heart Association class, thoracic aorta surgery, urgency, and concomitant surgery. Other components included body surface area (BSA), baseline hemoglobin level, baseline platelet count, and atrial fibrillation (AF). Complete right bundle branch block (CRBBB) or complete left bundle branch block on electrocardiogram, history of permanent pacemaker (PPM) implantation, peak and mean pressure gradients (PGs), and aortic regurgitation (AR) grade 2 or higher were also included.
Procedures
MICS-AVR
Patients were placed in the left decubitus position, anesthetized, and endotracheally intubated. A skin incision was made forward from the right anterior axillary line. After dissecting the space under the pectoralis major muscle anteriorly, a thoracotomy incision was made along the third intercostal space. The right femoral or axillary vein was cannulated for cardiopulmonary bypass. The left ventricular venting tube was inserted from the right upper pulmonary vein. The aortic root cannula was placed, and the distal end of the ascending aorta was cross-clamped. All procedures were performed under direct vision with thoracoscopic assistance. The MICS-AVR was performed following the same approach as in median sternotomy AVR. 7
TF-TAVR
Patients were administered general anesthesia under full monitoring including transesophageal echocardiography. The procedure was performed in the standard manner, using a surgical cut-down to the iliofemoral vessels. Thereafter, the prosthetic valve was delivered using an introducer sheath set, following which aortic valve predilatation with a standard balloon was performed. 8 The prosthetic valve was deployed in the proper position during rapid ventricular pacing, followed by balloon dilatation to fit the valve. Aortography was then performed to evaluate the extent of AR and the patency of the coronary arteries.
Categorical and Continuous Outcomes
Patient clinical characteristics and outcomes were analyzed. Postoperative complications according to the Valve Academic Research Consortium-2 criteria were evaluated. 9 The outcomes included all-cause death, cardiac death, heart failure hospitalization (HFH), PPM implantation, stroke and cerebral hemorrhage, and aortic valve reintervention during the follow-up period. The mean follow-up period in all 450 patients was 30.36 ± 22.50 months. The clinical follow-up was performed via outpatient and telephone interviews. The perioperative outcomes were defined as operation time, valve size, overt bleeding requiring blood transfusion, repeat thoracotomy, new-onset AF, pulmonary failure, length of stay (LOS) in the intensive care unit (ICU), and hospital LOS. The echocardiographic outcomes included LVEF, left ventricular end-diastolic dimension, left ventricular end-systolic dimension, peak and mean PGs, and PVL during the perioperative and midterm periods.
Statistical Analyses
Categorical data are expressed as frequencies and percentages. The differences between the groups were assessed using the chi-square test or Fisher’s exact test. Continuous variables are expressed as mean ± standard deviation (SD) and compared between the groups using Student’s t tests. To minimize the effect of selection bias by indication and potential confounding, we used PSM for the patient baseline characteristics. The propensity score was obtained using a logistic regression model that included treatment approaches as the outcome variable and all potential confounders as explanatory variables. We matched the patients who underwent MICS-AVR and TF-TAVR on a 1:1 basis using the logit of a propensity score with a 0.2 caliper width.
Time-to-event data were demonstrated in Kaplan–Meier curves and compared using the log-rank test for the matched pairs. Survival curves were compared using a stratified log-rank test for the propensity score–matched cohort. Comparison of the clinical outcomes of the 2 treatment approaches was also performed using Cox regression analysis and reported as hazard ratios (HRs) and 95% confidence intervals (CIs). Since there could be risks of imbalance regarding some variables even in the matched cohort, multivariable Cox regression was performed in the matched cohort to confirm the robustness of the results obtained from simple Cox regression. The proportional hazards assumption was checked by inspecting log-log plots before using Cox regression analysis.
Because death was considered a competing risk when it occurred before outcomes of interests such as HFH, PPM implantation, stroke and cerebral hemorrhage, and aortic valve reintervention, we calculated the cumulative incidence of these events in the presence of competing risks. Regarding cardiac death evaluation, death by other causes was considered a competing risk. A competing-risk regression analysis 10 was performed and reported as subdistribution hazard ratios (SHRs) and 95% CIs. We also performed causal mediation analysis to evaluate the proportional contribution of prevalent PVL severity, which was categorized by a perileak AR grade ≥2, to the association between TF-TAVR and outcomes. 11 Regarding the mediation analyses, TF-TAVR, AR grade, and outcomes of interests were managed as independent, intervening, and dependent variables, respectively. The total effect was the causal effect of TF-TAVR on the outcomes without taking the intervening variable, AR grade, into account. The indirect or direct effect was defined as the effect of TF-TAVR on the outcomes with or without the AR grade, respectively. Mediation analyses were performed by a Stata command, Idecomp. 12 p values of <0.05 were considered statistically significant. Statistical analyses were performed using Stata Statistical Software: Release 16 (StataCorp, College Station, TX, USA).
Results
Baseline Characteristics
The preoperative baseline characteristics of the study population are presented in Supplemental Table 1. Compared with the MICS-AVR group, the TF-TAVR group included older female patients, lower BSAs, and higher surgical risk profiles. Table 1 summarizes the baseline characteristics before and after PSM. This PSM method identified 96 pairs of patients with similar background factors. Notably, both matched cohorts showed a similar intermediate surgical risk (logit of EuroSCORE II: −3.46 ± 0.38 in the MICS-AVR group vs −3.51 ± 0.44 in the TF-TAVR group, p = 0.41), mild anemia (hemoglobin: 11.7 ± 1.7 in the MICS-AVR group vs 11.6 ± 1.4 in the TF-TAVR group, p = 0.87), and small body size (BSA: 1.47 ± 0.18 in the MICS-AVR group vs 1.47 ± 0.18 in the TF-TAVR group, p = 0.95). No factors had absolute standardized differences >0.2, which indicated severe imbalance, and none of the variables showed a statistically significant difference between the 2 matched cohorts.
Baseline Characteristics of Patients With Severe Aortic Stenosis Undergoing MICS-AVR or TF-TAVR in the Unmatched and Matched Cohorts.
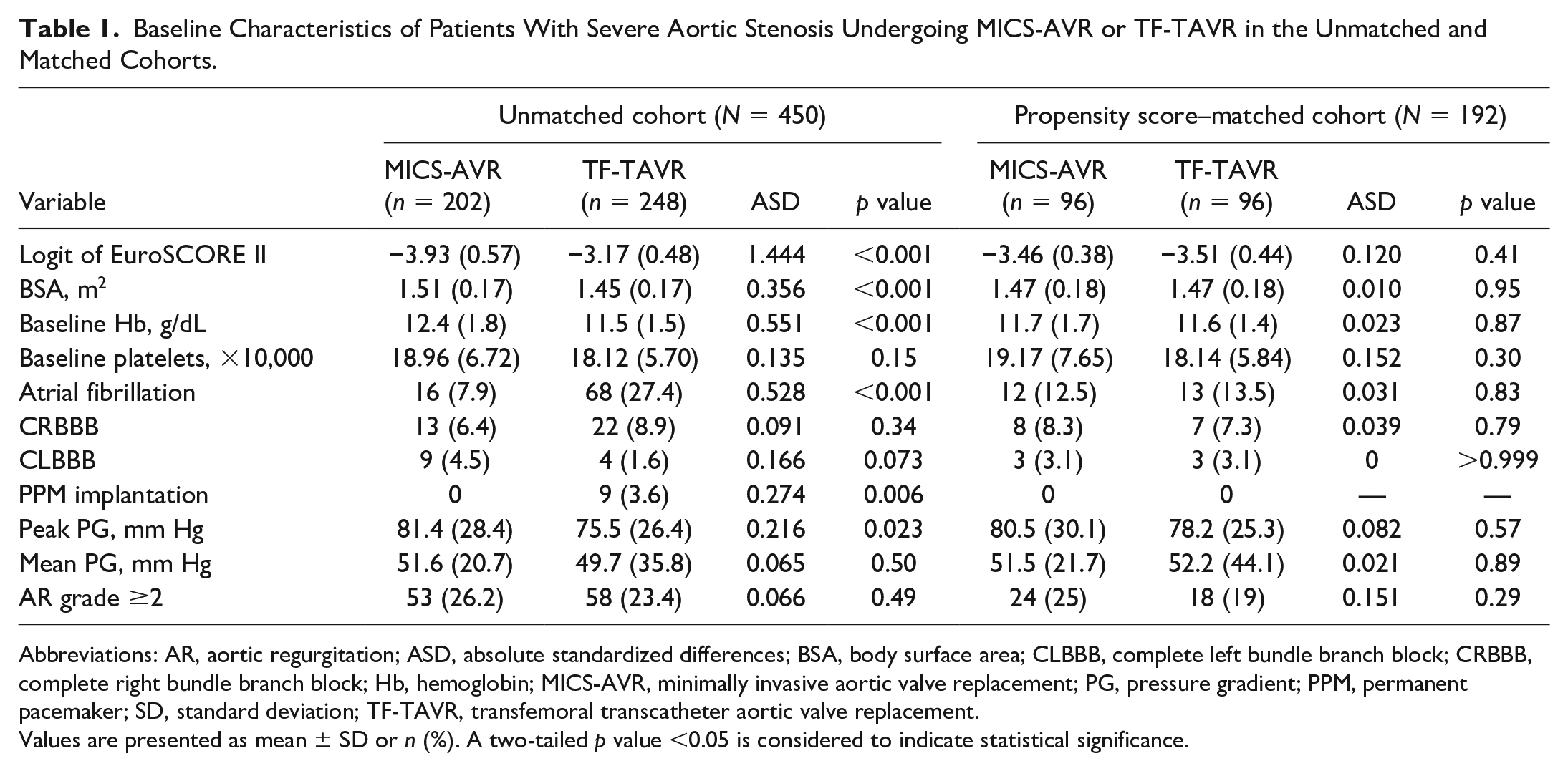
Abbreviations: AR, aortic regurgitation; ASD, absolute standardized differences; BSA, body surface area; CLBBB, complete left bundle branch block; CRBBB, complete right bundle branch block; Hb, hemoglobin; MICS-AVR, minimally invasive aortic valve replacement; PG, pressure gradient; PPM, permanent pacemaker; SD, standard deviation; TF-TAVR, transfemoral transcatheter aortic valve replacement.
Values are presented as mean ± SD or n (%). A two-tailed p value <0.05 is considered to indicate statistical significance.
Outcomes
Table 2 summarizes the outcomes of the 2 matched cohorts. The MICS-AVR cohort received sutured prostheses (74 of 96, 77.1%), sutureless prostheses (20 of 96, 20.8%), or mechanical prostheses (2 of 96, 2.1%). The TF-TAVR cohort received the following implanted prostheses: Edwards (Irvine, CA, USA) SAPIEN III (51 of 96, 53.1%), Edwards SAPIEN XT (16 of 96, 16.7%), Medtronic (Dublin, Ireland) Evolut PRO (13 of 96, 13.5%), Medtronic Evolut R (15 of 96, 15.6%), or Medtronic CoreValve (1 of 96, 1.0%).
Clinical Outcomes in the Propensity Score–Matched Cohort (N = 192).
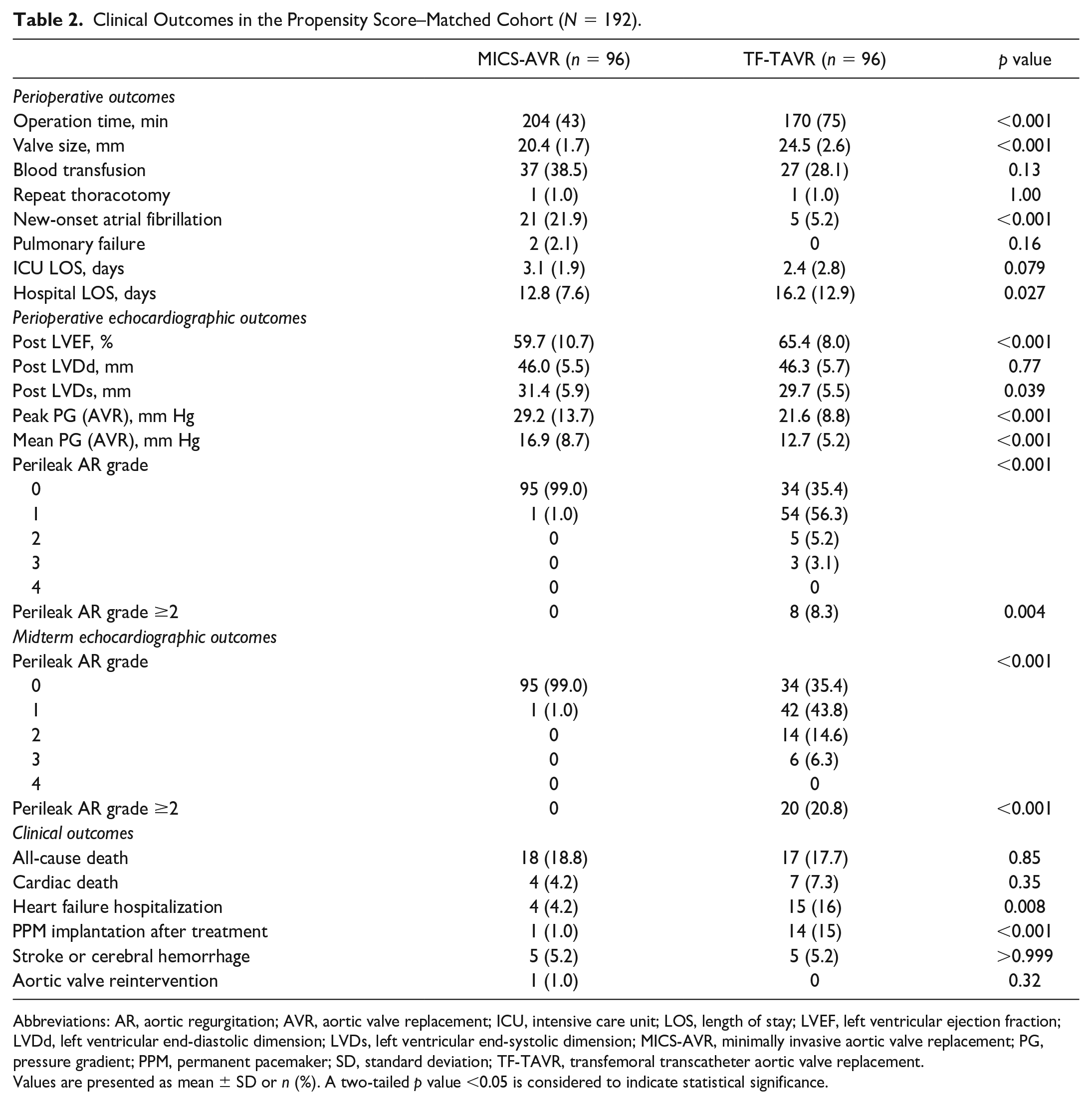
Abbreviations: AR, aortic regurgitation; AVR, aortic valve replacement; ICU, intensive care unit; LOS, length of stay; LVEF, left ventricular ejection fraction; LVDd, left ventricular end-diastolic dimension; LVDs, left ventricular end-systolic dimension; MICS-AVR, minimally invasive aortic valve replacement; PG, pressure gradient; PPM, permanent pacemaker; SD, standard deviation; TF-TAVR, transfemoral transcatheter aortic valve replacement.
Values are presented as mean ± SD or n (%). A two-tailed p value <0.05 is considered to indicate statistical significance.
Perioperative Outcomes
The MICS-AVR cohort had a significantly longer operation time compared with the TF-TAVR cohort (204 ± 43 min vs 170 ± 75 min, p < 0.001). Despite the comparable values of BSAs in both cohorts, the size of the prosthetic valve was significantly larger in the TF-TAVR cohort (24.5 ± 2.6 mm vs 20.4 ± 1.7 mm, p < 0.001). The incidence of new-onset AF was significantly higher in the MICS-AVR cohort (21.9% vs 5.2%, p < 0.001). Although there was a trend toward a longer ICU LOS, the hospital LOS was significantly shorter in the MICS-AVR cohort (12.8 ± 7.6 days vs 16.2 ± 12.9 days, p = 0.027). The major vascular complications were few and comparable between the 2 cohorts (data not shown).
Prosthetic Valve Performance
Perioperative echocardiography revealed that both the peak PG (29.2 ± 13.7 mm Hg vs 21.6 ± 8.8 mm Hg, p < 0.001) and mean PG (16.9 ± 8.7 mm Hg vs 12.7 ± 5.2 mm Hg, p < 0.001) were significantly higher in the MICS-AVR cohort. LVEF was lower in the MICS-AVR cohort (59.7% ± 10.7% vs 65.4% ± 8.0%, p < 0.001). PVLs were more frequently observed in the TF-TAVR cohort (p < 0.001). Most PVLs were trace to mild during the perioperative period. However, the ratio of more than mild PVLs increased over time. No structural valve deterioration was observed in either cohort.
Midterm and Long-Term Clinical Outcomes
All-cause and cardiac deaths, stroke/cerebral hemorrhage, and aortic valve reintervention were comparable between both cohorts. However, the TF-TAVR cohort required PPM implantation significantly more often (15% vs 1.0%, p < 0.001); the prosthetic valves implanted in the TF-TAVR cohort requiring PPMs were balloon-expandable valves (BEVs) in 64% and self-expandable valves (SEVs) in 36% of the patients. In addition, the TF-TAVR cohort more frequently required HFH (16% vs 4.2%, p = 0.008).
Time-to-Event and Cox Regression Analyses
Time-to-event and Cox regression analyses showed no significant differences between the 2 cohorts considering the clinical outcomes including all-cause death, cardiac death, stroke/cerebral hemorrhage, and aortic valve reintervention (Table 3, Fig. 1). However, the superiority of MICS-AVR over TF-TAVR was observed using Kaplan–Meier survival analyses for HFH and PPM implantation after treatment (Fig. 1c-d). The MICS-AVR procedure was associated with a significantly lower incidence of HFH using univariable Cox regression analysis (HR = 0.154, 95% CI: 0.050 to 0.474, p = 0.001). Significance persisted even using multivariable-adjusted Cox regression analysis (HR = 0.129, 95% CI: 0.038 to 0.445, p = 0.001). Similarly, the MICS-AVR procedure was associated with significantly lower PPM implantation rates using univariable Cox regression analysis (HR = 0.065, 95% CI: 0.009 to 0.498, p = 0.008). In addition, this significant association persisted even using multivariable-adjusted Cox regression analysis (HR = 0.050, 95% CI: 0.006 to 0.409, p = 0.005). Notably, the trend of significance was remarkably recognized during the procedural and perioperative periods, with the curves of the 2 groups then remaining parallel to each other (Fig. 1d).
Cox Regression Analyses for the Comparison of Clinical Outcomes in the Matched Cohort (N = 192).
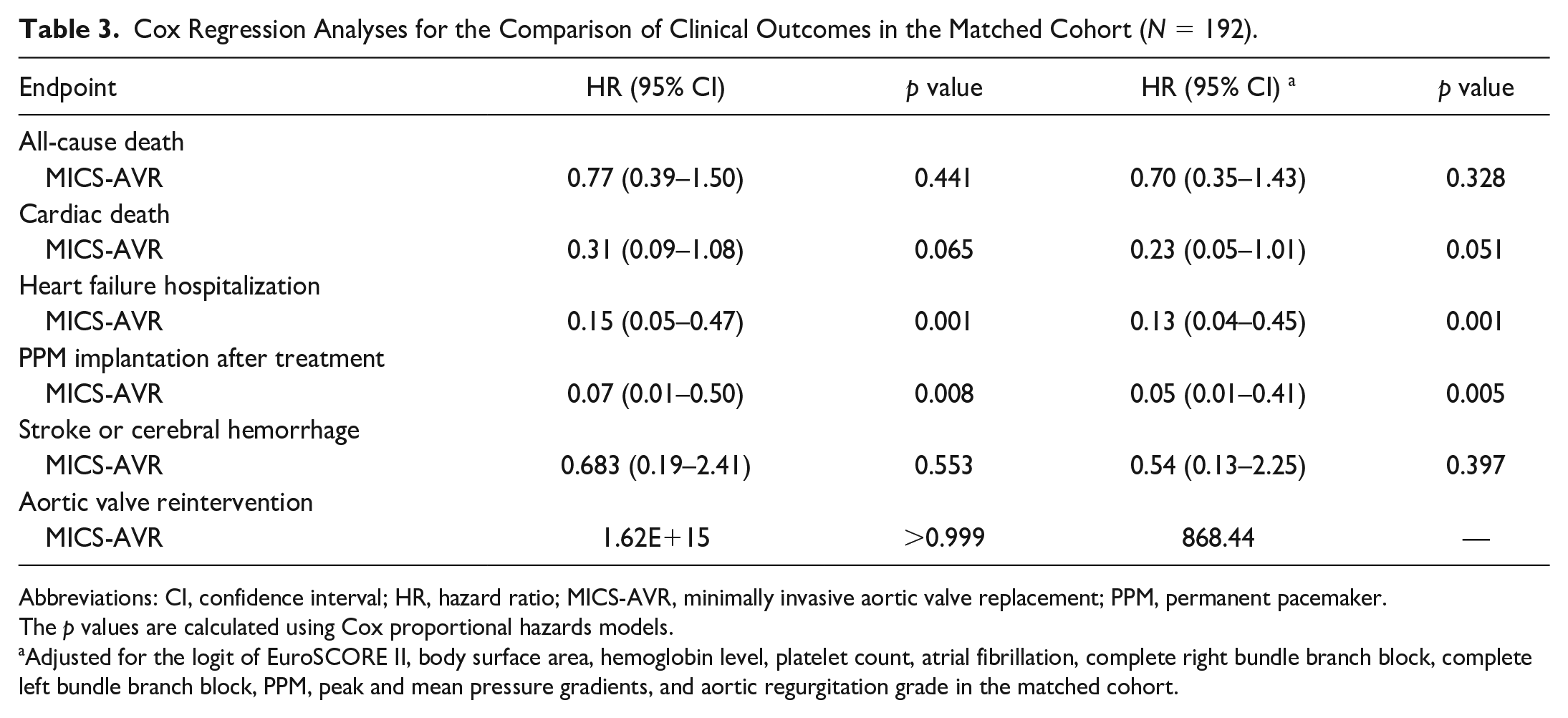
Abbreviations: CI, confidence interval; HR, hazard ratio; MICS-AVR, minimally invasive aortic valve replacement; PPM, permanent pacemaker.
The p values are calculated using Cox proportional hazards models.
Adjusted for the logit of EuroSCORE II, body surface area, hemoglobin level, platelet count, atrial fibrillation, complete right bundle branch block, complete left bundle branch block, PPM, peak and mean pressure gradients, and aortic regurgitation grade in the matched cohort.
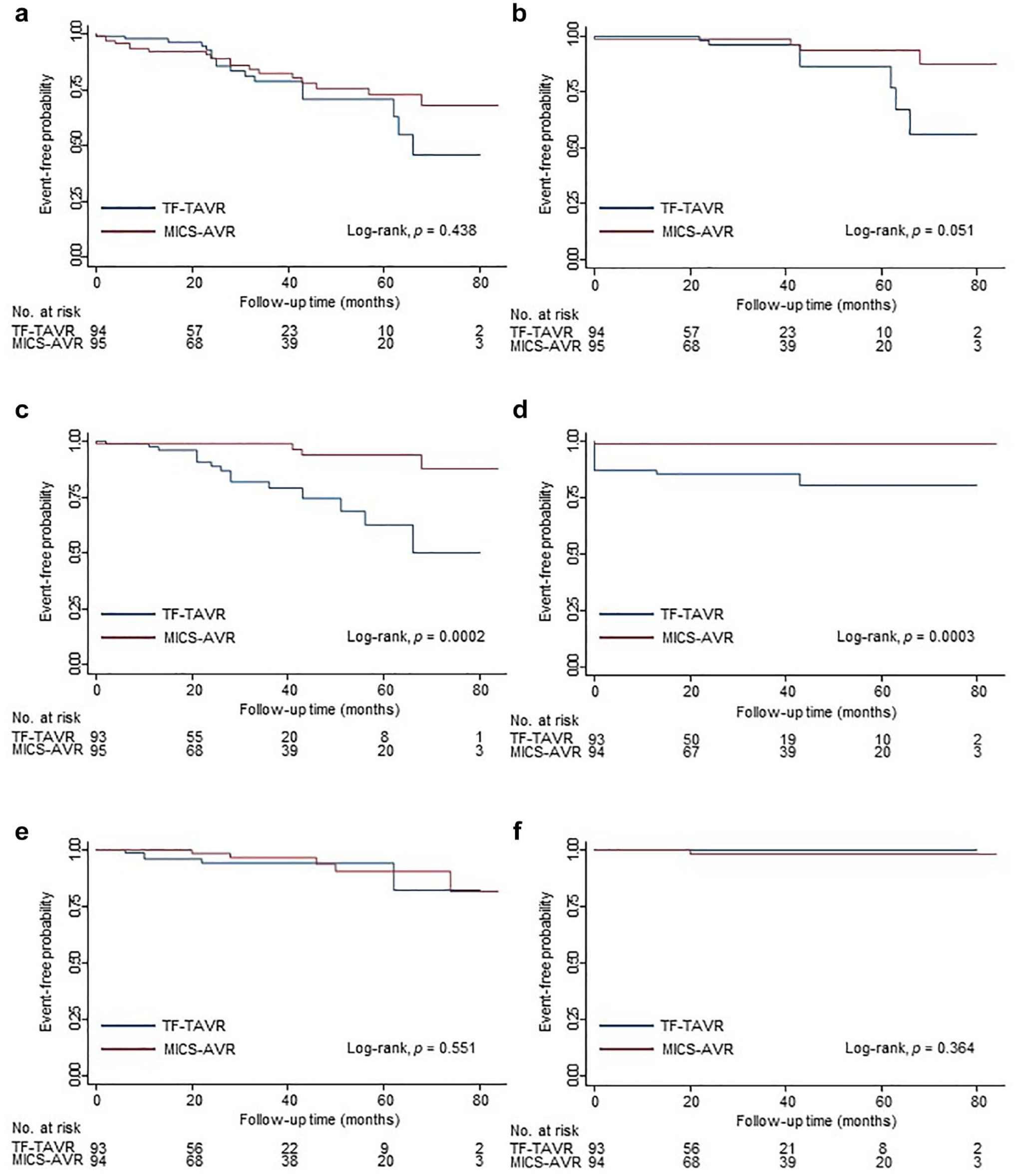
Kaplan–Meier survival curves for clinical outcomes in propensity score–matched patients treated with MICS-AVR versus TF-TAVR. (a) All-cause death, (b) cardiac death, (c) heart failure hospitalization, (d) PPM implantation after treatment, (e) stroke or cerebral hemorrhage, and (f) aortic valve reintervention. The p values were calculated using Cox proportional hazards models. The numbers of participants at risk are below the horizontal axis. MICS-AVR, minimally invasive aortic valve replacement; PPM, permanent pacemaker; TF-TAVR, transfemoral transcatheter aortic valve replacement.
Competing-Risk Regression Analyses
Time-to-event curves for HHF and PMI implantation revealed that TF-TAVR was associated with a higher rate of events compared with MICS-AVR (Supplemental Fig. 1). Competing-risk regression analyses confirmed similar findings (Table 4, Fig. 2), such that the MICS-AVR cohort was 81% less likely to experience HFH (SHR = 0.190, 95% CI: 0.063 to 0.569, p = 0.003) and 92.9% less likely to require PPM implantation (SHR = 0.071, 95% CI: 0.009 to 0.545, p = 0.011).
Competing-Risk Regression for the Comparison of Clinical Outcomes in the Matched Cohort (N = 192).
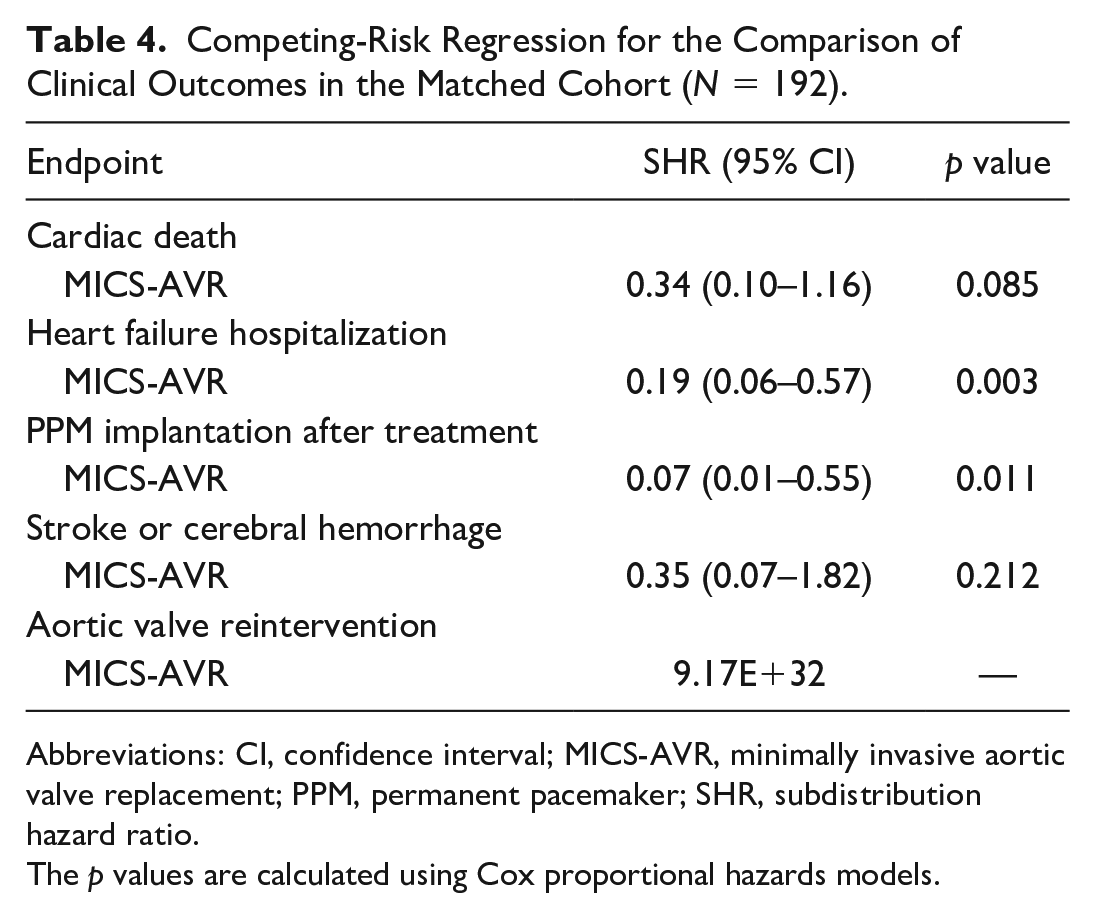
Abbreviations: CI, confidence interval; MICS-AVR, minimally invasive aortic valve replacement; PPM, permanent pacemaker; SHR, subdistribution hazard ratio.
The p values are calculated using Cox proportional hazards models.
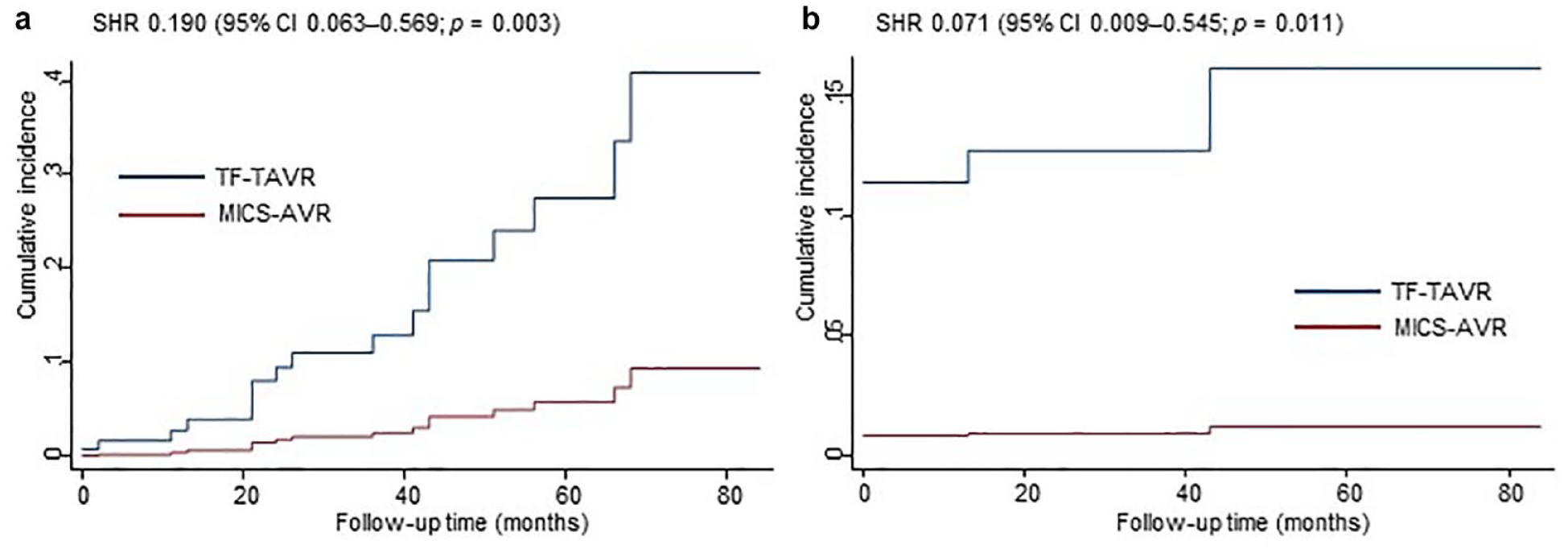
Competing-risk regression of (a) heart failure hospitalization and (b) PPM implantation after treatment. Patients undergoing TF-TAVR were more likely to develop heart failure hospitalization and require postprocedural PPM implantation. CI, confidence interval; MICS-AVR, minimally invasive aortic valve replacement; SHR, subdistribution hazard ratio; PPM, permanent pacemaker; TF-TAVR, transfemoral transcatheter aortic valve replacement.
Mediation Analyses
Table 5 summarizes the results of the mediation analyses between TF-TAVR and clinical outcomes through more than mild PVLs. The direct effect was statistically significant on the association between TF-TAVR and HFH (OR = 4.349, 95% CI: 1.429 to 13.235, p = 0.010) and between TF-TAVR and PPM implantation (OR = 14.858, 95% CI: 4.360 to 50.639, p < 0.010). However, indirect effects through perileak AR grade ≥2 were not statistically significant in all analyses on the association between TF-TAVR and clinical outcomes, suggesting that the presence of significant PVLs might not significantly affect clinical outcomes in the present study.
Mediation Analyses Between TF-TAVR and Clinical Outcomes Through Perileak Aortic Regurgitation Grade ≥2 in the Matched Cohort.
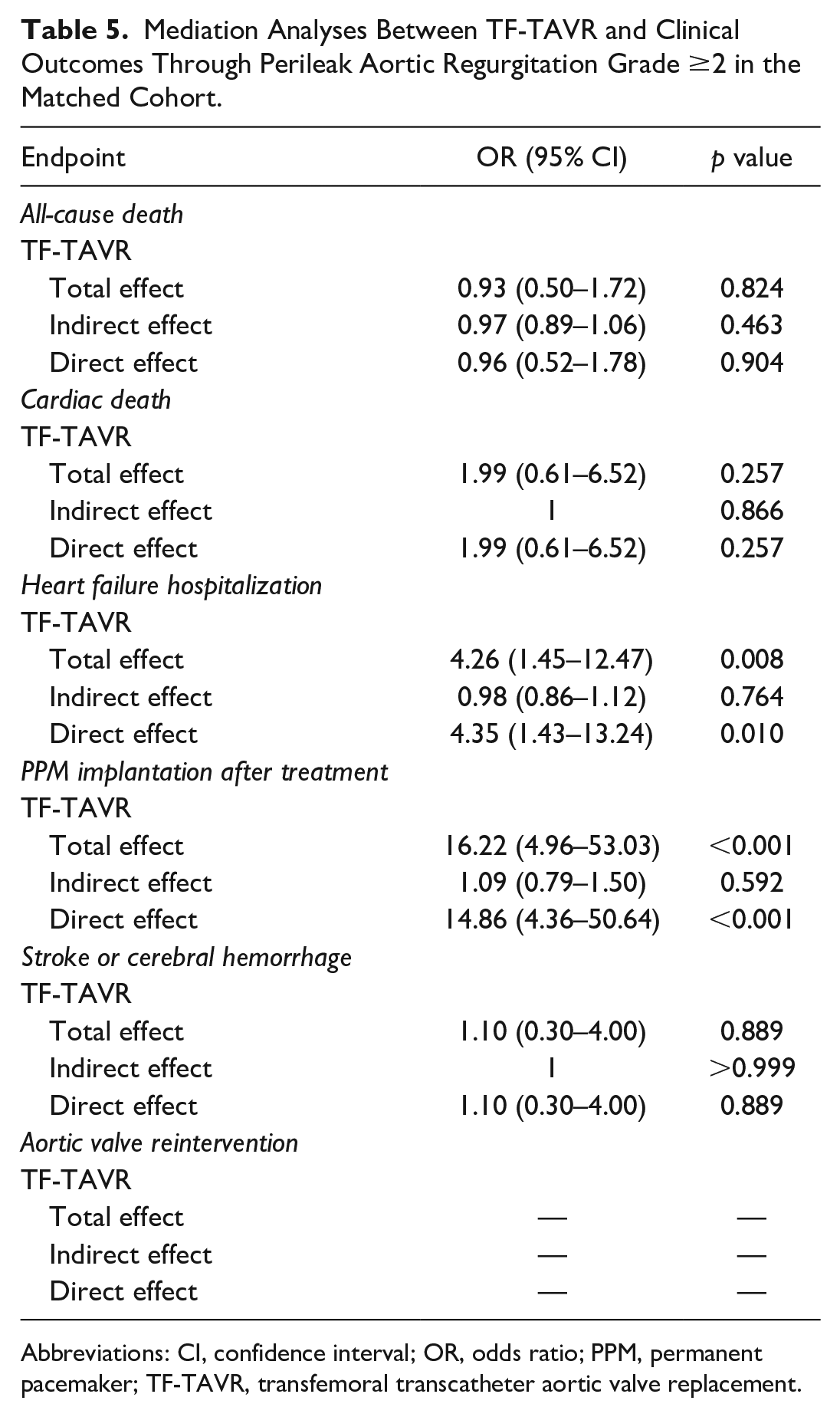
Abbreviations: CI, confidence interval; OR, odds ratio; PPM, permanent pacemaker; TF-TAVR, transfemoral transcatheter aortic valve replacement.
Discussion
To the best of our knowledge, this is the first retrospective, observational, single-center study of the outcomes of MICS-AVR versus TF-TAVR in Japan. Five primary findings can be summarized. First, both propensity score–matched cohorts in this study presented with a small body size. Second, mortality and major adverse cerebrovascular event rates were comparable between the 2 groups throughout the follow-up period. Third, TF-TAVR was associated with higher postprocedural PVL and PPM implantation rates. Fourth, the incident rate of HFH favored MICS-AVR. Finally, the TF-TAVR procedure was associated with longer hospital LOS.
After PSM, both cohorts had a small BSA of 1.47 m2. According to the OCEAN-TAVI registry, including the majority of Japanese patients undergoing TAVR, Japanese patients with AS, representing Asian countries, comprise very older women with smaller body sizes (BSA: 1.40 to 1.46 m2) compared with those in Western countries (BSA: 1.75 to 2.17 m2).13,14 Generally, a patient with a smaller BSA has a smaller aortic ring size, vascular access, and left ventricle. Thus, patients with small body sizes undergoing TF-TAVR are at potential risk for significant procedure-related complications including valve annulus rupture due to prosthetic valve oversizing and vascular complications due to mismatch between the sheath size and femoral artery diameter. 15 Only a few studies have been reported focusing on the small body–sized Japanese patients undergoing TAVR.13,14 To our knowledge, this is the first study to compare the outcomes in Asian patients undergoing MICS-AVR versus TF-TAVR.
Second, mortality and major adverse cerebrovascular event rates were similar between the 2 procedures. A recent systematic review and meta-analysis showed that short-term and intermediate-term all-cause and cardiac mortality rates were similar in low-risk and intermediate-risk patients with AS undergoing TAVR versus SAVR. 16 Subanalysis by the access route showed a trend in favor of survival following TF-TAVR compared with SAVR, without the advantage of transapical TAVR. Furthermore, a meta-analysis of TAVR versus MICS-AVR showed similar short-term all-cause mortality (risk ratio [RR] = 1.00, 95% CI: 0.55 to 1.81, I2 = 0%) but higher midterm all-cause mortality after TAVR (RR = 1.93, 95% CI: 1.16 to 3.22, I2 = 0%). 6 Stratified analysis according to the access route showed no significant difference between TF-TAVR and MICS-AVR (RR = 1.71, 95% CI: 0.86 to 3.43, I2 = 0%). Despite the longer follow-up period in our study, all-cause mortality and cardiac mortality rates were comparable in both groups, suggesting the same therapeutic potential of MICS-AVR as that of TF-TAVR. A meta-analysis comparing TAVR and MICS-AVR also showed that neurologic event rates were not significantly different between the 2 approaches. 5 Similarly, in the present study, there were similar rates of postoperative stroke and cerebral hemorrhage with an acceptable frequency between the 2 groups.
Third, the incident rates of PPM implantation and PVLs were higher in the TF-TAVR cohort. A meta-analysis of RCTs showed higher rates of PPM implantation in patients undergoing TAVR than in those receiving SAVR (HR = 2.31, 95% CI: 1.47 to 3.64, p < 0.001). 17 The mechanisms underlying TAVR-related high-degree atrioventricular blocks are complex and multifactorial. 18 Preexisting CRBBBs and the use of a first-generation CoreValve are well-recognized risk factors for TAVR-related PPM implantation. Another meta-analysis of RCTs revealed that the differences in PPM implantation frequencies were due to the TAVR valve design (HR = 3.44, 95% CI: 2.27 to 5.20, p = 0.001 in the SEVs; HR = 1.23, 95% CI: 0.99 to 1.52, p = 0.056 in the BEVs). 17 In our study, the PPM implantation rate was relatively high (15%) despite the high use of new-generation BEVs (67%).
Moreover, in our study, 20.8% of the TF-TAVR cohort had more than mild PVLs during the midterm period (mild grade, 14.6%; moderate to severe grade, 6.3%). A meta-analysis of RCTs demonstrated that PVLs favored SAVR over TAVR (HR = 6.06, 95% CI: 1.96 to 18.78, p = 0.002). 19 Furthermore, moderate or severe PVLs after TAVR were associated with mortality.20,21 Therefore, our study evaluated the impact of more than mild PVLs on clinical outcomes using a mediation analysis. However, no significant mediation effects among them were observed. Although the exact mechanism of PVLs remains unclear, the possible factors might include incomplete attachment of prosthetic valves owing to a highly calcified volume or eccentric calcification of the aortic annulus. 22 Although more than mild PVL has been commonly reported after TAVR, the diagnosis and evaluation of PVL remain challenging. The incidence of PVL after TAVR varies widely (7% to 70% for mild PVL; 0% to 24% for moderate or severe PVL).20,23 This large variation may be attributed to the differences in study populations, procedural approaches, implanted prosthetic valves, and timing and methods to evaluate PVL. Notably, the incidence of moderate or severe PVL after TAVR has decreased markedly over the years due to the use of newer-generation valves with technological advances. Moreover, a study from the large Transcatheter Valve Therapy (TVT) registry on SEVs reported that the incidence of more than mild PVL in the matched populations significantly decreased from 8.3% with first-generation valves to 5.4% with second-generation and 3.4% with third-generation valves (p = 0.032). 24 Therefore, the older-generation valves implanted during the early study period might have influenced the total incidence rates of PVL after TAVR in the present study. Future prospective evaluations, including the timing and methods to evaluate PVL, are warranted. Given the excellent low incidence of PPM implantation and PVLs following MICS-AVR in the present study, the MICS-AVR procedure might have advantages. These could include avoiding direct mechanical stress on the atrioventricular conduction system, carefully removing diseased valve leaflets and annular calcification, and appropriately positioning an optimal prosthetic valve, thereby considerably reducing these risks.
Fourth, the TF-TAVR cohort more frequently required HFH throughout the follow-up period. Although previous reports have indicated that PVLs were involved in HFH, 21 the causal mediation analysis suggested that PVLs were unlikely related to HFH in the present study. Limited to the retrospective chart review or interviews, our study included coronary ischemia and pulmonary hypertension as the primary causes of HFH in the MICS-AVR group. Whereas comorbidities including preexisting AF, hypertension, and renal failure or infections were involved as the primary causes of HFH in the TF-TAVR group.
Finally, the TF-TAVR procedure was associated with longer hospital LOS. Previous RCTs have revealed an advantage of TAVR in terms of the ICU LOS and hospital LOS over SAVR, supporting the benefit of avoiding surgical exposure and trauma.2,3 Similarly, a meta-analysis comparing TAVR with MICS-AVR using a PSM analysis showed that TAVR significantly led to faster postoperative recovery and shorter hospital LOS. 8 However, the TF-TAVR cohort required longer hospital LOS in our study (12.8 ± 7.6 days in the MICS-AVR group vs 16.2 ± 12.9 days in the TF-TAVR group, p = 0.027). The following factors might explain our results: the differences between internists and surgeons regarding their perspectives on hospital LOS and the prudence of postoperative follow-up, patient backgrounds, and other possible factors that may inhibit being discharged home. A systematic review with meta-analysis using a PSM analysis reported on hospital LOS with both MICS-AVR and TAVR groups as per US standards, suggesting a much shorter duration than that in Japan, although high heterogeneity was observed. 5 In fact, the Japanese multicenter prospective OCEAN-TAVI registry reported a longer median hospital LOS of 13 days for conventional approach TAVR, which is comparable to that noted in the present study. 25 Globally, Japan has the highest percentage of the older population aged ≥65 years, and the number of older individuals living alone is steadily increasing. Furthermore, Japan’s health care system, characterized by universal health insurance, covers the entire population living in Japan and secures access to affordable health care services. Presumably, the longer hospital LOS in both procedural approaches in Japan can be explained by the differences in the rates of aging of the population, proportions of individuals living alone, health care systems, and traditions compared with those based on US standards.
Based on the above findings, the present study strongly suggests that MICS-AVR and TF-TAVR may be equally efficient and feasible in Asian patients with AS at intermediate risk. Although recent improvements in the TF-TAVR system have made the technique easier and reduced procedure-related complications, there are certain cases anatomically unsuitable for receiving the TF-TAVR procedure, such as those with a highly tortuous aorta or iliac arteries, heavily calcified or small-caliber vessels, or long-segmental occlusions. Therefore, MICS-AVR may be the preferred alternative in such cases. Given the progressive expansion of TAVR to patients with AS at any risk and the expected application to younger patients in near future, the long-term impact of TAVR-related complications and the durability of prosthetic valves are highly likely to be new crucial issues for future research. Therefore, an individualized optimal procedural approach is warranted, depending on each patient’s background.
Study Limitations
Our study has several limitations. Since this study was a retrospective observational analysis at a single center, the patients were not randomized to a specific procedure. Although PSM analysis was performed to balance preoperative backgrounds in the present study, unmeasured confounders could have been imbalanced. The sample size was small. The difference in learning curves for each procedure may have influenced our results. In addition, the older-generation prosthetic valves represent another limitation, since newer ones with better profiles are now available.
Conclusions
This is the first retrospective observational single-center study to compare MICS-AVR and TF-TAVR in terms of outcomes in Asian patients with AS. Our study suggested that MICS-AVR may be a feasible and effective alternative approach to TF-TAVR. The TF-TAVR cohort had a considerable incidence of PPM implantation and PVLs, whereas the MICS-AVR cohort had a negligible incidence of them throughout the follow-up period. MICS-AVR could overcome the initial disadvantages observed in patients receiving SAVR and maintain the advantages of AVR in the longer outcomes, thereby showing the potential to be comparable to TF-TAVR. Future prospective RCTs are warranted to validate our results.
Supplemental Material
sj-jpg-2-inv-10.1177_15569845231205587 – Supplemental material for Minimally Invasive Surgical Versus Transcatheter Aortic Valve Replacement: A Retrospective Observational Single-Center Study in Japan
Supplemental material, sj-jpg-2-inv-10.1177_15569845231205587 for Minimally Invasive Surgical Versus Transcatheter Aortic Valve Replacement: A Retrospective Observational Single-Center Study in Japan by Hiroyuki Yamamoto, Taisuke Nakayama, Naoki Ishii and Yoshitsugu Nakamura in Innovations
Supplemental Material
sj-pdf-1-inv-10.1177_15569845231205587 – Supplemental material for Minimally Invasive Surgical Versus Transcatheter Aortic Valve Replacement: A Retrospective Observational Single-Center Study in Japan
Supplemental material, sj-pdf-1-inv-10.1177_15569845231205587 for Minimally Invasive Surgical Versus Transcatheter Aortic Valve Replacement: A Retrospective Observational Single-Center Study in Japan by Hiroyuki Yamamoto, Taisuke Nakayama, Naoki Ishii and Yoshitsugu Nakamura in Innovations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
Data collection and analysis were performed under the approval by the Institutional Review Board of Chiba-Nishi General Hospital (IRB No. TGE02118–025; January 25, 2023). Written informed consent was obtained from all patients for the publication of the study data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
