Abstract
Objective:
Extracorporeal life support (ECLS) for circulatory and/or respiratory failure is improving. Currently, invasive sternotomies or rib-spreading thoracotomies are used for central cannulation of the heart and great vessels. Although peripheral cannulation of the extremities is often used, this approach may result in immobility and unintentional dislodgement. Less invasive methods for central cannulation are needed to achieve long-term ECLS. The objective of this study was to develop 2 different minimally invasive approaches for central thoracic cannulation.
Methods:
Porcine hearts were positioned in a plastic thoracic model. An endoscopic camera and multiple endoscopic instruments were used. Both access points, uniportal (lateral) and subxiphoidal, were simulatively investigated. A novel cannulation method using purse string sutures, a custom-made endoscopic puncture tool, guidewires, and dilator-assisted cannulas was developed. Simulations were tested in a closed circuit regarding leak tightness.
Results:
The uniportal approach allowed a cannulation of the aorta, inferior vena cava, right atrium, and main pulmonary artery. Cannulation of the right branches of the pulmonary artery and vein was also possible. From the subxiphoid approach, cannulation of the aorta, main pulmonary artery, and both atria were possible. Subsequent evaluation and leakage tests revealed no damage to the surrounding structures and tightly sealed cannulation sites. The uniportal approach was also successfully performed in a human cadaver to connect the aorta and right atrium with cannulas from the subxiphoidal space.
Conclusions:
Both uniportal and subxiphoid central cannulation of potential sites for ECLS were feasible. This study encourages further investigation and potential clinical translation of minimally invasive central organ support.
Keywords
Central Message
Less invasive methods for central cannulation are needed to achieve long-term ECLS with high flow rates and safe ambulation. Minimally invasive cannulation of potential sites for ECLS were feasible from a uniportal or subxiphoid access. This study encourages further investigation of minimally invasive central organ support.
Introduction
Chronic heart and lung diseases can significantly impair quality of life1,2 and are within the top 3 causes of death worldwide. 3 Organ transplantation significantly improves quality of life and survival.4,5 However, the number of patients awaiting organ transplantation greatly exceeds the number of suitable donors.6,7 In recent years, heart and lung assist devices have improved, enabling their use in this population.5,8
Connection methods for extracorporeal life support (ECLS) devices are numerous and have varying disadvantages.9,10 Cannulas in peripheral ECLS are inserted in the extremities, which can cause immobility and create a risk of unintentional dislodgement. 11 Central thoracic cannulation is favorable to achieve long use under high flow rates, which has been demonstrated with extracorporeal ventricular assist devices (VAD). 12 However, conventional methods for central cannulation are limited to invasive sternotomy or rib-spreading thoracotomy approaches. 13
Video-assisted thoracic surgery (VATS) is performed via small incisions without rib spreading and thereby damaging surrounding structures. Therefore, VATS is associated with less pain, fewer complications, and shorter hospital stay.14,15 In thoracic surgery, uniportal VATS is commonly used for lung surgery with lateral access 16 or thymectomies with subxiphoid access. 17 Ambulation for patients who undergo VATS is even possible on the day of surgery. In specialized centers, VATS is often used. 18
Both approaches are of special interest for central cannulation of the heart and great vessels to reduce surgical trauma and facilitate a longer duration of ECLS. Furthermore, central cannulation could be used more often in existing awake and mobile patients undergoing ECLS strategies for cardiac and/or respiratory failure to reduce complications related to mechanical ventilation, sedation, and immobilization. 19
Hence, our objective for this study was to develop a minimally invasive approach for central ECLS cannulation that can be used from either right lateral uniportal or subxiphoid thoracic access. Our goals were to (1) develop a reliable cannulation method for 2 different thoracoscopic access points, (2) conduct and evaluate the method for all potential cardiac structures and great vessels in an in vitro model with porcine hearts, and (3) perform a leakage test of all cannulated structures.
Methods
A novel cannulation method was conceptualized for thoracoscopic use with minimally invasive access. The method was developed using an in vitro model with porcine hearts (including lungs) in a plastic thoracic simulator with minimally invasive access points (Fig. 1). In addition, hearts and great vessels were connected to a continuous circuit with water. Cannulation of all potential cardiac structures and great vessels were investigated from either the right lateral uniportal or subxiphoid access (Fig. 2). All trials were conducted with the same cannulation method described in the “Surgical Procedure” section. Trials were recorded for evaluation, and successfully cannulated structures were individually extracted for a leakage test.
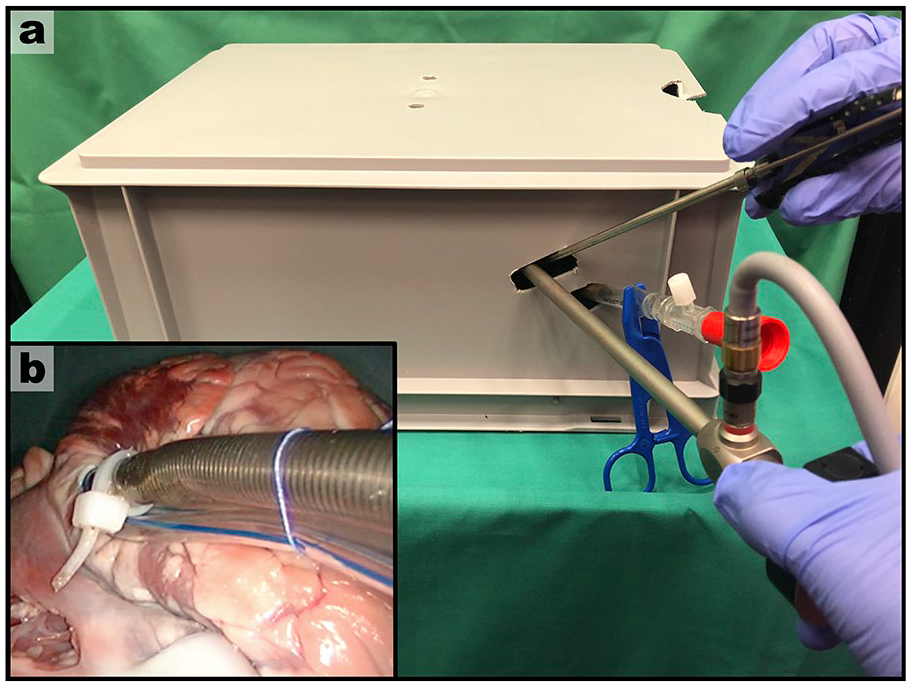
In vitro model for a novel cannulation approach: (a) thoracoscope and thoracoscopic instruments are used from a minimally invasive port; (b) successful cannulation of the right atrium.
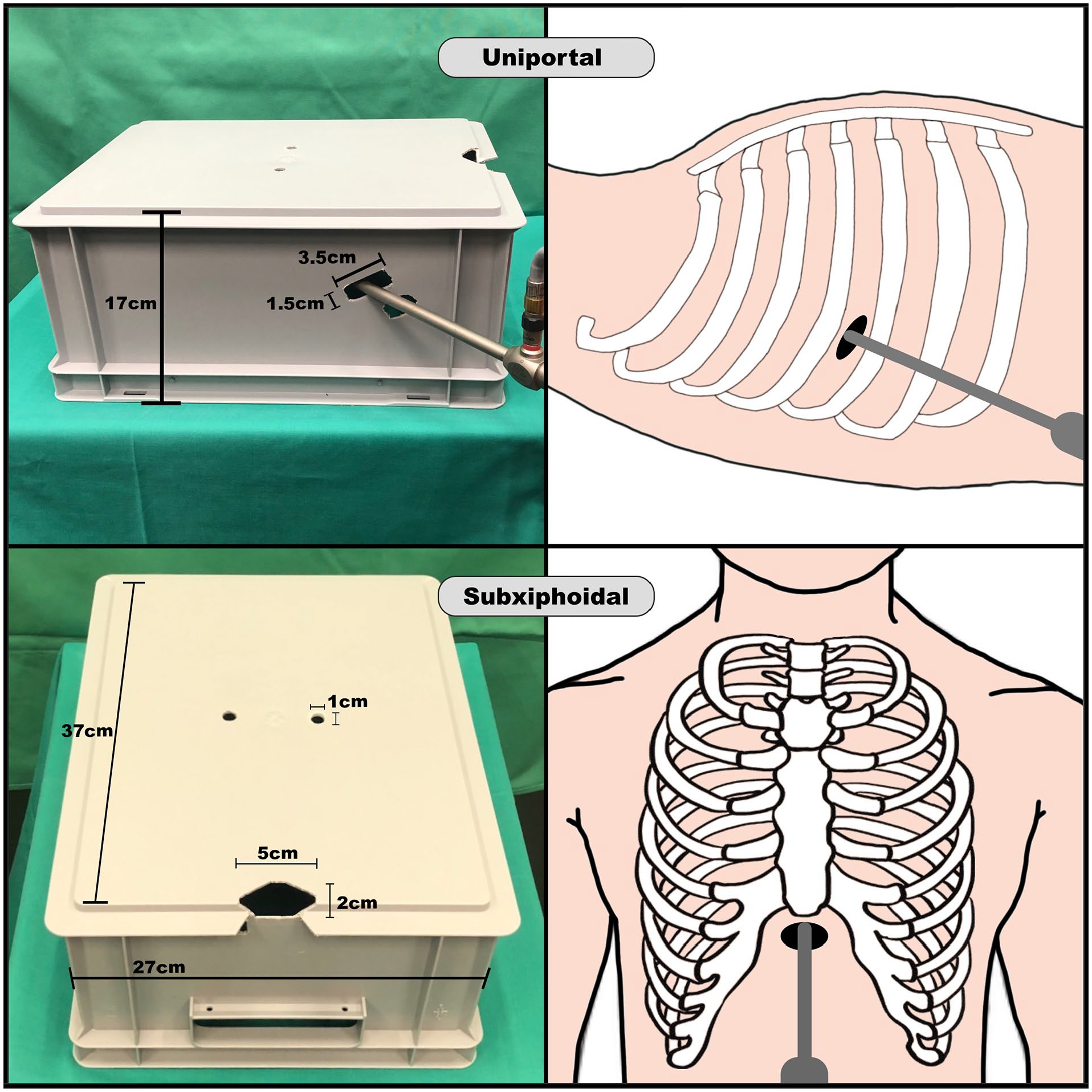
Thoracic simulator. Two minimally invasive ports are shown: a “uniportal” opening in the fourth right intercostal space and a “subxiphoid” opening.
In Vitro and Human Cadaver Model
The thoracic simulator had 2 access points that mimicked a subxiphoid opening and a lateral (non–rib spreading) uniportal opening in the fourth right intercostal space (Fig. 2). To lead the cannula outside the body, another opening under the subxiphoid and uniportal access was created. In addition, 2 openings that mimicked a right and lateral parasternal access were added for use if necessary. The simulator box dimensions were a length of 37 cm, width of 27 cm, and depth of 17 cm. The subxiphoid opening had a length of 5 cm and width of 2 cm, the uniportal opening had a length of 3.5 cm and width of 1.5 cm, and both parasternal openings had a length of 1 cm and width of 1 cm. The openings for cannulas had a length of 2 cm and width of 1.5 cm. After exploration of different cannulation sites from the uniportal and subxiphoid port, the surgical method was tested in a fresh human cadaver (Fig. 3).
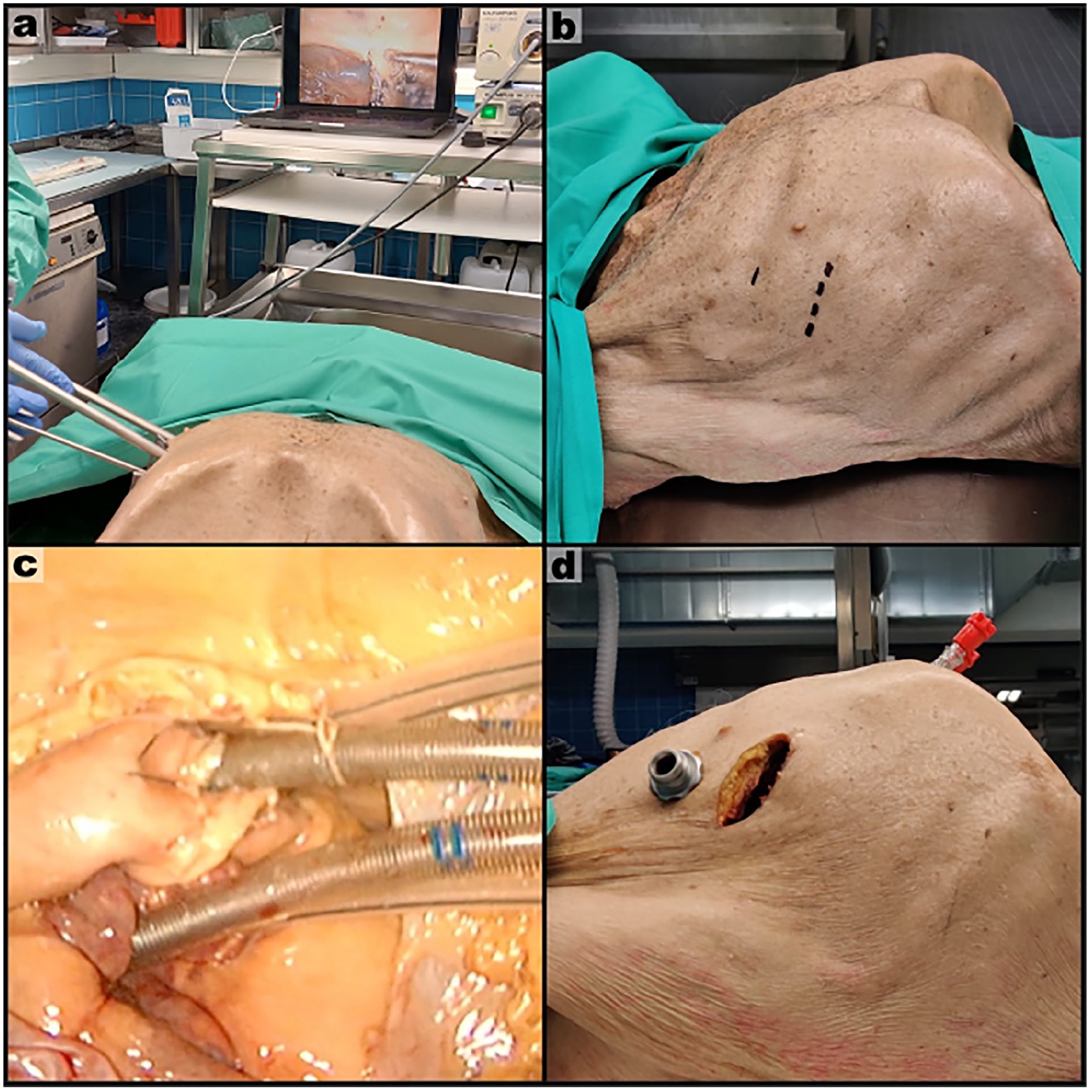
Human cadaver trial: (a) thoracoscopic setup; (b) incision lines in fourth and fifth ICS; (c) successfully cannulated aorta (top cannula) and right atrium (bottom cannula); (d) cannulas leaving the thoracic cavity via the subxiphoidal space. The fourth ICS was used for the camera port and the fifth ICS for endoscopic instruments, without the use of any rib spreaders. ICS, intercostal space.
Our thoracoscopic setup included a 30° endoscopic optic (10 mm diameter) with light source and commercially available endoscopic instruments. For surgical preparation of 1 cardiac structure or vessel, 2 polypropylene sutures (4-0, with an 18 mm needle), 2 tourniquets, and 2 artery forceps were used. For cannulation of 1 cardiac structure or vessel, we used a 23-Fr HLS cannula (Maquet, Cardiopulmonary AG, Hirrlingen, Germany), a J-tip guidewire (0.038 inches × 100 cm), and a puncture needle 1.27 × 70 mm or an aspiration needle 2 × 30 mm. Both needles were curved. To permanently secure the cannula, we used 3 suture strings (Vicryl 3-0, 45 cm) and 2 rubber pieces (0.5 × 0.5 cm).
A novel endoscopic puncture tool was designed for this study (Fig. 4). One of the curved puncture needles was connected to a flexible plastic tube, which allowed us to perform the Seldinger technique endoscopically on distances of 10 to 30 cm. In addition, cable ties that were endoscopically graspable were attached to the cannula.
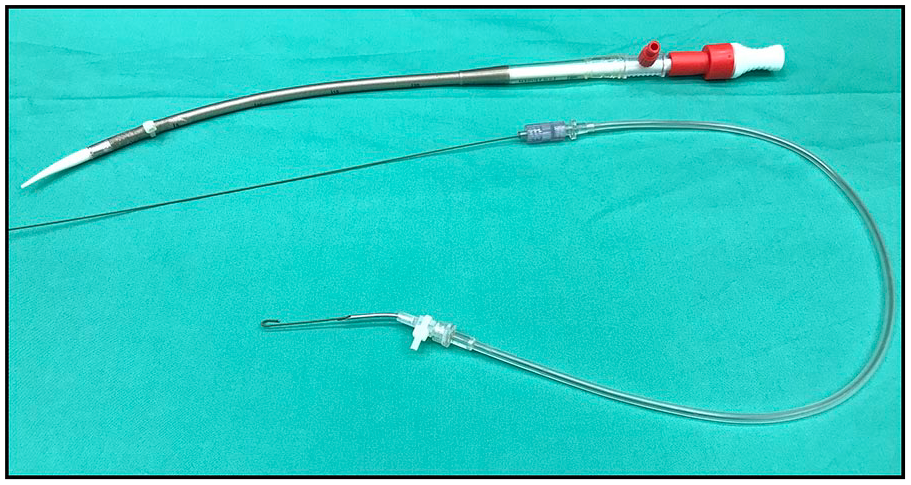
Endoscopic puncture tool containing a J-tip guidewire and (above) a cannula; both are equipped with graspable cable ties.
Surgical Procedure
The same surgical procedure was used in all cannulated structures, with the exception of the right pulmonary branch vessels, because these vessels did not require pericardial dissection but instead individual dissection of surrounding tissue. Therefore, the general steps of the right atrial cannulation via uniportal access are presented as a representation of the cannulation technique for all other structures.
The first step was to perform a central bottom-up incision of the pericardial sac without damaging the lateral pericardial vessels and nerves (Fig. 5a). We identified the right atrium and placed 2 purse string sutures at the proximal side of the right atrial appendage (Fig. 5b). Two tourniquets were inserted (Fig. 5c). The end of both tourniquets stayed outside the thorax and were clamped with 2 forceps (Fig. 5d).
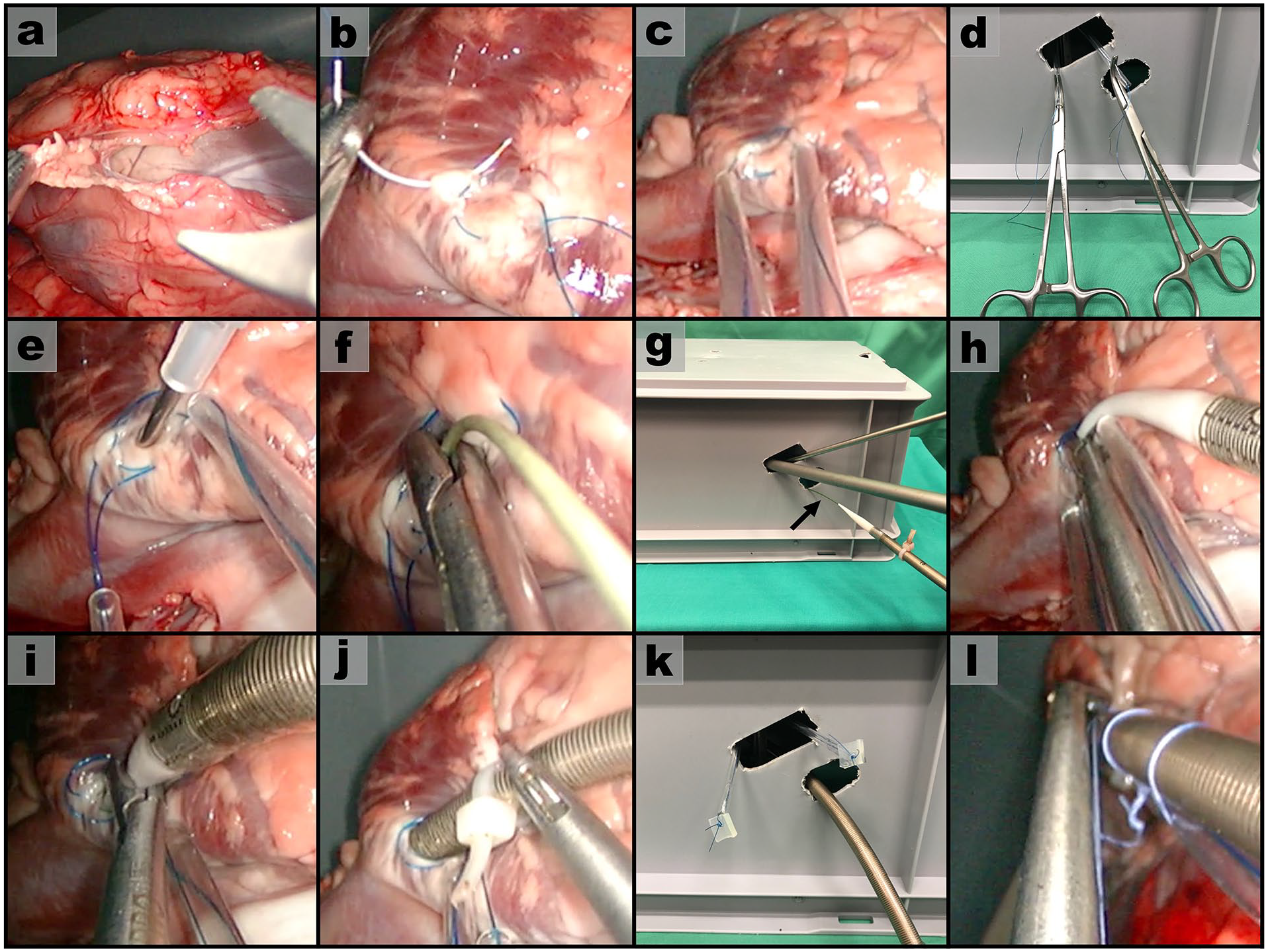
Surgical preparation: (a) incision of pericardium; (b) placing 2 purse string sutures on right atrium; (c) (d) inserting 2 tourniquets; (e) puncturing of the right atrium; (f) removing the needle and securing the guidewire; (g) (h) inserting the cannula via the guidewire, which is indicated with an arrow; (i) (j) cannula is advanced into right atrium; (k) permanently tightening of tourniquets with rubber pieces; (l) securing of cannula with 3 endoscopic knots.
The right atrium was punctured inside the purse string sutures (Fig. 5e). For all cannulations, an accessible space of 3 to 5 cm within the heart or great vessels is needed. Therefore, the curved needle is pointed toward the superior vena cava (SVC). The J-tip guidewire was advanced via the plastic tube of the needle and pushed cranially into the SVC. After a 5 cm insertion of the guidewire, we removed the needle slowly and secured the guidewire with an endoscopic instrument (Fig. 5f). The needle was removed out of the thorax, and then the guidewire was led out through an additional access in the fifth intercostal space. From this access, we inserted the cannula (with dilator) via the guidewire until it reached the securing endoscopic instrument (Fig. 5g–h).
The endoscopic instrument released the guidewire so that the cannula could be inserted into the right atrium (Fig. 5i). A practical tip for smooth insertion is to moisten and twist the cannula. The cannula was completely advanced with the help of an endoscopic instrument and graspable cable ties (Fig. 5j). We removed the dilator inside the cannula, clamped the end of the cannula, and tightened both tourniquets. The sutures of the tourniquets were tied to rubber pieces so that the purse string sutures were permanently tightened (Fig. 5k). Tourniquets were inserted into the thoracic cavity and positioned next to the cannula. To secure the cannula permanently, we used 3 knots that attached the cannula to both tourniquets (Fig. 5l). Eventually, the tip of the cannula was located at the beginning of the SVC, and drainage from the water circuit was observed.
Results
We performed 6 and 4 different cannulations of cardiac structures from the uniportal (Table 1) and subxiphoid (Table 2) access, respectively. Every single uniportal and subxiphoid trial is described and evaluated in 2 separate sections. The evaluation assessed the wall characteristics, blood pressure, distance to thoracic access, visualization, free area for suturing, moving structures, secure puncturing, and whether an additional access was needed.
Cannulations From the Uniportal Access.
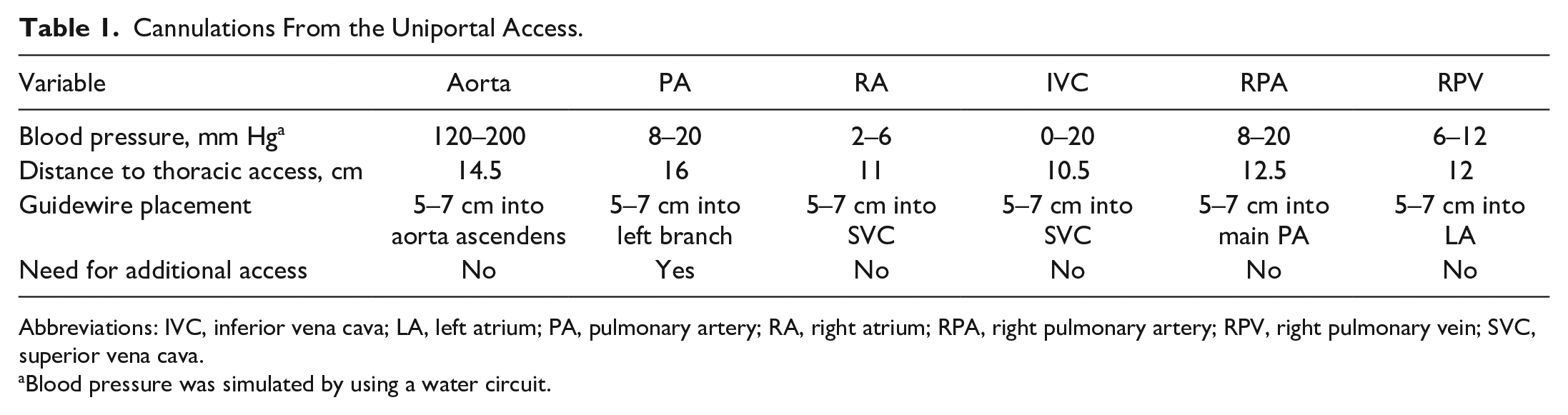
Abbreviations: IVC, inferior vena cava; LA, left atrium; PA, pulmonary artery; RA, right atrium; RPA, right pulmonary artery; RPV, right pulmonary vein; SVC, superior vena cava.
Blood pressure was simulated by using a water circuit.
Cannulations From the Subxiphoidal Access.

Abbreviations: PA, pulmonary artery; RA, right atrium; RPA, right pulmonary artery; RPV, right pulmonary vein; SVC, superior vena cava.
Blood pressure was simulated by using a water circuit.
Uniportal Access
From the uniportal approach, we successfully cannulated the aorta, inferior vena cava, right atrium, and main pulmonary artery. In addition, we performed cannulations of the right branch of the pulmonary artery and vein.
Aorta
Visualization of the aorta was limited because of the right atrial appendage. Sufficient space for suturing was available on the anterior and lateral side of the aorta. The aortic wall was thick and resilient, which facilitated the suturing step. High water pressure (120 to 200 mm Hg) was adjusted on the aorta; therefore, temporary tightening of the first purse string suture was necessary to minimize water leakage during puncturing. The guidewire could easily be pushed 5 to 7 cm into the ascending aorta.
Inferior vena cava
Good visualization was possible because of the short distance to the uniportal access. Great caution was necessary for pericardial vessels and nerves because they are located nearby. Suturing of the inferior vena cava was difficult because of a small area and thin wall. Nevertheless, stitches did not penetrate the vessel, and we recorded no fluid leakage. Puncturing was performed carefully without damaging the structures inside the vessel lumen. The guidewire was advanced easily into the SVC, and cannulation was uncomplicated.
Right atrium
Good visualization was possible because of the short distance to the uniportal access. Again, caution was needed for pericardial vessels and nerves. Suturing was performed at the proximal side of the right atrial appendage. The atrial wall was vulnerable and had a varying thickness. Puncturing and guidewire advancement was directed to the SVC, and the cannulation was uncomplicated.
Main pulmonary artery
The distance to the uniportal access was relatively high (16 cm). Only a small portion of the proximal pulmonary artery (PA) was accessible, which impaired visualization and suturing. The PA wall was moderately thick and distensible. Suturing was uncomplicated without water leakage. Puncturing and guidewire advancement was difficult because of the limited accessibility. Small adjustments were needed to direct the guidewire into the right PA branch. For cannulation, the additional parasternal access was needed, and the dilator was also advanced into the right PA branch.
Right pulmonary artery and right pulmonary vein
No bottom-up incision in the pericardial sac was needed. Access to both branches was possible by small incisions and preparation of pericardial tissue posterolateral to the SVC without damage to the pericardial vessels and phrenic nerve. Visualization of the right PA (RPA) was less optimal because it is located postero-cranially to the right pulmonary vein (RPV). Suturing of both vessels was possible. However, the RPV was more difficult to suture because of a thin wall. Puncturing was uncomplicated, and the cannulas could be advanced easily into the main PA from the RPA and left atrium (LA) from the RPV.
Subxiphoid Access
From the subxiphoid approach, a cannulation of the aorta, main PA, right atrium, and LA was possible.
Aorta
Visualization of the aorta was adequate, and sufficient space for suturing was available on the anterior side. The aortic wall was thick and resilient, which facilitated suturing. High water pressure (120 to 200 mm Hg) was adjusted on the aorta. Therefore, temporary tightening of the first purse string suture was necessary to minimize water leakage during puncturing. The guidewire can easily be pushed 5 to 7 cm into the aorta ascendens, which enabled a fluent cannulation.
Main pulmonary artery
Visualization of the PA was adequate, and sufficient space for suturing was available on the anterior side. The PA wall was moderately thick and distensible. Suturing was uncomplicated without water leakage. The guidewire and subsequently the dilator were advanced into the right branch without complications.
Right atrium
Good visualization of the anterior appendage was possible. Again, caution was needed for pericardial vessels and nerves. Suturing was performed at the proximal side of the right atrial appendage. The atrial wall was vulnerable and had a varying thickness. Puncturing and guidewire advancement were directed to the SVC, and the cannulation was uncomplicated.
Left atrium
Good visualization and suturing on the anterior LA was possible. The atrial wall was vulnerable and had a varying thickness. During puncturing, it was difficult to advance the guidewire into the left pulmonary vein. After multiple attempts, we successfully placed the guidewire and cannulated the LA. The additional parasternal port was used to assist guidewire placement and cannula advancement.
Leakage Test
To find potential tissue damages or inadequate suturing, cannulated cardiac structures were extracted, and a leakage test was performed in a continuous circuit operated with water (Fig. 6a). The cannula and cardiac structure were connected between a DP2 pump (Medos Medizintechnik AG, Heilbronn, Germany) and a reservoir to form a closed circuit. Two pressure sensors were placed before and after the DP2 pump. All cannulated cardiac structures and vessels were tested under the upper limits of its physiological blood pressure during 10-min intervals, and no leaking water was found (Table 3). To ensure the reliability of the results, sufficient extension of the heart and vessel walls was observed in all cannulated structures during the leakage test (Fig. 6b,c). The RPV could not be extracted properly and thus was not tested.
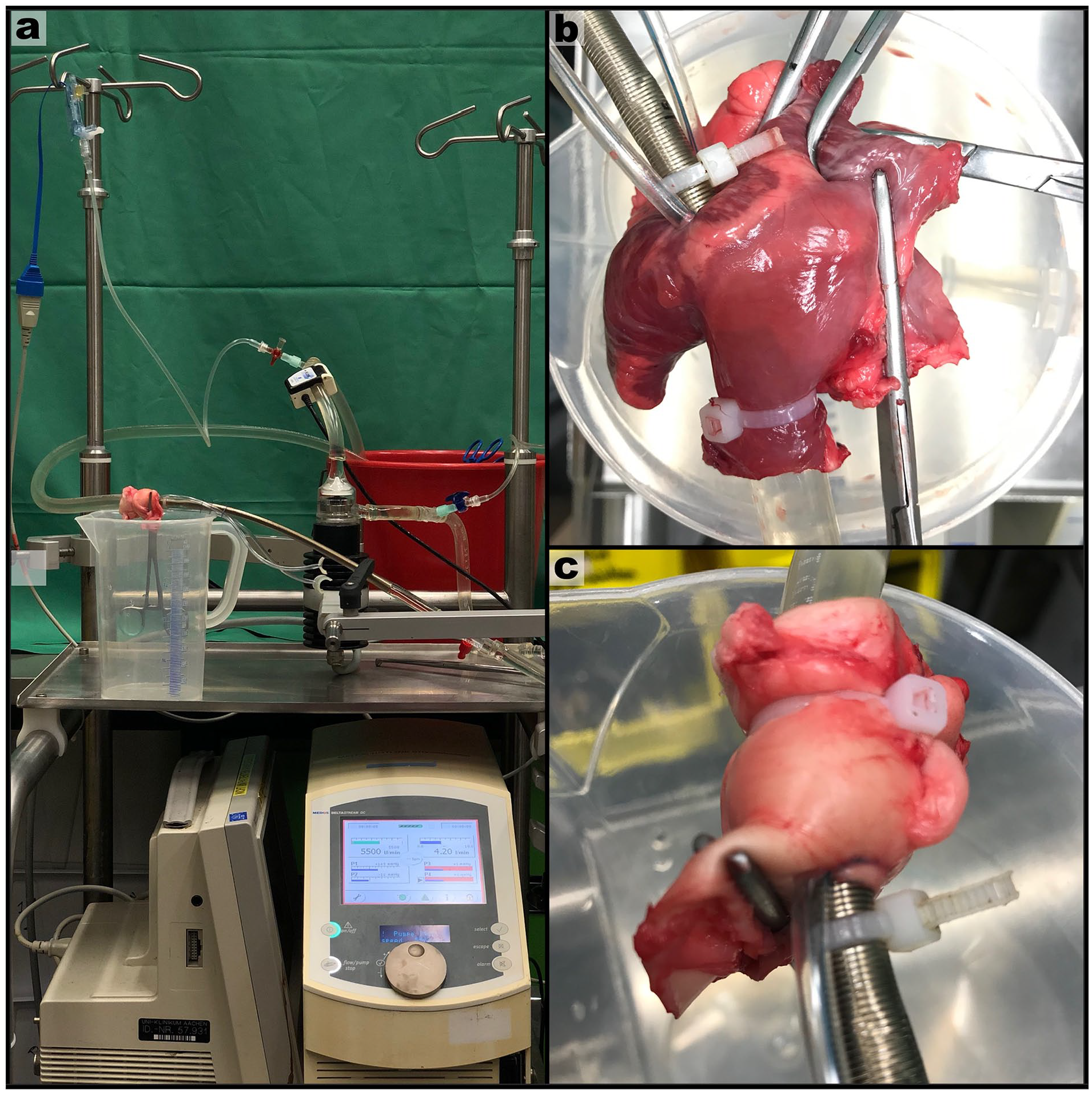
Leakage testing in a continuous water circuit: (a) the set up of our pressure test included a water pump, 2 pressure sensors, and a reservoir; (b) a pressure test of a cannulated right atrium with water inflow from the SVC and outflow via the cannula; (c) pressure test of a cannulated aorta with water inflow from the cannula and outflow via the aorta.
Leakage Testing During 10-Min Intervals.
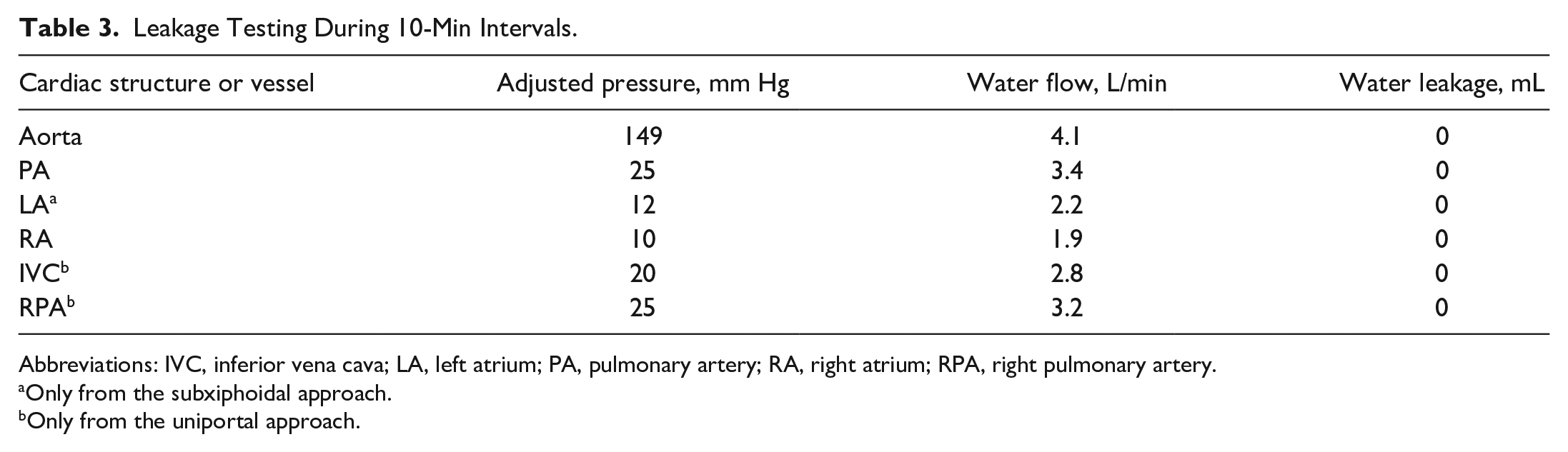
Abbreviations: IVC, inferior vena cava; LA, left atrium; PA, pulmonary artery; RA, right atrium; RPA, right pulmonary artery.
Only from the subxiphoidal approach.
Only from the uniportal approach.
Cadaver Trial
Feasibility was further examined in a human cadaver (Fig. 3a). The uniportal approach was performed to cannulate the aorta and right atrium using a 2 cm and 6 cm incision in the fourth and fifth intercostal space, respectively (Fig. 3b–c). Cannulas were inserted into the thorax at the subxiphoidal space, similar to conventional mediastinal chest tubes (Fig. 3d). The surgical procedure was successfully performed, and all steps were recorded and summarized in a video (Supplemental Video).
Discussion
In this experimental study, a novel method for minimally invasive central cannulation from a uniportal or subxiphoid thoracic access was developed. This approach is meant for different types of ECLS (veno-arterial or veno-venous) or VAD. The initial idea was to find a suitable method for future artificial lung devices or oxygenators that can be used for long recovery periods or even as destination therapy in hypoxic or hypercapnic patients. For example, in patients awaiting transplantation for whom adequate physical conditioning is a prerequisite for transplant candidacy, this method may be useful. The patient selection can be expanded to circulatory assist devices, including selective configurations such as LA to aorta or subclavian artery. Pulmonary hypertension is another possible indication. To date, pulmonary artery-to-LA configurations were possible only with invasive sternotomy. An important issue is that some patients who are already unstable are not suitable for any surgery, especially including procedures with single-lung ventilation. A possible solution would be to stabilize heart and lung function with peripheral ECLS and subsequently perform minimally invasive central cannulation in patients who show promising clinical improvement. This conversion to central cannulation would allow greater activity levels and facilitate recovery.
Application of this novel method also offers possible benefits for current cannulation strategies. A main advantage of central versus peripheral cannulation is good venous drainage and arterial return in antegrade fashion, especially in patients with atherosclerosis in peripheral vessels. 20 This approach can prevent complications such as the Harlequin or watershed phenomenon, upper body hypoxemia, left ventricular distension, and pulmonary edema.20–22 In longer therapy periods, mobilization—especially early mobilization—plays an important role. 23 However, movement restriction and cannula dislodgement risk are well-known problems in peripheral cannulation, especially in femoral vessel configurations. 24 Furthermore, physical rehabilitation requires multiple health professionals who carefully assist these patients. This assistance is not feasible in many ECLS centers and was recognized as a challenge in recent clinical trials about this issue. 25 In contrast, centrally connected VADs can be used without these problems, and even ambulatory rehabilitation is feasible. 26 In another regard, central ECLS is associated with higher transfusion requirements and revision surgery for postoperative bleeding. 27 Furthermore, a significant risk exists for thrombosis in the lungs if a full diversion from pulmonary circulation is achieved. Previous studies compared central and peripheral cannulation for ECLS, but the outcomes were controversial.21,27 However, these studies included postcardiotomy patients, a population that is ineligible for our novel method. Furthermore, the current invasive approach for central cannulation may have caused higher mortality rates. Literature on minimally invasive central cannulation compared with peripheral cannulation in non-postcardiotomy patients do not exist, but such data could provide new insights.
Studies on minimally invasive central heart cannulation are limited. Small thoracotomies in the intercostal space are commonly used in minimally invasive cardiac surgery with the possibility of ECLS cannulation. 28 In some centers, VADs are implanted via a hemisternotomy or a right minithoracotomy. 10 Furthermore, a case report by Singh et al. described long-term ECMO support with a minimally invasive central ECMO approach as bridge to heart and lung transplantation. 29 In this study, a 57-year-old patient with chronic pneumonitis and idiopathic cardiomyopathy was cannulated via a small anterior thoracotomy at the third intercostal space. This approach allowed the patient to participate in full physical mobilization and maintain physical strength for 50 days until transplantation. However, these thoracotomy approaches used retractors or chest spreaders. Moreover, significant incisions to the pericardium were performed. Our thoracic access points were not rib spreading, and we performed VATS, which is associated with less postoperative pain and better quality of life compared with a thoracotomy. 30 In addition to these advantages, incisions to the pericardium can be minimalized and closed after removal of the cannulas. Thus, this approach may be beneficial in terms of pericardial and right ventricular function.
Implementation of preexisting techniques or tools could further improve our approach. The subxiphoid access is known as a minimally invasive approach for thymectomies and lung resections or even for specific bypass surgeries.31,32 Abdellateef et al. summarized many tips and specialized techniques that are useful in subxiphoid VATS. 32 For example, they used long and curved instruments (grasper, forceps, and needle holder) or thoracoscopes with a variable range of view. 32 Suturing and cannulation of both atria will be difficult if performed in pumping hearts. Therefore, a suction device that ensures immobilization of targeted areas could be helpful. New methods are also developed from our working group, such as catheter-based devices that create a stent-based opening on vessels, which can be connected to cannulas outside.
Of particular interest are the pulmonary vessel cannulations from the uniportal access because they are the least invasive approaches. Cannulation of both the RPA and RPV was feasible without surgical operation at the heart. Furthermore, there is no need for a bottom-up incision in the pericardium, and only minor dissections of pericardial tissue posterolateral to the SVC were made. Cannulas were advanced into the main PA (from RPA) and LA (from RPV). This placement allows complete blood drainage in the PA and return via the LA. In patients awaiting isolated lung transplantation, preservation of heart function is important, and unnecessary damage should be avoided. Moreover, pulmonary vessels may nevertheless be dissected for a subsequent transplantation surgery.
Although our results add further knowledge to a new field of research, our study has limitations. Regarding experimental design, we used only isolated porcine hearts and a human cadaver. Therefore, reproducibility with heparinized blood and beating heart models must be examined. Nevertheless, high pressure levels were simulated, and Figure 6 shows more than enough extension of cardiac tissue. With regard to clinical application, the first limitation is that this procedure requires advanced VATS skills. In addition, complications during or after surgery are more difficult to handle from a minimally invasive access point. Some complications such as subacute bleeding can be resolved by VATS, which is commonly used in experienced centers.33,34 In difficult situations or severe complications, invasive sternotomy will be necessary. Furthermore, temporary peripheral cannulation could be used if oxygenation or circulation is critical. The feasibility of minimally invasive decannulation was not investigated in this study, but exploration of and experience with this technique is crucial for further development. A possible method for minimally invasive decannulation is the use of tunneled vascular prosthesis and closure systems to remove a cannula.
Conclusions
Our study is a systematic exploration of minimally invasive methods to cannulate heart and great vessels for circulatory and/or respiratory ECLS. A novel cannulation method was conceptualized, and it can be performed from 2 different access points: by a lateral uniport that is frequently used for VATS and by subxiphoid access, which is uncommon in the field of cardiothoracic surgery but offers unique advantages. Minimally invasive central cannulation of potential ECLS sites was feasible in an in vitro model. In addition, the uniportal approach was tested in a human cadaver to connect the aorta and right atrium with cannulas from the subxiphoidal space. This study encourages further investigation and potential clinical translation of minimally invasive central organ support for patients with respiratory and/or circulatory failure.
Supplemental Material
Visual abstract – Supplemental material for Minimally Invasive Central Cannulation for Extracorporeal Life Support: The Uniportal and Subxiphoid Approach
Supplemental material, sj-pptx-1-inv-10.1177_15569845221137299 for Minimally Invasive Central Cannulation for Extracorporeal Life Support: The Uniportal and Subxiphoid Approach by Koray Durak, Dana Rizk, Janina Emunds, Felix Vorwold, Sebastian Kalverkamp, Ulrich Steinseifer, Lasse Strudthoff, Jan Spillner and Flutura Hima in Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery
Footnotes
Acknowledgements
Many thanks to Earn Esten, Amine Yahiaoui, Zenel Shehu, and Gerrit Konin for alternately assisting during the surgical trials.
^
Parts of this study were presented at the ISMICS 2021 Annual Scientific Meeting from 18-20 June 2021.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the German Research Foundation (DFG): Project-Nr. 347325614. Endoscopic instruments were lent to our research group from InGraMed (Vlotho, Germany).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
