Abstract
Objective:
Transcatheter aortic valve replacement (TAVR), previously reserved for patients of intermediate to prohibitive surgical risk, has now been expanded to patients of any surgical risk with severe aortic stenosis. Bioprostheses are prone to structural valve degeneration (SVD), a progressive and multifactorial process that limits valve durability. As the population undergoing TAVR shifts toward a lower-risk and younger profile, long-term durability is a crucial determinant for patient outcomes. Our objective was to determine the incidence and risk factors of SVD at midterm follow-up in a veteran TAVR population.
Methods:
Patients undergoing TAVR at our federal facility were retrospectively evaluated for SVD and other endpoints with standardized consensus criteria. Multivariable Cox proportional hazards analysis was performed to evaluate risk factors for mortality and SVD.
Results:
From 2013 to 2020, 344 patients (median age, 78 years) underwent TAVR. Survival from all-cause mortality was 91.3% at 1 year, 75.1% at 3 years, and 61.7% at 5 years. Cumulative freedom from SVD was 98.2% at 1 year, 96.5% at 3 years, and 93.7% at 5 years. All 13 patients with SVD met hemodynamic criteria, and 1 required intervention. Median time to hemodynamic SVD was 1.04 years. Independent risk factors for SVD included age (hazard ratio [HR] = 0.92, 95% confidence interval [CI]: 0.86 to 0.99) and valve size (HR = 0.19, 95% CI: 0.04 to 0.89).
Conclusions:
SVD was evident at a low but detectable rate at 5-year follow-up. Further understanding of TAVR biomechanics as well as continued longer-term follow-up will be essential for informing patient-specific risk of SVD.
Central Message
TAVR durability remains unknown. We showed that structural valve degeneration after TAVR occurred at a low rate at midterm follow-up. Whether elevated mean gradients resolve with or without anticoagulation will be important to understand TAVR durability and inform valve choice for specific patients.
Introduction
Transcatheter aortic valve replacement (TAVR), previously reserved for patients of high or prohibitive surgical risk, was approved by the United States Food and Drug Administration for patients of any surgical risk with severe aortic stenosis (AS) in August 2019. 1 As the population undergoing TAVR shifts toward younger and lower-risk patients, understanding valve durability has become crucial. TAVR in low-risk AS has proven to be effective with an excellent short-term safety profile.2–4 However, both TAVR and surgical AVR (SAVR) bioprostheses are prone to structural valve degeneration (SVD), a degenerative process mediated by leaflet calcification that leads to valve dysfunction and limited durability. 5 TAVR valves require mounting bioprosthetic valve leaflets into an expandable stent for vascular delivery, and they differ from SAVR valves. Thinner TAVR leaflets with variable anatomic deployment can theoretically potentiate SVD.6,7 Rates of SVD in the SAVR literature, which vary depending on definitions and follow-up periods, range from 2.4% to 31%.8–10
Although TAVR was initially described in 2002, limited midterm durability data exist due to the very low survival rates of the original elderly cohorts. 11 Only since 2017 have standardized definitions of SVD been introduced for both TAVR and SAVR. 12 Veterans Affairs Medical Centers (VAMC) do not submit TAVR outcomes to the Society of Thoracic Surgeons (STS)/American College of Cardiology (ACC) Transcatheter Valve Therapy (TVT) registry, instead reporting outcomes to Veterans Affairs Clinical Assessment, Reporting, and Tracking (CART) system for interventional cardiology and VA Surgical Quality Improvement Program (VASQIP) for surgery. We have previously demonstrated successful outcomes of TAVR procedures at our federal facility during the early evolution of our program.13–15 Thus far, TAVR clinical outcomes of mortality and rehospitalization at VAMCs compared favorably with benchmark outcome data from the TVT registry. 16 Given the limited TAVR SVD and durability data, particularly in the VAMC population, our objectives were to evaluate the incidence of SVD in our TAVR patients according to standardized definitions 12 and examine risk factors that predict SVD.
Methods
The study population comprised 344 consecutive patients who underwent transarterial TAVR from November 2013 to June 2020 at our federal facility, excluding only transapical TAVR patients. We reviewed baseline, procedural, and follow-up data focusing on outcomes, survival, and durability. Patient and procedural characteristics were obtained from the Veterans Affairs Computerized Patient Record System, including cardiac catheterizations, echocardiograms, notes, and surgical operative reports. Preoperative and procedural characteristics were collected and coded according to the STS/ACC TVT Registry. Postoperative outcomes were reported according to the updated Valve Academic Research Consortium–3 (VARC-3) criteria. 17 SVD and bioprosthetic valve failure (BVF) were determined according to the European Association of Percutaneous Intervention consensus definitions. 12 SVD was defined as moderate or severe hemodynamic versus morphologic SVD. Moderate SVD includes mean transprosthetic gradient ≥20 mm Hg and <40 mm Hg or change from baseline of ≥10 mm Hg and <20 mm Hg, or moderate intraprosthetic aortic regurgitation (AR), new or worsening (>1+/4+) from baseline. Severe SVD includes mean transprosthetic gradient ≥40 mm Hg or change from baseline of ≥20 mm Hg, or severe intraprosthetic AR, new or worsening (>2+/4+) from baseline. Morphologic SVD includes leaflet integrity abnormality (tear or flail with AR), leaflet structure abnormality (thickening or calcification with AS or AR), leaflet function abnormality (impaired mobility causing AS/AR), or strut/frame abnormality (fracture). BVF was defined at autopsy with bioprosthetic valve dysfunction likely related to cause of death, valve-related death, repeat intervention (valve-in-valve implantation, paravalvular leak [PVL] closure, or SAVR) after confirmed bioprosthetic valve dysfunction, or severe hemodynamic SVD. Given that the study began with a high-risk/inoperable cohort and expanded to patients of any surgical risk, earlier TAVR patients from November 2013 to May 2016 were compared with recent TAVR patients from May 2016 onward. This study was approved by the Institutional Review Boards at the University of California San Francisco and San Francisco VAMC (CHR #11-06811, approval #271623, January 25, 2020).
Statistical Analysis
Continuous variables are reported as mean ± standard deviation (SD) or median (interquartile range [IQR]). Categorical variables are reported as number and percentage of total. Comparisons for categorical variables were analyzed using the Fisher’s exact test. Comparisons for continuous variables were analyzed using the t test for variables with normal distribution and the Mann–Whitney U test for variables that were not normally distributed. The Anderson–Darling test was used to determine whether the sample of data fit a normal distribution. Univariate Cox proportional hazards analysis was used to identify the factors associated with cumulative mortality and SVD. A P value <0.05 was considered statistically significant. In the multivariable Cox proportional hazards analysis, only risk factors that demonstrated statistical significance from the univariate analysis were selected. Hazard ratios (HRs) from the multivariable analysis are reported with 95% confidence intervals (CIs). Survival from death and from SVD were assessed using the Kaplan–Meier method, with curves plotted with the 95% CI. All analyses were performed using MATLAB R2019b computing environment (MathWorks, Natick, MA, USA).
Results
Patient and Procedural Data
From November 2013 to June 2020, 344 veterans (97.7% male) who underwent TAVR at the San Francisco Veterans Affairs Health Care System were retrospectively analyzed. Follow-up ranged from 168 to 2,557 days. The baseline and procedural characteristics of the study population are summarized in Table 1. The mean age at TAVR procedure was 78.1 ± 8.6 years. A large proportion were symptomatic, with 303 patients (88.1%) exhibiting New York Heart Association (NYHA) Functional Class III or IV symptoms. Mean STS mortality risk score for the entire cohort was 3.9% as calculated using the 4.20 version of the STS calculator. A total of 229 patients (66.6%) were considered low risk (<4%), 78 patients (22.7%) were intermediate risk (4% to 8%), and 37 patients (10.8%) were high risk (>8%). Baseline pre-TAVR permanent pacemakers or implantable cardioverter defibrillators were present in 40 patients (11.6%). Implanted valves included 16 (4.7%) CoreValve (Medtronic, Dublin, Ireland), 50 (14.5%) CoreValve Evolut R (Medtronic), 16 (4.7%) CoreValve Evolut Pro (Medtronic), 14 (4.1%) SAPIEN (Edwards Lifesciences, Irvine, CA, USA), 31 (9.0%) SAPIEN XT (Edwards Lifesciences), and 217 (63.1%) SAPIEN 3 (Edwards Lifesciences; Table 2). One patient underwent a simultaneous bioprosthetic aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction (BASILICA) during the TAVR procedure. Valve-in-valve procedures made up 9.0% of TAVRs (n = 31), of which 30 were TAVR-in-SAVR and 1 was TAVR-in-TAVR.
Patient Demographics.
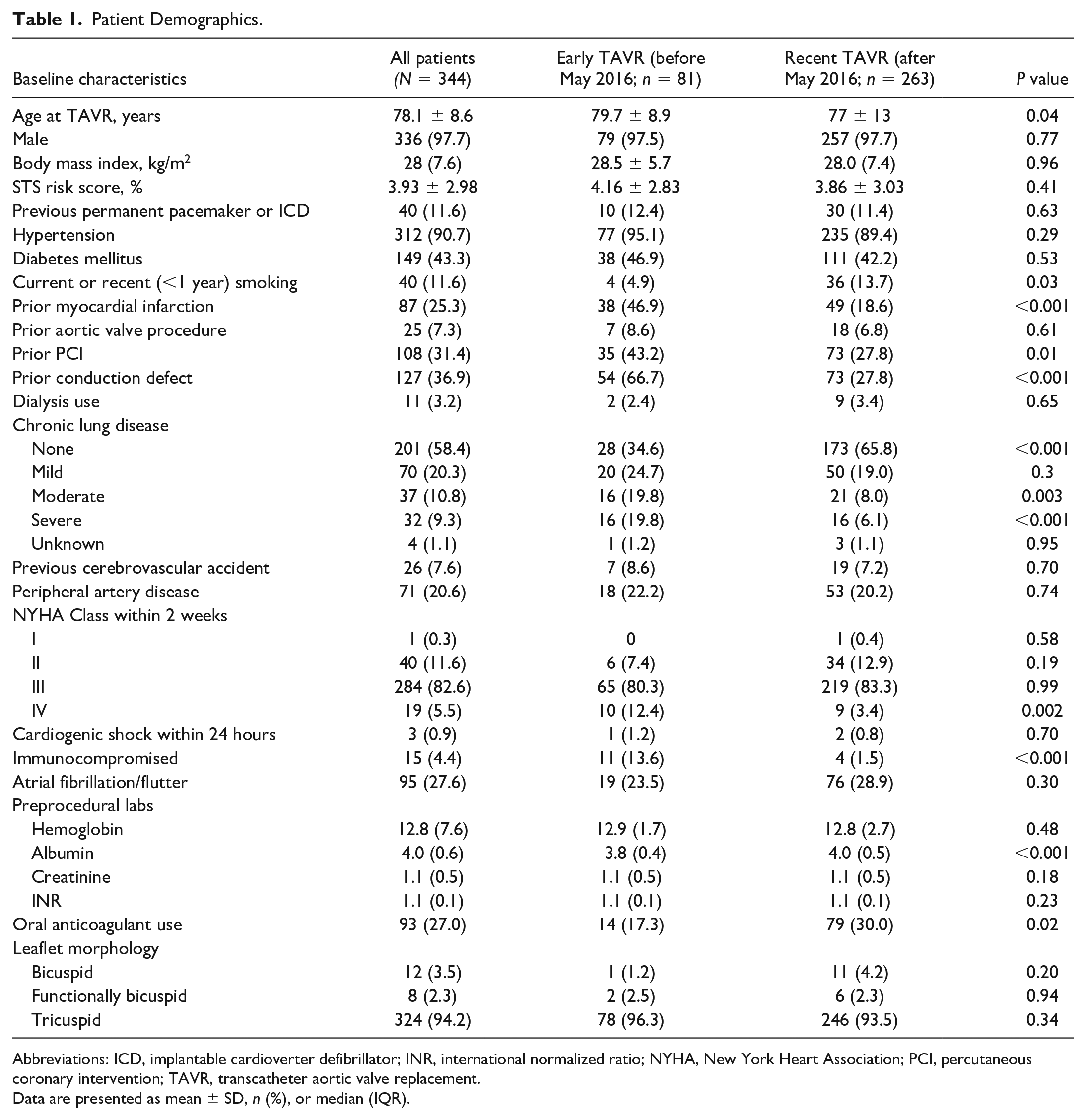
Abbreviations: ICD, implantable cardioverter defibrillator; INR, international normalized ratio; NYHA, New York Heart Association; PCI, percutaneous coronary intervention; TAVR, transcatheter aortic valve replacement.
Data are presented as mean ± SD, n (%), or median (IQR).
Procedural Characteristics.
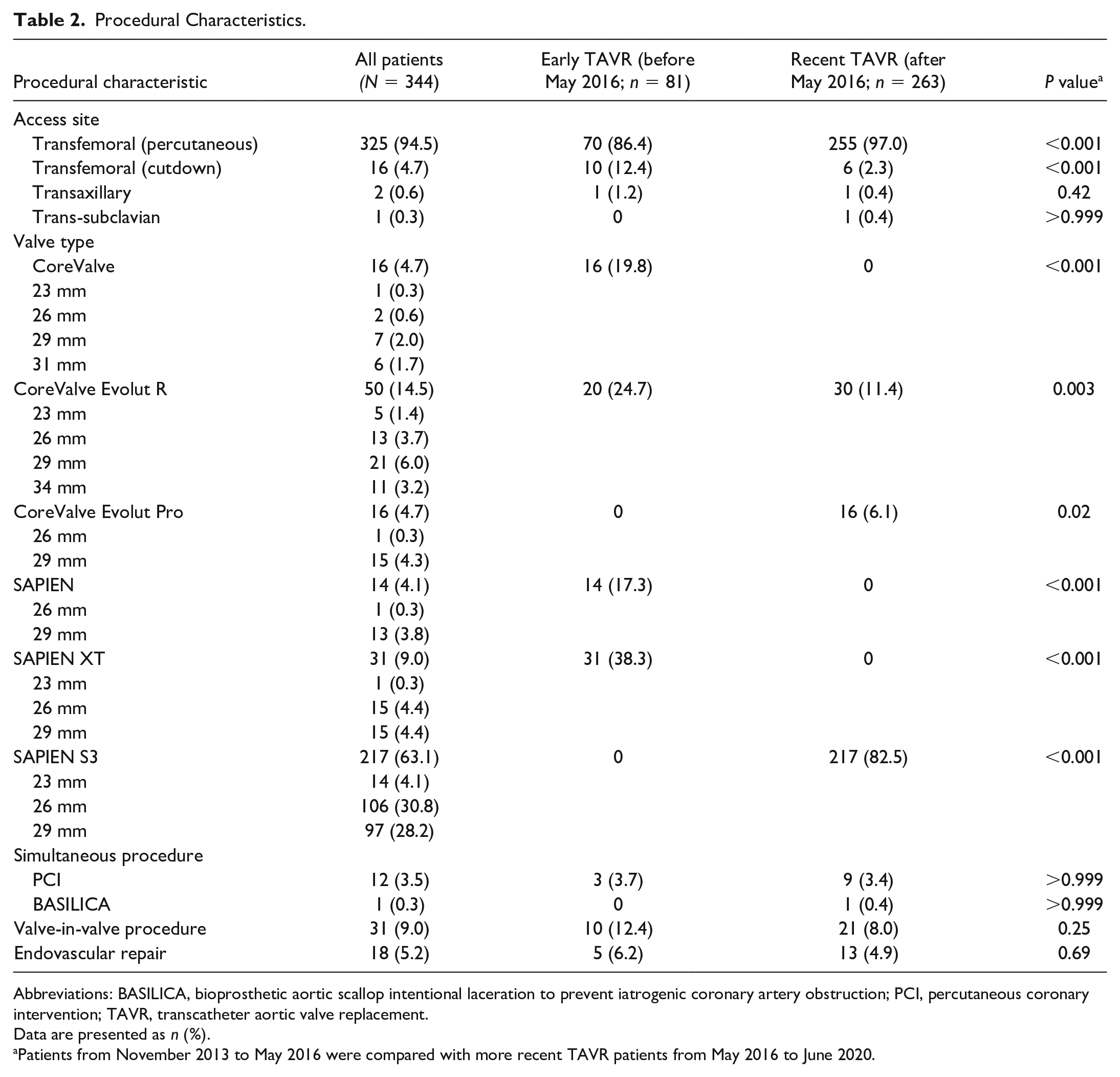
Abbreviations: BASILICA, bioprosthetic aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction; PCI, percutaneous coronary intervention; TAVR, transcatheter aortic valve replacement.
Data are presented as n (%).
Patients from November 2013 to May 2016 were compared with more recent TAVR patients from May 2016 to June 2020.
First- and second-generation valves (CoreValve, CoreValve Evolut R, SAPIEN, and SAPIEN XT) were used in early TAVR, and the second- and third-generation valves (CoreValve Evolut R, CoreValve Evolut Pro, and SAPIEN 3) were used in recent TAVR. Compared with earlier TAVR patients from November 2013 to May 2016, recent TAVR patients from May 2016 onward had significantly lower percentages of prior myocardial infarction (P < 0.001), percutaneous coronary intervention (P = 0.01), moderate (P = 0.003) or severe chronic lung disease (P < 0.001), severe NYHA classification (P = 0.002), and immunocompromised state (P < 0.001). Compared with the early TAVR patients, the recent cohort also had lower preprocedural albumin (P < 0.001) and higher use of oral anticoagulation (P = 0.02). The recent TAVR procedures had a statistically significantly higher percentage of percutaneous transfemoral access (P < 0.001) and lower percentage of surgical cutdown transfemoral access (P < 0.001) than the early TAVR procedures.
Outcomes
Midterm outcomes are summarized in Table 3. Death within 30 days occurred in 4 patients (1.2%), which occurred on post-procedure days 8, 11, 15, and 24 (mean of 14.5 days). The most common complication within 30 days was a conduction disturbance (n = 109, 31.7%). There were 54 patients (15.7%) who required a permanent pacemaker within 30 days among whom 8 had self-expanding valves (14.8%) and 46 had balloon-expanding valves (85.2%). VARC-3 vascular complications within 30 days occurred in 11 patients (3.2%), all of which were minor and not major. Cardiac structural complications occurred in 5 patients, all of which were major including left ventricular perforation (n = 3, 0.9%), right ventricular perforation (n = 1, 0.3%), and new pericardial effusion with tamponade (n = 1, 0.3%). Aortic reintervention was required within 30 days for 2 patients (0.6%); 1 patient required a redo TAVR for device migration, while the other required a redo valve-in-valve TAVR for severe PVL.
Outcomes of Veterans Undergoing TAVR at the San Francisco Veterans Affairs Medical Center.
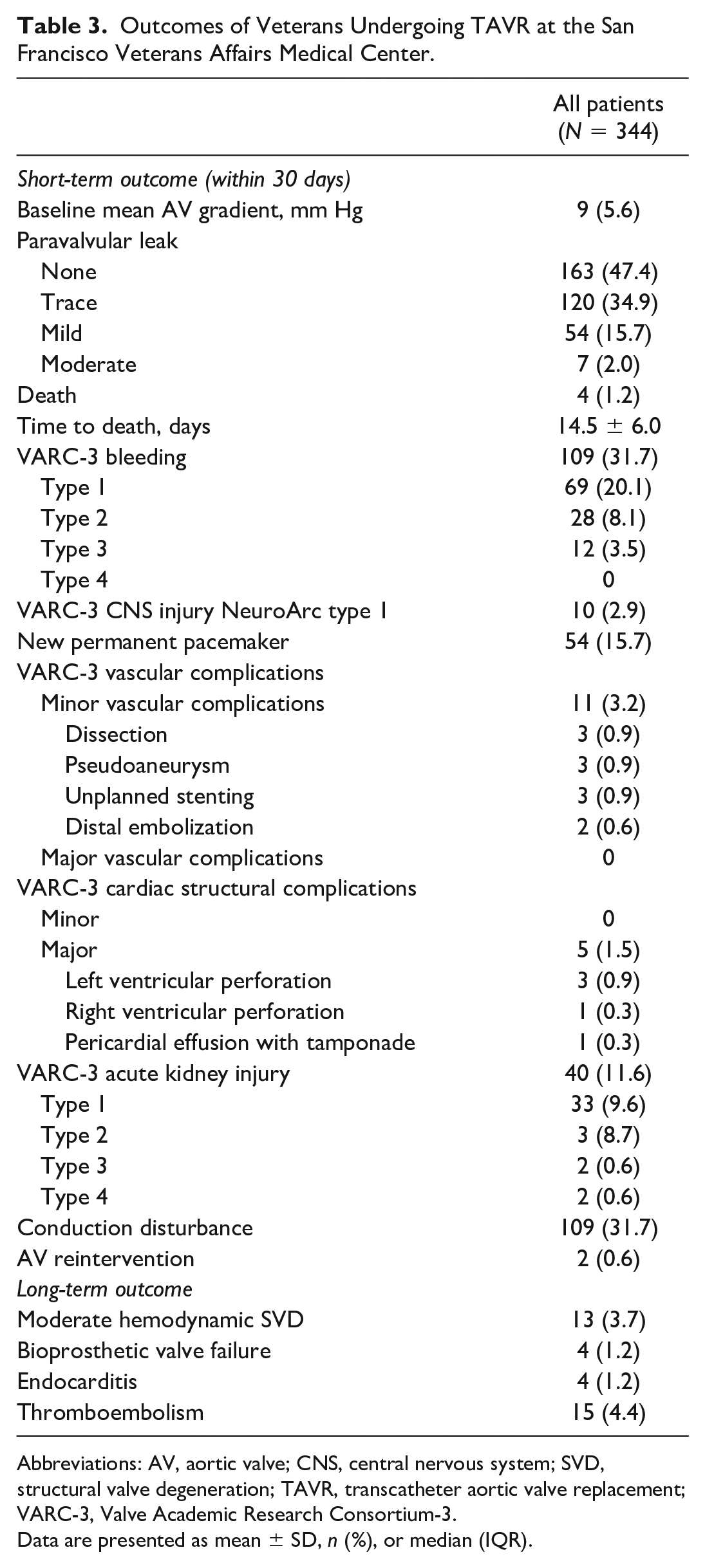
Abbreviations: AV, aortic valve; CNS, central nervous system; SVD, structural valve degeneration; TAVR, transcatheter aortic valve replacement; VARC-3, Valve Academic Research Consortium-3.
Data are presented as mean ± SD, n (%), or median (IQR).
Follow-Up
Follow-up echocardiography was performed in 322 patients (93.6%) using STS/ACC TVT recommendations. Kaplan-Meier survival curves for all-cause mortality and SVD are shown in Figure 1 and Figure 2, respectively. Survival from all-cause mortality was 91.3% (95% CI: 88.7% to 94.5%) at 1 year, 75.1% (95% CI: 69.8% to 80.3%) at 3 years, and 61.7% (95% CI: 54.5% to 68.9%) at 5 years. Cumulative freedom from SVD was 98.2% (95% CI: 96.8% to 99.6%) at 1 year, 96.5% (95% CI: 94.2% to 98.7%) at 3 years, and 93.7% (95% CI: 89.0% to 98.3%) at 5 years.
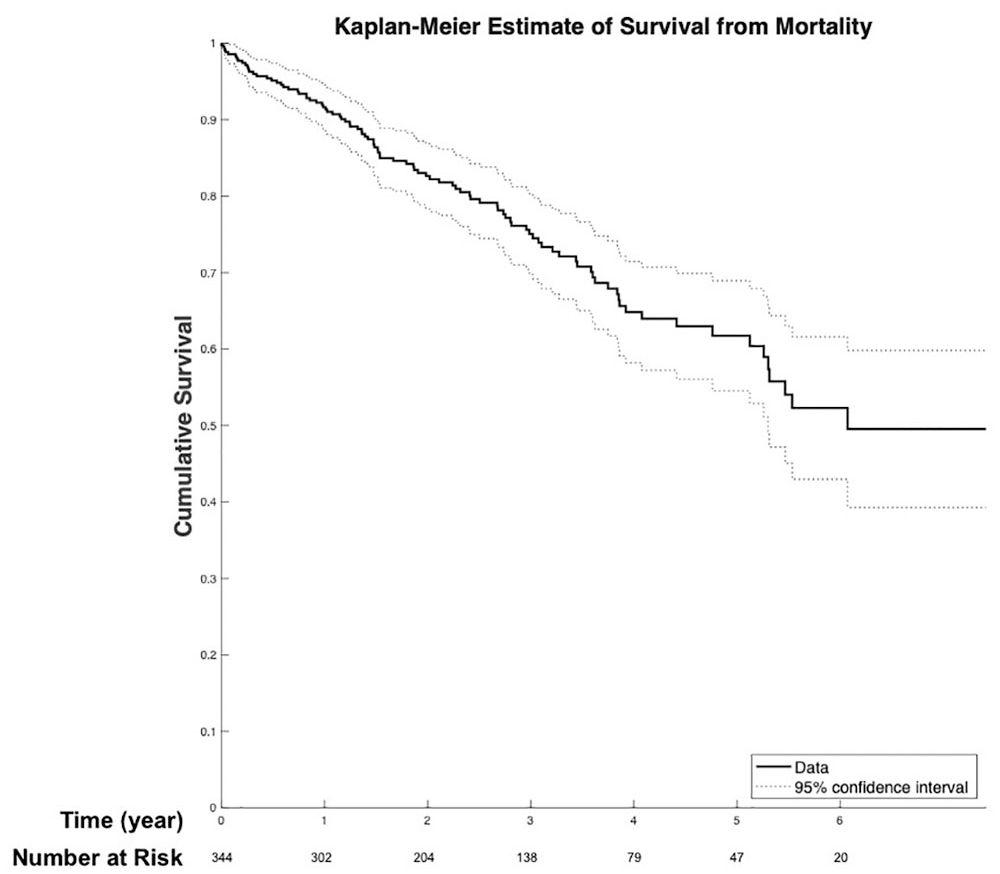
Survival from all-cause mortality was 91.3% (95% CI: 88.7% to 94.5%) at 1 year, 75.1% (95% CI: 69.8% to 80.3%) at 3 years, and 61.7% (95% CI: 54.5% to 68.9%) at 5 years. CI, confidence interval.
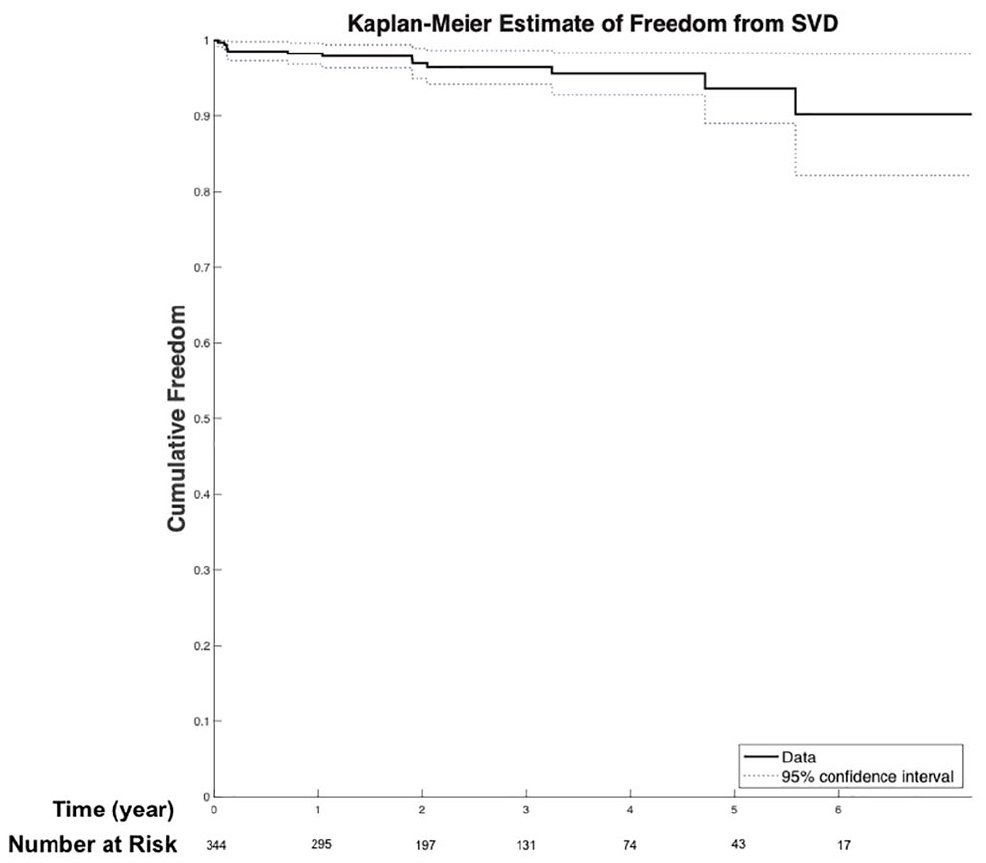
Cumulative freedom from SVD was 98.2% (95% CI: 96.8% to 99.6%) at 1 year, 96.5% (95% CI: 94.2% to 98.7%) at 3 years, and 93.7% (95% CI: 89.0% to 98.3%) at 5 years. CI, confidence interval; SVD, structural valve degeneration.
Durability
Thirteen patients (3.8%) met criteria for SVD, all of whom met hemodynamic criteria, with mean gradients exceeding 20 mm Hg. The median time to SVD was 1.04 (IQR, 1.9) years, with mean aortic gradients increasing in the first 5 years of follow-up (Fig. 3). Among patients who developed SVD, the mean age was 72.1 years (range, 57 to 84 years) at the time of TAVR. Among the 13 patients with SVD, 5 were implanted with first-generation valves (4 SAPIEN and 1 CoreValve), 2 with second-generation valves (both Corevalve Evolut R), and 6 with third-generation valves (all SAPIEN S3). In total, 1 valve was 23 mm, 10 valves were 26 mm, 1 valve was 29 mm, and 1 valve was 31 mm. Among patients who had SVD, 10 had balloon-expandable valves (76.9%) and 3 had self-expandable valves (23.1%). Among 13 patients with SVD, 4 (31%) underwent valve-in-valve (TAVR-in-SAVR) procedures, and the remaining 9 patients (69%) underwent native-valve TAVR. Among 331 patients who did not have SVD, 252 had balloon-expandable valves (76.1%) and 79 had self-expandable valves (23.9%), with no significant difference between the groups (P = 0.948). Among the 13 patients with SVD, 5 had evidence of patient-prosthetic mismatch (PPM), with 4 patients having moderate PPM (iEOA 0.66 to 0.85) and 1 patient having severe PPM (iEOA <0.65). The remaining 8 patients had no/mild PPM (iEOA >0.85). In the 4 valve-in-valve cases, 1 had no/mild PPM, 2 had moderate PPM, and 1 had severe PPM. The valve-in-valve implantations included the following: 26 mm Edwards SAPIEN in a 25 mm Carpentier-Edwards Paramount bovine valve, 23 mm Medtronic CoreValve Evolut R in a 23 mm Sorin Mitroflow valve, and 26 mm Medtronic CoreValve Evolut R in a 25 mm Carpentier-Edwards MagnaEase valve. The patient with the SAPIEN TAVR-in-TAVR eventually required reoperative SAVR with TAVR removal to correct SVD using an Edwards 25 mm Magna surgical bioprosthesis.
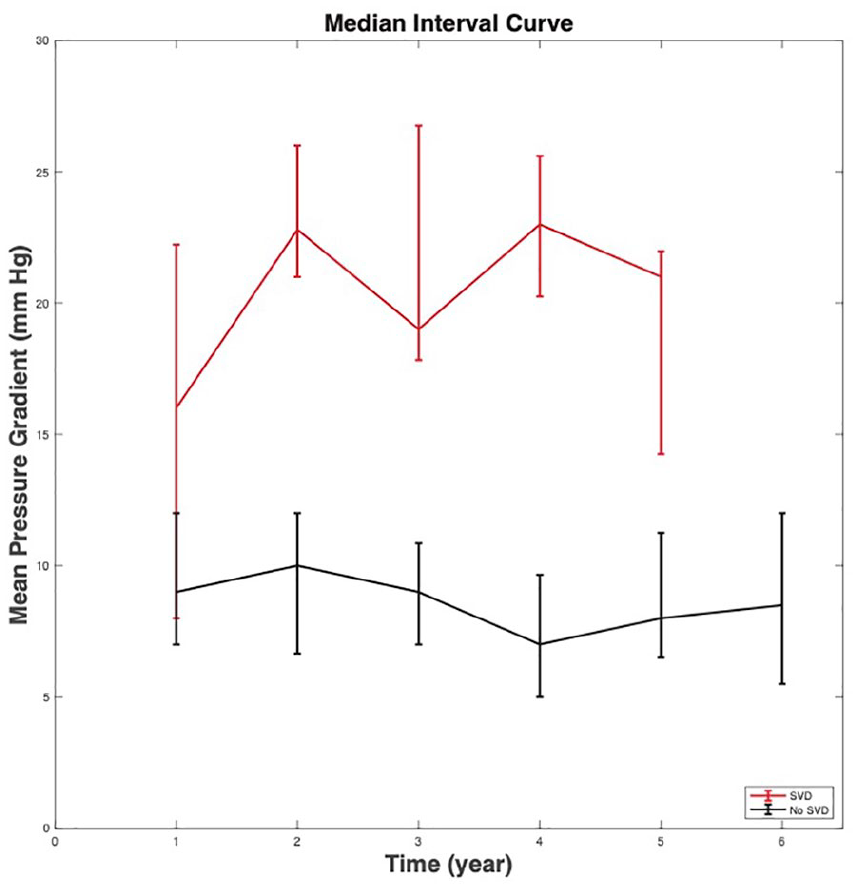
Median and interquartile range of the mean aortic valve gradient of patients with structural valve degeneration (n = 13) compared with patients without structural valve degeneration (n = 331) over time. Echocardiographic data were censored for patients who received treatment or intervention.
Three patients with SVD died during follow-up, 1 from complications of hepatocellular carcinoma and decompensated cirrhosis, 1 from ventricular fibrillation cardiac arrest, and 1 from complications of a stroke. The 9 remaining living patients without surgical reintervention have not had clinical sequelae and continue to be followed with their elevated gradients. One patient subsequently developed a decrease in gradients due to worsening heart failure.
Of the 13 patients with SVD, 2 were on oral anticoagulation at the time they met criteria for SVD (1 on apixaban and 1 on warfarin), and the remaining 11 patients were either on aspirin only (n = 7) or aspirin plus clopidogrel (n = 4). Two patients had clinical valve thrombosis (CVT) diagnosed on 4-dimensional electrocardiogram-gated computed tomography angiography (CTA) and were started on anticoagulation. One of these patients also required percutaneous treatment of moderate PVL; however, with chronic warfarin anticoagulation, the mean aortic valve gradient decreased from 36 mm Hg to 12 mm Hg. The other patient had CVT and began anticoagulation but has yet to have a follow-up echocardiogram.
A total of 4 patients (1.2%) met criteria for BVF, 2 of whom also met criteria for SVD. Of those meeting criteria for both BVF and SVD, 1 met criteria for severe hemodynamic SVD and ultimately required SAVR, and 1 developed moderate SVD and significant PVL requiring redo TAVR-in-TAVR. Among the remaining 2 BVF patients, 1 developed severe PVL requiring percutaneous closure, and 1 had superior dislodgement of a self-expandable valve requiring snaring of the migrated valve and a redo TAVR. In total, 2 had self-expanding valves and 2 had balloon-expanding valves.
Risk Correlates
Table 4 and Table 5 show the patient risk factors associated with all-cause mortality and SVD. On univariate analysis, preoperative risk factors of age, body mass index, conduction defect, peripheral arterial disease, atrial fibrillation/flutter, chronic lung disease, NYHA class, hemoglobin, and albumin were associated with mortality, but when included in the multivariable analysis, only atrial fibrillation/flutter (HR = 1.72, 95% CI: 1.06 to 2.78), chronic lung disease (HR = 1.26, 95% CI: 1.03 to 1.54), and preprocedural albumin (HR = 0.34, 95% CI: 0.19 to 0.60) were independent risk factors. On univariate analysis, age, valve-in-valve implantation, and valve size were factors associated with SVD, but in the multivariable analysis, only age at TAVR (HR = 0.92, 95% CI: 0.86 to 0.99) and valve size (HR = 0.19, 95% CI: 0.04 to 0.89) were independent risk factors.
Association of Risk Factor Variables with Mortality.
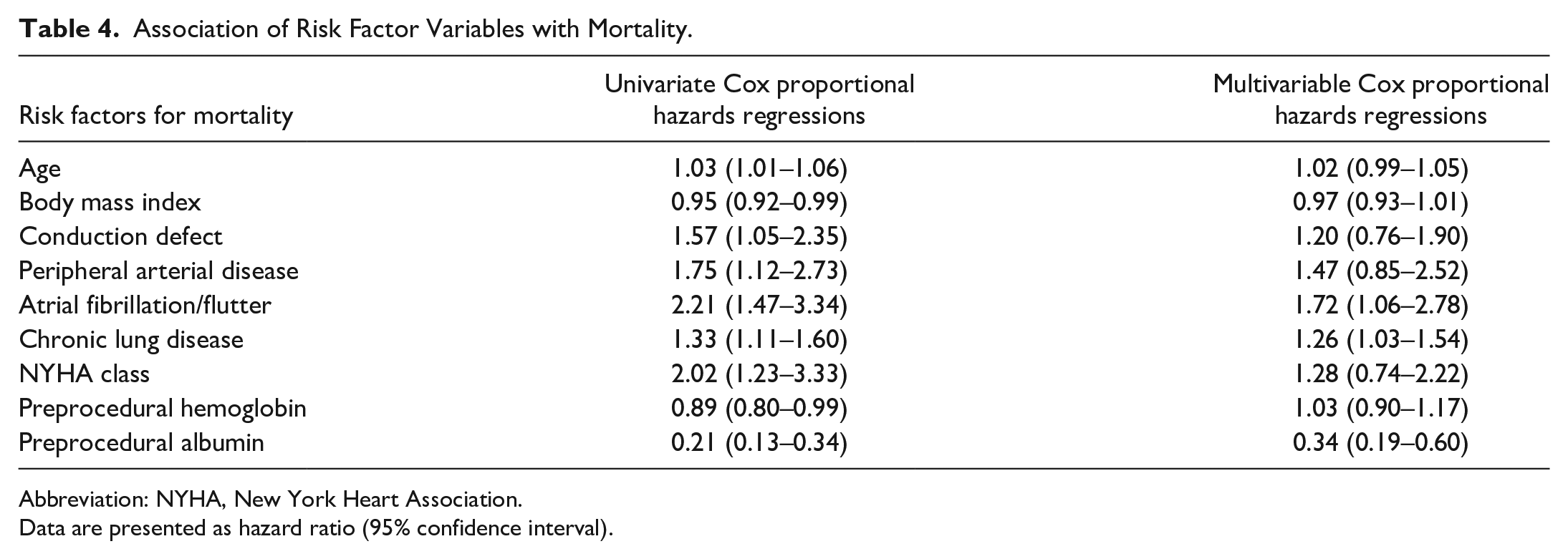
Abbreviation: NYHA, New York Heart Association.
Data are presented as hazard ratio (95% confidence interval).
Association of Risk Factor Variables with SVD.
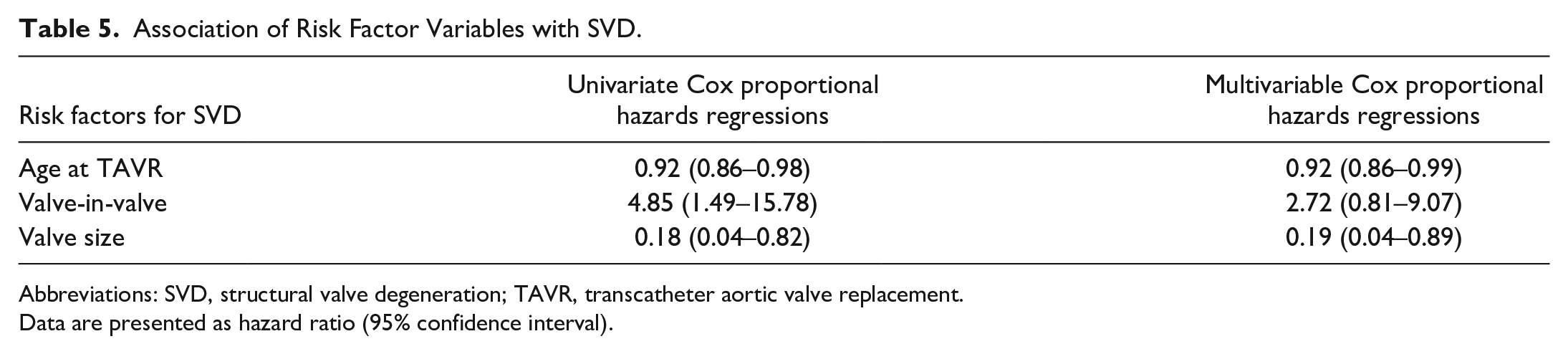
Abbreviations: SVD, structural valve degeneration; TAVR, transcatheter aortic valve replacement.
Data are presented as hazard ratio (95% confidence interval).
Discussion
In this analysis of patients at a single-center federal facility undergoing TAVR, we found that the incidence of SVD occurs at a low but detectable rate within the 5-year time frame. Rates of PVL in this cohort are similar to those reported in the literature. 18 There was a high degree of baseline preprocedure conduction disease in our cohort (36.9%), resulting in a somewhat higher rate of post-TAVR permanent pacemaker implantation. Our patient population began with high-to-prohibitive-risk patients and, following the Placement of AoRTic TraNscathetER Valves (PARTNER) II trial, was expanded to include intermediate-risk and then low-risk patients. For a population that includes both high and low surgical risk patients, our data show a favorable 5-year survival (61.7%) as compared with 48% in a systemic review of 31 studies. 19 Many prior studies report poor long-term survival and exclusion of younger, lower-risk patients as crucial limitations for evaluating TAVR SVD.20,21 Our low but detectable incidence of SVD, with a high 5-year survival rate and inclusion of lower-risk patients, positions us well to overcome those limitations.
Risk Factors for Mortality and SVD
We found that atrial fibrillation/flutter, chronic lung disease, and hypoalbuminemia were associated with higher mortality in our patients. Consistent with other studies showing moderate to severe chronic lung disease associated with higher mortality in TAVR patients, 22 our data confirm that higher mortality scales with increasing severity of chronic lung disease. Atrial flutter and fibrillation are known predictors of increased long-term mortality. 23 Our data also showed that hypoalbuminemia was associated with increased mortality in TAVR patients, likely a marker for frailty. 24 In our cohort, NYHA Class at the time of TAVR was not an independent risk factor for mortality on the basis of the multivariable Cox analysis. These risk factors for mortality may help inform preprocedural risk assessment and clinical management.
The background rate of SVD reported in SAVR depends on the definitions used for SVD and the type of SAVR bioprosthesis. In the PARTNER 2A trial, patients randomized to SAPIEN XT TAVR had higher rates of SVD, SVD-related BVF, and all-cause BVF than SAVR using VARC-3 criteria. For propensity-matched SAPIEN 3 versus SAVR (not randomized comparison), there were no significant differences in SVD, SVD-related BVF, or all-cause BVF. 25 On the other hand, using SVD definitions as used in our study for moderate SVD, the Nordic Aortic Valve Intervention (NOTION) trial found that the risk of SVD was lower after TAVR than after SAVR (13.9% vs 28.3%, P = 0.002), whereas the risk of BVF was similar (8.7% vs 10.5%, P = 0.61). 26 However, SAVR degeneration depends on type of bioprosthesis, with some valves demonstrating early failure (such as Mitroflow and now Trifecta), while others such as MagnaEase have shown very low long-term SVD.27,28 The weakness of the NOTION trial is that 34% of SAVR used these Mitroflow and Trifecta with demonstrated reduced durability. In general, SVD for SAVR has traditionally been considered a phenomenon occurring after 10 years, but our study at 5 years for TAVR as well as the discovery of subclinical leaflet thrombosis suggests that early SVD can occur and with longer follow-up may be important to determine if it is a harbinger for early BVF. 26 In our 13 patients with SVD, younger age and smaller valve size were associated with higher risk for SVD. Our data align with trends seen in other studies showing a higher risk of SVD with younger age, specifically the pediatric and <65 years of age populations. 29 Both SAVR and TAVR degeneration accelerate in younger patients; however, crucial questions remain regarding the comparative durability of SAVR versus TAVR in this population. Prosthesis-related factors, including valve size, have been shown to accelerate SVD.30,31 While other studies have also shown that a higher mean pressure gradient after TAVR is associated with higher risk of SVD, 32 in our study, valve size was an independent risk factor, possibly related in part to PPM. 30 While we found this to be true among the valve-in-valve patients who developed SVD (3 of 4 with moderate to severe PPM), it appeared to be a less important factor among those with SVD who underwent native-valve TAVR (2 of 9 with moderate PPM).
SVD Consensus Definitions and Timing
Many published series have assessed the long-term durability of TAVR and surgical valves based on survival without valve reintervention or explant for SVD.33,34 Consequently, we used standardized criteria for defining SVD with echocardiographic measures at a single time point, which allowed for longitudinal monitoring of elevated aortic gradients and defining SVD noninvasively. 12
Interestingly, our data showed that the median time to SVD in our population was 1.04 years. This finding brings into question whether early elevations in aortic mean pressure gradients truly indicate SVD, as degeneration is considered to be a more chronic process. Studies suggest that SVD occurring in a shorter-term period might be attributed to CVT, where anticoagulation therapy has led to regression of leaflet thickening and improvement of flow on imaging, in contrast to SVD from pannus formation or leaflet calcification. 35 We did not routinely acquire 4-dimensional electrocardiogram-gated CTA for thrombosis since many patients had their echocardiography follow-up with their outside referring cardiologists. However, based on these data, CTA should be considered when early increases in gradients are noted, to evaluate for CVT and consider treatment with anticoagulation. Continued follow-up for these patients with asymptomatic elevated gradients will provide valuable data about whether these gradients spontaneously resolve, remain stable, or progress to clinically significant SVD requiring reintervention. To date, most of our patients did not progress in SVD severity at midterm follow-up; however, the vast majority of our early SVD patients (70%) were treated medically with oral anticoagulation.
Valve-in-Valve Subpopulation
Notably, among the 13 patients with SVD, 4 patients (31%) underwent valve-in-valve (TAVR-in-SAVR) procedures, and the remaining 9 patients (69%) underwent native-valve TAVR. A total of 5 patients with SVD had evidence of PPM, with 3 of them derived from the 4 valve-in-valve cases (75%) as compared with 2 of the 9 native-valve cases (22%). Overall, valve-in-valve implantation accounted for approximately 10% of all TAVRs. This higher SVD risk is potentially related to the higher incidence of PPM among valve-in-valve patients and may be anticipated based upon the higher overall postprocedure gradient with valve-in-valve. Based on our experience as a member of the Valve-in-Valve International Data (VIVID) registry, we routinely anticoagulated for valve-in-valve implantation. Therefore, CVT was less likely. Instead, we posit that the degenerated surgical valve physically constrains TAVR expansion and ultimately reduces effective orifice area. This population should be carefully monitored for long-term outcomes of mortality and clinically significant SVD. Continuing to place successively smaller valves within degenerated valves could affect not only TAVR in degenerated SAVR but also TAVR in degenerated TAVR in the future.
While our patients with moderate SVD had minimal clinical sequelae at 5 years, ongoing surveillance will be important in understanding TAVR durability since clinically significant bioprosthetic degeneration requiring reoperation or intervention typically occurs after 8 to 10 years.
Limitations
Our retrospective single-center study at a VAMC reflects the predominantly older and male veteran population. Therefore, the findings may be less applicable to women and other civilian populations with different patient demographics and risk profiles. In particular, the rate of SVD may be somewhat underestimated, as men tend to have larger aortic annular sizes than women do, 36 and larger valves are less prone to SVD as compared with smaller valves.30,31 One limitation is the overall smaller number of TAVR patients with relatively few SVD events. However, our center is considered 1 of the top 3 high-volume TAVR centers within the Veterans Affairs system. Our population includes use of each generation of TAVR valve; however, no statistically significant associations were found between valve type and all-cause mortality. Furthermore, we have previously investigated the leaflet stresses of each generation of Edwards SAPIEN valves and found that leaflet stresses were greater in the third-generation SAPIEN 3 compared with prior first- and second-generation valves. 7 Higher leaflet stress could lead to faster degeneration in the newer generation leaflets, which is why this study includes patients with all generations of TAVR. Lastly, the incidence of SVD and other midterm outcomes could be underestimated due to the competing risk of death. However, our survival rate at 5 years was favorable compared with the current literature.
Conclusions
In veterans undergoing TAVR at a federal facility, we demonstrated that SVD was evident at a low but detectable rate in 5-year follow-up. SVD was associated with risk factors of patient age and implanted valve size. At midterm follow-up, only 1 patient required surgical reoperation for clinically significant SVD, and most patients with moderate SVD did not have clinical sequelae. Valve-in-valve implantation of TAVR within degenerated SAVR had a higher proportion of SVD than native TAVR, likely due to PPM. Clinical valve thrombosis was also a factor in early moderate SVD and was treated with oral anticoagulation. Future work will continue to follow this cohort. Given the variable mechanisms by which patients meet criteria for SVD, further understanding of TAVR biomechanics will inform patient-specific risks of SVD as well as guide follow-up and treatment decisions for elevated gradients.
Supplemental Material
Visual abstract – Supplemental material for Midterm Durability and Structural Valve Degeneration of Transcatheter Aortic Valve Replacement in a Federal Facility
Supplemental material, sj-pptx-1-inv-10.1177_15569845221123259 for Midterm Durability and Structural Valve Degeneration of Transcatheter Aortic Valve Replacement in a Federal Facility by Rachel Yang, Aaron F. Grober, Ramon Riojas, Vimala Ponna, Kendrick A. Shunk, Jeffrey M. Zimmet, Joshua Gustafson, Liang Ge and Elaine E. Tseng in Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Tseng and Dr. Ge disclose they are founders of ReValve Med, Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health Grant R01HL119857-01A1 (Tseng/Ge); the American Association for Thoracic Surgery Summer Intern Scholarship (Yang); University of California San Francisco Summer Explore Research Fellowship (Yang); and the VA Merit Award I01CX002365-01A1 (Tseng/Ge).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
