Abstract
Carcinoids of the left main bronchus are rare tumors of the bronchial system and patients often present with dyspnea, asthma-like symptoms, and pneumonia. Gold standard for therapy of carcinoids is surgical resection, but the surgical approach for segmental resection and anastomosis of the left main bronchus is a matter of discussion. With a left-sided approach the access to the bronchus is blocked by the aortic arch and the pulmonary vein. If a right-sided approach is performed, the problem of ventilation during resection and anastomosis of the bronchus occurs. We present a surgical approach from the right side using intraoperative extracorporeal membrane oxygenation to assure oxygen supply for resection of a typical carcinoid of the left main stem bronchus, and discuss the current literature.
Introduction
Low-grade neuroendocrine tumors of the bronchopulmonary system are rare malignancies comprising about 1% to 2% of all primary lung cancers. 1 Low-grade neuroendocrine tumors of the lung are classified as typical carcinoid tumors (TC; <2 mitoses per 2 mm2 and a Ki-67 proliferation index less than 5%) and atypical carcinoid tumors (AT; 2 to 10 mitoses per 2 mm2 of viable area of tumor and a Ki-67 proliferation index of more than 5% to 20%). 2 A carcinoid with more than 10 mitoses per 2 mm2 or a Ki-67 proliferation index over 20% is classified as a poorly differentiated high-grade large cell neuroendocrine carcinoma. 2,3 Low-grade neuroendocrine tumors are commonly located centrally in the bronchial tree. Tumor growth then causes stenosis of the bronchus, which often leads to nonspecific symptoms like dyspnea, pneumonia or asthma-like symptoms.
Diagnostic workup includes chest computer tomography (CT) as well as bronchoscopy with histological sampling. 4 Gold standard in treatment of low-grade carcinoid tumor is surgery resulting in 5-year overall survival of 90% for TCs and 70% for ATs. 3,4 The extent of surgical resection depends mainly on tumor location and tumor invasiveness; the low malignant potential of these carcinoids enables parenchyma-sparing resections, i.e. segmentectomies for peripheral tumors or bronchial sleeve resections for central lesions. 5
We discuss technical considerations for segmental resections of the left main bronchus using the available literature and present technical details of segmental resection of the left main bronchus.
Case Report
A 31-year-old female patient presented with recurrent pneumonia and worsening dyspnea in a secondary care hospital. A CT scan was performed and revealed an occluding lesion in the left main bronchus. The tumor was located centrally in the left main bronchus, 1.5 cm distally of the carina and obstructed the left main bronchus with concurrent atelectasis of the left lung (Fig. 1).
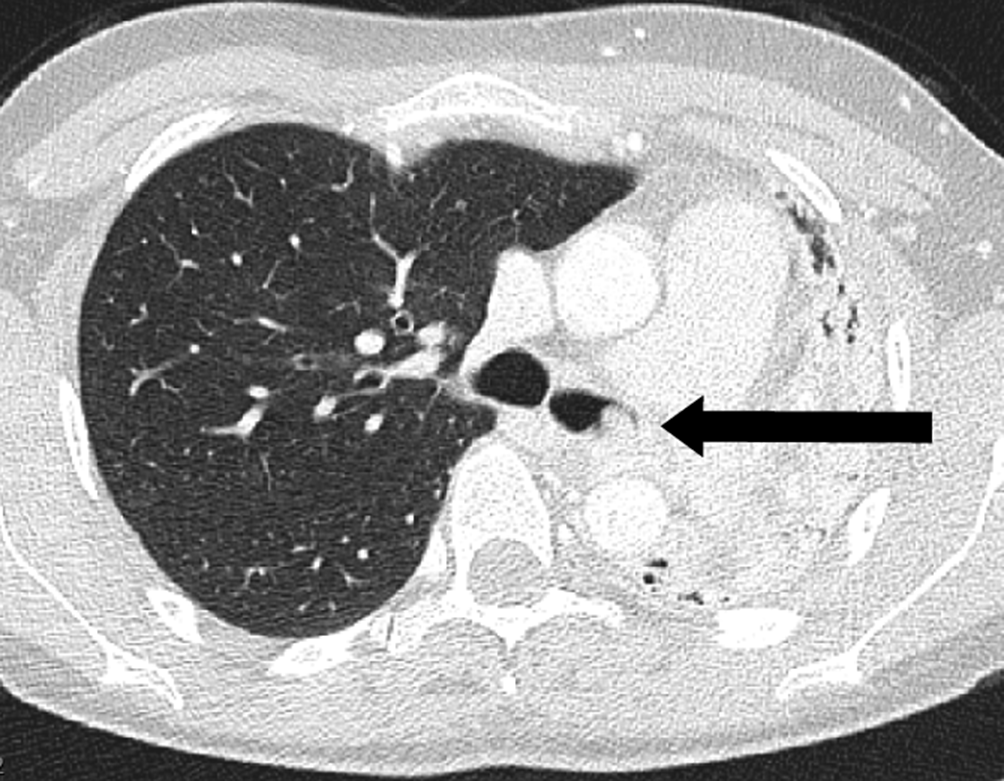
Computed tomography scan showing the atelectatic left lung due to the tumor in the left main stem bronchus.
Bronchoscopy was initiated for further diagnostic workup. Biopsies were taken and revealed a low-grade carcinoid tumor without any visible mitoses and a Ki-67 index <2%. To achieve recanalization of the bronchus, endoluminal laser resection (Nd-YAG) of the tumor was performed and a silicone stent was placed. There was no information about the completeness of resection. Also, the tumor was not described as purely endoluminal, indicating a possible complete laser resection. The patient was referred to our hospital for further treatment planning. Diagnostic workup with a 68Gallium-DOTATOC positron emission tomography scan 2 months post laser resection revealed persisting activity indicative for residual tumor and possible involved mediastinal lymph nodes. In the multidisciplinary tumorboard surgical resection was recommended.
Intraoperative oxygenation was ensured with single-lumen endotracheal intubation and a venoarterial extracorporeal membrane oxygenation (vaECMO) during resection and suturing of the anastomosis. During the first part of the procedure, the vaECMO was placed with the patient in a recumbent position using a 21Fr venous cannula and a 17Fr arterial cannula via the right femoral vessels and an additional line to assure perfusion of the right leg. The vaECMO was set to a flow rate of 4 L/min. Once the vaECMO was in place, the patient was turned into a lateral position with a partially retroflexed pelvis to avoid kinking of the ECMO lines.
Exposure of the carina and the central left main bronchus was then achieved using a right-sided muscle-sparing posterolateral thoracotomy via the fifth intercostal space. Dissection was started at the region of the right main bronchus, lymph nodes of the subcarinal region were resected, and the left main bronchus was exposed. There was no need to ligate the azygos vein for optimal access. Furthermore, lymph nodes of the right and left paratracheal region were sent to frozen section to assess the nodal status. After exposure, the central left main bronchus was cut and the stent removed (Fig. 2a). Then, 2.5 cm of the central left main bronchus was resected (Fig. 2b, Fig. 2c). Margins of the specimen were sent for frozen section to confirm R0 resection. Anastomosis was performed in a single-stitch fashion using absorbable monofilament threads. During the time of resection and anastomosis, the vaECMO provided adequate oxygenation while ventilation was stopped to achieve the deflation of the right lung and a stable operating field.
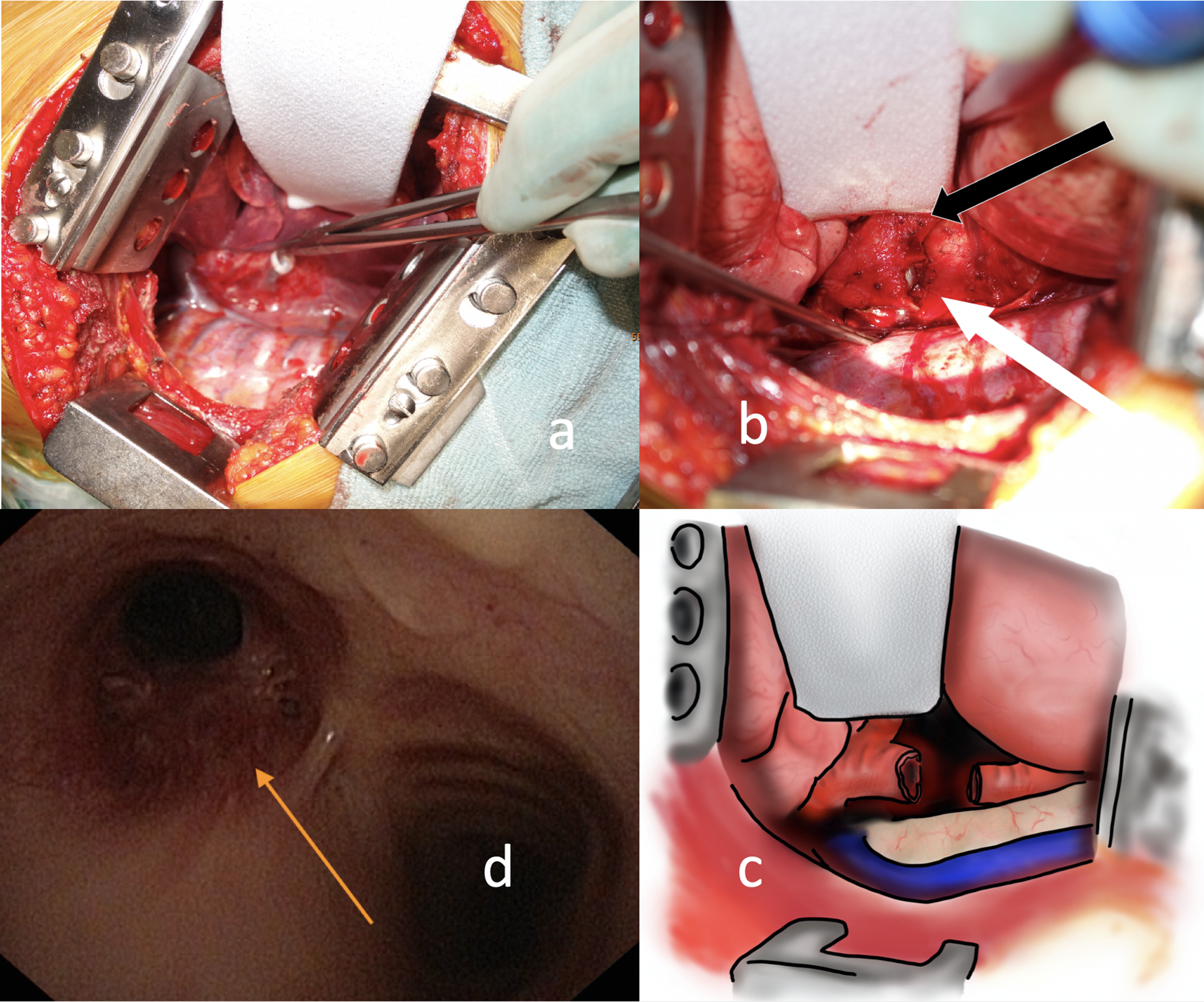
Extracorporeal membrane oxygenation support enables a clear view to the left main bronchus via a right-sided thoracotomy. (a) The left main bronchus is cut and the stent is removed. (b) A 2.5-cm portion of the central left main bronchus is resected close to the carina (white arrow: cut left main bronchus, black arrow: right main bronchus). The right lung and the mediastinal structures are retracted to gain sufficient exposure of the left main bronchus. (c) Schematic drawing of the intraoperative situs showing distal trachea, transected left main bronchus, and the lung retractor. (d) Control bronchoscopy at 7 months post-resection shows a patent anastomosis.
Once the anastomosis was completed and controlled for patency with an underwater test, ventilation via the single-lumen endobronchial tube was restarted. The chest was closed in a standard fashion. Finally, the patient was weaned from ECMO and the cannulas were removed. She was extubated in the operating room without any complications and transferred to the postoperative recovery room, where she was kept overnight. The postoperative course was uneventful; the patient was discharged on postoperative day 10.
On final pathologic workup, histology revealed no signs of malignancy in the resected bronchus, all 16 dissected lymph nodes were free of tumor. Control bronchoscopies 2 and 7 months after the operation revealed a well-healed anastomosis with no signs of tumor recurrence (Fig. 2d). Follow-up CT scan 36 months after the procedure confirmed the recurrence-free state of the patient and a well-expanded left lung. She is taking care of her two children and is back to part-time work as a land surveyor without any physical limitations.
Discussion
Low-grade neuroendocrine tumors of the lungs are rare. Tumor location often is within the central bronchial system leading to symptoms of pneumonia and dyspnea. 4 Due to its favorable tumor biology, parenchyma-sparing procedures should be attempted whenever feasible. Low malignant or benign lesions in the main bronchus without extrabronchial spread and a normal bronchial tree in the periphery can be cured with bronchial sleeve resection. 4,5 Pneumonectomy should be avoided whenever possible, as it would have a drastic impact on everyday life.
While resections of the right main bronchus can be managed with standard approach and left-sided double lumen intubation, the challenge in resecting a tumor of the left main bronchus is 2-fold: first, the sufficient exposure of the left main bronchus during resection up to the level of the carina and trachea; second, the adequate control of ventilation and oxygenation during resection and anastomosis. If a standard left posterolateral thoracotomy is performed, the central left main bronchus may not be easily approached due to the overlapping aortic arch, the pulmonary vessels, and the heart. Grillo describes a technique with extensive mobilization of the aorta to expose the left main bronchus via a left-sided thoracotomy. 6 Despite this advanced exposure, the carina might still be difficult to reach and the recurrent laryngeal nerve is at risk. There are 2 alternative approaches to the central left main bronchus: first, an anterior transpericardial approach via a median sternotomy; second, a right-sided posterolateral thoracotomy. According to Ragusa et al., the transpericardial approach is a challenging and complex procedure that adds considerable trauma to the patient. 7 Therefore, it is reasonable to assess the right-sided thoracotomy as a possible access. As Grillo admits, this approach is complicated by a more difficult management of ventilation during resection and anastomosis. However, it is described as the better approach to lesions in proximity to the carina. 6 The right lung needs to be deflated to grant access to the central structures. Grillo proposed an extra-long single-lumen tube positioned in the left main bronchus at the beginning of the procedure. Once the main bronchus is resected, intraoperative cross-field ventilation with a sterile endotracheal tube in the remaining left main bronchus might be used for oxygenation. To place the stitches during the anastomosis the tube can be removed for short times of apnea. The endotracheal tube can be reinserted for knot tying. 6 However, this may impair the approximation of the bronchial stumps. Alternatively, the right lung might be ventilated and retracted during knot tying. However, this might compromise visible control during this critical part of the procedure.
ECMO has been described as a safe tool for oxygenation during complex tracheobronchial procedures and enables an operating field without any additional lines or tubes. 8 Whether to use vaECMO or venovenous ECMO (vvECMO) is depending on the surgeon’s expectations on intraoperative interference of exposure and cardiac compression during retraction. It is possible to perform a carinal resection, where less exposure of the left main bronchus is needed, also using a less invasive vvECMO, as shown previously. 8 In our case, however, as we needed to retract the heart to gain sufficient access to the left main bronchus, we opted for a vaECMO, which not only provides oxygenation during apnea, but also offers circulatory support during cardiac retraction and compression.
In contrast to cardiopulmonary bypass, vaECMO avoids full anticoagulation, which might otherwise lead to severe intraoperative bleeding. Another advantage of this approach is an unobstructed view at the surgical field, without any additional tubes impairing the surgeon’s view. Moreover, it prevents desaturation periods that might occur during times of apnea.
Due to its more invasive nature, peripheral vaECMO results in more complex complications in comparison to vvECMO. 9 Potential complications of vaECMO include vascular complications at the insertion site, cardiac thrombosis due to retrograde blood flow in the ascending aorta and stasis of blood in the left ventricle, and coronary and cerebral hypoxia due to peripheral insertion and selective perfusion of the lower extremities with oxygenated blood. While vascular complications can occur in any patients, they are more likely in older patients with atherosclerosis; the overall rate is below 5%. Cardiac thrombosis in short-term intraoperative application of a vaECMO, monitored anticoagulation, and otherwise normal ventricular function seems unlikely, as it is usually reported to occur in patients with vaECMO due to myocardial infarction, primary graft-dysfunction after cardiac transplant, or worsening heart failure in cardiomyopathy. 10 Coronary and cerebral blood flow are closely monitored at our institution using intraoperative electrocardiogram and near-infrared spectroscopy for monitoring of cerebral oxygenation. Moreover, complications related to use of vaECMO can be reduced with high experience and a limited time of ECMO application. With close to 100 ECMO procedures a year at our institution and a close cooperation between the thoracic surgery, cardiac surgery, and anesthesia departments, all specialties involved seem experienced to reduce complication rates to a minimum. However, it highlights the importance of a standardized and well-established ECMO program to apply the technique to complex thoracic procedures.
Finally, the question can be raised, whether this case would have needed surgery at all due to the negative final pathology report. Laser treatment of pure endoluminal and centrally located endobronchial carcinoid tumors achieves impressive long-term results with high cure rates. 11 Workup of the patient after laser treatment, however, revealed a PET positive lesion, which guided our decision to opt for resection. An alternative approach in case of a negative PET after laser resection could be stent removal and postponing resection after repeated bronchoscopies reveal a visible tumor recurrence. Moreover, stent removal prior to surgical resection improves mucosal blood flow and anastomotic healing.
In conclusion, this is to the best of our knowledge the first report of a right-sided approach for segmental resection of the left main bronchus using a vaECMO. The availability of vaECMO in these complex tracheobronchial procedures leads to intraoperative ventilatory and circulatory stability and reduces the risk of desaturation, improves the view at the surgical field, and might reduce the complexity of crosstable ventilation as well as procedural time. The combination of a right-sided approach and vaECMO enables the resection and bronchoplasty of the central left main bronchus thus sparing the whole left lung in this rare kind of patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
