Abstract
Piriformis syndrome (PS) is a sciatic nerve entrapment condition caused by compression by the piriformis muscle, often presenting with symptoms that mimic lumbar radiculopathy and gluteal and buttock pain. This narrative review explores its diagnostic challenges and highlights the role of advanced imaging techniques in improving diagnostic accuracy. Literature suggests that PS may account for 5% to 8% of low back pain cases, frequently manifesting as buttock pain exacerbated by sitting or hip motion. Traditional imaging often fails to detect PS due to nonspecific findings, but magnetic resonance neurography can reveal sciatic nerve inflammation. A multimodal approach combining clinical assessment with targeted imaging enhances diagnostic precision and guides effective management of PS.
Keywords
Introduction
Piriformis syndrome (PS) is a neuromuscular condition characterized primarily by pain in the buttocks region, which often radiates to the leg. It is caused by irritation or compression of the sciatic nerve by the piriformis muscle (PM); pain is often made worse by specific motions or postures. It commonly affects individuals in their fourth or fifth decade of life, regardless of occupation or level of physical activity.11,14,20 Literature suggests that PS may account for 5% to 8% of low back pain cases, frequently manifesting as buttock pain exacerbated by sitting or hip motion.5,16,18 Women have a higher rate of PS, possibly due to their broader quadriceps femoris muscle angle (Q angle) at the os coxae. 16 Although hip dysplasia is not a direct cause of PS, both conditions can produce symptoms such as hip pain and altered posture, which may affect the diagnostic process. 15 Physicians often overlook PS due to the similarity in presentation with lumbar radiculopathy and sacroiliitis. 12 Furthermore, diagnosing PS has been challenging, primarily due to the lack of a reliable clinical examination or a single known reliable diagnostic test. 13 Although local anesthetic injection into the PM is accepted by some clinicians as a reference test, it still lacks strong validation and is not universally standardized, limiting its reliability.
For clinicians, the main challenge is how to spot the signs and symptoms of PS, so they can treat it properly. Diagnosis is typically supported by the patient’s description of their symptoms and by physical examination. 21 Given the overlapping symptoms and lack of standardized diagnostics, the aim of this review is to address the under-recognition of PS as a cause of gluteal pain and sciatica and to evaluate imaging as a noninvasive tool to improve PS diagnosis and clinical outcomes.
We conducted a narrative review of the literature by searching PubMed, Scopus, and Google Scholar using the following Medical Subject Headings and keywords: “piriformis syndrome,” “sciatic nerve compression,” “low back pain,” “buttock pain,” “diagnosis,” “MRI,” and “treatment.” All articles in English and published in the last 15 years were considered. Inclusion criteria comprised studies—such as observational research, reviews, case reports, and expert opinions—that focused on PS in adults, as well as articles discussing its diagnosis, imaging techniques, or treatment strategies. Non-English articles, unrelated topics, and pediatric studies were excluded. A total of 24 articles were selected.
Clinical Presentation and Diagnostic Challenges
The PM is attached proximally to the anterior aspect of the sacrum and inserts on the proximal aspect of the greater trochanter. A distinctive feature of PS lies in the anatomical relationship between the PM and the sciatic nerve. Typically, the sciatic nerve exits the pelvis beneath the PM; however, anatomical variations—such as the nerve passing through or above the muscle—can predispose individuals to nerve compression. These variations range from normal configurations to more complex arrangements in which the nerve splits or traverses the muscle belly, increasing susceptibility to entrapment. Trauma to the buttock area is a common antecedent, with about 50% of PS patients reporting a history of direct contusion or torsional injury. 9 Additionally, surrounding structures like the sacroiliac joint, sacrotuberous ligament, and gluteal musculature may contribute to altered biomechanics or inflammation, further exacerbating sciatic nerve irritation and pain syndromes. When patients attempt to get out of bed, remain seated for a long time, or turn or bend their hips, the pain often becomes more noticeable. 9 Patients often display symptoms that are similar to those of sciatica, which makes it hard to distinguish PS from other spine disorders. The discomfort may begin at the back of the thigh and travel down into the lower leg along the L5 or S1 dermatomes. Upon physical examination, findings that are important for diagnosis include: a tender or contracted PM, a positive flexion, adduction, and internal rotation (FAIR) test, or a reproduction of pain with maneuvers such as the Pace sign and Freiberg sign 6 (Table 1). Additionally, H-reflex latency prolongation during hip flexion and internal rotation has been proposed as an electrophysiological marker for PS. 3
Physical Examination Tests and Signs of Piriformis Syndrome.
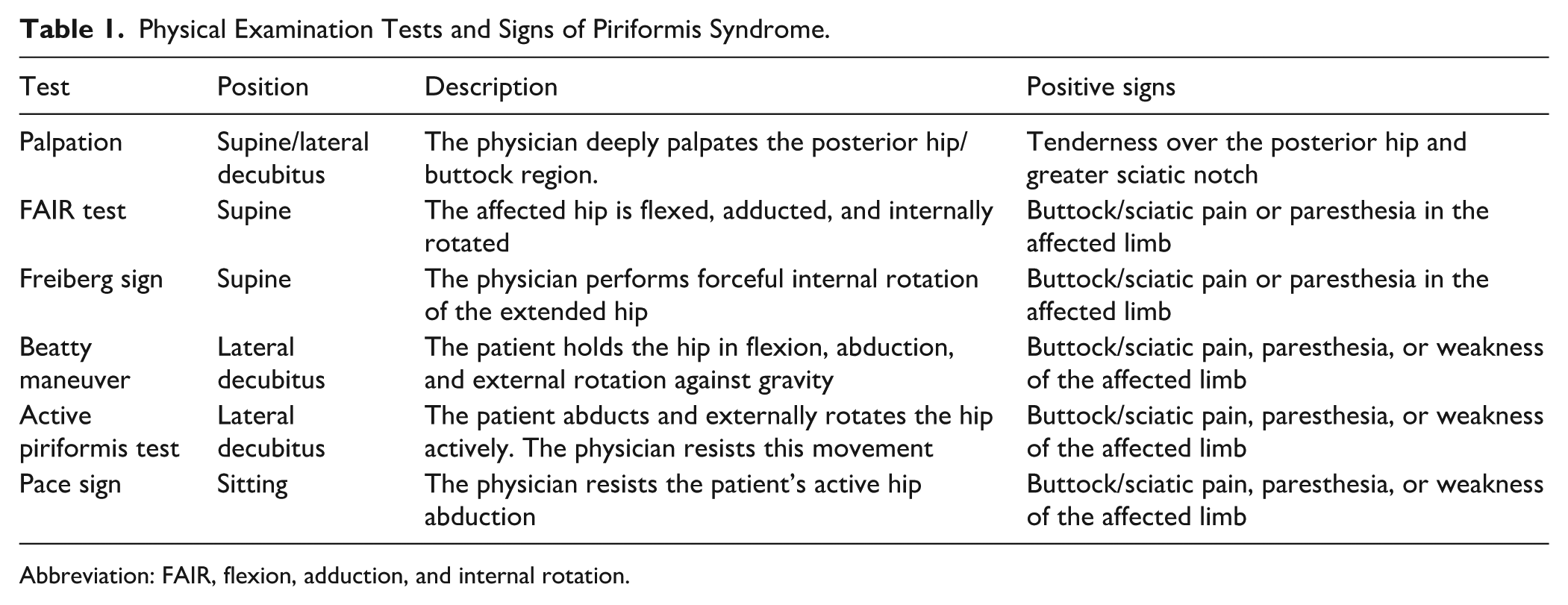
Abbreviation: FAIR, flexion, adduction, and internal rotation.
In his review article, Cass highlights the difficulty of diagnosing PS due to the absence of consistent neurological deficits and the normalcy of most imaging and neurodiagnostic tests. 4 Cass explains that using the FAIR test and palpating the piriformis can help make a diagnosis during a physical exam. Conservative treatment, including physical therapy and corticosteroid injections, is typically effective, though some cases require a botulinum toxin (BT) injection or surgical intervention. 4 Diagnostic challenges arise from symptom overlap with lumbar spine disorders and limited utility of imaging, necessitating careful clinical and electrodiagnostic evaluation. 4 Sharma et al highlight the significant clinical challenges in diagnosing PS, particularly due to its symptomatic overlap with other neuropathic and musculoskeletal causes of lower back pain and sciatica. 19 The authors of the latter study noted that PS is commonly mistaken for other ailments. The proximity of the piriformis to the sciatic nerve may often lead to an assumption of PS when patients report buttock or posterior thigh pain. However, true piriformis-related sciatica is relatively rare, with PS estimated to account for only 0.3% to 0.6% of lower back pain or posterior thigh pain cases and ~2.4 million cases annually. 19 The study underscores the absence of a validated diagnostic gold standard, noting that PS is typically a diagnosis of exclusion.
Limitations of Imaging and Neurodiagnostic Tools
Imaging studies such as magnetic resonance imaging (MRI) and computed tomography primarily serve to exclude other pathologies such as lumbar disc herniation and facet arthritis. Imaging modalities such as magnetic resonance neurography (MRN) have shown promise in detecting asymmetrical piriformis morphology, but even here, sensitivity remains low at 46%, with a specificity of 66%. 19 Intraoperative findings have also documented that other short external rotator muscles (eg, obturator internus) may be responsible for sciatic nerve impingement in some patients diagnosed with PS. 19 Moreover, anatomical variations affect clinical representation. For instance, in 17% of individuals, the sciatic nerve partially or fully traverses the PM (Beaton’s type B variation), complicating the diagnostic picture.17,19 Given this overlap, Sharma et al advocate for abandoning the umbrella term PS in favor of more anatomically inclusive descriptors that promote a more precise, evidence-based approach to diagnosis and management. 19
Advances in Imaging
PS is increasingly recognized as an overlooked cause of radiculopathy and buttock pain, often mimicking sciatica due to disc herniation. Imaging techniques, especially MRI, have become essential tools for diagnosis and to assess treatment efficacy. Al-Al-Shaikh et al conducted a retrospective case-control study evaluating the morphological changes of the PM following BT injections in patients diagnosed with PS. 1 Their study included 20 patients, divided into treated (n = 12) and untreated (n = 8) groups; it focused on quantitative MRI measurements—including PM thickness (PMT) and volume—as well as qualitative grading of fatty infiltration using Goutallier’s classification. The findings demonstrated a significant reduction in PMT (mean difference −4.22 mm, P < .001) and volume (mean difference −74.4 mm3, P < .001) post-BT treatment, coupled with increased fatty degeneration (P = .0002). In the untreated group, there was no significant difference in volume or thickness. Importantly, the degree of muscle atrophy correlated strongly with the number of BT injections (R = −0.77, P < .001) and the time elapsed before follow-up MRI, suggesting a dose- and time-dependent effect of BT on PM morphology. Clinically, this morphological improvement aligned with significant pain relief, evidenced by reduced Visual Analog Scale scores for both buttock and sciatic pain post-treatment. Research shows that BT injections are effective in reducing symptoms associated with PS. 1 These findings support the use of image-guided BT injections, which can be helpful in both the diagnosis and treatment of PS.
Diagnosing and Monitoring Treatment Response Using MRN
Imaging methods and electrophysiological testing are used to confirm the diagnosis of PS. One promising method is MRN—an advanced imaging modality that specifically visualizes peripheral nerves and their surrounding soft tissues. 10 Unlike standard MRI, MRN uses special sequences to highlight nerve signal abnormalities and structural changes, making it particularly valuable in detecting nerve entrapments like PS. Yang et al demonstrated the diagnostic and therapeutic role of MRN in a case report documenting treatment of a 64-year-old man experiencing major gait issues and symptoms typical of PS. 23 MRN findings included an inflamed sciatic nerve and a swollen lumbosacral plexus. MRI revealed that the left piriformis was larger than usual, and the sciatic nerve looked swollen on the T2 image. The results of electrophysiological testing on the patient’s left tibia demonstrated a 5.66 msH-reflex prolongation during FAIR, which supported the diagnosis of nerve compression. One week after ultrasound guided injection of 100 units of BT type A, the patient was able to walk short distances indoors without an aid. At 2 months, he could walk independently outdoors for >500 m and MRN revealed reduced swelling of the sciatic nerve and a reduction in volume of the PM. At 4 months, the pain had resolved, and the patient was able to complete all activities of daily living. Latency of the H-reflex returned to normal, and MRN documented resolution of inflammatory changes in the sciatic nerve. In this case, MRN was a useful bioindicator confirming a diagnosis of PS and provided imaging that documented successful resolution of sciatic nerve and piriformis abnormalities, accompanied by electrophysiological improvement. 23
MRI-Based Diagnostic Markers
Hip MRI (H-MRI) is increasingly used to diagnose PS. A recent study by Lim et al used T1-weighted H-MRI to compare the PMs of 30 patients with PS to those of 30 healthy individuals. 12 Two anatomical markers were evaluated: PMT and PM cross-sectional area (PMCSA). Both markers were significantly greater in the patients with PS, with mean PMCSA values of 736.88 mm2 in the PS group and 564.36 mm2 in controls, and PMT values of 15.99 and 13.83 mm, respectively. Diagnostic performance was assessed using the receiver operating characteristic analysis. PMCSA had an area under the curve (AUC) of 0.81, with 75% sensitivity and specificity at a cutoff of 611.67 mm2. PMT had a slightly lower AUC of 0.78. These results indicate that PMCSA is a more accurate marker for the diagnosis of PS. Moreover, both parameters showed strong reliability between different observers. The authors concluded that incorporating PMCSA into routine H-MRI protocols can help distinguish PS from other causes of sciatica, especially in non-disc-related cases. 12 Thus, H-MRI provides a non-invasive, objective tool to enhance PS diagnosis and guide treatment decisions.
Anatomical and MRI Correlation in Diagnosis
PS goes undiagnosed in many patients since its symptoms often overlap with lumbar radiculopathy. A study carried out by Goidescu et al combined cadaveric dissections with MRI imaging. The authors found that the PM, the sciatic nerve, and the internal pudendal neurovascular bundle have important connections. 8 In this study, the sciatic nerve was most commonly located beneath the PM. However, the nerve was found to pass both above and below the muscle in about 16% to 22% of the cases. Bartret et al suggested that these variants can lead to sciatic nerve compression leading to nerve pain and symptoms like those of discogenic sciatica. 2 Imaging documented that impingement is likely to occur in the regions of the ischial spine and the greater sciatic notch. The authors concluded that MRI clarifies the anatomical variations and improves the clarity of diagnosis, allowing more targeted treatment for gluteal and back pain caused by PS. 8
Diagnostic Musculoskeletal Ultrasound
Musculoskeletal ultrasound (MSK US) has also been used to define the pathoanatomy of PS. Dynamic viewing through high-precision ultrasound makes it possible to check soft tissues in real time, offering a fast and less expensive method, that is, as reliable as an MRI.7,22,24 Manske et al noted that once the diagnosis is confirmed, treatment can be provided by ultrasound-guided injections as part of the ultrasound procedure. 13 In general, MSK US helps improve diagnoses, allows early treatment, and lowers the chance of chronic complications caused by delayed diagnosis.
In conclusion, PS remains a challenging diagnosis, due to its symptom overlap with other lumbosacral conditions.4,19 Buttock and hip pain in PS can worsen when sitting or doing certain hip activities, although these symptoms may be nonspecific and resemble other neuropathic or musculoskeletal ailments. 19 Due to inconsistent neurological symptoms and unreliable imaging tests, misdiagnosis is a common problem. 16 Advanced imaging techniques make it easier to diagnose problems. H-MRI markers such as the PMCSA and PMT of the PM demonstrate potential to improve diagnostic accuracy. Lim et al reported a PMCSA threshold of 611.67 mm2 as highly accurate, with 75% sensitivity and specificity (AUC = 0.81). 12 MRN is comparable in revealing nerve inflammation and swelling, and it is closely linked with clinical and neurophysiological test results. 23 MSK US is showing promise as a convenient and affordable alternative to MRN and permits real-time visualization of the pathoanatomy and progression to treatment by guided injection. The currently reported studies are all observational with small sample sizes and concerns regarding generalizability suggesting additional, higher quality studies are needed.1,10,19
Supplemental Material
sj-pdf-1-hss-10.1177_15563316251392059 – Supplemental material for Piriformis Syndrome Is Often Overlooked as a Cause of Gluteal Pain and Sciatica: Diagnostic Challenges and the Role of Imaging—A Narrative Review
Supplemental material, sj-pdf-1-hss-10.1177_15563316251392059 for Piriformis Syndrome Is Often Overlooked as a Cause of Gluteal Pain and Sciatica: Diagnostic Challenges and the Role of Imaging—A Narrative Review by Hamza M. Alrabai, Yousef Alrashidi, Rakan Abdulkarim A. AlEtebi, Taher Mohammed A. Mufti, Mohammed Yousef A. Alali and Abdulaziz Saad A. Alnazhan in HSS Journal®
Footnotes
Ethical Considerations
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration.
Consent to Participate
Informed consent was not required for this narrative review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article as supplemental material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
