Abstract
Background:
Compressive neuropathy of the common fibular nerve (CFN) is increasingly recognized as an etiology for foot drop and falls. Electrodiagnostic (EDX) studies are widely used to evaluate this condition, but such tests are invasive and costly. As with carpal and cubital tunnel syndromes, there may be patients with characteristic symptoms of CFN compressive neuropathy but normal EDX studies in which ultrasound may aid in decision-making.
Purpose:
We sought to examine the association between ultrasound and nerve conduction studies (NCS) and electromyography (EMG) in the diagnosis of compressive neuropathy of the CFN.
Methods:
We performed a retrospective review identifying 104 patients who underwent CFN decompression from January 1, 2015, to June 30, 2023. Patients were included if they had both ultrasound and NCS/EMG prior to CFN decompression for compressive neuropathy and if they were older than 18 years at time of surgery. Patients were excluded if they had entrapment secondary to trauma, iatrogenic injury, or if they had had superficial fibular decompression alone without CFN decompression. After applying exclusion criteria, 17 patients remained in the cohort.
Results:
Mean ultrasound cross-sectional area and side-to-side (STS) ratios were significantly higher in those with abnormal compound muscle action potential (CMAP) amplitudes versus those with normal CMAP amplitudes. The probability of having an abnormal CMAP amplitude when STS ratio was abnormal was 18 times greater compared with those with normal STS ratio. With each unit increase in STS ratio, CMAP amplitude was reduced by 2.79 mV.
Conclusions:
This retrospective review found that ultrasound may provide complementary diagnostic information to EMG/NCS for compressive neuropathy of the CFN. Further study is needed to examine the relationship between ultrasound findings for CFN compressive neuropathy and results of surgical decompression.
Introduction
Compressive neuropathy of the common fibular nerve (CFN) is increasingly recognized as an etiology for foot drop [17,24,31] and falls [5,26]. Surgical decompression of the CFN at the fibular neck can lead to improvement in muscle strength [3,5,24], sensory symptoms [10,27], and pain relief [2,13] with better outcomes compared with nonoperative management [20]. Accordingly, there has been an increase in electrodiagnostic (EDX) evaluation of compressive neuropathy of the CFN [11].
In addition to establishing a diagnosis of compressive neuropathy of the CFN, EDX testing can be used to determine the severity of compression, which may help in counseling patients about their prognosis after treatment [28]. However, EDX testing can be uncomfortable for patients and entail additional cost [9,29]. Furthermore, EDX studies and physical examination cannot identify anatomical causes of CFN neuropathy, such as scarring or tethering, that occur with knee movement [11]. Therefore, a less invasive diagnostic test, such as ultrasound, is desirable to assist with diagnosis and prognosis. Ultrasound has served a similar diagnostic role in compressive neuropathies such as carpal tunnel [15,30] and cubital tunnel syndromes [7,14,18,19] and it has been shown to correlate with electromyography (EMG) [16] and nerve conduction studies (NCS) [22] for those conditions. However, this relationship has not been explored for CFN entrapment. The purpose of this study was to examine the association between sonographic assessment of compressive neuropathy of the CFN and concurrently performed EDX. We hypothesized that patients with enlarged CFN cross-sectional area (CSA) and increased side-to-side (STS) ratio would have evidence of more severe compression on EDX in the form of reduced compound muscle action potential (CMAP) amplitudes, reduced recruitment, and the presence of active denervation.
Methods
Following approval from our institutional review board, we performed a retrospective review of patients who underwent CFN decompression from January 1, 2015, to June 30, 2023, at a single institution. Diagnosis and ensuing surgical intervention were derived from the cumulative findings of the physical examination, symptom history, and results of EDX testing. Patients with numbness, tingling, and pain in the distribution of the CFN and foot drop were suspected of CFN entrapment. Furthermore, there was concern for neuropathy of the CFN in patients with weakness in dorsiflexion, tenderness over the fibular head, or sensory disturbance in the distribution of the CFN. Patients with abnormal EMG or ultrasound findings were considered positive for CFN compressive neuropathy. Patients without any such evidence in the contralateral leg were ruled to have unilateral neuropathy. Each patient was examined by 1 of 2 board-certified orthopedic surgeons, who made the decision on the appropriateness of surgical intervention. Patients were excluded if they were less than 18 years of age, did not have a preoperative ultrasound, did not have preoperative EDX studies, or had CFN that was iatrogenic or secondary to trauma.
We initially identified 104 patients who underwent CFN decompression. Of these, we excluded 39 patients for entrapment secondary to trauma, 1 patient for iatrogenic injury secondary to total knee arthroplasty, 1 patient for being <18 years old at time of surgery, 14 patients for having superficial fibular decompression alone without CFN decompression, 29 patients for not having a preoperative ultrasound, and 3 for not having preoperative EDX. After applying these exclusion criteria, 17 patients remained in the cohort (Fig. 1): 6 (35.3%) men and 11 (64.7%) women, with a mean age of 53.8 (±13.8) years and body mass index of 30.7 (±6.82). All included patients were White and non-Hispanic.
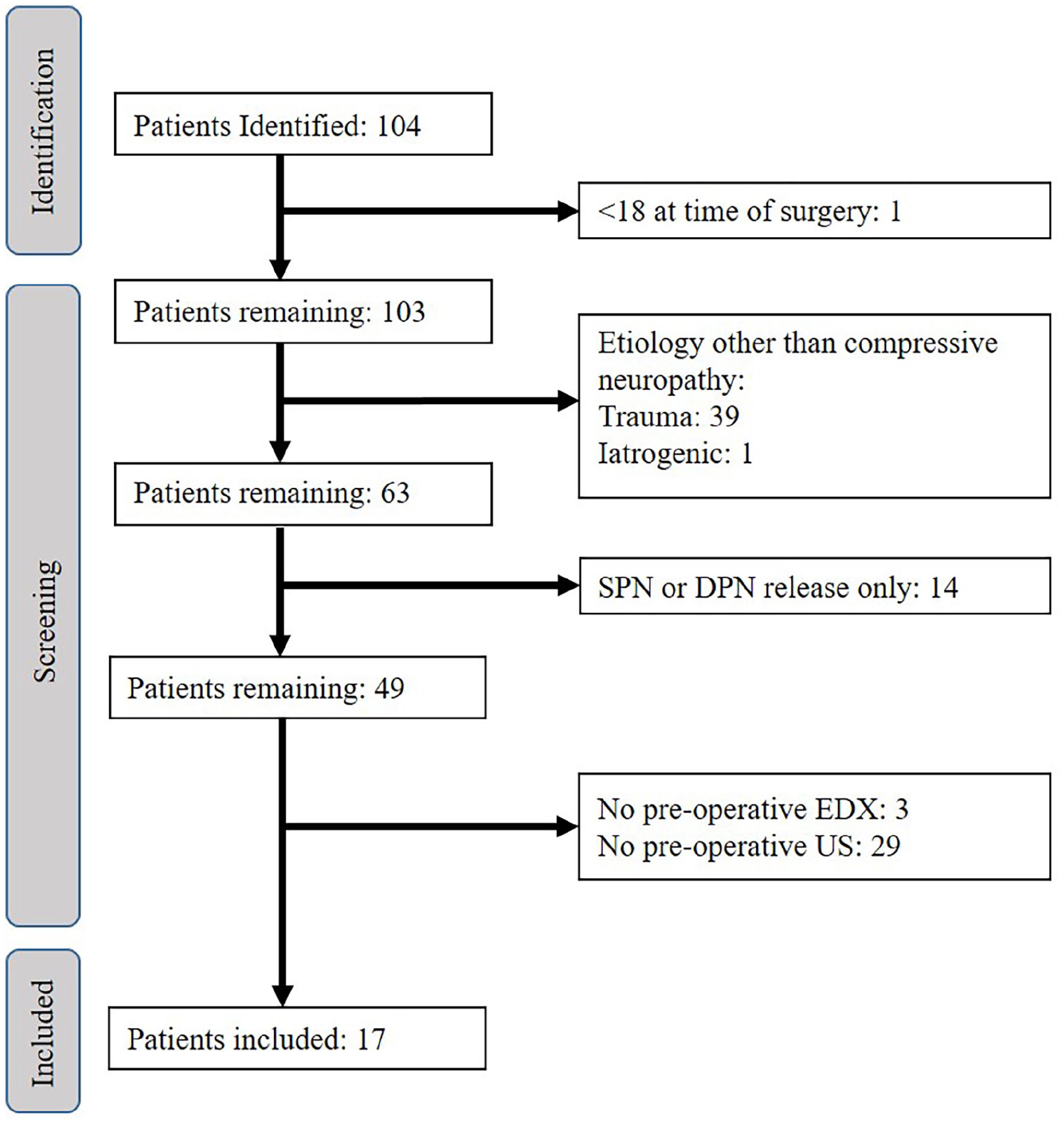
Identification of atraumatic CFN release patients with preoperative EDX and ultrasound studies.
As part of standard clinical care, ultrasound images were collected by 2 physiatrists with certification in musculoskeletal ultrasound using the Sonosite X-Porte, linear 6-15 Hz probe. The distal sciatic nerve and the branch point of the CFN and tibial nerve were identified in short axis in the popliteal fossa. The transducer was then moved distally, keeping the nerve visualized in short axis, tracing the CFN to the fibular head. The maximum CSA was then measured in standard fashion, tracing just on the inside inner edge of the hyperechoic epineurium (Fig. 2). Ultrasound values were recorded as the largest CSA on the affected and unaffected sides, as well as the STS ratio. Patient NCS were evaluated for motor amplitudes to the extensor digitorum brevis at the ankle. The EMG results from the tibialis anterior were evaluated for active denervation (fibrillations and positive sharp waves).
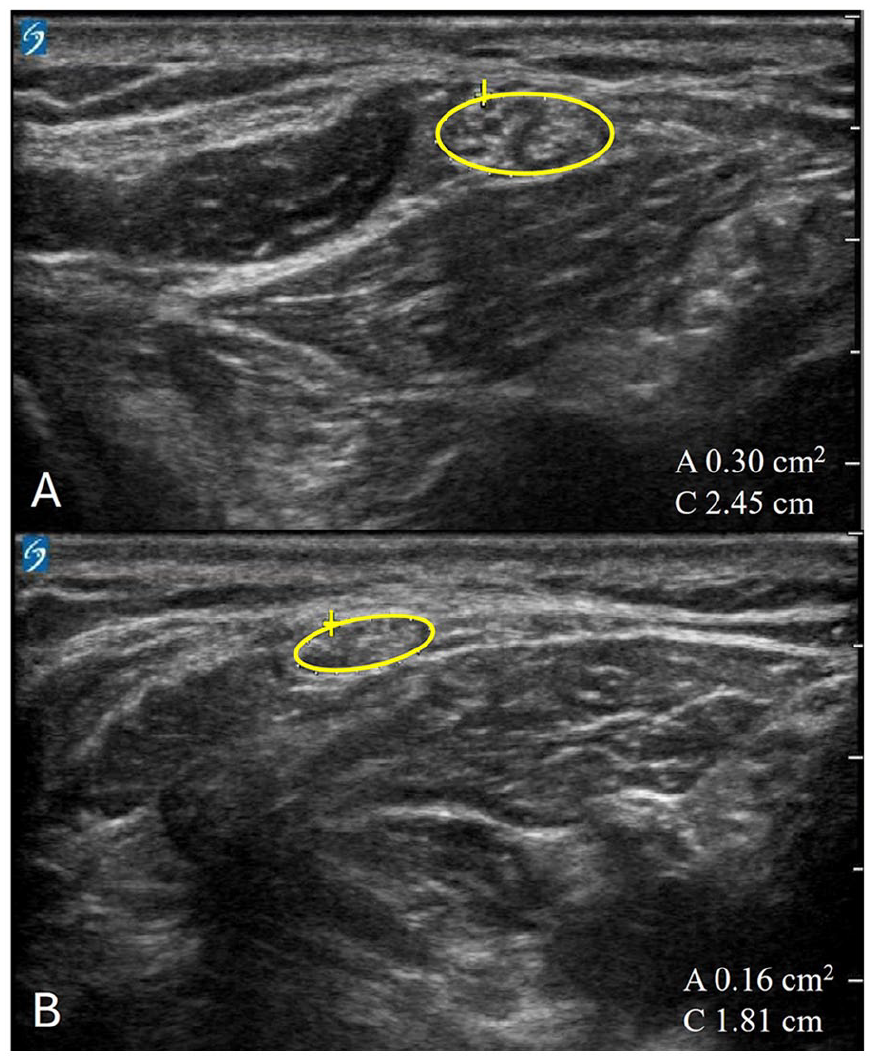
Common peroneal nerve ultrasound of affected (A) and unaffected (B) limbs based on nerve cross-sectional area. Yellow circle indicates area of the CFN.
Statistical Analysis
Descriptive statistics were reported as mean ± standard deviation for continuous variables and frequency (percentage) for categorical variables. Ultrasound measurements of the CSA at the CFN were considered normal at ≤20 mm. The STS ratio was considered normal at <1.5 affected side:unaffected side ratio [6]. The CMAP amplitudes to the extensor digitorum brevis were considered normal if >1.3 mV at the level of the ankle and abnormal if ≤1.3 mV as established during initial setup and calibration of the EDX testing machine in our diagnostic suite. The CFN active denervation on EMG was considered present if either fibrillations or positive sharp waves were recorded. We performed a correlation analysis between CSA and CMAP amplitude, as well as STS and CMAP amplitude. Linear regression was also used to examine the relationship between ultrasound characteristics and CMAP amplitude. Relationships between mean CSA of patients with normal and abnormal CMAP amplitudes were analyzed using Student independent t test.
Results
In evaluating ultrasound and CMAP amplitude, we found the mean CSA was significantly higher in patients with abnormal CMAP amplitudes (CSA 25.7 mm2) compared with those patients with normal CMAP amplitudes (CSA 18.5 mm2; P = .04). When analyzing the STS ratio as a binary measure (normal/abnormal), there was a significant difference in CMAP amplitude values for those with a normal STS (n = 6; mean CMAP amplitude = 3.77) compared with those with an abnormal STS (n = 8; mean CMAP amplitude = 0.49) (P = .039). Unaffected-side CSA values were not recorded for 3 (17.6%) of our patients, precluding the calculation of STS ratio for those patients.
In a logistic regression model, the probability of having an abnormal CMAP amplitude when STS ratio was abnormal was 18 times greater than for those with a normal STS ratio (P = .0344) (odds ratio = 18.3; 95% confidence interval = 1.24–271.3). When analyzing STS as a continuous variable, there is a statistically significant correlation between STS and CMAP amplitude, and for each 1 unit increase in STS ratio, there was a 2.79 mV decrease in CMAP amplitude (Fig. 2).
When setting CMAP amplitude as the diagnostic standard of CFN compressive neuropathy, we found ultrasound CSA was determined to have a sensitivity of 70.0% and specificity of 71.4%. Ultrasound STS ratio was more sensitive and specific than CSA, with a sensitivity of 87.5% and a specificity of 83.3% (Table 1).
CFN decompression patient preoperative ultrasound findings in relation to preoperative EMG readings.
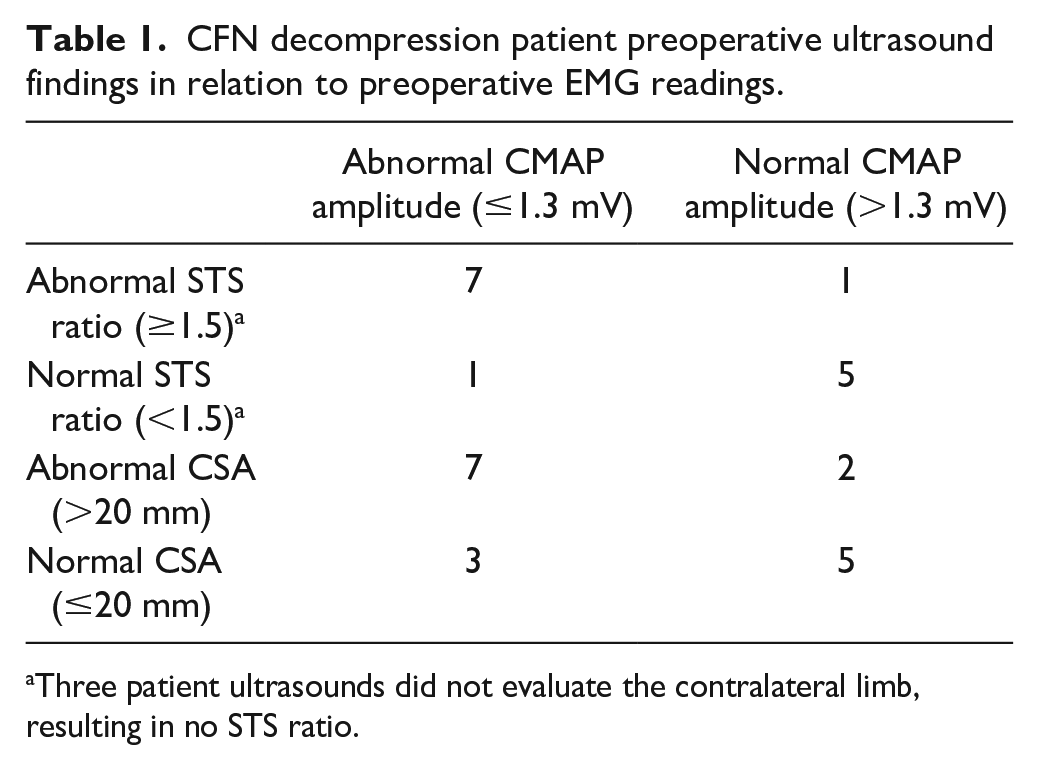
Three patient ultrasounds did not evaluate the contralateral limb, resulting in no STS ratio.
In evaluating ultrasound and EMG, there was no difference in mean CSA (23.4 mm2) in those with active tibialis anterior (TA) denervation than those without (CSA 22.2 mm2; P = .740). There was no difference in mean STS (1.94) in those with active TA denervation and mean STS (1.40) in those without TA denervation (P = .098).
Discussion
The purpose of our study was to examine the association between sonographic assessment of compressive neuropathy of the CFN and concurrently performed EDX. We found that patients with signs of axonal loss (as measured by CMAP amplitude) have significantly higher CSA of the CFN at the fibular neck on ultrasound. We also found that there was a significant correlation between STS ratio on ultrasound and CMAP amplitude. These findings suggest that ultrasound may be a reliable predictor of disease severity for CFN compressive neuropathy, potentially decreasing the need for EDX in its diagnosis.
Our study is limited by the potential for operator dependency in the use of ultrasound. Two physicians with advanced certification in musculoskeletal ultrasound performed all ultrasound studies included in this analysis, which may limit the generalizability of our results. However, the expertise of the performing sonographer likely has a role in maximizing diagnostic utility. Our study is also limited by the exclusion of patients with traumatic peripheral nerve injury. Given the distinction in the pathophysiology of compressive neuropathy compared with traumatic nerve injury, the findings from our study may not be generalizable to evaluation of traumatic CFN injury.
There has been recent enthusiasm and expert consensus on the combined utility of ultrasound and EDX in the diagnosis of cubital tunnel syndrome [23], aligning with our findings that adding ultrasound may improve the diagnostic accuracy regarding CFN compressive neuropathy [4] (despite the inherent differences in the pathophysiology of the 2 conditions). Furthermore, given that enlargement of nerve CSA on ultrasound is not exclusive to regional entrapment, we view ultrasound and EDX as complementary tests as opposed to stand-alone assessments. If using ultrasound, our findings suggest that adding the STS ratio results in improved sensitivity and specificity compared with CSA alone. Because of this, we have modified our diagnostic protocol to routinely assess both limbs sonographically to allow calculation of the STS ratio.
These findings are consistent with studies of other compressive neuropathies, in which ratio values are used in combination with CSA for the diagnosis of cubital tunnel syndrome [8,12] and ulnar neuropathy, with comparable sensitivity and specificity [25,33]. Ultrasound CSA was also noted to correlate more strongly with EMG characteristics when observed as a ratio in different planes/positions, further demonstrating that abnormal nerve enlargement alone is less diagnostic than multiple measurements [32,34]. These findings and our results indicate that ultrasound of the unaffected limb (to have the STS comparison) helps to maximize performance characteristics. It is possible that evaluating CSA in different degrees of knee flexion, as has been done by Yoon et al [34] in the upper extremity, could aid in improving the diagnostic accuracy of CSA alone. In cases of suspected bilateral CFN neuropathy [21], CSA measurements at various points along the ipsilateral CFN may serve as a potential alternative to STS ratio, as demonstrated in upper extremity entrapment neuropathies [1]. In the absence of such cases and until further information is available for evaluation at different degrees of knee flexion, we recommend using an STS comparison when evaluating the CFN at the fibular neck.
In conclusion, our results suggest that abnormal ultrasound findings, specifically STS ratio, may aid in the diagnosis of compressive neuropathy of the CFN. Future studies should focus on the utility of ultrasound in diagnosing traumatic CFN injury and on the relationship between ultrasound and patient outcomes to evaluate its potential use as a predictor for improvement after CFN decompression.
Supplemental Material
sj-docx-1-hss-10.1177_15563316241285898 – Supplemental material for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve
Supplemental material, sj-docx-1-hss-10.1177_15563316241285898 for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve by Stephen J. DeMartini, Amanda M. Faust, Nathan P. Olafsen, David M. Brogan and Christopher J. Dy in HSS Journal®
Supplemental Material
sj-docx-2-hss-10.1177_15563316241285898 – Supplemental material for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve
Supplemental material, sj-docx-2-hss-10.1177_15563316241285898 for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve by Stephen J. DeMartini, Amanda M. Faust, Nathan P. Olafsen, David M. Brogan and Christopher J. Dy in HSS Journal®
Supplemental Material
sj-docx-3-hss-10.1177_15563316241285898 – Supplemental material for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve
Supplemental material, sj-docx-3-hss-10.1177_15563316241285898 for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve by Stephen J. DeMartini, Amanda M. Faust, Nathan P. Olafsen, David M. Brogan and Christopher J. Dy in HSS Journal®
Supplemental Material
sj-docx-4-hss-10.1177_15563316241285898 – Supplemental material for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve
Supplemental material, sj-docx-4-hss-10.1177_15563316241285898 for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve by Stephen J. DeMartini, Amanda M. Faust, Nathan P. Olafsen, David M. Brogan and Christopher J. Dy in HSS Journal®
Supplemental Material
sj-docx-5-hss-10.1177_15563316241285898 – Supplemental material for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve
Supplemental material, sj-docx-5-hss-10.1177_15563316241285898 for Ultrasound as a Complementary Tool to Electrodiagnostics in the Evaluation of Compressive Neuropathy of the Common Fibular Nerve by Stephen J. DeMartini, Amanda M. Faust, Nathan P. Olafsen, David M. Brogan and Christopher J. Dy in HSS Journal®
Footnotes
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christopher J. Dy, MD, MPH, has relationships with Sonex Healthcare, Springer, Orthocell, Johnson and Johnson, and Checkpoint Surgical. The other authors declared no potential conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Human/Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2013.
Informed Consent
Informed consent was waived for all patients included in this study by the IRB of Washington University.
Level of Evidence
Level IV, retrospective review.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article as supplemental material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
