Abstract
This study explored the knowledge and approaches of researchers in studies involving children. The qualitative analysis carried out in line with the suggestions of the Ethical Research Involving Children guidance revealed that the participating researchers, who completed their graduate-level research involving children, addressed some fundamental ethical considerations. These included obtaining institutional review board approval, securing informed consent from the caregivers, and ensuring the confidentiality of the collected data. However, they overlooked certain specific requirements, such as getting consent from the child, monitoring for signs of dissent, ensuring privacy by removing caregiver interventions during data collection, and creating an incentive strategy. The results also highlighted the necessity of the institutional review board's role in the effective guidance of researchers by considering the specific requirements of their research involving children.
Keywords
For every child, every right.
Introduction
Social research traditionally encompassing disciplines such as psychology, sociology, education, and anthropology (Morrow & Richards, 1996) viewed children as passive and conforming objects of an insufficient, incomplete, and unilateral socialization process (James & Prout, 1997; Waksler, 1991). According to Carter (2009), children were marginalized because studies were designed around meaningful data collected from adults who represented those children (Harris & Udry, 2016; Van Voorhis et al., 2013). However, the theory of socialization (Waksler, 1991), approaches in developmental psychology (Erikson, 1958; Piaget, 1964), and advances in the children's rights domain (United Nation's Children Fund-UNICEF, 1989) are crucial (Kirk, 2007) during the process of reshaping childhood. Additionally, contrary to the belief that children are mere recipients of societal ideas (Waksler, 1991), a new and modern approach has emerged. This approach recognizes children as actors in their own lives and engaged research participants (Carter, 2009). Reflections of this new approach are evident in the emergence of contemporary and innovative research methodologies (Christensen & James, 2017) and in the official documents of world organizations, such as the United Nation’s Children Fund (UNICEF).
The Convention on the Rights of the Child (UNCRC; UNICEF, 1989) mandates that all children are unconditionally “entitled to the rights and freedoms of respect, dignity, equality, expression, nondiscrimination, life, and civil participation” (Beazley et al., 2009, p. 368). Accordingly, researchers must focus not only on studies with adults but also with children. Such research may include critical issues, such as children's rights, needs, past experiences, positions in society, diversity (e.g., age, gender, ethnicity, and disability), and inherently unequal and hierarchical power relations with adults (Punch, 2002; Thomas & O’Kane, 1998). In this respect, Carter (2009) identified the need for ensuring “controls, constraints, risk management, and protection” (p. 859) and providing inclusion in any research with children. Conversely, Smith (2018) stressed that every child has the right to be properly and decently researched and proposed human rights-based approaches to meet this need. Such requirements have instigated discussions on finding ways to receive consent from children (David et al., 2001; Hurley & Underwood, 2002) or using techniques that enhance children's active participation (Alderson, 2012; Broström, 2012). Punch (2002) addressed some ethical and methodological issues, stating that any research involving children must consider the potential differences stemming from the child and the cultural, social, political, and financial factors that may shape the studies. Christensen and Prout (2002), sharing similar concerns, highlighted the negligence of requirements in research involving children. Giorgini et al. (2015) found that researchers lack knowledge about ethical guidelines and described such guidelines as “idealistic responses to situations in a best-case scenario context” (p. 8).
The human rights-based approach is considered a core condition researchers use to attain transparency, equality, and inclusiveness in their studies (Smith, 2018; The Swedish International Development Cooperation Agency, 2015) and is directly related to codes of ethics. Any research regarded as ethical prevents wrong applications and harm to children (Morrow & Richards, 1996) and highlights the need to consider core principles to protect the rights and best interests of the child at all stages. These core principles, specific to research involving children, have been compiled directly or indirectly from various sources (Save the Children, 2004; The American Counseling Association, 2014). While these sources typically reference the UNCRC (UNICEF, 1989), they differ in their content and do not adequately propose ethical frameworks to address children's vulnerabilities and developmental needs in research. The evolution of research involving children indicates that although medical research ethics standards have been established since World War II (Council for International Organizations of Medical Sciences-CIOMS Ethical Guidelines, 2016), social research ethics have lagged behind. This may be due to the perception that such studies do not pose risks or entail significant health and social impacts (Bell, 2008; Morrow, 2009). Even though children are now recognized as active participants in certain studies, the sparse ethical guidelines reviewing social research have ignored children's vulnerabilities, treating them merely as objects of research (The Social Research Association Ethical Guidelines, 2003).
The context of Türkiye reflects the same deficiencies related to the ethical issues of children's involvement in scientific research. Two of the leading documents, “The Higher Education Institutions Ethical Behavior Principles” and “The Directive on Scientific Research and Publication Ethics,” were issued in 2014 and 2016, respectively, by the Council of Higher Education (CoHE, 2014, 2016), the regulatory body responsible for all higher education institutions in Türkiye. These documents contained no text on any ethical issues related to research involving children. A quick review of the documents and processes of the Institutional Review Boards (IRBs), the establishments of which are mandated by the Council of Higher Education in all universities, revealed that they use standard application forms and protocols for all types of studies and provide no guidance for any ethical requirements specific to research involving children. Professional ethics codebooks also provide little guidance on the requirements unique to research with children. For example, the research section of the codebook of the Turkish Psychological Association (2004) includes items on receiving consent and debriefing, offering incentives, protecting participants from adverse consequences, and reporting research results; however, none are specific to child participation. In the recently revised codebook of the Turkish Psychological Counseling and Guidance Association (2024), two items address the specifics of child participation in the research ethics section: “Considering their sensitive developmental periods, researchers pay special attention to studies with children and adolescents. It is right to get parental consent if the child is 15 years old or younger, however, they must be treated as independent individuals” (Item 8.3.1.1.) and “Receives written and oral consent from all participants before study. If the participants are not eligible, receives consent from their legal caregivers. It's mandatory to get consent from the families or legal caregivers in research with children under the age of 18.” (Item 8.5.1).
Drawing upon these international and local discussions, “The Ethical Research Involving Children” (ERIC guidance) (Graham et al., 2013) was selected for research purposes. It was developed specifically for studies involving children and differs from other sources owing to its contemporary, inclusive, and comprehensible content and practical case samples. The ERIC guidance was developed by a team from the UNICEF Innocenti Office of Research to conduct research involving children that was compatible with human dignity, regardless of context. These guidelines include respect, benefit, and justice as their core principles. Respect in research recognizes every child's experiences and cultural structures within the agenda of protection and participation rights, and beneficence refers to identifying clear benefits by minimizing possible risk factors. The principle of justice in research requires children's meaningful participation in the decision-making process on related topics and ensures a researcher–child relationship that is not built on power imbalances.
The ERIC project has conducted a comprehensive review of extant research related to ethical issues centering on children. It has also mapped out existing “Charters and Guidelines,” analyzed ethics systems in different countries from a multidisciplinary perspective, and reviewed governance systems and implementations across various local and disciplinary contexts (Graham et al., 2013). Alongside the UNCRC, the ERIC guidance addresses the ethical issues of research involving children under 18. It frames the ethical dilemmas that researchers may encounter in four sections: harms and benefits; informed consent; privacy and confidentiality; and payment and compensation. Harms and benefits concern a researcher's measures and preparations to ensure child safety. However, contrary to the traditional view that obtaining consent from parents or caregivers is sufficient, informed consent refers to researchers gaining consent directly from the child by selecting the most suitable method (e.g., oral, child-friendly, visuals or videos, games, and thumbprint) and reviewing that consent through all stages of research. Privacy and confidentiality are meticulous measures taken for the data collection processes from the child and involve safeguarding the data and providing flexibility for any challenges that may arise in the research context. Finally, in any research that provides rewards or returns for involvement, payment and compensation highlight planning decisions regarding when and how the payment will be made. Therefore, it would not be wrong to define the ERIC as a facilitating source for practitioners in research involving children.
Although the existing guidelines address ethical issues in research involving children, researchers have not yet reached a strong consensus. While children are considered incompetent to give consent owing to their inability to comprehend the procedures and their rights in a study (Masty & Fisher, 2008), research on children's competence for consent shows that age and ability are unrelated (Miller et al., 2004). In another study, a neutral coworker negotiated consent with the children to reduce power imbalances and potential coercion or embarrassment (Loveridge et al., 2024), whereas other studies have discussed the possible risks of payment to the child and its influence on their choice of participation (Taplin et al., 2019). Against these complexities and uncertainties, the ERIC guidance goes beyond the procedural ethics approach, in which only ethical requirements are followed. Instead, it adopts an ethics-in-practice approach by providing researchers with sample cases (Graham et al., 2014). The ERIC approach aims to equip researchers with the three pillars in their studies involving children—reflexivity, rights, and relationship—where reflexivity represents the ethical mindfulness and attitude of the researchers; rights refer to researchers’ responsibilities to respect children's rights, well-being, and dignity; and relationship emphasizes the multiple dialogs between the researchers, children, and others involved (Graham et al., 2015). In short, the ERIC compendium (Graham et al., 2013) advocates a list of activities: “… no package of resources or ethics review process that can ultimately mandate and/or monitor …” (p. 167); it provides “… a rich repository of evidence-based information and resources to guide and improve research involving children …” (p. 167), with “… a deep respect for human dignity and a desire to advance the status of children in the way called for by the United Nations Convention on the Rights of the Child” (p. 2).
Few studies have used the ERIC guidance, and the ones reviewed by the authors adhered to only a limited number of its recommendations. Receiving consent from caregivers and the children (Lortoğlu & Kurtulmuş, 2020; Nguyen et al., 2016; Prendergast et al., 2021) is one of those ethical practices. In their research with children having a parent with cancer, Helseth and Ulfsaet (2003) practiced the following principles suggested by the compendium: explaining the content of the research and articulating the expectations to the child regarding their developmental competencies, receiving consent not only once but for every session with the child, conducting interviews in familiar settings where children feel safe, having experience and training in researcher–child communication, designing follow-up sessions for an occasional emotional crisis intervention, and referring the child to mental health professionals if required. Having followed such a long list of suggestions, this study appropriately followed the ERIC methodology.
The notable paucity of research that adheres to ethics codes specific to the nature of child studies raises questions about IRB processes. Receiving approval from ethical review boards established within universities or independent institutions may not necessarily ensure that the researchers meet the ethical requirements or standards of conduct in their studies. While applying for approval, researchers generally concentrate on the requirements of ethical review boards and fail to review the existing guidelines that provide standards or requirements specific to their research (Bell, 2008). In such cases, instead of supporting ethical research and preventing poor research (Alderson & Morrow, 2011), the functionality of IRBs is reduced to protecting basic human rights only (Tsan & Nguyen, 2019). Moreover, inconsistencies in ethical guidelines complicate IRB functioning. For example, while ethical guidelines related to health research (CIOMS, 2016) state that “children and adolescents who are legally minors cannot give legally valid informed consent, but they may be able to give assent” (p. 67), the ERIC guidance suggests that children are capable of providing consent if they are properly informed and if the adapted consent processes are utilized.
Considerable uncertainty remains about children's rights, benefits, and well-being because of the lack of recent studies on researchers’ understanding and approaches to ethical guidelines in research involving children. Owing to its significance, this qualitative study sought to explore Turkish researchers’ knowledge and approaches related to suggestions in the ERIC guidance (Graham et al., 2013) in their research involving children.
Method
This qualitative study utilized interviews to explore researchers’ knowledge of and approaches toward ethical guidelines in research involving children, following the recommendations of the ERIC guidance. The following sections summarize the participants’ characteristics, data collection, and analytical processes.
Participants
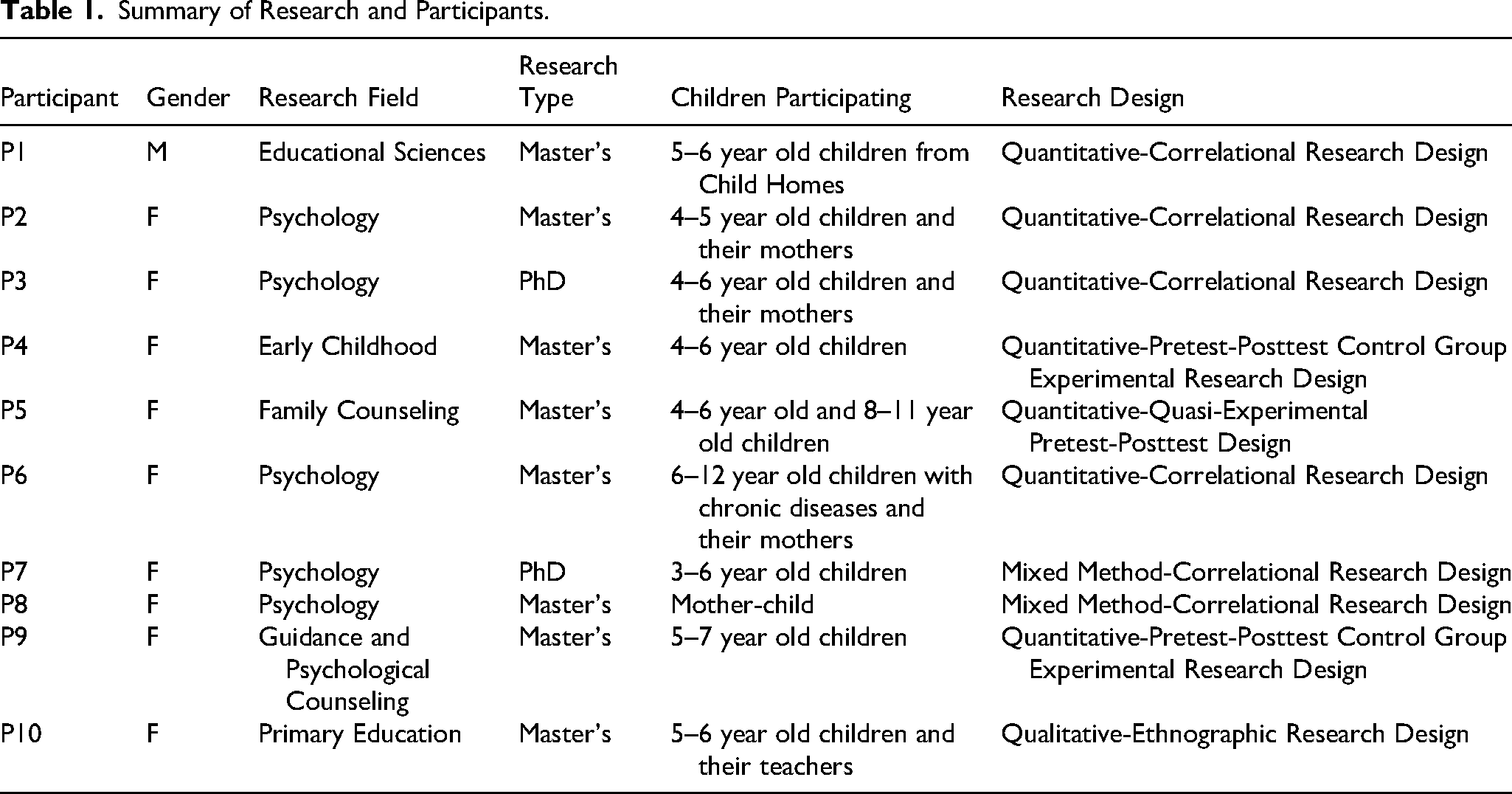
The sample comprised researchers who had completed their master's or doctoral research involving children in Türkiye. A search of the Higher Education Council Thesis and Dissertation Center was conducted to locate the related research by using the keyword “child,” which revealed 18,318 studies. After excluding studies in the health sciences, the authors of the remaining 178 studies in social sciences were sent an invitation by e-mail. Ten researchers accepted the invitation and were interviewed via Zoom meetings, with each lasting approximately 45 min. The demographic characteristics of the participants are presented in Table 1.
Summary of Research and Participants.
Data Collection and Analysis
Prior to the research, TED University Institutional Review Board approval (No. 2023/01) was received, and informed consent was obtained from the participants. This qualitative study used interviews to gain better insights into the participants’ approaches to ethical rules in their research on children. A semi-structured interview form was developed for this purpose. The design of the form was based on the scope, ethical approaches, and critical issues addressed in previous studies. After the participants’ consent to participate, a semi-structured form with 12 main and 18 probe questions was used to interview the researchers on topics such as their research purposes; the resources they used on children's rights and the methodology of doing research involving children; how they detected the risks, benefits, limitations, and sensitive issues during data collection; the ethical principles they followed; what they experienced during the process; and their suggestions for other researchers. The Zoom sessions were conducted in Turkish, and all interviews were recorded for subsequent transcriptions.
The transcribed interviews were coded according to the main themes in the ERIC guidance, and subthemes were subsequently generated. Two researchers with experience in qualitative analysis reached full agreement on the coding process, which ensured the reliability of this research. The researchers examined the transcripts independently, exchanged codes, and discussed and resolved conflicts. The codes and accompanying sample statements were then translated into English by another researcher with an excellent command of both languages. The researchers maintained trustworthiness through credibility, transferability, dependability, and confirmability. Credibility was ensured by receiving expert opinions in the development of the interview protocol; recording and transcribing the interviews; adopting coding procedures and generating themes based on related literature; ERIC guidance being handled by researchers with expertise in ethics; and using participation validation to complete missing data. Studies comprising children were identified through purposeful sampling to ensure transferability. The utilization of an external researcher to review the related literature, ERIC Guidance, and qualitative analysis method ensured dependability. Finally, confirmability was ensured by reflecting on the theoretical details of the codebook, participants’ verification of the coded data, and a literature review.
Results
Based on the ERIC guidance, the findings were organized under four main themes and several related subthemes.
Harms and Benefits
Considering the main theme of harms and benefits, the participants’ accounts were grouped under the subthemes of preparation, data collection, and presentation of study results.
Preparation
There was common sense among the participants regarding the necessity of conducting research with children. One participant believed in hearing the child's voice, saying, “We wanted children to be active participants in this study. Even though they are immature, I believe they have the right to speak. Being young is not an obstacle to reflection; we have suitable methods. […] In short, we prefer to conduct research with children, not on children.” Another researcher commented on collecting data directly from the child, “There is a version of the creative thinking skills scale, scores of which are collected from the teachers. I think when you collect data from the child, you are face-to-face with the data. The other way is to collect data from a transmitter. For several reasons, when collecting data from a parent, as in other studies, by instinct, the parent may involuntarily try to show the child in a better light. Alternatively, the teachers may appear unconcerned. Unfortunately, they don’t want to complete many questionnaires and may make an effort to show that their classes are better, or they may not be objective or holistic. This is because the behaviors of children at home and in school are different. I preferred to work with children because I believed you could collect more accurate data from the child when you were alone with them, asking more open-ended items instead of true–false tests. I knew it would be harder.”
Accessibility was greatly valued when selecting children for research. As one participant commented, “I examined the preschools subsidiary to primary schools; however, the conditions of independent preschools were better. Classroom setting, size, and all available conditions, such as suitability for scale implementation, were examined. […] As a result, I selected the best school and even the best classes.” The researchers stated that they preferred classes or children already enrolled in their institutions; however, a few researchers reported that they selected child samples from other institutions, such as Love Houses (a sort of childcare center) or hospitals. For example, one interviewee said, “[…] First we collected data from the children in hospitals because we wanted to match their demographics with those of healthy children.”
The participating researchers expressed various perspectives on risk assessment and management during data collection. The participants listed their possible risk conditions as children not being accessible because of illnesses or disabilities, children showing noncompliance with out-of-routine practices, and/or children being traumatized during data collection. Regarding the caregivers’ reluctance to join, one of the participants remarked, “Now, in our e-book study, we bring together the children and tablets, and many parents might not appreciate this.” Moreover, the researchers’ risk management varied, including learning children's routines beforehand, excluding children with mental or physical disabilities, and/or referring traumatized children to an expert. Regarding risk management, one of the participants commented, “Considering children's ethical values, we tried not to harm or cancel any routines such as sleep time or lunchtime. We planned the design accordingly.”
A common perspective of the interviewees was that they all received the required ethics committee permission before the study. The permissions were obtained from the Ministry of National Education, the Ministry of Family and Social Services, hospitals, and therapy centers where the research was implemented. As an interviewees remarked, “Before data collection, we got permission from the Ministry of Family and Social Services. In fact, because of this sensitive issue, they granted permission with the minister's approval.”
Considering researchers’ competence, most participants stated that they needed no training for implementation, and a few mentioned training on specific techniques and scale implementation. Regarding this issue, an interviewee said, “Already being an active preschool teacher, I did not have any training for the activities. I had scale training.” Another interviewee commented, “I had both training and reading on play therapy approaches. I am a practitioner of four play therapy approaches. Before completing my dissertation on child-centered play therapy, I passed through both training and supervision processes.”
When it came to the preparation phase, every participant stated that they had examined every source that was accessible for use. Some researchers listed sources on research with children, such as the Declaration of the Rights of the Child, the UNCRC, and book chapters on research ethics with children. Referring to one participant's comment, “I used Alison Clark again. I reviewed the books and articles on active participation of two other authors, Heart and Sheer. I asked questions such as ‘What do children do in those processes?’” and according to another account, “I paid attention to the Ministry of National Education (MoNE) curriculum while working with the children. Accordingly, I prepared for these activities. I started by considering their ages and developmental stages.”, the researchers used various sources to help in conducting research with children.
Regarding the final issue of the preparation stage, some participants reported that they had made significant efforts to prepare the physical setting. Others stated that no preparation was necessary as the data would be collected from students who were already enrolled in their classes or counselees from therapy sessions. However, most researchers ensured children's adaptations by introducing the data collectors beforehand, explaining the procedure using games or metaphorical stories or connecting it with warm-up activities. One researcher stated, “[…] Later, I regularly visited the hospital to meet children with chronic illnesses. Most importantly, I grew acquainted with this setting because of regular visits. Knocking on the door to ask for permission to enter was essential for the children interviewees and the families and children who shared that room. […] However, before obtaining consent, I went through the steps of introduction, establishing relationship, and introducing oneself.” Another researcher added, “Before the scale implementation, I went to the children's school, introduced myself, and tried to win their confidence. They saw me that way. We played games and socialized. We established a warm relationship before scale implementation because the scale was slightly longer. I wanted them to approach me with no hesitation, open themselves to me comfortably, and give honest and accurate responses. That's why I went to their classes without any notice and introduced myself to make them think that a teacher came to their class just to play some games with them. This way, they wouldn’t see me for the first time during the scale implementation or pretest.”
Data Collection
The findings regarding the data collection process centered around using various data collection methods and challenges met and managed by the researchers. Most participants reported using valid and reliable instruments, and a few developed their own instruments for data collection. For example, one participant stated, “If you’re asking what I did with the children in the sessions, we used a structured form, like which toys or themes they choose for each session. As I was the therapist, we filled in forms using my notes on each session. We proceeded with my observation forms.” Accordingly, the participants reported that they primarily relied on observation, and some researchers used art activities, interview transcriptions, standard tests, and projective tests (e.g., Draw-a-person Test). For example, one researcher said, “We used the Doll Completion Story Task. In the mother sensitivity observation, we mainly observed the child's setting; however, we also looked at the mother's reactions.” Another researcher remarked, “[…] I administrated the Draw-a-person Test, instructing the students to draw a person, indicate where they would like to be at present, and sketch a family.” While some participants reported challenges related to research content and resistance to participation, a few stated that they experienced no challenges at all. One individual stated, “Because some children did not want me in their rooms, they kicked me out of the room. It was important not to force them and to give them their presents even if they didn’t participate.” Conversely, another participant commented, “I didn’t notice their sensitive issues during data collection. I discovered their need for clinginess. I restrained myself from adding another dimension.” Highlighting the need for the presence of parents while collecting online data during the pandemic, one researcher recollected, “Sensitive issues … I anticipated many sensitive issues during therapy sessions. During the pandemic, we needed the parents to be accessible during the child sessions. Parents should not be present during in-person sessions. However, during online sessions, we (researchers) could not intervene promptly in urgent situations with the child. Therefore, we kept the parents in an easily accessible position to respond to our calls. There were no other risks.”
Presentation of Study Results
One interviewee reported no significant contributions to the child at the macro or micro levels, “To be honest, neither the children nor the parents benefitted from the study. I’m happy for them if they enjoyed it.” However, one researcher mentioned the contribution to the field by recounting, “[…] I thought working with children would greatly contribute to the field. In our country, child research is conducted with teachers, but I disagree with only relying on the teacher's perspective.”
Informed Consent
The participants’ comments were grouped under the subthemes of informing and obtaining consent.
Informing
Although many researchers mentioned that they did not inform the children about the study, several noted that the research content was thoroughly shared with the parents or caregivers only. One participant commented, “But I can’t measure their conditions openly with children as it was necessary to evaluate these through the caregivers. Therefore, I designed the activity with the parents. The children were too young and not yet at the formal operational stage, and their mode of communication was through games and toys. They were not mature enough.” A sizeable number of interviewees stated that they told children that they would play games, as illustrated in the following comment, “Simply put, they believed they were just playing games with us. If we called it a test, it could sound frightening. Even though it didn’t sound ethical, the children were unaware that they had participated in a research. […] In fact, we must discuss the restrictions on collecting data from children. Unlike data collection from adults, where researchers are required to inform participants, we did not do the same with children.”
Obtaining Consent
Most participants stated that they had obtained consent from the parents, and, in a few cases, the child's consent was asked for. In most cases, the child's assent was assumed. Regarding this, one of the researchers commented, “I approached them with questions such as ‘Would you like to join this?’, ‘Would you like to help?’, or ‘Let's do this together, shall we?’ They said yes because I was already their teacher. There was no opportunity for them to decline, making the extent of their free thought questionable.” The parents signed written consent forms, while the children gave either oral or written consent. Talking about the consent from the child, one participant commented, “It was a form of images and written statements. We also provided an oral explanation of the questionnaire. Instead of requiring a signature, we used boxes to allow students to indicate their choices of participation or nonparticipation. They ticked the boxes labeled ‘I want’ or ‘I don’t want’.” In one of the few longitudinal studies, consent was obtained only at the beginning, whereas in another study, consent was obtained for every session. For example, one researcher revealed, “Later, I obtained consent from the child. I repeated this process every week until the end of the project.”
In cases of dissent from either the parents or the child, the researchers acknowledged that they excluded the children from the study. For example, one participant recounted, “With some children, their mothers gave consent; however, if the children did not want it, we stopped instantly.” In a few cases, the researchers stated that they convinced those children to participate or that some dissenting students were self-convinced. As one interviewee stated, “I told them, ‘Alright, if you do not want to, you do not have to.’ When the other children saw this and began talking among themselves, they wanted to join in. Once they did, I included them in the activity. In other words, I considered their participation to be voluntary.” Regarding the management of consent withdrawal, many participants reported no instances of withdrawal, and,. For example, one interviewee said, “I would prefer not to work with them if they did not want to or if they had pain or just had an operation.” Another interviewee recounted, “There could be some parts the child doesn’t consent to. […] Sometimes questions may cause discomfort or trigger other things. At that point, if the child wanted to leave, we didn’t continue. I didn’t think about the data at that time. […] You can force the child to collect data by rewarding them. This is also important to me—not to force the child and harm the productive value of the data collected from that child.”
Privacy and Confidentiality
We grouped participants’ concerns regarding privacy and confidentiality under the subthemes of data collection and securing the collected data.
Privacy During Data Collection
The participants reported that they ensured privacy during data collection by preparing the setting and managing the risks. They stated that they chose empty and quiet rooms to ensure that the children felt comfortable. Two comments on this issue were as follows: “It changed from one school to the other; the aim was to find a place that was empty and quiet all the time” and “We did not want to use the child's room; we preferred another room where the child spent most of their time. They could transfer a few toys that they liked there. In this way, we selected a standard place where the child would not be distracted or feel alienated.” Most researchers mentioned no risks threatening privacy during data collection; however, they said that in some instances, external interventions were carefully handled with warnings. Two participants illustrated this issue, “[…] Hence, we informed the mother beforehand that the 15 min must not be interrupted; if possible, no one else must be present at home; if there was another child who needed care, somebody else must be present at home to give that care. We had no problem with that” and “We informed the parents about the online setting and setup before the online process and what they should be careful about regarding confidentiality and other issues. Later, when the child arrived for the session, we told them, ‘We’ll do the same activities we did in the room but online. We’ll ensure privacy just like when we kept the door closed so that others cannot hear you.’”
Securing Collected Data
Regarding the confidentiality of the participants’ identities, all researchers except one stated that they used code names or hid their identities. For example, one researcher commented, “We created codes during data collection. We used codes to track the questionnaires; they were highly confidential, and we destroyed them directly.” Another one recounted, “The consent form was stored away from other forms and drawings. No one else had access to the consent forms or other documents except me. To calculate inter-rater reliability, some of the drawings were shared with the coders for blind review by hiding participant identities.”
The participants stated that they took measures to ensure the privacy of the collected data by keeping them encrypted on a computer or external hard drive, storing them in a locked closet, or destroying them. As one researcher stated, “[…] After sending the recordings, the therapists removed them from their individual computers. The research team stored these recordings on a private hard drive accessible only to the research team.” Regarding sharing the collected data with third parties, the researchers commented that they did not share the raw data but shared specific results in most cases. One researcher said, “Parents never requested data sharing; I never shared. The teachers wondered about their students’ results, and I kindly turned them down. This is because they were not entitled to the data.” Another researcher commented, “That was also my private space, which could be seen by anyone reading my thesis—our relations and studies become visible. They don’t know the children, but they know me. It doesn’t look good in terms of confidentiality; however, there are such study samples, and such studies should be conducted to make the child's voice heard.”
Payment and Compensation
In their accounts of the events surrounding payment and compensation, the researchers chose to either present gifts or rewards to the participating children or not. For example, one researcher reported, “In our e-book study, we gave books to the children as a gift. I remember that we gave stickers in the robot study.” By contrast, another researcher commented, “No, I didn’t give anything. Rewarding a child in therapy is not recommended, so it didn’t happen.” Only in a few cases did the researchers report announcing the time of payment at the beginning of the study or presenting gifts to all the children upon the study's completion. One comment illustrated this issue: “[…] Even though they didn’t participate, it was crucial to give them gifts. When the study concluded, we gave all the children, including the research participants and children in the room, a drawing book and crayons because I think these are important.”
Discussion
The ERIC guidance seeks “to ensure that the human dignity of children is honored and their rights and well-being are respected in all research, regardless of context” and aims to strengthen “critical reflection, dialogue, and collegiality across the international research community” by supporting all stakeholders who potentially influence research with children (Graham et al., 2013, p. 2). While researchers, from early on in their career to those with more experience, and research institutions are supposed to benefit from this compendium in their projects from conceptualization to post-dissemination impact planning, the research ethics committees will find it useful in reviewing and approving submitted projects by considering children's backgrounds, vulnerabilities, diverse contexts, and assumptions about the child. The results of our study are discussed below using the guidelines for four themes that the ERIC compendium offers to solve these problems.
Considering the harms and benefits, the ERIC guidance suggests that researchers should maintain a strong understanding of the need to conduct research involving children and provide sound justifications for including or excluding them, starting from the planning phase. Additionally, researchers should weigh the benefits for child participants during and after the research process (Graham et al., 2013). Regarding the harms and benefits, the findings of our study were not encouraging. The researchers failed to explain the necessity of conducting their research with children and provided little information about any direct or indirect contribution to the child participants. However, they stressed the significance of collecting data directly from the child for the research methodology, reliability, and validity of the study results. When supporting the child after the research, statements showed no follow-up monitoring stage planned as part of their study. Therefore, the researchers did not fully pursue children's benefits in the planning, data collection, and monitoring stages. To minimize harm, the ERIC guidance advocates assessing potential risks, incorporating measures to eliminate adverse consequences, and designing protocols to assist children during and after the study. The researchers highlighted the lack of a risk and harm assessment before the research process, and their arrangements of support for children under stress (e.g., keeping the parent accessible during online sessions with the child) showed the need for more expertise and skills to address those issues and concerns. Regarding obtaining the required permissions, receiving training, and reviewing the sources in the preparation stage to prevent any harm to the child, few researchers identified training (e.g., on special techniques or scale implementation). However, the interviewees shared the perspective about reviewing the necessary sources on doing research with the child (e.g., Declaration of the Rights of the Child, UNCRC, or some book chapters on research ethics with children). All researchers declared that they received the required ethics committee permissions, which included the Ministry of National Education, the Ministry of Family and Social Services, hospitals, and therapy centers where the research was implemented. In addition, following the “do no harm to the child” principle, most participants stated that they ensured the psychological readiness of the participating children before the study and used valid and reliable data collection instruments in their research. As an interesting finding, several researchers said that they did not need a preparation process, including debriefing the child about their participation in the study, designing the physical environment, or having an acquaintance meeting with the child.
The second section of the ERIC guidance focuses on informed consent, which can be examined under two titles: (i) informing the child about the purpose of the research and their involvement and (ii) obtaining the consent. The compendium is “designed for research with children and young people under the age of 18 years” (Graham et al., 2013, p. 26). While no practical distinctions are provided for different age groups (e.g., preschoolers and adolescents), it strongly recommends informing the child and gaining their consent to participate and offers some flexibility in practices owing to legal, social, and cultural contexts. The compendium suggests that researchers should clearly explain the purpose of the research to the child and inform them about expectations during the participation process. A common element among the participating researchers was that they did not inform children about their participation in the study. In a few instances, they told the children that they would play a game. Instead, the researchers explained the research purpose, content, and processes to the parents and assumed that it was adequate. The researchers waiting for the children's voluntary participation might confirm that the children were not adequately or clearly informed about the research, its purpose, or expectations. These accounts of researchers showed that they challenged “the ethical principles of respect, justice, and honesty” (Graham et al., 2013, p. 67) by withholding information from the children. Instead, they should have followed the conclusion drawn by the compendium: “Children are entitled to see, receive, and impart information; they have the right to know what the research is about and what it involves. Adults have a responsibility to ensure that the information makes sense to children and the research does not place them at harm” (p. 69). Furthermore, in contrast to the traditional practice of receiving only children's assent, the guidelines highlight the significance of receiving consent from all participating children in developmentally appropriate ways. The researchers should accept all decisions of the child, including their reservations and objections, and “assist them to exercise their power and decline participation should they wish” (Graham et al., 2013, p. 63). A recurrent theme in the interviews was that the researchers considered obtaining the children's assent; therefore, they only obtained consent from the parents. This finding reflects the researchers’ adoption of a traditional approach in which children are considered passive participants, as opposed to the current approach in which children are respected for their autonomy, voice, and right to take an active role in their individual development. Interestingly, among the few participants who obtained consent from the child, only one obtained consent separately at each stage of implementation. Furthermore, they ignored the norm of tailoring the consent procedure appropriate to children's evolving capacities. The compendium suggests alternative ways such as “using photographs or video vignettes to decrease reliance on written consent forms” (Graham et al., 2013, p. 61) during the consent process with children, enhancing the reception and understanding of the information by the child. However, in the present study, consent was obtained either orally or, as in the case of parental consent, only in written form. Finally, ERIC suggests the sensitive observation of the child's behavioral (e.g., silence, uneasiness, multiple yawns, and avoiding eye contact) and verbal (e.g., “I’m tired,” repeated responses such as “I don’t know,” or irrelevant answers) signs of dissent, thereby acting according to the child's unwillingness to participate. An overwhelming majority of researchers reported no child dissent in their studies. This could indicate that such behavioral or verbal signs went unnoticed during the research process or that the power relations between the adults and children in the Turkish context might have compelled the children to comply with requests, reflecting the emphasis on obedience in Turkish culture (Sunar & Okman Fisek, 2005). However, a few statements on ending data collection with children unwilling to participate could be considered an ideal researcher behavior as suggested in the compendium.
The section on privacy and confidentiality in the compendium states that researchers should be concerned about and sensitive to privacy during data collection and information dissemination. Some researchers’ statements indicated that they were alone with the child during data collection; however, contrary to the ideal practice suggested by the compendium, children were not involved in choosing where the research would take place. Additionally, many researchers provided accounts of the parental interventions while collecting data from the participants’ homes. Although the parents were informed about the data collection process and the consequences of various interruptions in a written directions form beforehand, the researchers claimed that they had to issue oral warnings again. These findings highlighted the difficulties faced by researchers in ensuring privacy. Various perspectives were expressed regarding the confidentiality of the collected data. While most researchers commented on storing data using code names to ensure participants’ anonymity, only one interviewee mentioned performing split-half protection, which stores individual and collected data separately. Additionally, as suggested by the compendium, almost all participants commented that they stored the data in a soft or hard copy in locked storage or an encrypted computer file, and other comments concerned the disposal of collected data after use. These results suggested that the collected data were safeguarded and that there was no serious threat to privacy and confidentiality. However, making the collected data accessible to the child whenever they wanted and without the presence of another person was not a method known to or practiced by the researchers in the present study. Moreover, considering the sharing of study results with third parties, the ERIC guidance suggests respecting children's autonomy and the right to decide how much data should be accessible and by whom. Considering the social and cultural contexts, the emphasis on maintaining anonymity helps prevent harm.
In the present study, although it seemed promising to discover that the researchers did not share the raw data with a third party, one researcher's comment on not maintaining the child's anonymity may not be considered a good practice. Moreover, the negligence of the child's participation in deciding what to share with whom conflicts with the purpose of having the child's voice heard in the study, which can harm children's autonomy. As the compendium suggests, decisions about data sharing should be disclosed during the consent process and clearly understood by the children and their parents. However, as mentioned in the Harms and Benefits section, sharing remarkable findings as summary data could indirectly contribute to the child. The participating researchers reported that they used the summary data only upon request. Consequently, children may miss out on information that could benefit their development or well-being.
The final section of the guidelines focuses on payment and compensation. The reservations of the participants about offering a payment or prize reflect the discussions in the literature on the use of incentives (Taplin et al., 2019). It is possible that the participants who chose not to offer incentives, such as gifts or prizes, were worried about detracting from the purpose of the research. Conversely, Morrow (2009) highlighted that “payments can be made to reimburse expenses; to compensate for time, inconvenience, and possible discomfort; to show appreciation for participants’ help; or to pay for people's help” (p. 8). Accordingly, the participants who offered gifts (i.e., drawing books and crayons) as incentives related to the study's content and scope might indicate the researchers’ preference for reimbursement because the children used their own materials during research, or the gifts were a simple token of gratitude for their assistance. The appreciation of children's participation is also evident in another researcher's account of giving books to children as gifts. Considering the limited accounts on payment and compensation, the participating researchers might not have been competent in designing an incentive strategy and might have overlooked the recommendations on compensating for time, inconvenience, and possible discomfort or exploitation in their studies with children. The researchers might have failed to consider the local social and cultural context when establishing the nature of any payment or reward for children's participation (Graham et al., 2013). Nevertheless, offering incentives to all children in the research context, irrespective of their participation, is an exceptional practice aligned with the ERIC principles.
This study explored researchers’ knowledge and approaches in their studies involving children based on the ERIC guidance. Although not all participants followed the best practices in their studies, the results showed several practices related to each compendium section. All participating researchers applied for ethical approval from IRBs and, in some cases, received permission from governmental offices such as ministries. The findings of numerous incompatible practices in research involving children were particularly prominent in this study. There are three possible explanations for these results. First, IRBs receive uniform applications from every researcher, and, in most cases, these forms rely on several codes suggested in the research ethics guidelines. Consequently, rather than focusing on the research and its specific requirements, the researcher is scrutinized, and the approval falls short of addressing children's needs and rights (Bell, 2008). Fisher and Goodman (2009) stressed the need to oversee ethical rules specific to the context and requirements while working, especially with vulnerable groups such as children. However, IRBs may hover between the necessity of child protection and inclusion in a study. This “protection–inclusion dilemma” may result in IRBs adopting a protective approach toward child participants and, in some instances, may lead to the exclusion of children from research (Friesen et al., 2023). Moreover, the acknowledgment of researchers and society on children's status and perspectives, as noted by Graham et al. (2015), invites a deeper consideration of individual and collective factors. In collectivistic cultures such as Türkiye, where traditional norms often emphasize adult authority and collective decision-making over individual autonomy (Sunar & Okman Fisek, 2005), frameworks aligned with the ERIC guidelines recognizing children as active participants can be developed. A notable and persistent issue in such contexts is the insufficiency of developmentally appropriate, child-friendly informed consent processes and limited efforts to ensure that children fully comprehend their rights and the extent of their participation. To address these shortcomings, it is crucial to encourage culturally responsive ethical practices that integrate ERIC principles within local cultural and legal frameworks. This approach may include the formulation of localized guidelines that integrate ERIC's universal standards with the sociocultural dynamics of Türkiye, the provision of tailored resources for IRBs and researchers, and the active engagement of children and their families in developing research protocols. Such measures could effectively reframe the protection–inclusion dilemma, ensuring the adequate safeguarding of children and their active engagement in research endeavors. Second, in their evaluation of applications, IRBs do not allow researchers to guide their specific research requirements properly. Graham et al. (2015) stated that, unlike other ethics guides offering “formulaic ‘one-size-fits-all’ guidelines” (p. 337), ERIC provides a foundation that facilitates researchers in navigating key ethical considerations specific to their studies. By including sample cases supplemented by suggestions for specific ethical requirements, the ERIC guidance offers proper assistance in engaging the complexities of research involving children. The IRBs should follow the same approach to guide researchers and consider specific ethical issues in different research contexts. Third, after receiving their approvals, researchers can benefit from continuing reviews performed in collaboration and cooperation with IRBs, including the screening of the research process by evaluating remedial measures against complications (Tsan & Nguyen, 2019). The recommendation of several practices by Nguyen et al. (2016) helps researchers better address ethical issues when working with children. These include tracking the expiration dates of the IRB approval, training IRB members and researchers, ensuring the timely submission of continuing reviews for IRB approval, or stopping all research activities.
This research provides evidence of the knowledge and approach of researchers in their studies with or involving children. The results suggested that the participating researchers addressed some basic ethical considerations, such as receiving institutional approval, obtaining informed consent from caregivers, and ensuring the confidentiality of the collected data. However, because they did not use the ERIC guidance in their studies involving children, the researchers seemed to miss several considerations, such as planning a follow-up monitoring stage, receiving consent from the child, observing signs of dissent, ensuring privacy by eliminating caregiver interventions during data collection, and designing an incentive strategy. The results also underscored the necessity of the IRB's role in effectively guiding researchers by considering the specific requirements of their research involving children. These results reflect the researchers’ struggle to adhere to the ethical principles of child research for several reasons, such as their lack of knowledge of and experience with ethical considerations in research with children and the paucity of ethics codes and guidelines specific to child research in IRBs’ review forms and processes. This may lead to shortages in providing the necessary guidance and support.
These results offer insights into the practical value of using a compendium in research involving children. With the UNCRC, the core international document pertaining to children's rights (UNICEF, 1989), as a starting point, the related articles of that convention can be easily traced throughout the ERIC guidance, and the suggestions illustrated in the research findings and case studies from different cultures and contexts can help researchers allow the child's rights to be recognized, respected, and voiced in research. The compendium has the potential to contribute further to its associated website (http://childethics.com) and the reflexive approach it supports regarding ethical decisions and subsequent actions throughout the research process.
Best Practices
This study confirms the need to extend the role of IRBs beyond providing approval to include continuing the review, in which, by following an evidence-based practice of ethics review (McDonald & Cox, 2009), they can better guide researchers through the research processes. However, as Pickworth (2000) highlighted, adding a monitoring role could increase the existing heavy workload of committees and affect researcher and committee relationships. As the aim is not to intimidate researchers but to provide them with the necessary support to better address children's rights in their studies, the IRB forms and expectations can be revised to integrate the suggestions of the ERIC guidance. As stated by Wassenaar and Slack (2016), changes to ethics review systems and ethics guidelines take time and effort because “research ethics is a dynamic area driven by ongoing conceptual and empirical work” (p. 311). Therefore, IRBs can publish best practices, sample consent forms with expected details, flowcharts illustrating risk management strategies and methods, or a list of requirements for research involving children on their websites. Additionally, researchers frequently complain that ethics review committees disregard their discipline-specific research requirements (Malouff & Schutte, 2005). One way to address this criticism could be to have a member who is knowledgeable about or skilled in the research (social sciences or health-related) under review (Wassenaar & Mamotte, 2012). Another option would be to place ethics assessments within departments or providing in-school appraisals. As suggested by Wassenaar and Slack (2016), before submission, researchers may communicate with the IRB about the complexities or controversies of their studies to receive early advice and input.
Research Agenda
Future studies should investigate researchers’ ethical decision-making skills or behaviors regarding the contextual factors specific to their research. These factors greatly influence ethical decision-making (Decker et al., 2011). For this purpose, context-specific vignettes or cases, such as the dilemmas in the ERIC guidance, can be developed. They can be applied to obtain quantitative and/or qualitative data regarding researchers’ decision-making strategies in studies involving children. The results of such research could be used to design a training framework, which can be implemented to explore its impact on researchers’ knowledge of and approaches toward ethical considerations.
Educational Implications
This study identified several ethical considerations that aligned with the suggestions of the ERIC guidelines. The four dimensions presented in the compendium may be used as a framework for designing training programs for basic- and graduate-level students who have the potential for conducting research with children (Marcos Miguel & Noy, 2023; Wassenaar & Slack, 2016) or as a separate course or module integrated into existing research course content. Similar to the Ethics Training Model of Stenmark and Miller (2021), a model specific to children's research could be developed to guide future endeavors. In existing research and related studies (Taplin et al., 2019), researchers call for better guidance in making ethical decisions, which indicates the need for IRB training on requirements specific to research involving children.
Limitations
Several limitations exist owing to the context of this study and the nature of qualitative research. First, the participants were selected from among researchers who had completed their master's or doctoral studies involving children in Türkiye. Second, the results of this study are limited to the self-reported details provided by the 10 participants during the interviews. Finally, regarding the qualitative design of this research, the generalizability of the results is limited in terms of drawing conclusions about the prevalence of ethical considerations in research involving children.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
