Abstract
Deciding to participate in clinical research requires patients to evaluate various trial characteristics. This survey study measured the influence of trial characteristics (visit volume and open data sharing) on willingness to participate in patients with Parkinson's disease. Patients were randomized to either evaluate two trials (joint condition) or a single trial (separate condition). For patients who evaluated both trials, willingness to participate was greater in the restricted science protocol with fewer visits (7.60 vs. 7.12; mean difference, −0.48; 95% CI, −0.83 to −0.14). Patients who evaluated both trials were less willing to participate in the study involving more visits than patients who only evaluated the study involving more visits (7.12 vs. 8.14; mean difference, 1.02; 95% CI, 0.30 to 1.74). Patients have difficulty evaluating information about open science and visit burden when information is presented separately. Although views toward open science were favorable, preferences for fewer visits dominated decisions.
Introduction
When invited into research, patients must evaluate characteristics of trials and weigh them to determine whether to participate (Agoritsas et al., 2011; Gayet-Ageron et al., 2020; Noirmain et al., 2020; Vellinga et al., 2020). For example, a patient might enroll in a trial to access a new therapy, despite the burdens of added research visits. Decisions to participate in research may also involve trial characteristics that are less familiar to patients. This may include open data sharing, which typically involves circulating patient data for other scientific investigations. Open data sharing is increasingly common in research, and has proven especially attractive in disease areas like Parkinson's disease, where patient heterogeneity makes access to large open datasets valuable for discovery and replication (Marek et al., 2011; Riley & Schekman, 2021; Smolensky et al., 2020). Although patients are generally sympathetic to open data sharing (Bergmann et al., 2022; Broes et al., 2020; J. Kim et al., 2019; Mello et al., 2018), attitudes have only been investigated in surveys, where responses are non-binding and where open science (OS) is evaluated in isolation from other trial characteristics. Moreover, the socially desirable nature of endorsing data sharing may bias survey responses. The extent to which open data sharing influences willingness to participate is largely unexplored.
Informed consent is the main vehicle for helping patients make decisions about enrollment. During consent, patients are presented with attributes about a trial. These include procedures, risks, and benefits. The assumption is that patients can evaluate these attributes and arrive at decisions that align with their preferences. However, preferences and decision-making are heavily impacted by extraneous factors like how risks and benefits are framed (S. Y. Kim et al., 2015; Tversky & Kahneman, 1981) or anchoring (Chapman & Johnson, 2002; Tversky & Kahneman, 1974).
Another, less widely appreciated challenge for informed consent is evaluability (Chatterjee et al., 2009; Hsee, 1996; Hsee & Zhang, 2010; Xu, 2020; Zikmund-Fisher et al., 2004). During consent discussions, patients are often given information that is difficult to evaluate because it is abstract or difficult to interpret. For example, patients with life-threatening diseases might become attached to a treatment option when they are told that 30% of patients in studies of the treatment responded. Such attachments may dissolve if patients discover that other treatments in trials produced responses in 60% of patients. Absent comparative information, patients may have no basis for judging whether 30% is a lot or a little. Similarly, open data sharing is widely practiced and is often included in consent discussions. However, OS is poorly defined, and its implications for patients may not be well understood.
Patients have difficulties understanding important concepts in informed consent (e.g., randomization) and the benefits and burdens of research participation (Behrendt et al., 2011; Bergenmar et al., 2011; Coyle & Gillies, 2020; Davies et al., 2023; Gad et al., 2018; Hillersdal et al., 2022; Koonrungsesomboon et al., 2022; Nishimura et al., 2013; Tilch et al., 2022). Ascertaining how patients understand and evaluate different trial attributes is methodologically difficult. Research exploring informed consent and willingness to participate often uses surveys and discrete choice methods (Agoritsas et al., 2011; Gayet-Ageron et al., 2020; Noirmain et al., 2020; Vellinga et al., 2020). Such studies present patients with a series of choices to elicit preferences. Because such approaches provide participants with a comparison, they artificially include more information about trial attributes than what patients would encounter in consent discussions. Therefore, such approaches do not ascertain the extent to which patients would actually consider these attributes in real-life participation decisions (Agoritsas et al., 2011; Hsee et al., 1999; Hsee & Zhang, 2010; Noirmain et al., 2020). A discrete choice method might simply ask patients to choose between participating in a trial involving open data sharing or restricted data sharing approaches. This methodological approach does not resolve whether patients find the attribute of open data sharing easy to evaluate, or whether patients find open data sharing scientifically beneficial when asked about it in a more natural context. Standard approaches for studying willingness to participate are inadequate for understanding which trial attributes matter most to patients in real-world clinical trial participation decisions.
In the present study, we used a different approach to measure willingness to participate. Our approach involves randomizing participants to view and rate their willingness to participate for a single clinical trial (“separate mode”) or two trials (“joint mode”) (Hsee, 1996; Hsee et al., 1999; Hsee & Zhang, 2004, 2010). The advantage of this approach is that it exposes the contingency of patient preferences to the presence of comparative information. By using this approach, we can determine which trial attributes are more evaluable, and therefore, more likely to dominate decision-making when patients have a basis for interpreting attributes. In particular, this approach can measure preferences of hard-to-evaluate attributes, such as open data sharing. Given that patients do not have much contextual information about medical research conduct, preferences about OS may be sensitive to comparative information, such as knowledge that data in most studies are not shared widely (“restricted science” [RS]). If correct, one would expect to observe that participation preferences may change when patients become aware of the possibility of both OS and RS options.
This pilot study is part of a broader suite of investigations aimed at understanding how prospective study participants evaluate trial characteristics and assign value to make participation decisions. Many studies, like those involving tissue biobanking, disease progression tracking, or phase 1 trials, ask patients to endure inconvenience and risk without any meaningful prospect of direct benefit. A key question is: how much burden and inconvenience are patients willing to endure to contribute to science? It seems likely that, for many patients, scientific value is somewhat difficult to identify and evaluate, since most patients have no way of assessing a study's potential scientific impact. If so, one would anticipate that a patient's willingness to participate in a study would be sensitive to contextual information that helps them better evaluate a study's scientific impact. In this experimental study using randomized vignettes of clinical trials, we asked participants to evaluate the burdens of extra clinic visits against the prospect of contributing to medical science by having their data widely shared through OS.
Methods
Overview
Our primary objective was to assess how patients evaluate their preferences about OS and study visits in a hypothetical trial. The main part of our study involved two vignettes and two evaluation conditions. One group of patients was randomized to see one trial vignette in separate mode and asked to rate their willingness to participate (Figure 1). A second group was randomized to see a different trial vignette in separate mode. The third group was randomized to see both trial vignettes in joint mode. One trial involved high burden (8 visits) and high scientific value (open science). The other trial involved low burden (2 visits) and low scientific value (restricted science) (Figure 1). We investigated whether patients who saw the first vignette alone would rate it higher than patients who were presented both vignettes. Observing a higher preference for a trial with a smaller number of visits in the joint mode would suggest that patients are much more enthused about research participation once they realize there are less burdensome trial options. Observing a higher preference for an OS trial under the joint mode would suggest that patients are much more enthused about OS once they become aware that some research restricts data access. Despite greater burdens, this pattern would also suggest that patients are willing to accept more burden if they understand the scientific opportunities of OS approaches.

Study Schematic Showing Randomization of Survey Participants to Different Versions of the Survey Involving Different Vignettes. The Clinical Trial Vignettes are Presented with Burden Attribute Statements Underlined and Societal Benefit Attribute Statements Italicized. Vignette 1 Involves High Burden and High Societal Benefit, While Vignette 2 Involves Low Burden and Low Societal Benefit.
Participants
The target population included patients with Parkinson's disease (PD), aged 18 or older, and were recruited from one of two patient registries, the Quebec Parkinson Network (Gan-Or et al., 2019) and the Canadian Open Parkinson Network (Cressatti et al., 2024). These comprise patients with PD who have expressed interest in participating in research. We focused on PD because of access to a large database for potential research participants at an institution, the Montreal Neurological Institute, which has a policy of supporting open science (Poupon et al., 2017). Participants were contacted by email with a link to the consent form and survey, which was available in either English or French.
Survey Instrument and Administration
We developed a survey instrument using randomized vignettes on Qualtrics. Based on methodological survey creation literature, we designed questions to be clear, simple, and neutral (Boynton & Greenhalgh, 2004; Gillham, 2007; Oppenheim, 2005). We also reviewed items for relevance, redundancy, and wording. To minimize social desirability bias, we formulated questions to be as neutral as possible, and respondents were masked to the manipulation of study factors and the study purpose. We modelled vignettes after other studies using vignettes and the separate versus joint evaluation paradigm (Hsee, 1996; Hsee et al., 1999; Zikmund-Fisher et al., 2004). We piloted surveys among colleagues for clarity, four neurologists for feasibility and plausibility, healthy volunteers online using Amazon MTurk, and six patients with PD who would have been eligible to participate in our study. Following pilot testing, we refined the vignettes.
The first part of the survey presented patients with one (separate mode) or two (joint mode) vignettes. Patients were then asked to use a 10-point scale (1 = not at all willing; 10 = extremely willing) to rate their willingness to participate in each trial. In joint mode, patients rated both trials simultaneously. The trials described in the vignettes varied based on type of drug, number of visits, and presence of open data sharing. In this section, participants also rated trial attributes on their levels of burden and benefit.
In the second part, we surveyed patients on their perceptions towards clinical research (using the Research Attitude Questionnaire (Rubright et al., 2011), altruism (Webb et al., 2000), and open science (Mello et al., 2018). “Open science” was defined in the survey as “all data (except for information that could identify you) becomes broadly shared with other researchers who may use data for other research purposes.” “Restricted science” was described as “data would be used by research team but would not be shared with other researchers.” The final section included participant demographics (e.g., age, gender, education). These sections and data items were based on previous studies measuring willingness to participate (Agoritsas et al., 2011; Gayet-Ageron et al., 2020; Noirmain et al., 2020).
Data from patients were collected through Qualtrics from September 2023 to October 2023. The piloting results (Figure S1, Table S1, Figure S2, Figure S3, Figure S4, and Figure S5), joint version of the survey, and the consensus-based checklist for reporting of survey studies (CROSS) (Sharma et al., 2021), are included in the supplementary material.
Sample Size
We developed a simulation based on a medium effect size of 0.4 for the main effects testing whether participants rate two vignettes (one set) differently and whether participants give different ratings based on presentation condition (separate versus joint). We also used a large effect size of 0.8 for interaction effects between the main effects of the vignette and presentation mode. To achieve 80% power, we calculated we would need 50 participants for each group for a total of 150 participants. Given that the survey was short, we expected minimal drop-out (10%). We therefore aimed to enroll at least 165 patients.
Data Collection and Analysis
Survey data was collected and analyzed in R (version 4.3.1). For our primary analysis, we compared the mean willingness to participate in each vignette. We calculated 95% confidence intervals on the mean difference in ratings using a student's t-distribution. For calculation of confidence intervals, we used paired or unpaired t-statistics as appropriate depending on the sample.
Ethics
This study received ethical approval at McGill University Health Centre and all participants provided informed consent before study participation. This study was conducted in accordance with the Tri-Council Policy Statement (2022).
Results
Sample
Of 1306 patients who were invited to participate, 335 responded and were randomized (26% response rate). Of the 335, 268 responses were considered complete and eligible for inclusion in the analysis (Figure 2). Survey responses that were incomplete or had missing data were not included.
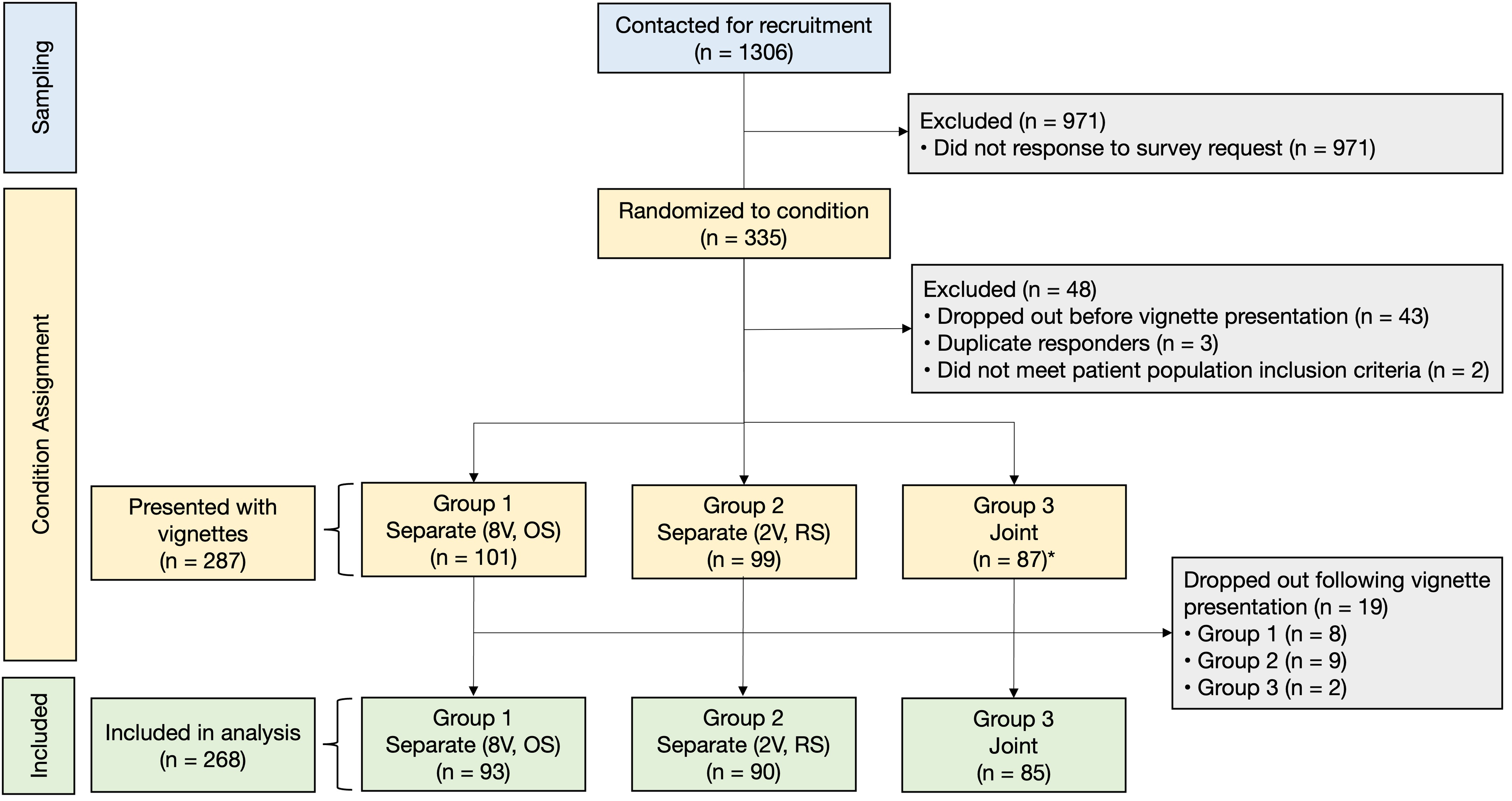
Participant Flow Diagram.
The average age of respondents was 67 years with a range of 50–89 (Table 1). Most respondents (59%) had completed at least a 4-year college/university degree. Sixty-five percent of respondents were not participating in clinical research at the time of survey completion, but 58% of all respondents reported previously participating in research.
Sample Characteristics.
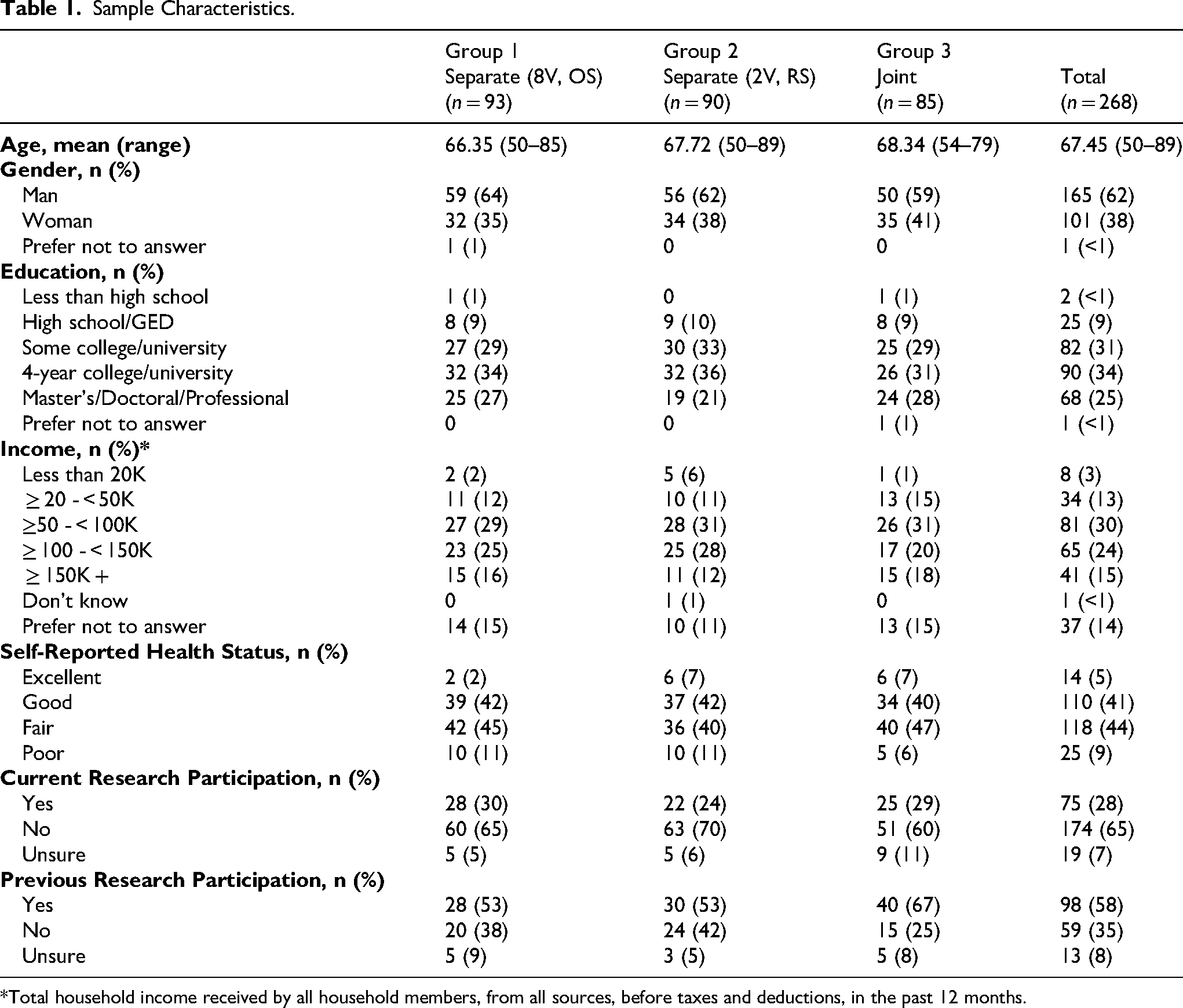
*Total household income received by all household members, from all sources, before taxes and deductions, in the past 12 months.
Impact of Context on Willingness to Participate Ratings
The average willingness to participate rating for respondents who only saw vignette 1 (8 visits, OS) was 8.14 (SD, 1.90) (Figure 3). The average willingness to participate rating for respondents who only saw vignette 2 (2 visits, RS) was 7.83 (SD, 2.38) (Figure 3). The mean difference in preference ratings for the vignettes in the separate mode was 0.31 (95% CI, −0.33 to 0.94) (Figure 4A). When respondents saw both vignettes together, the average willingness to participate ratings for vignette 1 (8 visits, OS) and vignette 2 (2 visits, RS) were 7.12 (SD, 2.81) and 7.60 (SD, 2.36), respectively (Figure 3). The mean difference in willingness to participate ratings for these two vignettes was −0.48 in the joint mode (95% CI, −0.83 to −0.14) (Figure 4A). Participants in the joint condition gave lower ratings to vignette 1 compared to participants in the separate condition (8.14 vs. 7.12; mean difference, 1.02; 95% CI, 0.30 to 1.74). By contrast, the difference for vignette 2 (2 visits, RS) was smaller (7.83 vs. 7.60; mean difference, 0.23; 95% CI, −0.48 to 0.94). Overall, preferences for vignette 1 versus vignette 2 alone—irrespective of presentation mode—did not impact preferences, but preferences varied more within individual vignettes depending on presentation mode (Table S2). Similar willingness to participate preferences were observed in post hoc sensitivity analyses for respondents who said patients “benefit a lot” from open science (Figure S6) and for respondents who are currently or have previously participated in clinical research (Figure S7).
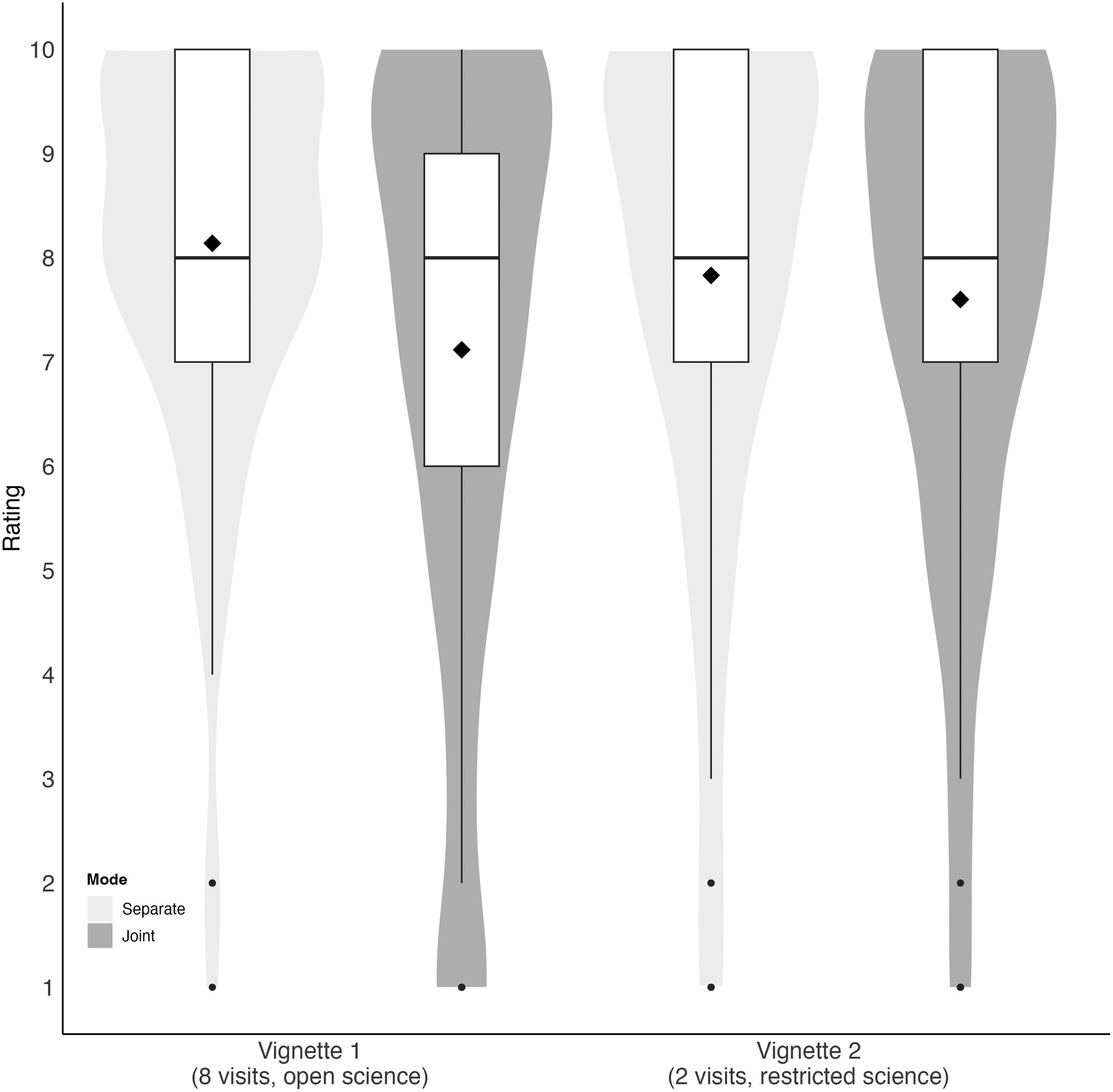
Willingness to Participate Compared Across Study Vignettes and Stratified by Mode of Presentation. Medians are Depicted by Boxplots and Means are Depicted by Black Diamonds (8.14, 7.12, 7.83, 7.60).
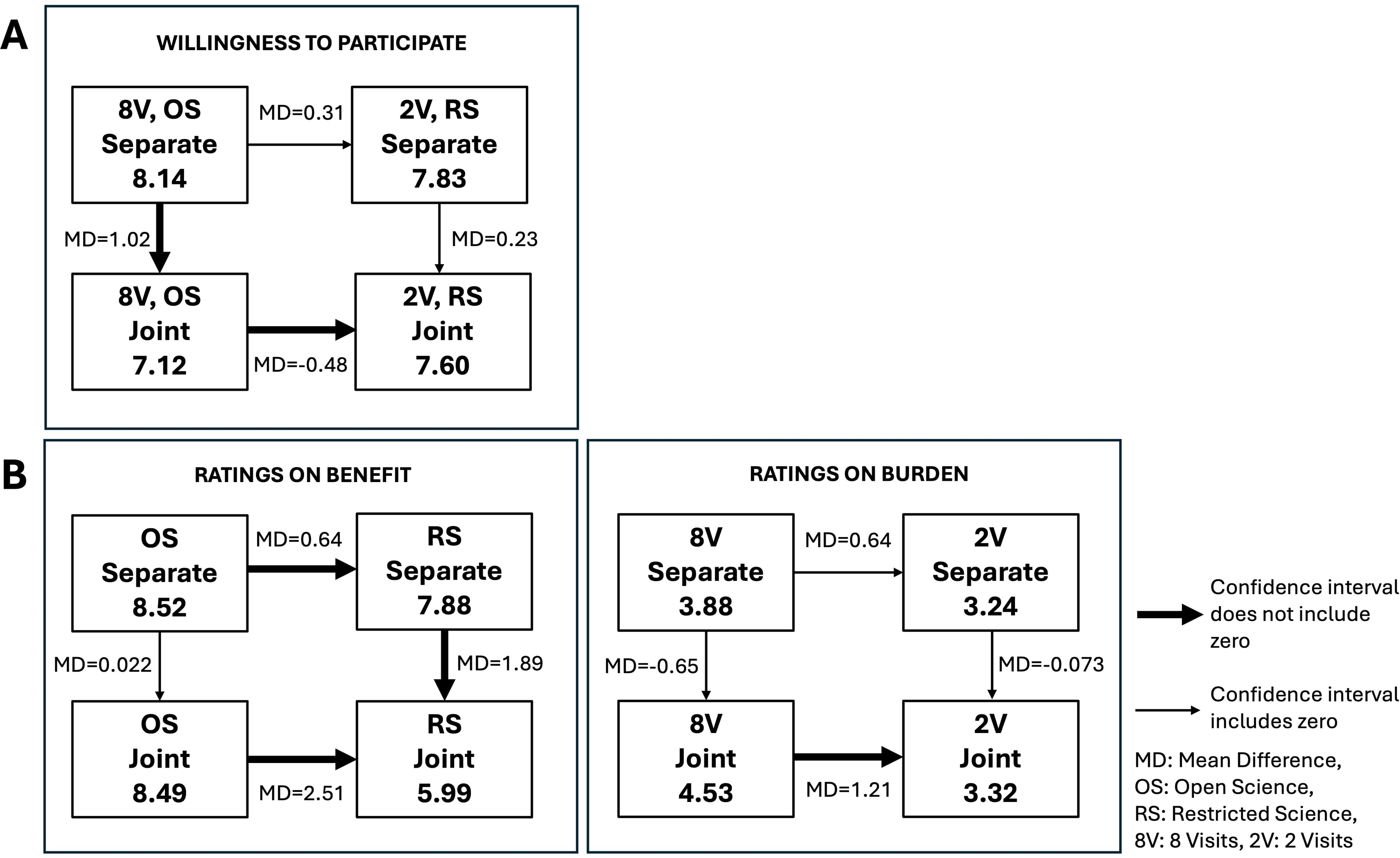
Diagram of Differences in Mean Willingness to Participate Preferences (Panel A) Comparing Separate and Joint Presentation Modes for Each Vignette (1 = not at all Likely to Participate and 10 = Extremely Likely to Participate). Panel B Shows the Significance in Changes of Ratings of Benefit for Open Science (OS) Versus Restricted Science (RS) and Burden for 8 Visits (8V) Versus 2 Visits (2V) (1 = not at all Beneficial/Burdensome and 10 = Extremely Beneficial/Burdensome) in Both Presentation Modes. Horizontal Arrows Show Mean Differences in Preferences or Ratings Between Vignette 1 (8V, OS) and Vignette 2 (2V, RS) in the Same Presentation Mode. Vertical Arrows Show Mean Differences in Preferences or Ratings Between Separate and Joint Mode for the Same Vignette.
Comparisons Between Perceptions of Benefit and Willingness to Participate
Willingness to participate and perception of benefit for OS were positively correlated (separate = 0.44 and joint = 0.35) (Table S3). However, willingness to participate and perception of benefit for RS in joint mode were weakly correlated (0.07), showing low predictability (Table S3). Ratings of the benefit of OS approaches were similar when the OS attribute was presented alone or alongside the RS attribute (mean difference, 0.022; 95% CI, −0.53 to 0.57) (Figure 4B). However, ratings of the benefit of RS approaches were much lower when the RS attribute was presented alongside the OS attribute in joint mode (mean difference, 2.51; 95% CI, 1.83 to 3.18) (Figure 4B). This suggests that OS preferences did not change when viewed jointly but preferences on RS did change. In other words, participants who were only presented with RS did not know that OS was an option, but when participants considered RS and OS options side-by-side, the benefit ratings for RS were lower. Meanwhile, perceptions of benefit related to OS were more stable. Participants who saw the OS option only and participants who saw both OS and RS options, rated the benefit of OS similarly high.
Comparisons Between Perceptions of Burden and Willingness to Participate
Greater perceived trial burden was associated with lower willingness to participate (Table S3). The burdens of eight visits versus two visits were rated similarly when presented in separate mode (3.88 vs. 3.24; mean difference, 0.64; 95% CI, −0.12 to 1.39) (Figure 4B). However, in joint mode, ratings of burden of eight visits were greater than burden ratings of two visits (mean difference, 1.21; 95% CI, 0.70 to 1.72) (Figure 4B). This suggests that participants only came to appreciate that eight visits were very burdensome once they became aware that some studies involved only two visits.
Attitudes Towards Clinical Research
Ninety percent of respondents expressed a general interest in clinical trial participation and 95% maintained a positive view about medical research in general (Figure 5A). Seventy-two percent of respondents believed that participating in medical research is safe. Two-thirds of respondents (67%) believed that research will find cures for major diseases in their lifetimes. Overall, respondents showed strong inclinations towards altruism. Ninety-one percent agreed that people should be more charitable toward others in society, while 79% believed that helping troubled people was important to them (Figure 5B).
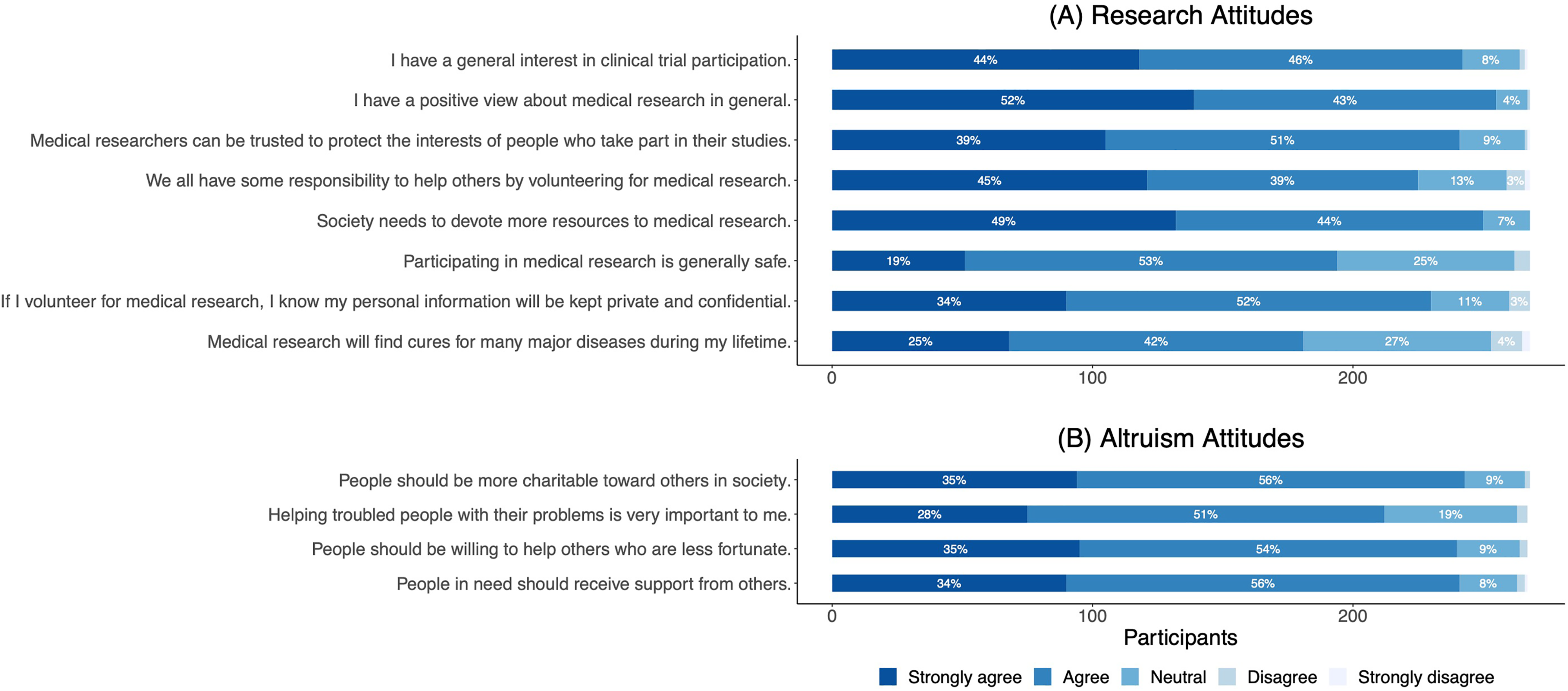
Attitudes Towards Research Based on the Research Attitude Questionnaire (RAQ-7) (A) and Attitudes Towards Altruism (B). Percentages Less Than 3% are not Displayed.
Attitudes Towards Open Science
Forty percent of respondents expressed concern about with whom and for what purposes their anonymous, individual research data would be shared (Figure 6A). However, most respondents trusted sharing their data with university scientists (92%), government agencies (76%), and pharmaceutical companies (61%). Less than half (45%) wanted their data shared with the public.
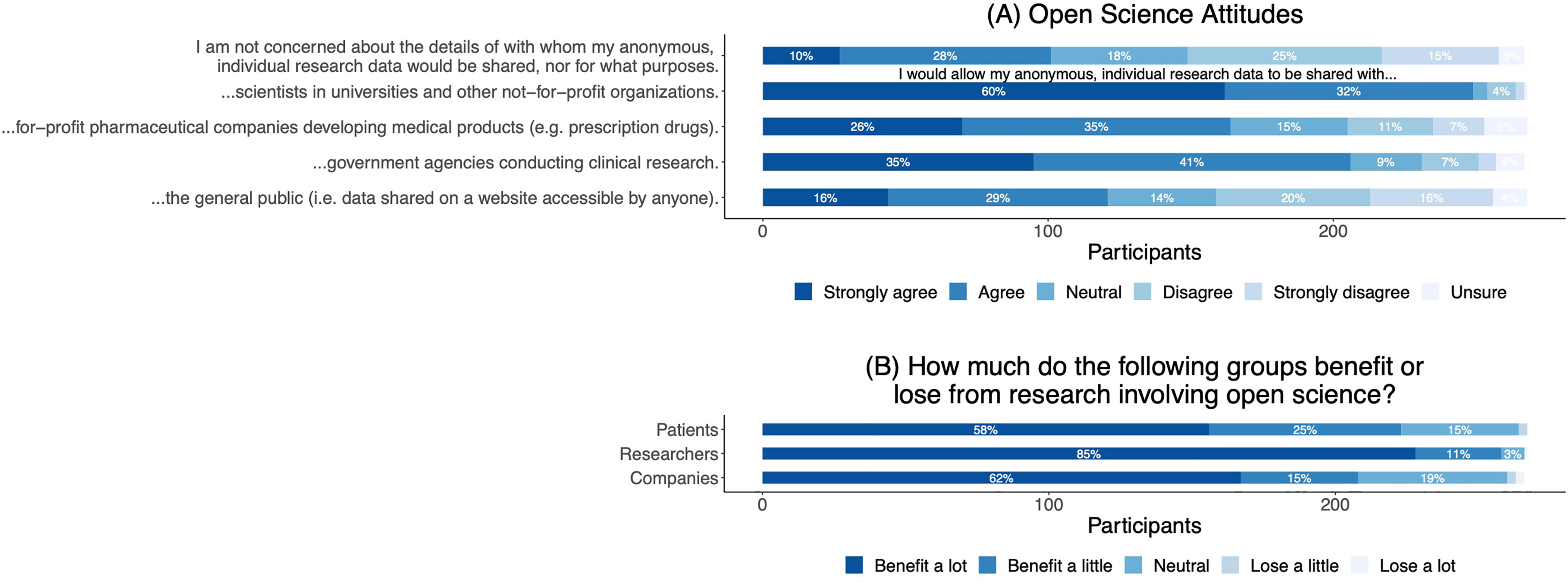
Attitudes Towards Open Science in Research (A) and Perceptions of Benefit or Less from Research Involving Open Science (B). Percentages Less Than 3% are not Displayed.
Respondents believed that researchers benefitted the most from research involving OS (96%), followed by patients (83%) and companies (77%) (Figure 6B).
When asked if participants would want their research data to be completely open (0) or completely restricted (100) on a scale of 0–100, participants (n = 262), on average, were inclined towards openness (22.4), with 213 (81%) providing scores below 50. Respondents were also asked about the benefits and risks of OS. The most common benefits of OS were that it “helps get answers to scientific questions faster using information that others have already gathered” (94%) and it “helps scientists check the accuracy of research results reported by other scientists of companies” (85%). The most common response regarding risks was that there were none (48%) or that information might be used in scientific projects that are not in line with their beliefs (26%).
Three-quarters of participants believed that, even in cases when informed consent could not be obtained, data without any identifying information could be shared. Finally, participants were asked to recall if the consent form that they read for the present study mentioned whether their data would be shared openly. Only 8% correctly recalled that their data would not be shared. Approximately half (53%) incorrectly remembered that the data from the study would be shared openly.
Discussion
This study piloted an approach for assessing how patients evaluate burden and scientific benefit when considering clinical trial participation. We studied how patients rate the burdens of research visits and the scientific value of OS. We anticipated that, because patients are unfamiliar with the scientific process but are generally inclined towards OS, they would have difficulty interpreting OS. We also hypothesized that, because the concept of visit burden is more familiar to patients, preferences about burden would not be affected when participants are given more contextual information.
Briefly, we found that despite patients harboring positive views of OS, they did not show greater willingness to attend more clinic visits for OS studies when they learned that some studies restrict data access. Indeed, preferences for fewer visits proved a much stronger influence. We observed a decline in willingness to participate when patients were presented with greater burden (8 visits instead of 2) in joint mode versus separate mode. This shift in preference suggests that, contrary to our expectations, patients may have difficulty evaluating research burden.
One explanation for our results is that patients do not consider OS to be beneficial. However, this is unlikely. Patients rated OS as being more beneficial than RS in both separate mode (8.52 vs. 7.88; mean difference, 0.64; 95% CI, 0.035 to 1.24) and joint mode (8.49 vs. 5.99; mean difference, 2.51; 95% CI, 1.83 to 3.18). This consistency and the survey responses at the end of the study suggest that respondents recognized the benefits of OS and did not need contextual information to appreciate them. In contrast, ratings of the benefit of RS varied substantially between the separate and joint modes, suggesting that patients may not have a good sense of the baseline assumptions they should make about data sharing requirements in research. Therefore, a more plausible explanation for our observations is that although patients are sympathetic to OS approaches, visit burden dominates preferences. Patients were willing to participate in the RS trial even though they viewed RS to be less beneficial than OS. This suggests that the burden attribute drives participation preferences.
This was a pilot study, and it has limitations. First, the methodological design limits external validity. Although vignettes were piloted in neurologists to test for feasibility and we employed the separate condition to mirror real-world consent discussions, the fact that the survey is hypothetical could have affected responses. Also, the separate versus joint methodology allows for only a small number of vignette variants and testable attributes with a given sample size. This approach is context-specific, and results may differ had other attributes or attribute levels been tested. The main value of the approach is in exploring the volatility of patient preferences, and the extent to which having comparative information might influence patient judgments. Second, our study is at risk of non-response bias since patients who completed the survey may differ in a non-random way from patients who started the survey but dropped out and those who were eligible to complete the survey but did not participate. Third, many respondents rated their willingness to participate as 10, potentially limiting the ability to detect effects due to ceiling effects. If the high-burden trial was made significantly more burdensome, larger effects might have been observed. These ceiling effects could also have been observed if participants perceive trial participation as having direct therapeutic benefit or if they viewed more visits as beneficial because it would enable more contact with doctors. Finally, this study used a sample of patients that may be atypical. Patients in our sample had higher incomes and were well-educated. Furthermore, the average age of patients in our sample indicates they were retired. This could explain high willingness to dedicate time to trial participation. Also, patients with PD may be less risk-averse than other patients (Clark & Dagher, 2014; Kobayashi et al., 2019; Trojano & Papagno, 2018). While studies using this approach commonly show how individual preferences are influenced by the mode of information presentation, we caution against generalization of our findings to other patient populations or to patients who may decide to participate in studies without reading consent forms. The point of our study is to establish the feasibility and value of the evaluability paradigm for assessing patient preferences during informed consent.
Best Practices
This investigation is the first to use this methodological approach to assess willingness to participate preferences of clinical trial participants. Our study found that patients view OS as more beneficial than RS, but only when comparing the two side-by-side. This suggests that patients have favorable attitudes toward OS but are unable to evaluate this attribute without more information. If these findings are confirmed, researchers should provide more information in consent discussions about whether they are using OS versus RS approaches, and why OS approaches might matter. Our finding that ratings of the same trial change when participants also consider another trial side-by-side–which is consistent with the evaluability literature—also has implications for how consent is studied. They suggest that preferences expressed in surveys may reflect the limited context for choices participants are presented with. Best practices for addressing improvements to consent can enable patients to make decisions that align with their values and preferences.
Research Agenda
The complexity of our results reaffirms the importance of exploring the role study attributes relating to risk and value play in patients’ willingness to participate in research. Future studies should build on this and explore other trial attributes, such as their rigor or scientific importance. While OS approaches did not appear to drive participation decisions in our study, other attributes that are difficult to evaluate might.
Educational Implications
Willingness to participate decreased in studies involving more clinic visits when patients had more information about other possibilities. These findings suggest the need to systematically evaluate how communicating time commitments to patients in a comparative manner might help participants make better decisions. Our study also affirms that participants are favorably inclined towards OS trial approaches, and perceptions of benefit strengthen when patients appreciate that some studies restrict data access.
Supplemental Material
sj-docx-1-jre-10.1177_15562646251384573 - Supplemental material for Weighing Open Science Against Research Participation Burden in Informed Consent: A Randomized Pilot Study
Supplemental material, sj-docx-1-jre-10.1177_15562646251384573 for Weighing Open Science Against Research Participation Burden in Informed Consent: A Randomized Pilot Study by Renata Iskander, Patrick Kane, Madeleine Sharp, and Jonathan Kimmelman in Journal of Empirical Research on Human Research Ethics
Footnotes
Acknowledgements
We would like to thank members of the STREAM lab and all individuals who helped to pilot the surveys before they were disseminated to patients.
Ethics Approval
This study was approved by the McGill University Health Centre Research Ethics Committee on January 11, 2022. Respondents gave written consent for review before completing surveys.
Consent for Publication
Not applicable.
Author Contributions
All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication. Conceptualization: All. Data curation: RI. Formal Analysis: RI, PK. Supervision: PK, MS, JK. Writing - original draft: RI, JK. Writing - review & editing: All.
Funding
This work was supported by grants from the Tanenbaum Open Science Institute (TOSI) and the Rossy Cancer Network.
Declaration of Conflicting Interest
Madeleine Sharp declares grants from The Neuro's Tanenbaum Open Science Institute, Fonds de Recherche Québec – Santé, Canadian Institutes for Health Research, McGill Healthy Brains for Healthy Lives, and the Michael J. Fox Foundation. Jonathan Kimmelman declares grants from Canadian Institutes for Health Research, Stem Cell Network, and Genome Canada. Jonathan Kimmelman serves on the advisory board for The National Institute of Allergy and Infectious Diseases Data Safety Monitoring Board. The authors declare no other potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Research data is available in the supplementary material.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
