Abstract
Background/Need
Uncontained power morcellation during laparoscopic gynecologic surgery risks intra-abdominal dissemination of benign or malignant tissue, a significant safety concern highlighted by FDA warnings. This has created a critical need for robust and reliable tissue containment systems that can be easily integrated into surgical workflows to mitigate this risk.
Methodology and device description
The LapBox Power Tissue Containment System is a single-use device featuring a dual-walled inflatable chamber designed to create a secure environment for morcellation. We conducted a Good Laboratory Practice (GLP)-compliant toxicology study in three female domestic pigs to assess its safety under simulated worst-case conditions. The device was inserted laparoscopically, and the internal dual-walled chamber of the device was inflated to a high pressure (∼160 mmHg) to simulate a localized worst-case compressive scenario, while the overall intra-abdominal insufflation was maintained at a standard 15 mmHg. Postoperative monitoring included clinical observation, bloodwork, and, at day 13, necropsy and histopathology.
Preliminary Results
All procedures were completed without mortality, morbidity, or device-related complications. The LapBox maintained full structural integrity. Postoperative clinical, hematological, and biochemical parameters showed no adverse effects. Gross necropsy and detailed histopathology confirmed the absence of device-related ischemia, necrosis, thrombosis, or foreign-body reaction.
Current status
This preclinical study demonstrates that the LapBox Power system has an excellent safety profile and biocompatibility, even under extreme conditions. The device is ready for the next stage of evaluation. These findings support its translational potential and warrant further investigation in human clinical studies to confirm its safety and efficacy.
Need
Minimally invasive gynecologic surgeries, such as laparoscopic hysterectomy and myomectomy, often require electromechanical power morcellators to remove large uterine specimens through small incisions. However, uncontained power morcellation carries a significant safety concern: the risk of disseminating benign or malignant tissues within the peritoneal cavity. The dissemination of benign tissue can lead to parasitic myomas, while the morcellation of an occult uterine sarcoma can upstage the disease and lead to a poorer prognosis.1-3
In response, the U.S. Food and Drug Administration (FDA) issued safety communications recommending that power morcellation be performed only with approved containment systems. While early containment systems demonstrated feasibility, they often suffered from cumbersome structure and risk of damage to surrounding tissues due to the changing inner volume that is caused by the “balloon like mechanism” that creates the pseudo-pneumoperitoneum working area. The lack of visualization and space between the tissue and the bag wall limits the operative field leading to encounters between the morcellator blade and the bag. Additional challenges are the multi-port structure requiring the extraction of several bag opening that are time consuming and cumbersome, leading to higher rates of bag failure and increased operative times.4,5
Technical Solution
The LapBox Power Tissue Containment System (Ark Surgical Ltd., Nazareth, Israel) is a single-use, sterile, non-degradable medical device designed to provide a secure environment for morcellation. It is composed of medical-grade metal, synthetic polymers, and elastomers. The LapBox Power is designed for seamless integration into laparoscopic workflows. The folded containment chamber, housed within a specialized delivery shaft, is introduced into the abdominal cavity via the umbilical incision. Introduction is achieved using a rotating motion and downward push until the shaft’s beveled edge is approximately 2 cm inside the cavity. Under direct visualization from a secondary 5 mm port, the chamber is deployed, the tissue to be extracted is placed inside, and the chamber’s double walls are inflated. The device’s neck (sleeve) is exteriorized through the umbilical incision. A specialized instrument port - tissue retractor that is fully open tube (with no valves) - is then placed inside the exteriorized sleeve. This configuration creates a “contained environment”, allowing the morcellator and auxiliary tools to be placed directly into the bag while preventing any contact between the morcellated fragments and the peritoneal cavity or the abdominal wall (Figure 1). Once deployed, the chamber is inflated to create a large, protected fixed size workspace around the target tissue. The dual-wall inflatable design provides a pressure resistant workspace that maintains its shape independently of intra-abdominal pressure and has the unique architecture that allow it to maintain its shape facing the pressure differential between the atmospheric pressure that exist in the operating room and the work space where the tissue is placed and the surrounding intraabdominal pressure on the outer side of the double walls. A significant advancement over passive pouches that provides a Pseudo-pneumoperitoneum cavity that changes in size as its inner pressure changes when the CO2 escapes through the power morcellator with the tissue that is extracted. This creates a large, protected environment that optimizes visualization, does not require pressure valves to keep the chamber size while providing superior protection due to the double wall barrier. Furthermore, its compatibility with standard workflows eliminates the need for the complex, non-intuitive maneuvers required by earlier retrieval systems. Illustration of the LapBox Power Tissue Containment System, showing (1) the inflatable chamber, (10) the large instrument port for the morcellator, and (11) the medium instrument port for laparoscopic graspers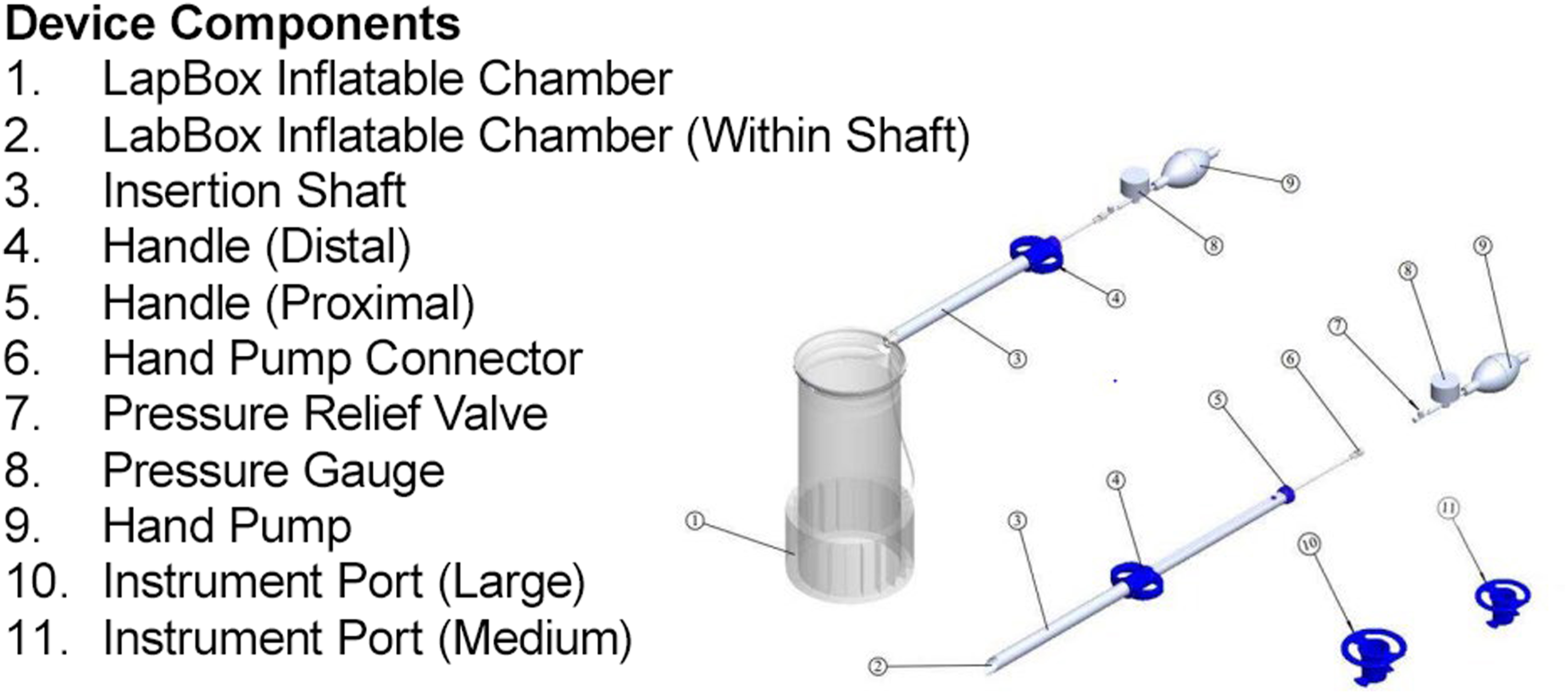
Proof of Concept
We conducted a Good Laboratory Practice (GLP)-compliant preclinical safety and toxicology study (in HBI Biotech Sciences, a Contract Research GLP-accredited Organization) in three female, nulliparous, domestic pigs (Landrace cross, ∼40 kg). The study was designed to assess the safety of the LapBox Power device under simulated worst-case intraoperative conditions. Following GLP and ISO 10993-2 standards, the device was inserted laparoscopically, and the device’s internal chamber was inflated to ∼160 mmHg. This high pressure was chosen to test the “upset tolerance” and localized compressive effects of the device far beyond its intended clinical range. In contrast, the intra-abdominal pneumoperitoneum was maintained at a standard clinical pressure of 15 mmHg. While the clinical device includes a pressure relief valve to prevent over-inflation, this was bypassed for the safety study to ensure maximal contact with abdominal organs.
Animals were monitored for 13 days postoperatively with clinical evaluations, body weight measurements, hematology, and serum biochemistry. On Day 13, animals were euthanized for complete gross necropsy and detailed histopathological evaluation of abdominal organs and tissues.
All procedures were completed without mortality, morbidity, or device-related complications. The surgical procedures were performed by an experienced laparoscopic operator to evaluate the device’s ergonomics and “hand-feel”. The system was successfully introduced, deployed, and extracted in all subjects (3/3) without any reported malfunctions, “stuck” bag scenarios, or delivery shaft failures. The deployment sequence was found to be intuitive, requiring no specialized instrumentation beyond standard laparoscopic tools. The device maintained structural integrity, with no visible malfunctions or breaches, even under high-pressure conditions (Figure 2). Intraoperative laparoscopic views demonstrating device placement and organ appearance. (A) Representative image from Animal #1 prior to introduction of the test device (Day 0). The large intestine is visible in the upper field, and the small intestine occupies the lower field. No gross abnormalities were observed. (B) Representative image from Animal #3 at the conclusion of a 45-minute interval with the test device inflated and filled under peritoneal insufflation (Day 0). The right side of the image displays the inner wall of the deployed containment chamber. Segments of small and large intestine are visible through the transparent base of the chamber. No abnormalities were noted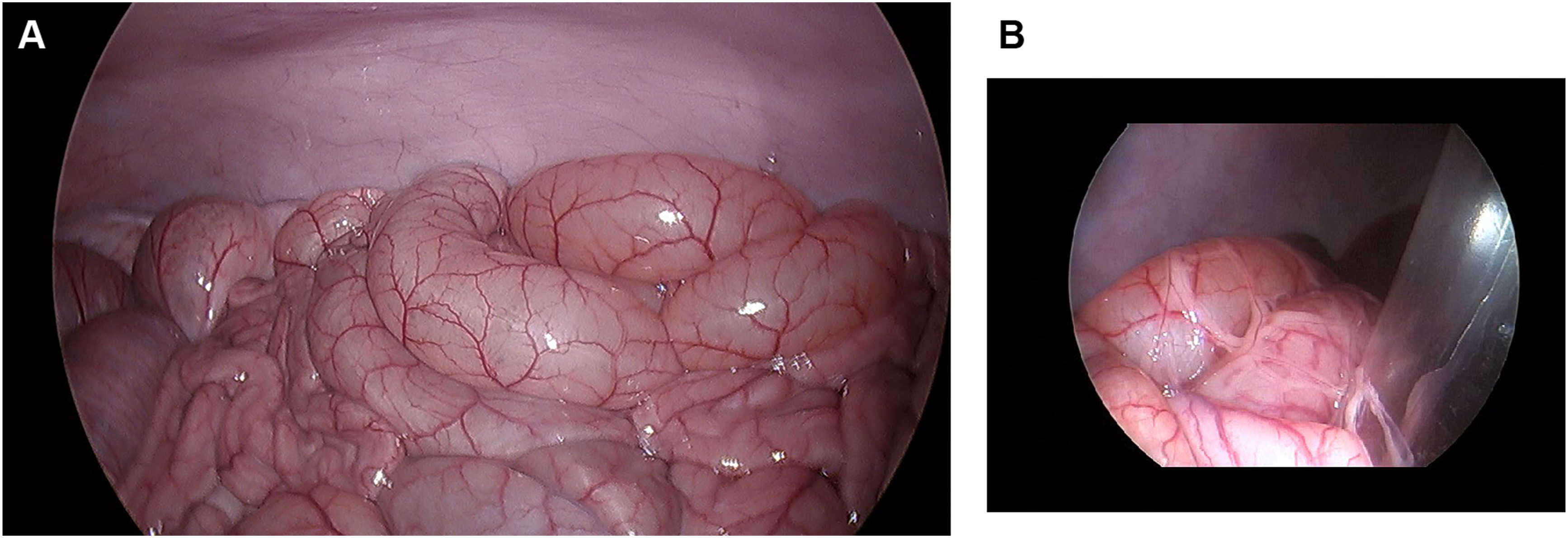
Postoperative recovery was normal. Hematological and biochemical parameters remained within physiological ranges, with only minor, transient postoperative variations (eg, mild thrombocytosis on Day 13) consistent with a normal surgical response. Gross necropsy revealed no abnormalities related to the LapBox Power. No discoloration or signs of ischemia, thrombosis, or mechanical compression of organs were detected.
Histopathological evaluation confirmed the absence of any treatment-related lesions (Figure 3). Specifically, no necrosis, hemorrhage, thrombosis, or foreign-body reaction was identified in organs adjacent to the deployed device. Incidental findings, such as localized sub-chronic serosal inflammation and adhesions in one animal, were attributed to surgical trauma during abdominal closure, consistent with normal tissue repair processes, and did not reflect any adverse response to the device itself. Histopathological evaluation of gastrointestinal tissues from Animal #1. (A) Representative hematoxylin and eosin (H&E)-stained section of the cecum showing normal histological architecture with no abnormalities detected. (B) Representative H&E-stained section of the colon demonstrating preserved mucosal and submucosal structures without evidence of inflammation, necrosis, or other pathological changes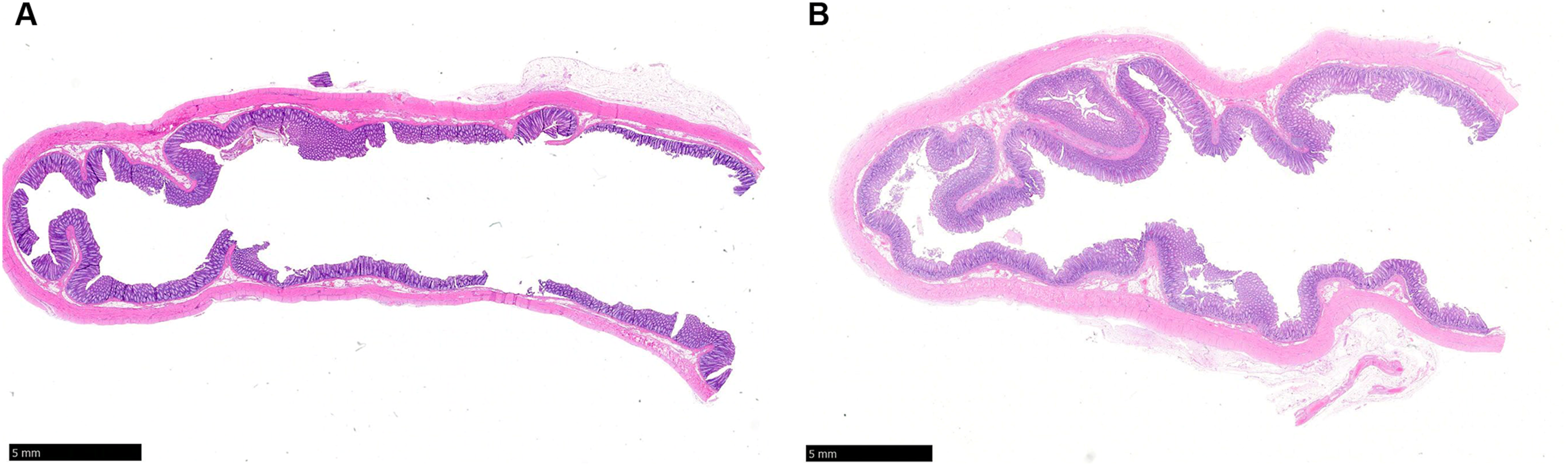
Next Steps
This preclinical toxicology study demonstrated safety, but it was limited by a small sample size (n = 3) and a 13-day follow-up, which may not capture rare complications or long-term outcomes like adhesion formation. Furthermore, while we simulated worst-case pressure and duration, actual in vivo morcellation was not performed. Therefore, potential issues related to blade contact, heat effects, or fragment containment during the morcellation process were not assessed. Future studies should include comparative testing against other containment systems and, most importantly, evaluation in human clinical studies. While this study confirms the basic usability and mechanical safety of the system, future human clinical trials will include formal human factors evaluation. These studies will involve multiple clinical surgeons to assess the ease of introduction across varied patient habitus and the efficiency of specimen loading into the chamber. Parallel to these trials, a formal Health Technology Assessment (HTA) will be conducted. This HTA will employ top-down modeling to quantify the system-wide savings achieved by reducing operating room time and avoiding the substantial clinical and financial burdens of tissue dissemination complications, such as parasitic myomas or upstaged sarcomas.
Conclusion
The LapBox Power Tissue Containment System demonstrated excellent short-term biocompatibility and structural stability in a porcine model, even under simulated worst-case conditions of high localized chamber pressure (∼160 mmHg). The maintenance of standard 15 mmHg pneumoperitoneum, combined with the lack of histopathological evidence of venous obstruction or distal ischemia, confirms that the device does not compromise systemic or local circulation even if accidentally over-inflated. The device behaved as an inert chamber, causing no local or systemic adverse effects, ischemia, or histopathological changes. These findings support its safety and translational potential. By enabling contained morcellation without introducing new device-related risks, the LapBox has the potential to strengthen the safety profile of minimally invasive gynecologic surgery. By addressing the traditional trade-off between containment security and surgical ergonomics, the LapBox Power offers a more robust and user-friendly alternative to existing bags. Its ability to maintain structural integrity under extreme conditions ensures that the surgeon can perform morcellation with higher confidence and reduced technical complexity. Ultimately, the LapBox Power aims to provide a “better and cheaper” solution by combining a superior safety profile with an efficient surgical workflow that supports the continued use of cost-effective minimally invasive techniques.
Footnotes
Ethical Considerations
All animal procedures were conducted in accordance with the Animal Welfare Law (Experiments in Animals, 1994) and approved by the Institutional Animal Care and Use Committee (IACUC), National Permit Committee for Animal Science, under permit number NPC bt - IL - 2310 - 546 – 4.
Author Contributions
Yuval Ramot: Writing - Original Draft, Writing - Review & Editing; Raphael Lioz: Methodology, Investigation, Data curation, Writing - Review & Editing; Tal Levin-Harrus: Methodology, Investigation, Data curation, Writing - Review & Editing; Abraham Nyska: Conceptualization, Methodology, Supervision, Histopathological analysis, Writing - Review & Editing. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Ark Surgical Ltd, Nazareth, Israel.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
