Abstract
Objective
This study aimed to evaluate the safety and effectiveness of an intraluminal fecal diverting device (FDD), COLO-BT™ as an alternative to defunctioning ileostomy in patients who declined stoma formation following proctectomy.
Methods
We retrospectively reviewed the medical records of patients who underwent proctectomy followed by placement of an intraluminal FDD (COLO-BT™) as a temporary substitute for ileostomy at a single tertiary care center between September 2017 and September 2021. Safety was assessed based on postoperative and anastomotic complications, as well as endoscopic findings of colonic changes at the FDD fixation site. Effectiveness was evaluated by the rate of permanent stoma formation.
Results
Among 91 patients, 8 (8.8%) required surgical re-intervention. Of these, five patients experienced anastomotic complications necessitating stoma formation: rectovaginal fistula (n = 2), delayed anastomotic leakage (n = 2), and rectourethral fistula (n = 1). One patient developed severe ileus requiring surgery, which also resulted in stoma formation. Overall, six patients (6.6%) underwent permanent stoma formation during a mean follow-up period of 24 months. Endoscopic follow-up identified colonic luminal deformities in 15 patients (16.5%), most of which were asymptomatic mucosal edema or erosion (n = 12). Three patients (3.3%) had non-symptomatic ulcers at the FDD fixation site. All luminal deformities had resolved on follow-up sigmoidoscopy performed 1 year later.
Conclusion
The intraluminal FDD (COLO-BT™) may offer a feasible alternative to defunctioning ileostomy following proctectomy in selected patients, with acceptable rates of postoperative complications and permanent stoma formation. Further studies are needed to confirm long-term safety and efficacy.
Background
Anastomotic leakage (AL) is one of the most serious postoperative complications following proctectomy, often leading to pelvic sepsis and, in severe cases, life-threatening outcomes. 1 To mitigate the consequences of AL, fecal diversion has become a key preventive strategy, with defunctioning ileostomy currently regarded as the standard approach.2,3 However, ileostomy creation is associated with various complications, including skin irritation, stoma prolapse, parastomal hernia, and dehydration. In addition, it imposes a significant psychological and economic burden on patients, adversely affecting their quality of life and self-esteem. 4
In response to these limitations, alternative methods to avoid stoma formation have been explored. Among them, intraluminal fecal bypass devices have garnered interest as a less invasive approach to achieve fecal diversion. Since the 1980s, several such devices have been developed to replace defunctioning stomas. For example, Coloshield showed promising results in animal models and early-phase clinical trials, reporting no cases of AL in pilot studies. However, it has not reached clinical adoption due to limited availability. Similarly, the C-Seal demonstrated an AL rate of 10.4%, which was inferior to that of the control group in comparative studies.5-8
COLO-BT™ (Colorectal Balloon Tube; JSR Medical Co, Ltd, Daegu, Korea) is an intraluminal fecal bypass device developed as a potential alternative to defunctioning ileostomy. It is the finalized and commercially available version of the fecal diverting device (FDD), which has been under development since 2005. While preclinical and early clinical trials have reported favorable outcomes regarding its safety and efficacy,9-12 evidence from real-world clinical settings remains limited.
Therefore, this study aimed to evaluate the safety and effectiveness of COLO-BT™ in patients undergoing proctectomy who declined stoma formation, using data from routine clinical practice.
Methods
Patients
Patients who underwent proctectomy at a single tertiary care hospital between September 2017 and September 2021 were retrospectively reviewed. Among them, patients who received the COLO-BT™ procedure instead of a temporary loop ileostomy and were over 20 years of age were included. Patients who had previously participated in COLO-BT™–related clinical trials were excluded from the study.
Device Description: Components of COLO-BT™
COLO-BT™ comprises four components: the FDD tube, BT cover, BT bar, and BT band (Figure 1). The FDD tube serves as the main device for fecal diversion and is inserted transanally into the colonic lumen. It includes an irrigation port that allows for the infusion of water to dilute fecal matter, facilitating drainage and minimizing the risk of tube obstruction. The BT cover and BT bar assist with the initial insertion of the FDD tube into the anal canal by maintaining proper angle and alignment. The BT band, composed of absorbent material, anchors the FDD tube in place externally by securing it to the bowel wall for a defined period. Components of COLO-BT™ Kit. (A) FDD Tube, (B) BT Cover, (C) BT bar, (D) BT Band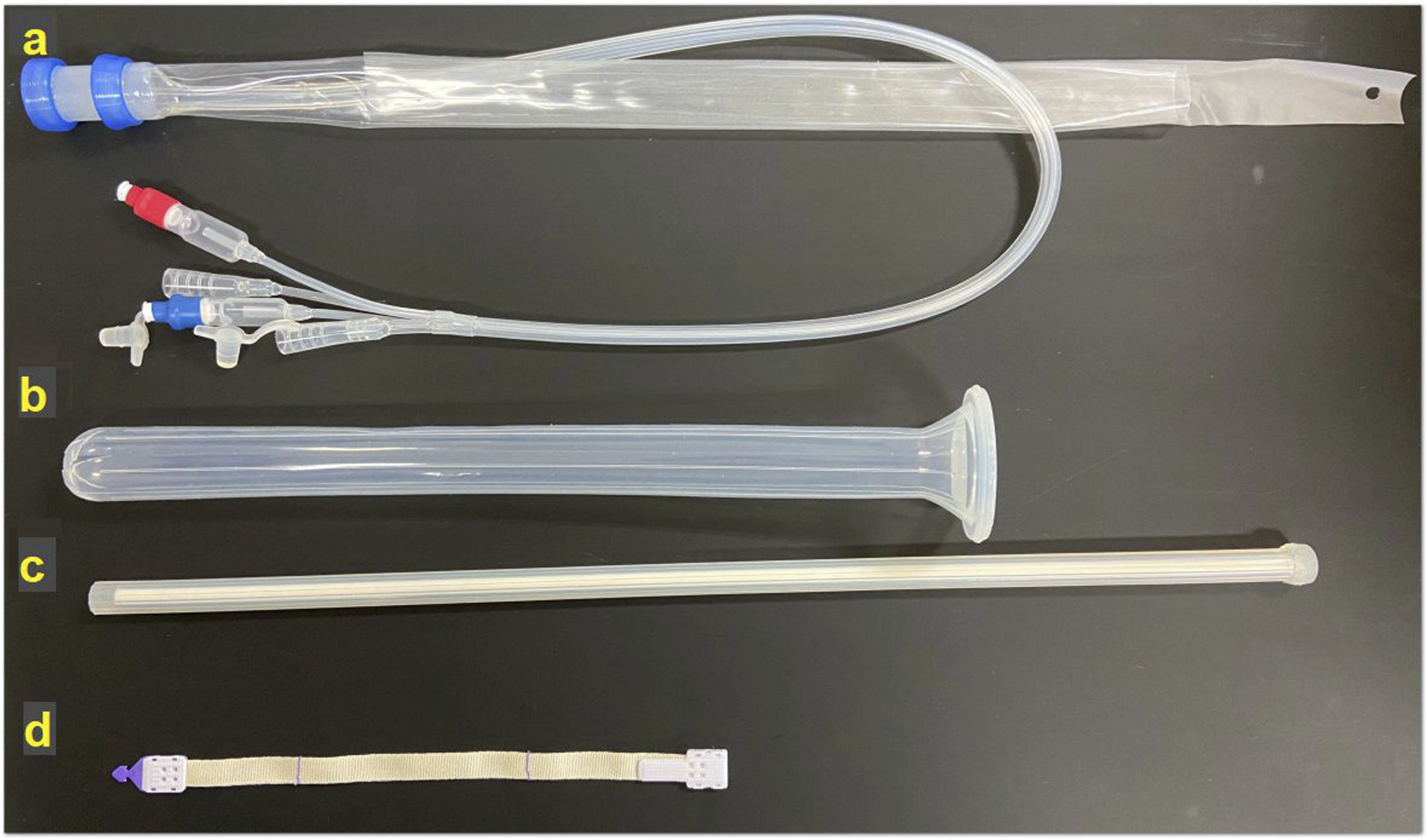
Surgical Technique and COLO-BT™ Insertion
All surgeries were performed using open, laparoscopic, or robotic techniques by board-certified colorectal surgeons. Preoperative bowel preparation was conducted in most patients, except those with bowel obstruction. Prophylactic antibiotics were administered preoperatively to all patients. In patients with rectal cancer, preoperative chemoradiotherapy was administered according to institutional protocols. Intraperitoneal drain placement was routinely performed intraoperatively.
The COLO-BT™ procedure was indicated for patients requiring fecal diversion after proctectomy who declined the formation of a defunctioning stoma. All patients provided informed consent following detailed explanation of the procedure. There were no differences in the application or management of the device among laparoscopic, robotic, and open surgical approaches, as the intraluminal insertion technique was standardized across all modalities.
The BT band was first temporarily mounted on the mesentery adjacent to the colonic wall, approximately 10-20 cm proximal to the anastomosis. The COLO-BT™ device—composed of the BT cover, FDD tube, and BT bar—was assembled outside the surgical field. The BT bar was inserted into the FDD tube and the assembly enclosed within the BT cover. Adequate lubrication was applied to facilitate smooth insertion and future removal.
Prior to insertion, both internal and external balloons were inspected for integrity. After completion of the anastomosis, the COLO-BT™ was inserted transanally and advanced carefully beyond the anastomotic site. The tube was positioned so that the inter-balloon segment aligned with the previously mounted BT band. The BT cover was removed, and the BT band was secured in an annular configuration. The suture was loosely tied to avoid excessive pressure on the colonic wall and to prevent ischemic injury. The outer balloon was then inflated with a water-soluble contrast agent (Gastrotropin) to anchor the FDD tube, and the BT bar was subsequently removed.
Postoperative Management
During the postoperative hospital stay, patients were instructed by medical staff on the proper use of the FDD tube. Typically, fecal excretion through the tube was performed twice daily. This was facilitated by bowel irrigation using a method similar to a water enema.
The process involved inflation of the inner balloon, followed by the infusion of approximately 1 liter of water through the irrigation port. If the patient experienced abdominal fullness or discomfort during infusion, the procedure was temporarily paused. After several minutes, the patient was advised to sit on the toilet and deflate the inner balloon, allowing the diluted stool to be expelled naturally (Figure 2). Pictorial Representation of Fecal Irrigation Using an FDD Tube. (A) Inflation of the Inner Balloon. (B) Water Infusion via the Irrigation Port. (C) Dilution of Fecal Material. (D) Deflation of the Inner Balloon and Fecal Excretion Through the Tube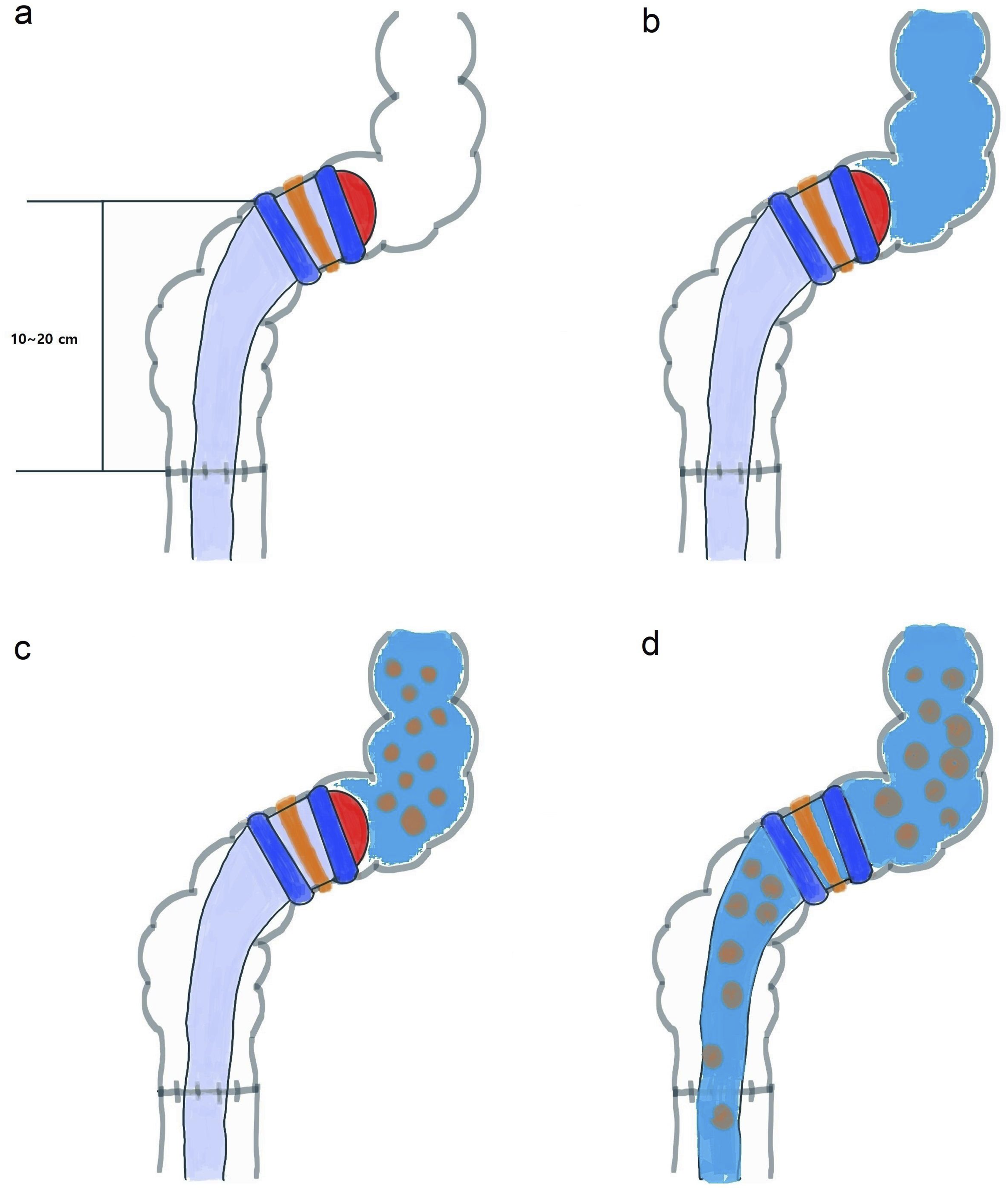
The position of the FDD tube was monitored postoperatively using plain abdominal radiography, as the contrast-filled outer balloon was radiopaque (Figure 3). Additionally, all patients underwent abdominal computed tomography (CT) approximately 1 week after surgery to assess the anastomotic site. In the absence of anastomotic leakage (AL) on a water-soluble contrast enema, the FDD tube was typically removed 3 weeks postoperatively. Removal was performed by deflating the outer balloon and gently extracting the tube through the anus. Subsequent sigmoidoscopy was conducted to evaluate the fixation site and the anastomosis. Final Fixation Status of FDD Tube (A) Intraoperative View. (B) External Appearance. (C) Abdominal Radiograph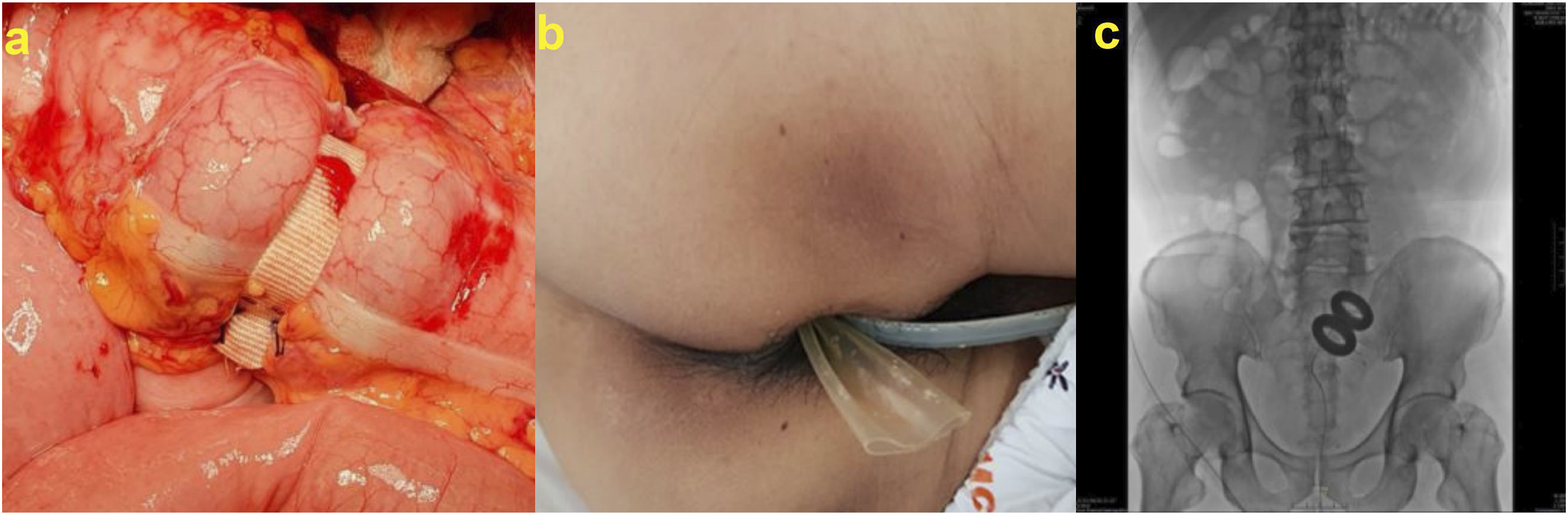
If AL was identified within the first 3 weeks, the FDD tube was maintained for an extended period, as the BT band is designed to retain tension for up to 6 weeks after application. Following tube removal, patients were monitored according to the institution’s standard postoperative protocol for proctectomy.
Outcomes of Interest
The primary outcomes of this study were perioperative complications associated with the COLO-BT™ procedure. To evaluate safety, the incidence of anastomotic complications and colonic luminal deformities at the FDD fixation site were assessed. Effectiveness was evaluated by the rate of permanent stoma formation. Postoperative complications were classified using the Clavien–Dindo classification system, 13 and AL was graded according to the criteria defined by the International Study Group of Rectal Cancer. 14
Results
A total of 91 patients were included in the study, with a mean follow-up duration of 24.2 ± 13.3 months. The mean age was 62.7 ± 12.1 years, and 71 patients (78.0%) were male. Radical surgery for malignancy was performed in 88 patients (98.7%), and 17 patients (18.7%) received preoperative chemoradiotherapy. The mean anastomotic height was 5.9 ± 2.5 cm.
Baseline Characteristics (n = 91)
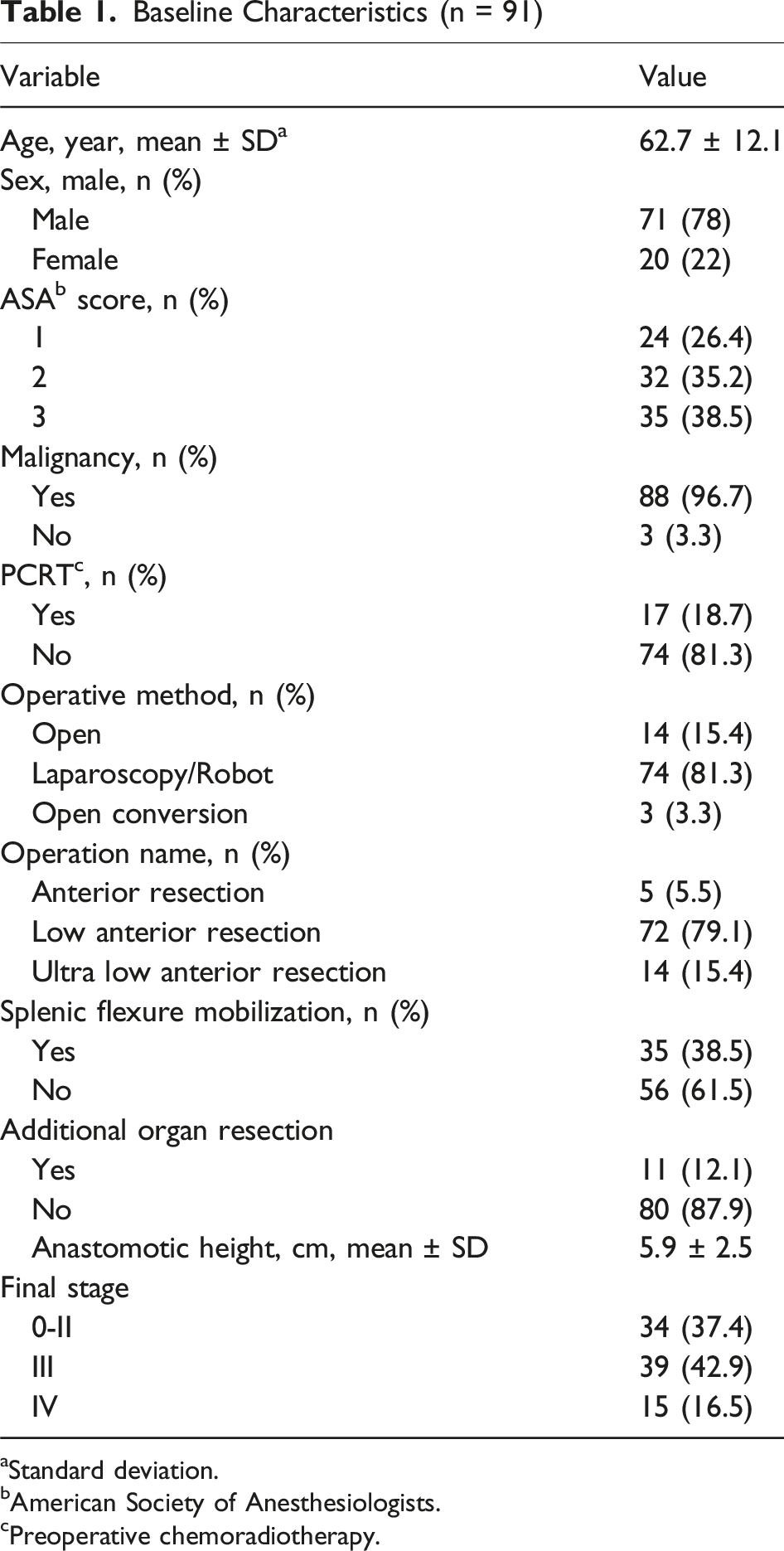
aStandard deviation.
bAmerican Society of Anesthesiologists.
cPreoperative chemoradiotherapy.
Clinical And/or Radiologic Anastomotic Leakage Grade According to International Study Group of Rectal Cancer
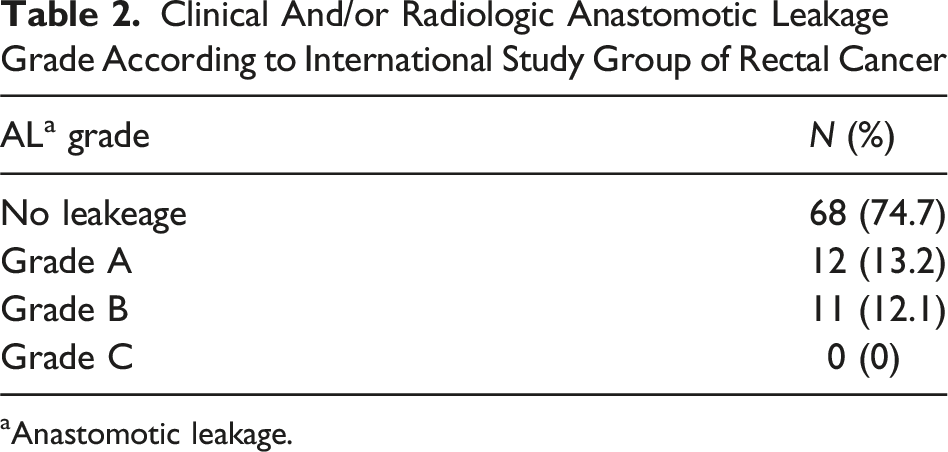
aAnastomotic leakage.
Patients Who had Complication Requiring Surgical Management
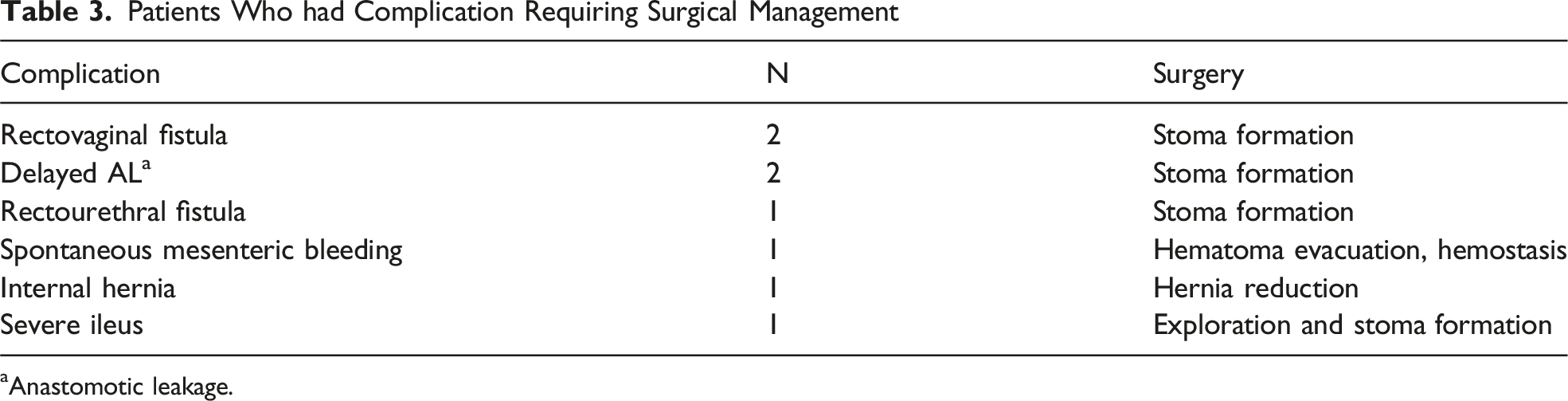
aAnastomotic leakage.
FDD Tube Fixation Site Complications on Sigmoidoscopy, n (%)

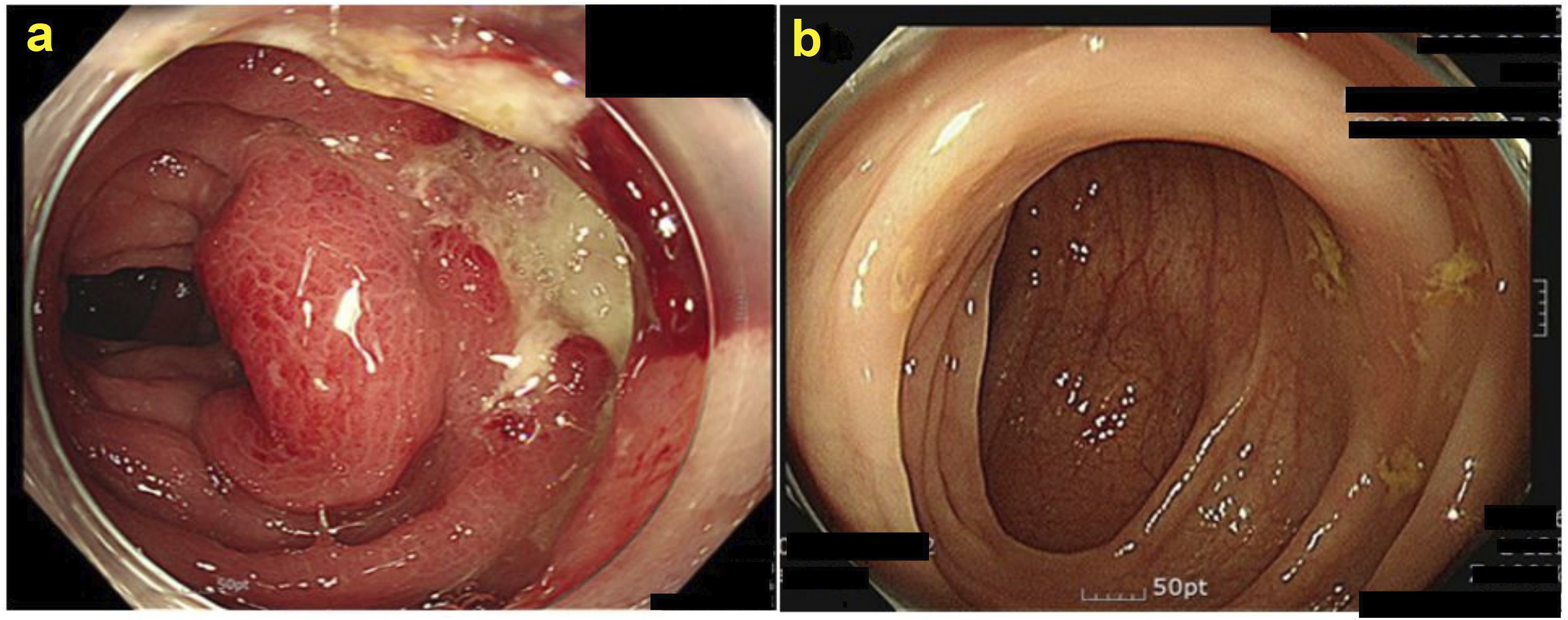
Luminal Deformity on FDD Tube Fixation Site. (A) Sigmoidoscopy Finding on FDD Tube Removal Day, (B) Sigmoidoscopy Finding on 1-Year Follow-up
Discussion
No patient willingly desires a stoma such as an ileostomy, as it is well known to negatively impact both quality of life and self-esteem.4,15 In addition to its psychological burden, ileostomy care entails ongoing financial costs due to the continuous need for appliances and supplies, and often requires subsequent reversal surgery. Furthermore, ileostomies are associated with a variety of complications, including peristomal skin irritation, stoma prolapse, parastomal hernia, and dehydration. 16 A recent meta-analysis reported that postoperative complications—excluding AL—were more frequently observed in patients with ileostomies than in those without. 17 Moreover, the clinical benefit of defunctioning ileostomy in reducing the incidence of AL remains controversial. 18 Despite these limitations, the creation of a defunctioning ileostomy continues to be the standard preventive strategy against AL-related complications following proctectomy.
The development of a procedure for substituting a temporary ileostomy has been attempted for several decades. The simplest way is to use a transanal drainage tube, which was believed to have a protective effect on anastomotic healing. 19 However, a recent large-scale randomized clinical trial found that transanal drainage tubes may not provide a significant benefit in preventing AL. 20 Other devices developed as substitutes for a defunctioning stoma include intraluminal tubes. Since the initial development of intraluminal tube, multiple devices—such as Colo-shield, 6 Valtrac, 8 C-seal, 5 and CG-100 7—have been introduced.
The COLO-BT™ is one of such devices but with some differences. In a multicenter randomized controlled trial, the C-seal failed to demonstrate efficacy in preventing AL compared to defunctioning ileostomy, with a higher incidence of AL requiring intervention observed in the C-seal. 5 Furthermore, a case report described stercoral ulcer perforation caused by fecal accumulation above the C-seal, 21 a complication potentially relevant to other intraluminal devices as well. In contrast, the COLO-BT™ possesses distinct features that set it apart from other intraluminal tubes, potentially providing advantages in preventing accumulation of hardened stool and associated complications. The COLO-BT™ has a fecal dilution system for preventing main tube luminal obstruction due to fecal materials. As a result, the COLO-BT™ had no problems excreting feces through the main intra-luminal tube for the planned 3 weeks or longer.
In this study, there was no grade C AL, which is encouraging for the effectiveness of the COLO-BT™ as a substitute for defunctioning ileostomy. However, the total AL rate was 25.2%, slightly higher than other reports.20,22,23 This result could be because the criteria for AL were strictly applied for patient safety and included all patients with fluid collection around the anastomosis on the CT 1 week after without any clinical symptoms. The previous report conducted by other investigators also revealed 40% AL rates, including 24% of clinical AL and 16% of radiologic AL. 24 Our study’s radiologic AL rate was 13.2%, comparable to the previous report.
The permanent stoma formation rate in this study was 6.6%. One study using data from two prospective studies also reported that the permanent stoma formation rate was 10% in the COLO-BT™ group and 13.7% in the defunctioning ileostomy group. 9 Furthermore, one study reported that the permanent stoma formation rate after rectal cancer surgery with defunctioning ileostomy is 9.5%. 25 Therefore, the permanent stoma formation rate of COLO-BT™ is comparable with the defunctioning ileostomy, according to the available data.
One of the concerns about applying COLO-BT™ to patients was that fixing FDD tube could lead to necrosis of the colonic wall since it was necessary to wrap the colon with a band and apply pressure to the colonic wall with balloons. Indeed, in our previous study, 68% of the patients who underwent the COLO-BT™ procedure had colonic wall abnormalities at the FDD tube fixation site. However, only two (4.0%) had severe complications. However, these two patients with severe deformity with wrap band intraluminal exposure showed no symptoms. Most luminal deformities disappeared after several months, followed by sigmoidoscopy. 9 In the present study, 83.5% of patients showed no evidence of colonic deformity at the FDD fixation site, and only three patients (3.3%) had asymptomatic mucosal ulcers. The reduction in complications may be attributed to improved technique in pressure control at the fixation site and accumulated procedural experience.
This study has some limitations due to its retrospective single-arm design. There may be some selection bias, including the following: first, the participants of this study were only those who wanted the COLO-BT™ procedure instead of stoma formation. Second, since the institution that conducted this study has experience conducting clinical studies on COLO-BT™, this may have affected the results of this retrospective study. To address this potential bias and further validate the usability of the procedure, future studies should consider developing a standardized questionnaire to assess surgeon satisfaction with COLO-BT™. Furthermore, there is currently a lack of efficient and objective data concerning the convenience of COLO-BT™ and patient satisfaction. Nevertheless, satisfaction appears high, attributed not only to the absence of stoma formation but also to the greater convenience of the COLO-BT™ compared with an ileostomy pouch, as it allows scheduled rather than urgent evacuation. Patient experiences reinforced these findings: during hospitalization, they received instruction from medical staff, and after discharge, most reported that self-management at home was feasible and not overly burdensome. Previous study has also shown that the FDD is more cost-effective than ileostomy, offering an additional advantage that further supports its clinical utility. 12 However, concerns remain regarding the need to maintain the device for approximately 3 weeks postoperatively. This practice is based on the healing characteristics of the anastomosis, as tensile strength is minimal during the first postoperative week—when the risk of leakage is greatest—and gradually increases to near-maximal levels within the first month. 26 Accordingly, patients were typically discharged about 1 week after surgery with the FDD in place, and management continued through outpatient follow-up and patient self-care until removal.
In conclusion, this retrospective study demonstrated encouraging results regarding the potential of the intraluminal FDD, COLO-BT™, as a substitute for defunctioning ileostomy following proctectomy, with acceptable rates of postoperative complications and permanent stoma formation. However, further multicenter studies are warranted, and a large randomized controlled trial is currently ongoing (ClinicalTrials.gov ID NCT05826743) to provide definitive evidence on the comparative effectiveness of COLO-BT™ vs diverting ileostomy.
Footnotes
Author Note
An abstract version of this article was presented at the 31st International Congress of European Association for Endoscopic Surgery (EAES) 2023.
Ethical Considerations
The Institutional Review Board of Yeungnam University Medical Center approved this study (IRB no. YUMC 2021-11-047-001).
Informed Consent
Informed consent was waived due to the retrospective nature of the study.
Author Contributions
H.Y. Lee and S.I. Kang contributed to the conceptualization of the study. H.Y. Lee conducted the investigation. S.I. Kang was responsible for developing the methodology and performing the formal analysis. The original draft of the manuscript was prepared by all authors. S.I. Kang and S.H. Kim contributed to the review and editing of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by non-profit grants from the 2021 Yeungnam University Research Grant program (221A580020). The funder had no role in the design, data collection, data analysis, and reporting of this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This article includes all data generated or analyzed during the course of this study. For supplementary information, please reach out to the corresponding author.
