Abstract
Background
Although there is evidence that indocyanine green angiography (ICGA) can predict mastectomy skin flap necrosis during breast reconstruction, consensus on optimal protocol is lacking. This study aimed to evaluate various technical factors which can influence ICG fluorescence intensity and thus interpretation of angiograms.
Method
Single institution retrospective study (2015-2021) of immediate implant-based breast reconstructions postmastectomy using a standardized technique of ICGA, controlling for modifiable factors of ambient lighting, camera distance and ICG dose. “Time to perfusion” assessment was defined as elapsed time from ICG administration to perfusion assessment. Intraoperative “absolute” and “relative” IGCA perfusion values of mastectomy flaps, taken at different time points (30, 60 and 90 seconds), were correlated with postoperative flap outcomes.
Results
There were 260 breast reconstructions with a 3.1% necrosis rate. ICGA perfusion values, when measured at 60 and 90 seconds, were significantly lower for cases that developed necrosis compared to cases that did not, and were both good predictors of necrosis (area under ROC curves, 0.84 and 0.85, respectively). Fluorescence intensity increased as “time to perfusion” assessment increased for flaps that did not develop necrosis (correlation coefficient, 0.9, P < 0.001). Perfusion value cut-off thresholds for predicting necrosis were higher for a longer “time to perfusion” assessment.
Conclusions
A standardized ICGA protocol is recommended as ICG fluorescence intensity increased with “time to perfusion” assessment, and ≤30 seconds did not allow for accurate perfusion analysis. Using a perfusion recording of 60 or 90 seconds, and the corresponding perfusion value cut-off, may optimize reliability of perfusion assessments.
Introduction
Skin perfusion in skin-sparing mastectomy (SSM) and nipple-sparing mastectomy (NSM) is critical to success of the breast reconstruction. Inadequate tissue perfusion can lead to mastectomy skin flap necrosis (MSFN) with reported prevalence of up to 24%.1,2 Early identification of skin flaps with potential to develop ischemia or postoperative necrosis can guide intraoperative decision-making. 3 Indocyanine green angiography (ICGA) involves real-time fluorescence imaging utilizing a near-infrared camera after intravenous administration of the fluorophore, indocyanine green (ICG), to produce angiograms.4,5
While studies have demonstrated that ICGA can help predict MSFN, consensus on protocols and data on factors that influence fluorescence intensity is lacking. Indocyanine green fluorescence intensity, and thus interpretation of angiograms, can be affected by various modifiable factors, including ambient lighting, distance between skin flap and camera, and ICG dosage.3,6 There is no consensus yet on the optimal technique, including “time to perfusion” assessment and which parameters best determine tissue at risk of necrosis, limiting its widespread use. 6 “Time to perfusion” being the elapsed time from ICG administration to perfusion assessment.
Previous studies mainly focus on the effect of ICGA in postmastectomy reconstructions.3,7 This study is of a large series of immediate implant-based breast reconstructions postmastectomy using a standardized technique of ICGA. Modifiable factors of ambient lighting, camera distance, and ICG dose were standardized. The aim was to evaluate factors influencing fluorescence intensity and which perfusion values best predict necrosis, to optimize angiogram interpretation.
Methods
Study Design and Participants
This was a single institution secondary analysis of retrospective data of patients who underwent immediate implant-based breast reconstructions postmastectomy using ICGA, from February 2015 to June 2021. Ethics was approved by the review board of the participating institution (Protocol No: X21-0054 & 2021/ETH00363). Baseline patient and procedural characteristics were collected including age, comorbidities, cancer staging, history of radiotherapy and smoking status. Exclusion criteria was patients with known contraindication to ICG, i.e., previous reaction to ICG, iodine allergy, chronic kidney disease stage 3, 4, or 5, or pregnancy. Patients with a history of diabetes, smoking or preoperative radiotherapy were excluded. Cases that involved axillary dissection were excluded as these tended to have longer skin incisions, wide subdermal dissection and sacrifices of surrounding skin perforator vessels. These factors might have had a negative effect on the mastectomy skin flap perfusion.
Intraoperative information was collected including ICGA perfusion analyses, tissue expander size and volume, and mastectomy breast weight. The “time to perfusion” assessment, defined as the elapsed time from ICG administration to perfusion assessment, at different time points (30, 60 and 90 seconds) was collected for each case. The ICGA perfusion values, corresponding to each time point, of the flaps was also collected. The incidence of postoperative complications, including necrosis, up to 90 days postoperative were documented. Mastectomy skin flap necrosis was defined as all-inclusive necrosis including: epidermolysis, partial-thickness/ superficial necrosis and full-thickness necrosis. Partial-thickness/ superficial necrosis was defined as loss of epidermis, partial dermal loss, and/ or eschar formation without subcutaneous fat exposure. Full-thickness necrosis was defined as loss of epidermis and dermis with exposure of subcutaneous fat, muscle, acellular dermal matrix (ADM) or implant. 8
Surgical Procedure
All patients underwent SSM or NSM followed by implant-based breast reconstruction performed by breast surgeons. All patients had drains placed before skin closure and received preoperative antibiotics. No local anaesthetic or epinephrine-containing injections were used before or during surgery. After completion of mastectomy, laparotomy sponges were placed in the breast pocket to fill the dead space and to allow skin flaps to lie flat. The skin was temporarily closed using staples. This allowed some degree of strain on the flaps that would mimic the pressure from a tissue expander or implant as perfusion analyses were completed. 9 A decision was made to either proceed with immediate reconstruction or to delay the reconstruction based on clinical assessment and the ICGA perfusion analyses. Clinical assessment included skin tissue colour, capillary refill, turgor, temperature, dermal edge bleeding and skin flap thickness. ICGA analysis involved using qualitative images and an “absolute” perfusion value of ≤14 units, at 90 seconds from ICG administration, as an indicator for poor perfusion. Qualitative assessment was undertaken by interpreting the grey scale images recorded. Ischemic flaps demonstrated reduced fluorescence intensity and this could be in the form of diffuse ischemia, geographic ischemia away from the mastectomy wound, incisional ischemia at the mastectomy wound, or nipple-areolar complex ischemia. 10 If the mastectomy flap was evaluated to have poor perfusion using ICGA, then the reconstruction would be delayed as this assessment took precedence over the clinical assessment result. Immediate breast reconstruction was universally abandoned at this stage if a determination of an unsatisfactory perfusion assessment was made.10,11
The implant-based reconstructions were completed using a pre-pectoral tissue expander (MENTOR® Tissue Expanders, Johnson & Johnson Medical Pty Ltd., Macquarie Park, Australia) with or without use of ADM (FlexHD® Acellular Hydrated Dermis, MTF/ Ethicon, Inc., Somerville, NJ; Veritas® Collagen Matrix, Synovis Life Technologies, Inc., St. Paul, Minn.). Tissue expanders were generally filled to 50-75% of total capacity at time of the operation. The alternative to ADM use was TiLOOP Bra mesh (PFM Medical, Germany) and the choice depended on the patient's specific circumstances. Direct-to-implant reconstructions were completed using pre-pectoral silicone implants (MENTOR® CPG™ Gel Implant, Johnson Medical Pty Ltd., Macquarie Park, Australia).
Indocyanine Green Angiography Protocol
Indocyanine green was supplied as a sterile water-soluble lyophilized powder (Infracyanine® 25 mg/10 mL, SERB, Paris, France). It was administered intravenously stat, at a weight-dependent dosage, just after the mastectomy was completed and prior to reconstruction. The dosage was 2 mL for patients weighing <50 Kg, 3 mL for patients weighing 50-100 Kg, and 4 mL for patients weighing over 100 Kg. This ICG dose was followed by a flush of 10 mL normal saline. Operating room lights were turned off and blinds over windows were closed to avoid any potential interference of ambient light with detection of ICG fluorescence. Fluorescence was excited by a near-infrared light source from the SPY Elite Fluorescence Imaging System (Novadaq Technologies ULC, a wholly owned subsidiary of Stryker Corporation, Burnaby, British Columbia, Canada). Distance between the camera imaging head and skin flap was positioned using the 2-dot laser-guided marker in the SPY system. Acquisition buffering was used to capture images for a pre-defined default time of 5 seconds before the operator began recording. This ensured that the initial few seconds of fluorescence were captured. Perfusion was then video recorded for at least 90 seconds after fluorescence was first detected in the mastectomy skin.
The vascularization of the skin flap was analyzed using SPY-Q Elite System software. Quantitative analysis, including “absolute” and “relative” perfusion values, were recorded. “Absolute” perfusion values are based off a fixed grey scale that is consistent from image to image. “Relative” perfusion values are based off a comparison with healthy tissue in the area. 12 Intraoperative ICGA perfusion analyses over 90 seconds and at 30, 60 and 90 second time points were retrospectively reviewed and correlated with skin flap outcomes.
Statistical Analysis
Continuous variables were presented as means with standard deviation (SD) or as median with interquartile ranges (IQR), and dichotomous and categorical data as frequencies with percentages. The cohort was categorized into two groups: those who developed skin flap necrosis and those who did not. Differences between means were analysed using Student’s t test. Categorical variables were analysed using Fisher’s exact test. The predictive power of ICGA was calculated based on the correlation of perfusion values to areas of necrosis observed clinically. Sensitivity and specificity was calculated for every perfusion reading associated with necrosis and results ranked in numerical order to determine a cut-off score for necrosis. 12 Pearson’s correlation coefficient was performed to evaluate the relationship between “time to perfusion” assessment and perfusion values. A P value <0.05 was considered significant. Receiver operating characteristic (ROC) curve analysis was performed to assess ability to predict postoperative necrosis. Statistical analysis was performed with RStudio, version 2024.09.0.
Results
Casemix Characteristics
Casemix Characteristics.
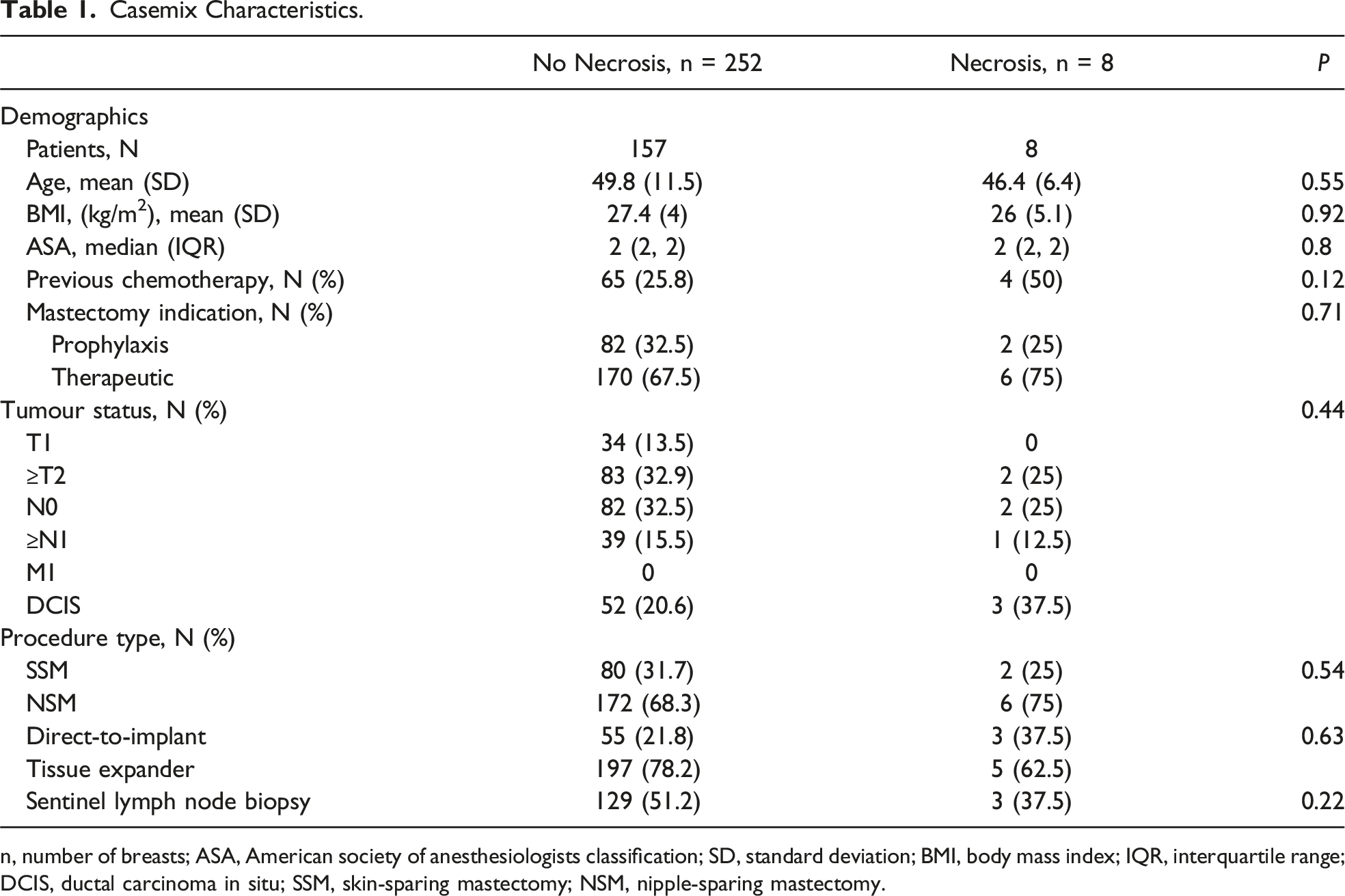
n, number of breasts; ASA, American society of anesthesiologists classification; SD, standard deviation; BMI, body mass index; IQR, interquartile range; DCIS, ductal carcinoma in situ; SSM, skin-sparing mastectomy; NSM, nipple-sparing mastectomy.
“Time to Perfusion” Assessment
Variables Associated With Mastectomy Skin Flap Necrosis.
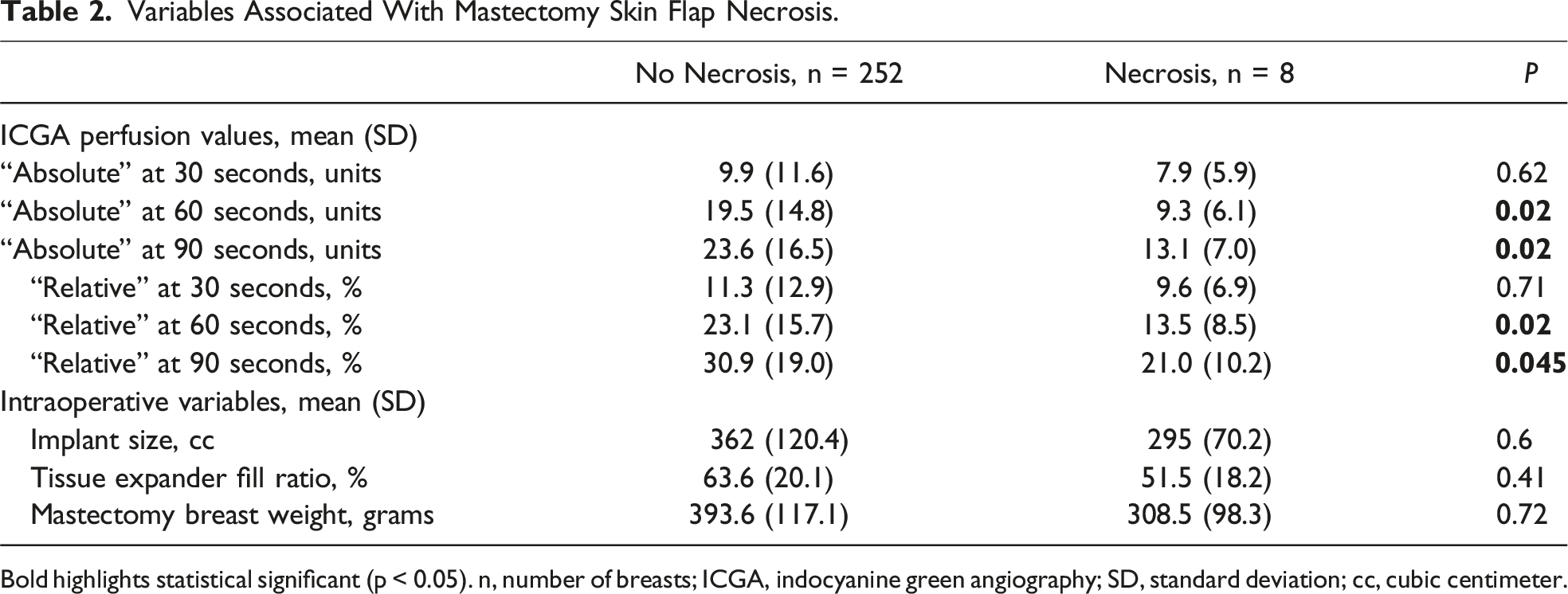
Bold highlights statistical significant (p < 0.05). n, number of breasts; ICGA, indocyanine green angiography; SD, standard deviation; cc, cubic centimeter.
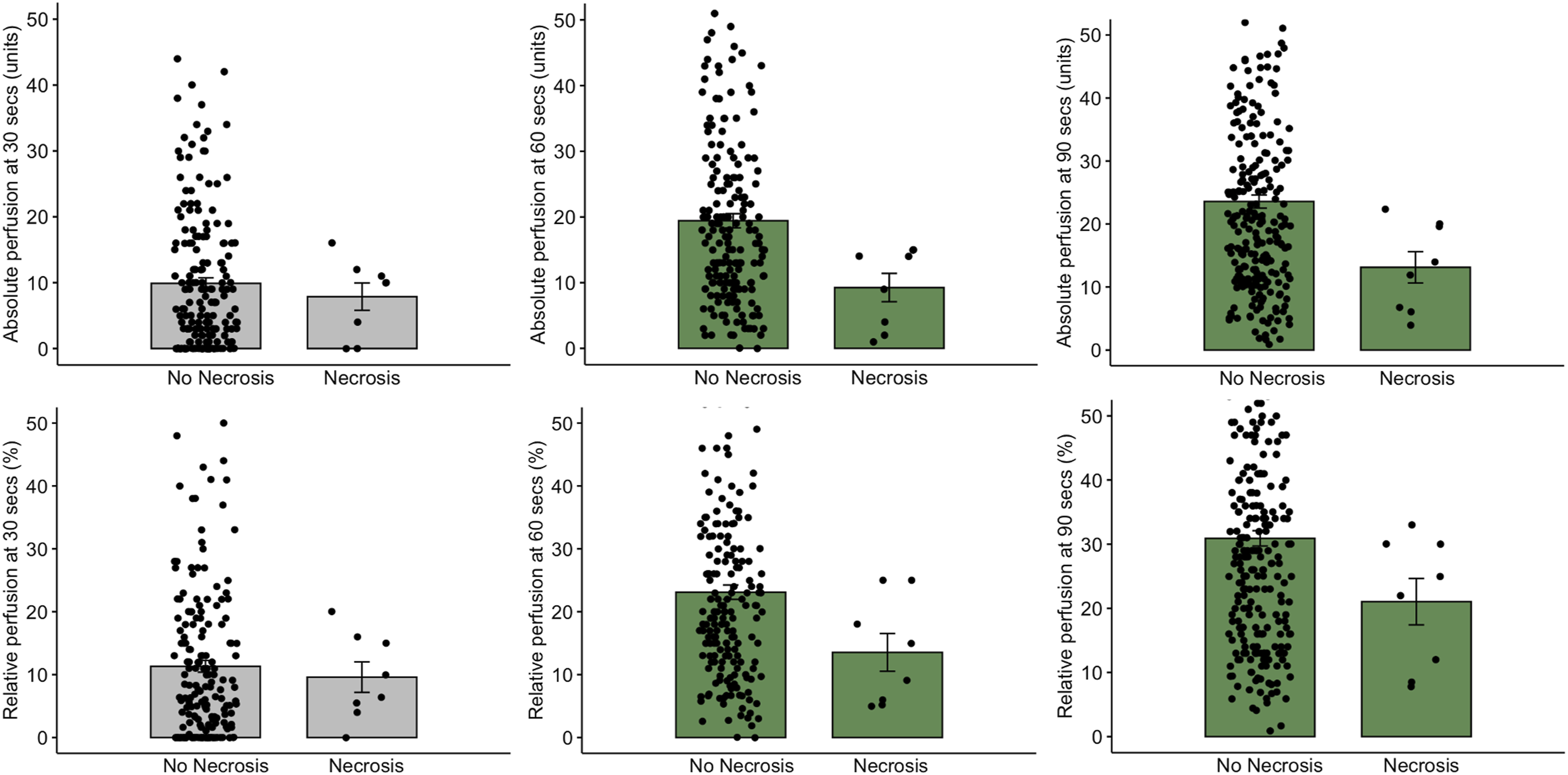
Intraoperative indocyanine green angiography perfusion values in skin flaps that did not develop necrosis and those that did. Clockwise from top left: “absolute” perfusion measured at 30 seconds; “absolute” perfusion measured at 60 seconds; “absolute” perfusion measured at 90 seconds; “relative” perfusion measured at 90 seconds; “Relative” perfusion measured at 60 seconds; “relative” perfusion measured at 30 seconds.
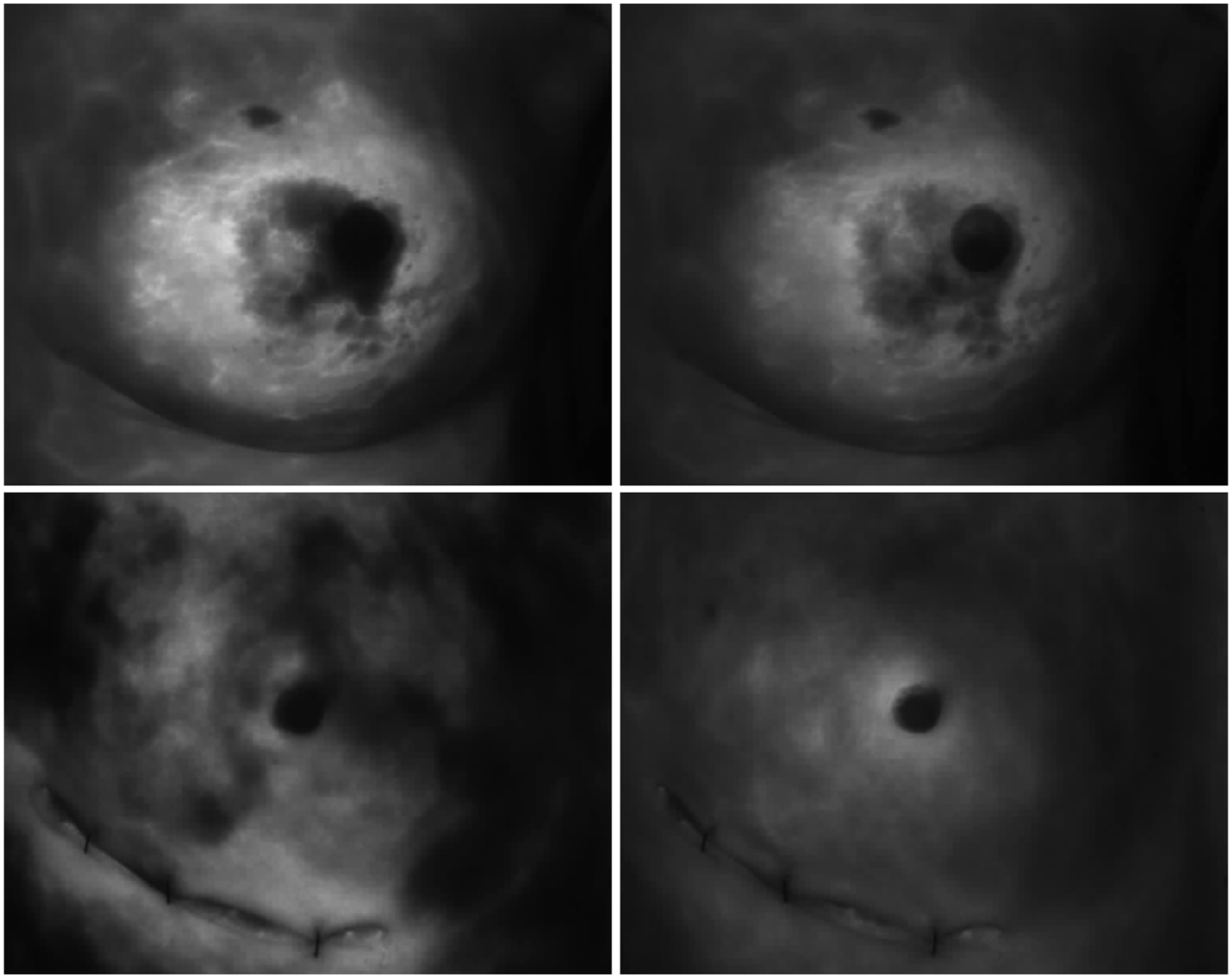
Intraoperative indocyanine green angiograms postmastectomy. Top left image: angiogram still image of a flap, that developed necrosis, recorded at 30 seconds showing poor perfusion; top right image: the same flap recorded at 90 seconds showing ongoing poor perfusion; bottom left image: angiogram still image of a flap, that did not develop necrosis, recorded at 30 seconds showing poor perfusion; bottom right image: The same flap recorded at 90 seconds showing good perfusion.
“Absolute” and “Relative” Perfusion Values
Cut-Off Indocyanine Green Angiography “Relative” Values for Predicting Skin Flap Necrosis, Measured at 60 Seconds.
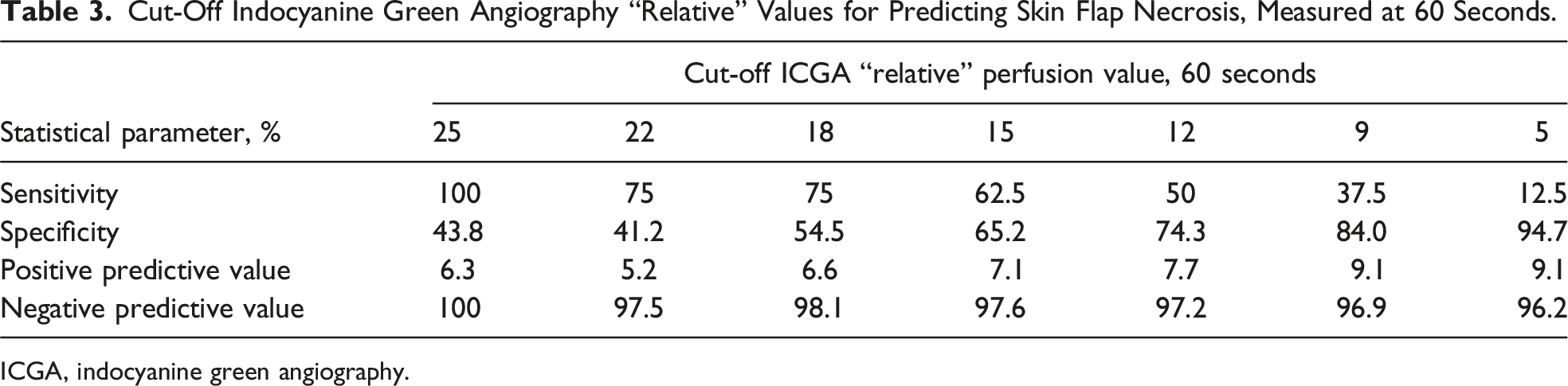
ICGA, indocyanine green angiography.
Cut-Off Indocyanine Green Angiography “Relative” Values for Predicting Skin Flap Necrosis, Measured at 90 Seconds.
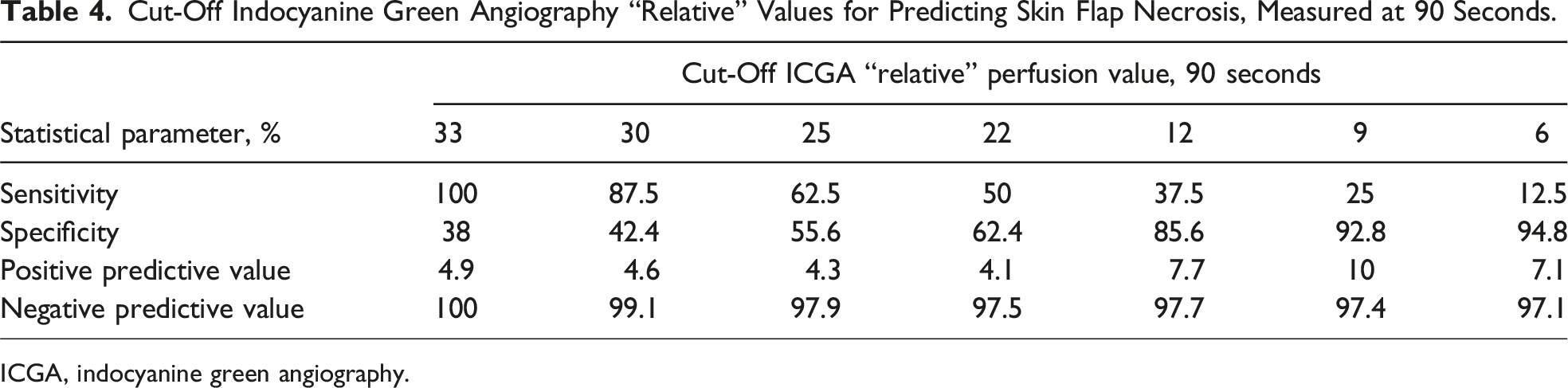
ICGA, indocyanine green angiography.
Relationship Between Perfusion Value and “Time to Perfusion” Assessment
Cut-off “absolute” and “relative” perfusion values for predicting necrosis were both different depending on the “time to perfusion” assessment. For example, when perfusion was analyzed at 60 seconds, a cut-off “relative” perfusion value of 25% had a sensitivity of 100% and specificity of 44% (Table 3). When perfusion was analyzed at 90 seconds, a higher “relative” perfusion value of 33% had similar sensitivity and specificity (Table 4). This was also the case for “absolute” perfusion values when cut-off thresholds were compared for 60 and 90 second time-point analyses (Appendixes 1 and 2). Both “time to perfusion” assessments at 60 and 90 seconds were good predictors of postoperative necrosis with demonstrated area under the ROC curve of 0.84 and 0.85, respectively.
Analysis was also performed on the relationship between perfusion value and “time to perfusion” assessment for individual skin flaps. For flaps that developed necrosis, there was no significant correlation between perfusion value and “time to perfusion” assessment. For flaps that did not develop necrosis, there was a positive correlation between perfusion value and “time to perfusion” assessments (correlation coefficient 0.9 and 0.84, P < 0.001 for “absolute” and “relative” perfusion values, respectively) (Figure 3). Correlation between indocyanine green fluorescence intensity and “time to perfusion” assessment in skin flaps that did not develop necrosis. Left: “absolute” perfusion measurement vs “time to perfusion” assessment. Right: “relative” perfusion measurement vs “time to perfusion” assessment. R, correlation coefficient.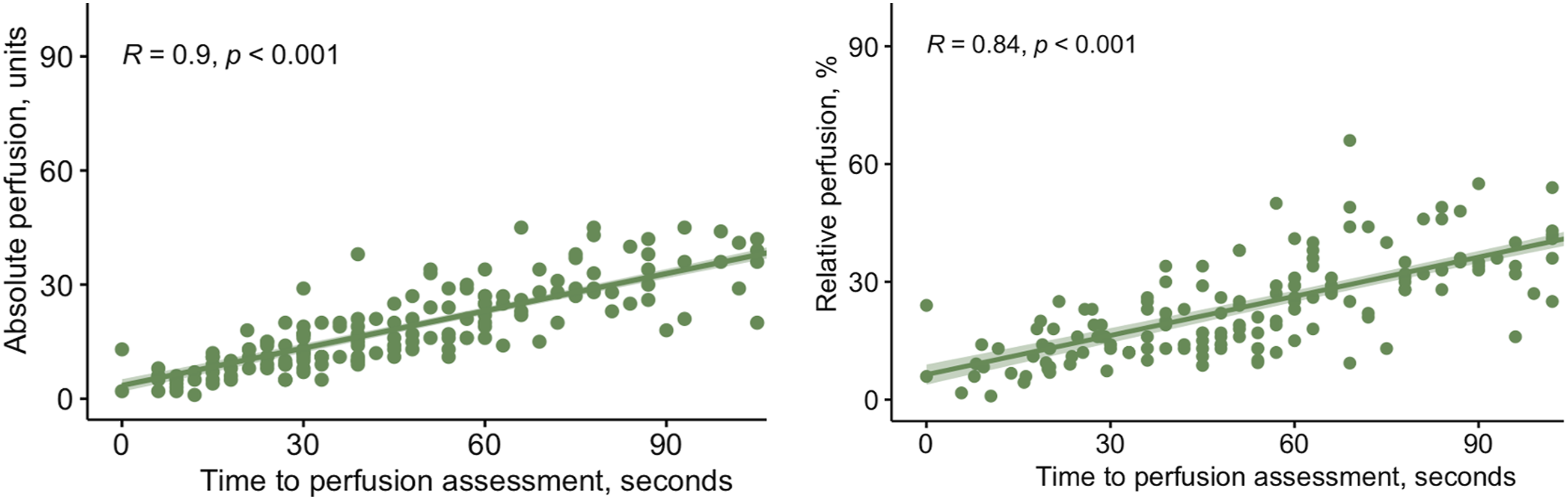
Discussion
Although recommendations for ICGA use have been previously reported, there is no consensus on the technical use of ICGA during postmastectomy reconstruction.6,13-15 ICGA protocols and factors that can affect fluorescence intensity are only briefly described in current literature.6,14 Previous studies mainly focus on outcomes of ICGA in relation to predicting MSFN.3,7 The practical application of ICGA is important because modifiable factors including ambient light during perfusion evaluation, distance between camera and skin flap, and ICG dosage have been shown to affect fluorescence intensity within the angiogram images recorded.3,15 The effect of “time to perfusion” assessment on fluorescence intensity and which perfusion values best predict necrosis, have yet to be fully elucidated. This current study found that “time to perfusion” assessment was an important factor for consistent angiogram interpretation.
The intravenous ICG dosage used in this study was weight-based and ranged from 5 to 10 mg. Common ICG dosages reported in the literature range from 10 to 25 mg and are mostly fixed dosages.8,12,13,16,17 There is no consensus regarding the optimal dose in flap surgery. Ex vivo experiments have demonstrated that ICG dose significantly influenced fluorescence intensity. The concentration of ICG can differ between patients as patients with a higher body weight have a larger blood volume.6,15 Utilizing a weight-based dose may be preferable over a fixed dose. There were also no adverse events to ICG in the 6 years of its use in this current study. Although there is no known relationship between dosage and adverse reactions, a smaller dose used in this study was sufficient to allow for angiogram assessment of flap perfusion with a low rate of postoperative necrosis.
Some studies on reconstructive flap surgery in the literature describe angiography recording directly following ICG administration, while other studies start recording 15 to 60 seconds after intravenous administration.18,19 In the current study, recording was started when the first sign of fluorescence was detected in the flap.12,20 When the recording is started, however, may not be as important as “time to perfusion” assessment. The “time to perfusion” assessment, defined as the elapsed time from ICG administration to perfusion assessment, has been documented to range from 30 seconds to 4 minutes in previous studies but its importance has not been investigated.8,12,13,17 In the current study, perfusion was measured over at least 90 seconds and perfusion analyzed at multiple time points. ICGA “absolute” and “relative” perfusion values when measured at 60 and 90 seconds were significantly lower for cases that developed necrosis compared to cases that did not. No significant difference was identified when perfusion values were measured at 30 seconds. Fluorescence is visible in blood vessels within 5 to 15 seconds after intravenous injection of ICG and fluorescence intensity gradually increased over the 90 seconds of recording. Measuring perfusion at 30 seconds after injection may not allow adequate time to elapse for an accurate assessment.
There was also a distinct relationship between fluorescence intensity and “time to perfusion” assessment for individual flaps. For flaps that did not develop necrosis, there was a positive correlation between “absolute” or “relative” perfusion values and “time to perfusion” assessment. The longer the time elapsed from ICG administration, the higher the measured perfusion value. However, for flaps that developed necrosis, there was no significant correlation. This could be related to ischemic flaps having poor overall perfusion, and hence less potential for improvement in fluorescence intensity. This suggests that the current iteration of the ICGA software could be improved with a more dynamic model that allows for assessment of the perfusion over time instead of perfusion measured at single time points.
There is no consensus on cut-off perfusion values for tissue at risk of postoperative necrosis in postmastectomy reconstruction when “absolute” or “relative” perfusion assessment is applied. Ex vivo experiments have shown that “absolute” perfusion measurements are affected by ICG dose, working distance and ambient light. 6 “Relative” perfusion may take into account some of these variables by using the patient as an internal control. In the current study, both “absolute” and “relative” perfusion functions were found to be relatively good predictors of postoperative necrosis. “Relative” perfusion assessment may be more reliable given it is less affected by external variables. It is however worth noting that cut-off “relative” perfusion values for predicting necrosis are different to those for “absolute” perfusion.
Sensitivity, specificity and predictive values can be calculated to develop thresholds to aid in intraoperative decision-making. In the current study, cut-off “absolute” and “relative” perfusion values were found to be different depending on the “time to perfusion” assessment. The ICGA perfusion units, measured at 60 and 90 seconds, were relatively good predictors of postoperative necrosis. Different cut-off perfusion values would need to be used depending on whether perfusion was measured at 60 or 90 seconds. A cut-off “relative” perfusion value of 25% was highly sensitive (100%) for predicting necrosis when “time to perfusion” assessment was 60 seconds. When perfusion was analyzed at 90 seconds, a higher “relative” perfusion value of 33% had the same sensitivity. A similar correlation was observed with “absolute” perfusion units. This suggests that “time to perfusion” assessment needs to be standardized as perfusion measurements can increase over time.
Limitations
A limitation of this study is that ICGA was not compared to clinical assessment of mastectomy flap perfusion. This comparison could more accurately identify predictors of necrosis and help develop strategies to reduce the risk of ischemic complications. Future studies investigating the behavior of fluorescence intensity in skin flaps at risk of necrosis and the effect of longer periods of “time to perfusion” assessment on fluorescence intensity would be useful to find an optimal threshold. More transparent and comprehensive reporting of ICGA methodology in future studies is advocated to provide reproducibility and allow comparison of methods between studies, as well as potential consensus to be reached to develop guidelines. Artificial intelligence-based quantitative analyses could potentially be applied in future to reduce inter-observer variability and allow for a more dynamic perfusion assessment over time rather than a single time point, to improve accuracy of perfusion interpretation.21-23
Conclusion
Although ICGA may be more objective than clinical assessment for evaluating flap perfusion, it still relies on subjective interpretation of the angiogram. It is thus important to develop a standardized ICGA protocol to best objectify the assessment. “Time to perfusion” assessment was found to significantly influence fluorescence intensity and hence affects accuracy of perfusion assessment. A “time to perfusion” assessment of ≤30 seconds did not allow for accurate perfusion analysis. Cut-off perfusion thresholds were different depending on “time to perfusion” assessment used. A suggested ICGA protocol involves using a perfusion recording of 60 or 90 seconds and the corresponding perfusion value cut-offs, to optimize reliability of perfusion assessments.
Supplemental Material
Supplemental Material - Developing an Indocyanine Green Angiography Protocol for Predicting Flap Necrosis During Breast Reconstruction
Supplemental Material for Developing an Indocyanine Green Angiography Protocol for Predicting Flap Necrosis During Breast Reconstruction by Chu Luan Nguyen, Nirmal Dayaratna, Neshanth Easwaralingam, Jue Li Seah, Farhad Azimi, Cindy Mak, Carlo Pulitano, and Sanjay Kumar Warrier in Surgical Innovation.
Footnotes
Author Contributions
Conceptualization: Chu Luan Nguyen, Sanjay Warrier. Methodology: Chu Luan Nguyen. Formal Analysis: Chu Luan Nguyen, Sanjay Warrier. Writing – Original Draft: Chu Luan Nguyen, Carlo Pulitano, Sanjay Warrier. Writing – Review & Editing: Neshanth Easwaralingam, Jue Li Seah, Farhad Azimi, Cindy Mak. Supervision: Cindy Mak, Carlo Pulitano, Sanjay Warrier. Project Administration: Nirmal Dayaratna.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary materials data is available on email request to the corresponding author. Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
