Abstract
Background
The Enhanced Recovery After Surgery (ERAS) protocol improved perioperative colorectal care. Although the protocol is firmly implemented across hospital settings, there are benefits to gain by actively involving patients in their recovery. The main objective of this study was to investigate whether compliance with selected items in the ERAS protocol could further improve by using a patient-centred mobile application.
Method
This multicentre, randomised controlled trial was conducted between October 2019 and September 2022. Patients aged 18 years or older who underwent elective colorectal surgery, and in possession of a smartphone were included. The intervention group used a mobile application combined with an activity tracker to be guided and supported through the ERAS pathway. The control group received standard care and wore an activity tracker to monitor their daily activities. The primary outcome was overall compliance with selected active elements of the ERAS protocol.
Results
In total, 140 participants were randomised to either the intervention (n = 72) or control group (n = 68). The use of the ERAS App demonstrated a significant improvement in overall compliance by 10%, particularly in early solid food intake by 42% and early mobilization by 27%. Postoperative or patient reported outcomes did not differ between groups.
Conclusion
The smartphone application ‘ERAS App’ is able to improve adherence to the active elements of the ERAS protocol for colorectal surgery. This is an important step towards optimizing perioperative care for colorectal surgery patients and enabling patients to optimize being in control of their own recovery. Trial registration: ERAS APPtimize, NTR7314 (https://trialsearch.who.int/Trial2.aspx?TrialID=NL-OMON29410).
Introduction
To optimize outcome for patients having to undergo colorectal surgery, the Enhanced Recovery After Surgery (ERAS) Study Group published the first evidence-based consensus protocol for colorectal surgery. 1 The ERAS protocol outlines the importance of a multidisciplinary and multimodal approach and consists of 24 core elements throughout the colorectal pathways. 2 All elements work together in an effort to reduce surgical stress, maintain postoperative physiological function, and enhance mobilization after surgery,3-7 resulting in a faster recovery, shorter hospital stay, and reduced rates of morbidity.8-10
High adherence to the ERAS protocol is significantly associated with markedly improved outcomes, such as shorter hospital stay, lower rates of postoperative complications, reduced 30-day morbidity, and lower readmission rates.11-14 However, local implementation of ERAS protocols differ across medical centres. Even when clinical pathways are based on the same ERAS guidelines, implementation of the protocol and outcomes vary. 15 Protocol adherence were 69%, 72% and 53% during the preoperative, perioperative and postoperative phase respectively. 16 ERAS protocol compliance may be most essential in the early postoperative phase, as it stimulate early mobilization and resumption of oral intake, avoid discharge delay and minimize the overall risk of complications. 16
ERAS elements can be categorized as requiring contribution from health care providers (passive elements), patients (active elements), or both (passive/active elements). 17 The provider-initiated part of the pathway include most ERAS elements which usually has high adherence. 3 The elements of the ERAS protocol that require patient involvement have the poorest compliance. There are benefits to gain here, as patient empowerment plays an essential part in improving patient adherence.18,19
In recent years, mobile health care applications (apps) and wearables have emerged as strategies to improve patients’ adherence to treatment.20-23 Apps can provide information, stimulate desired behaviour, enhance self-efficacy and empower patients allowing patients to take an active role in their own health care.24-26 Several apps for postoperative recovery have been described in literature, however, the level of evidence and outcomes were varying. 23 The “ERAS App” is an innovative app which combines stimulation of patient involvement in the ERAS with a personalised activity recovery program. The ERAS App offers an engaging approach to involve patients actively in their own care, providing timed information and recovery goals during the perioperative period. 27 This randomized controlled trial (RCT) was conducted to assess whether the use of a patient-centred app can significantly increase compliance with the active elements of the ERAS protocol in patients undergoing colorectal surgery.
Methods
Study Design
The ERAS APPtimize study is a multicenter RCT that was conducted between October 2019 and September 2022 at one academic hospital and four teaching hospitals in the Netherlands. The ERAS protocol was implemented into the care pathways at varying time points and accompanied by locally different adaptations in the centers. The study was approved by the local medical ethics committee of Amsterdam UMC (registration number NL63874.018.17). The study protocol has been previously published. 27 The trial was prospectively registered on International Clinical Trial Registry Platforms; registration number NTR7314. The study is reported according CONSORT-EHEALTH checklist and the RECOvER Checklist.28,29
Study Population
Patients were eligible if they underwent elective colorectal surgery for either malignant or benign disease, were aged 18 years or older, and were in possession of a smartphone running at least the operating systems iOS 9 or Android 8.0. Patients were excluded if they met any of the following criteria: • Palliative surgery or surgery performed after neoadjuvant radiotherapy or chemotherapy • Karnofsky Performance score ≤40 • Inability to understand thex dutch language • Visual impairment, unless well corrected with visual aids • Limitations in using mobile applications due to physical or mental impairments, • Wheelchair-restricted • Estimated pre-operatively if post-operative adherence to the ERAS protocol is not feasible • Resection of multiple organs
Group Allocation and Blinding
After informed consent, patients were randomly assigned (1:1) using Internet block randomization with block sizes of 2, four, and six to either the intervention or the control group. Randomization was stratified by disease (benign and malignant) and age (<50 years and >50 years). Participants, their involved health care professionals, and outcome assessors of study were not blinded to the treatment allocation. Participants were instructed not to tell other patients in their ward if they are assigned to the intervention or control group to avoid societal bias.
Procedures
The Presentation and Scoring of the Selected Active ERAS Elements.
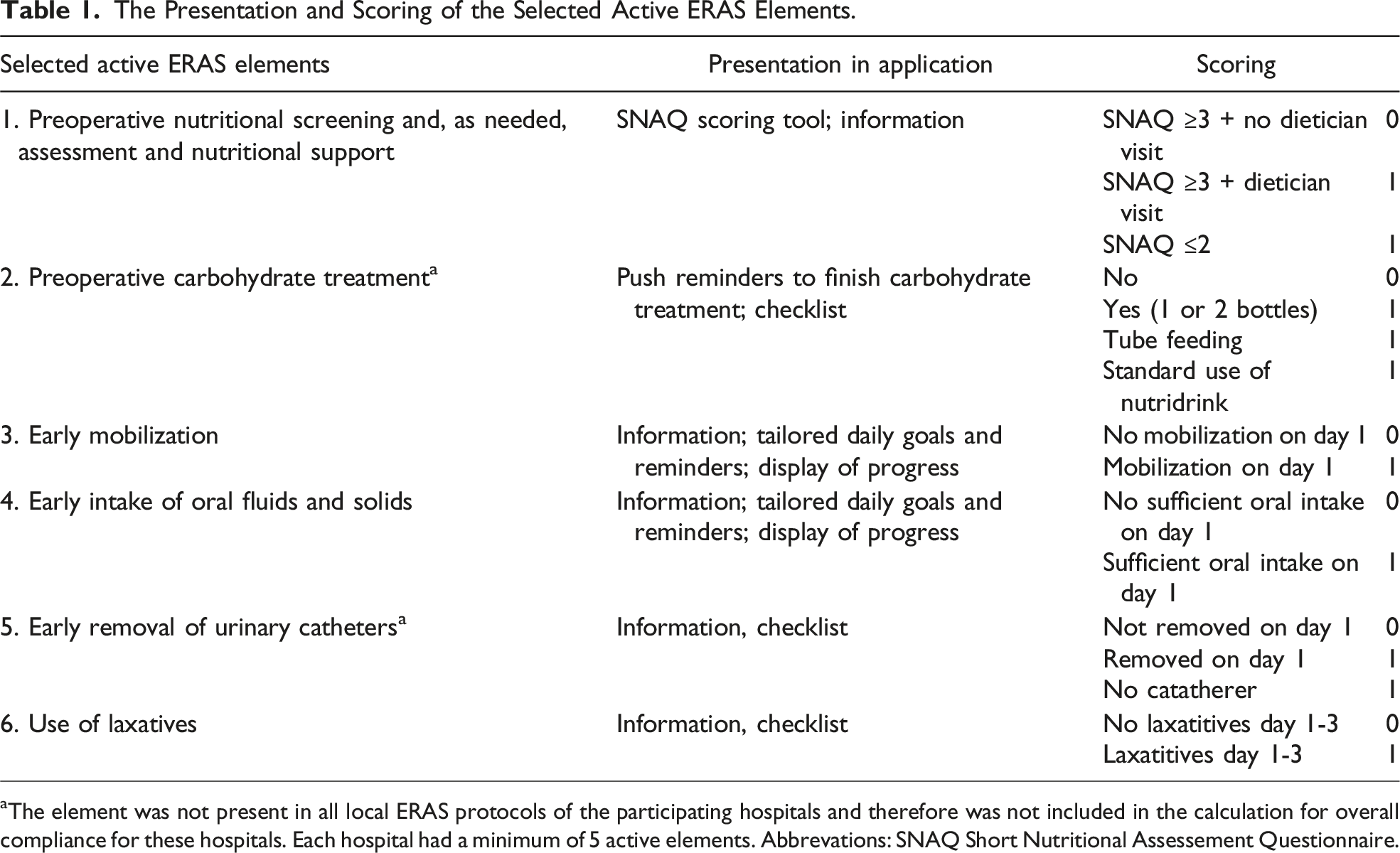
aThe element was not present in all local ERAS protocols of the participating hospitals and therefore was not included in the calculation for overall compliance for these hospitals. Each hospital had a minimum of 5 active elements. Abbrevations: SNAQ Short Nutritional Assessement Questionnaire.
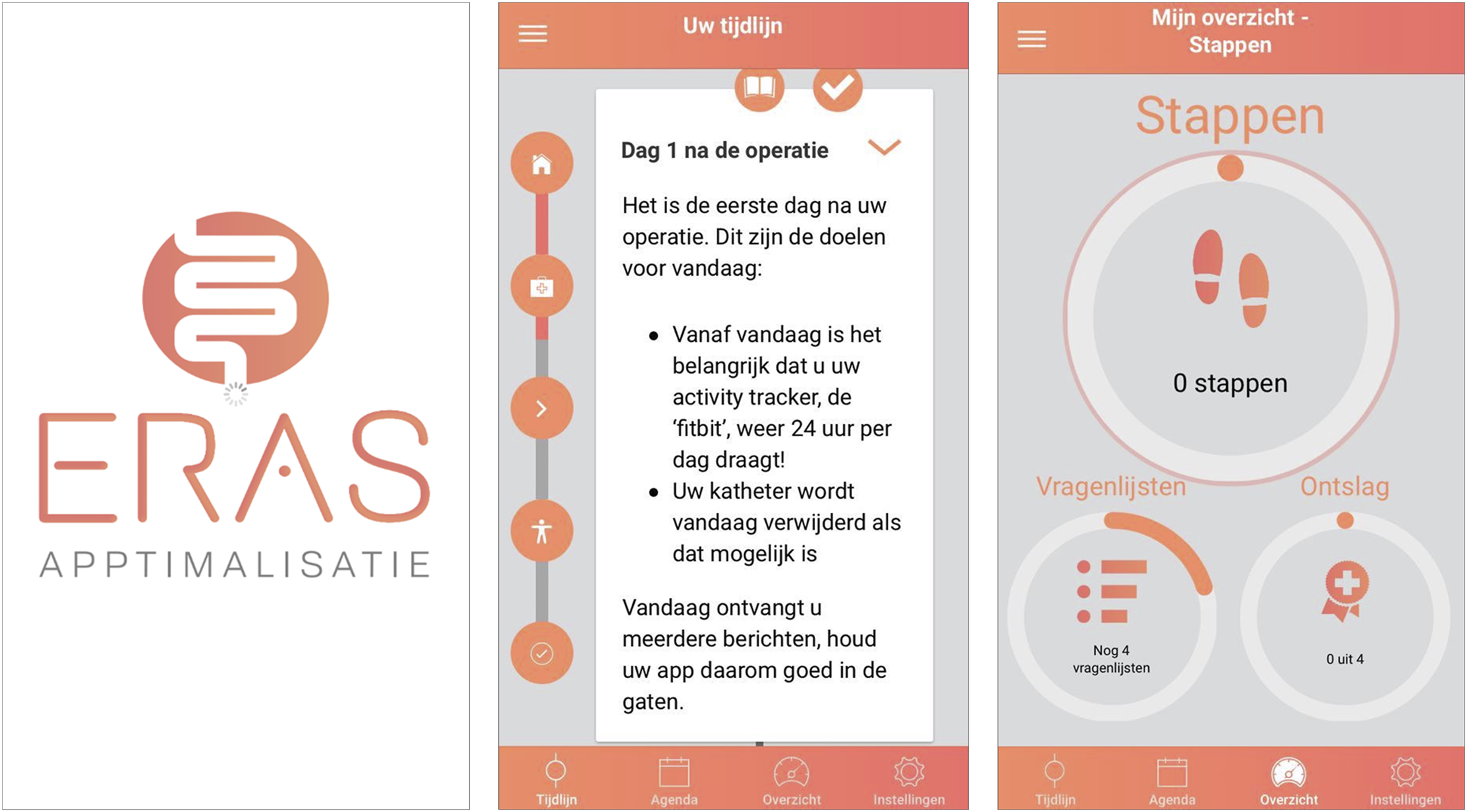
Screenshots of the ERAS App. The app is written in Dutch. First screenshot: Splash screen. Second screenshot: The app generates a timeline based on the operation date which provides information and daily goals to complete. The timeline gives patients an overview of their own care pathway and supports patients to prepare for surgery. If new information or goals are available, push notifications are sent to stimulate patients to adhere to the protocol. Third screenshot: The app’s ‘dashboard’ displays the completion of three subjects: 1) daily activity goal, 2) active ERAS elements, and 3) self-registered questionnaires throughout the entire study.
Participants assigned to the control group received the usual care following the local ERAS protocol and were given a blinded activity tracker to monitor activity. Participants received a paper booklet containing the ERAS elements completion checklist and questionnaires. They were instructed to complete the checklists once a day and the questionnaires according to the time points shown in Table S2. Figure 2 illustrates the study pathways for both groups. Flowchart of intervention and control group.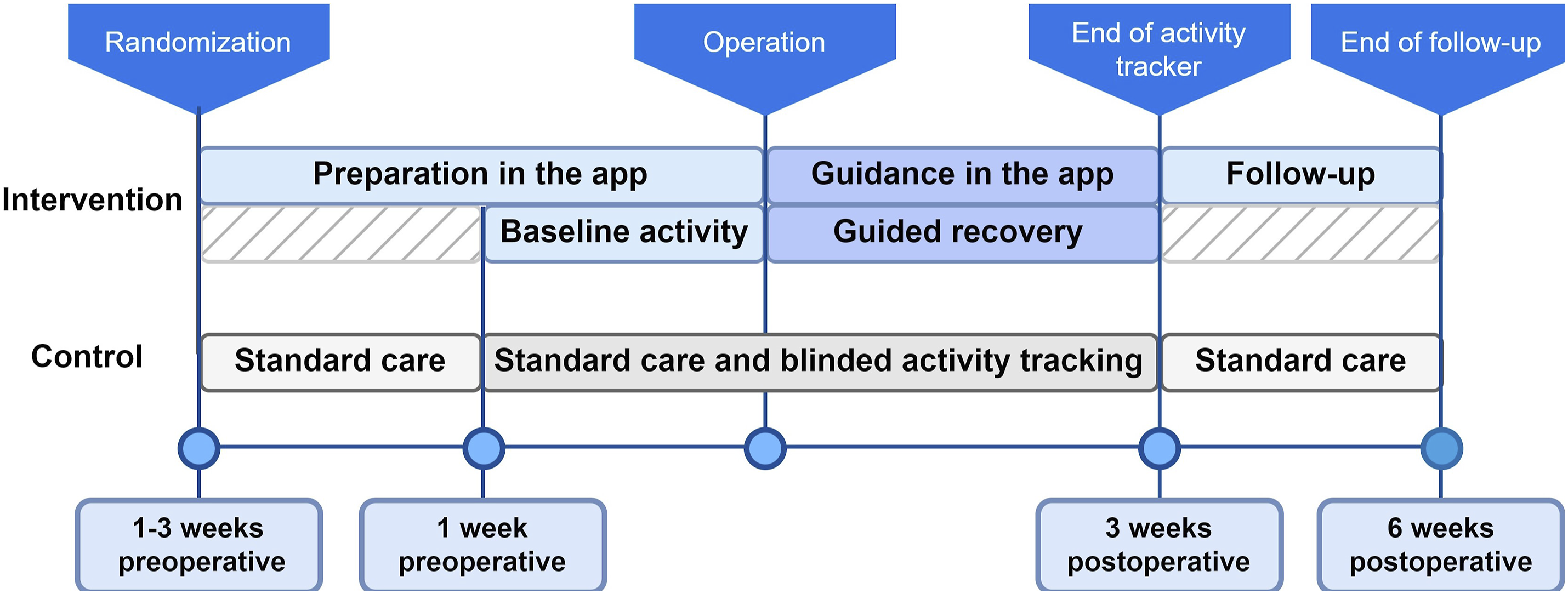
Outcome
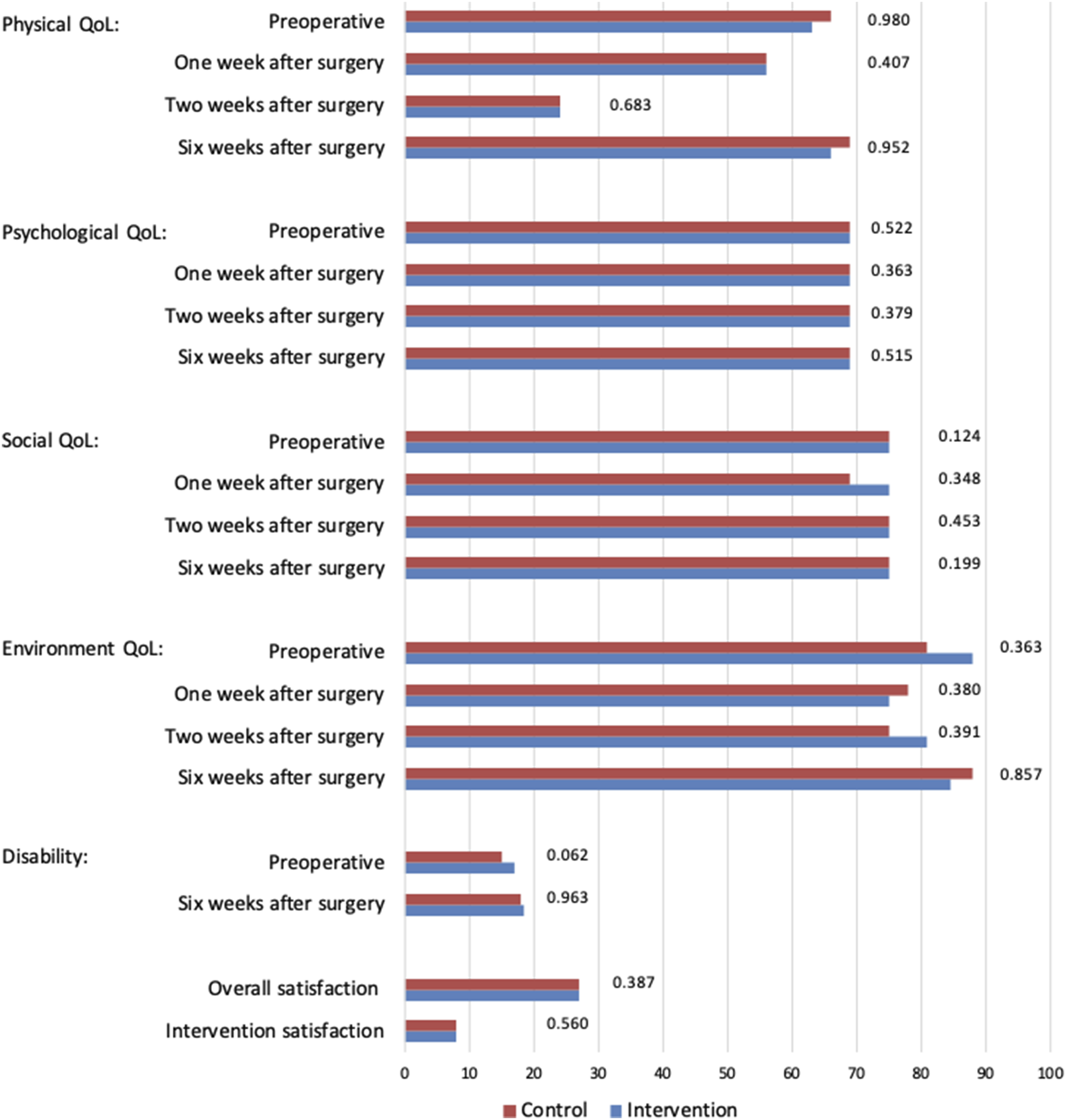
The primary outcome was overall average compliance with selected active ERAS elements (Table 1). To correct for cofounding on digital (il-)literacy, participants completed a questionnaire on use of apps and their mobile proficiency. Secondary outcome measures were postoperative outcomes, such as length of hospital stay (LOS), complications, readmissions, and reinterventions, as well as patient-reported outcomes (PROMs), including quality of life (measured with WHOQOL-BREF), disability (measured with WHODAS 2.0), and satisfaction with the app (measured using a self-developed questionnaire).31,32 Additionally, the activity was assessed from day −7 to surgery, until day 21 post-surgery.
Statistical Analysis
The sample size was calculated based on a compliance rate to active ERAS elements of 57% in a previous study and the hypothesis that the ERAS App would increase patient compliance to 62%. 17 Using a 2-sided alpha of 0.05, with 90% power and a standard deviation of 9, 140 patients were estimated to be required for the study. Data were analysed according to protocol analysis.
Statistical analyses were conducted using IBM SPSS version 28.0. Baseline characteristics were summarized using descriptive statistics and compared between the intervention and control groups and between the included and excluded patients. Continuous normally distributed variables were reported as mean ± standard deviation, and non-normally distributed continuous variables were reported as median and interquartile range (IQR). Distributions were evaluated using visual inspection of histograms. Categorical variables were presented as frequencies and percentages. Independent t-tests, Mann-Whitney U tests, Chi-squared tests, and Fisher’s exact tests were used to assess differences between groups as appropriate. A two-tailed P-value ≤0.05 was considered statistically significant.
The selected active ERAS elements were dichotomously scored as being fully complete or incomplete. The overall compliance is the average of all individual completion percentages. If a specific ERAS element was not present in the local pathway, it was not included into the calculation of overall compliance for these hospitals. Multivariate linear regression with stepwise backward selection was used to account for potential confounding and stratifying factors.
The extent of surgery was categorized as either being major or minor, with minor surgery defined as stoma creation/removal combined with an enterocutaneous fistula correction and major surgery including all the other operations. Postoperative activity was analysed using a Toeplitz linear mixed model.
PROMs were only included in the analysis if the patient completed >80% of the questionnaire per domain. Missing data were corrected using the participants’ mean of the (domain of the) PROM, if missing values were <20% within that scale.
Results
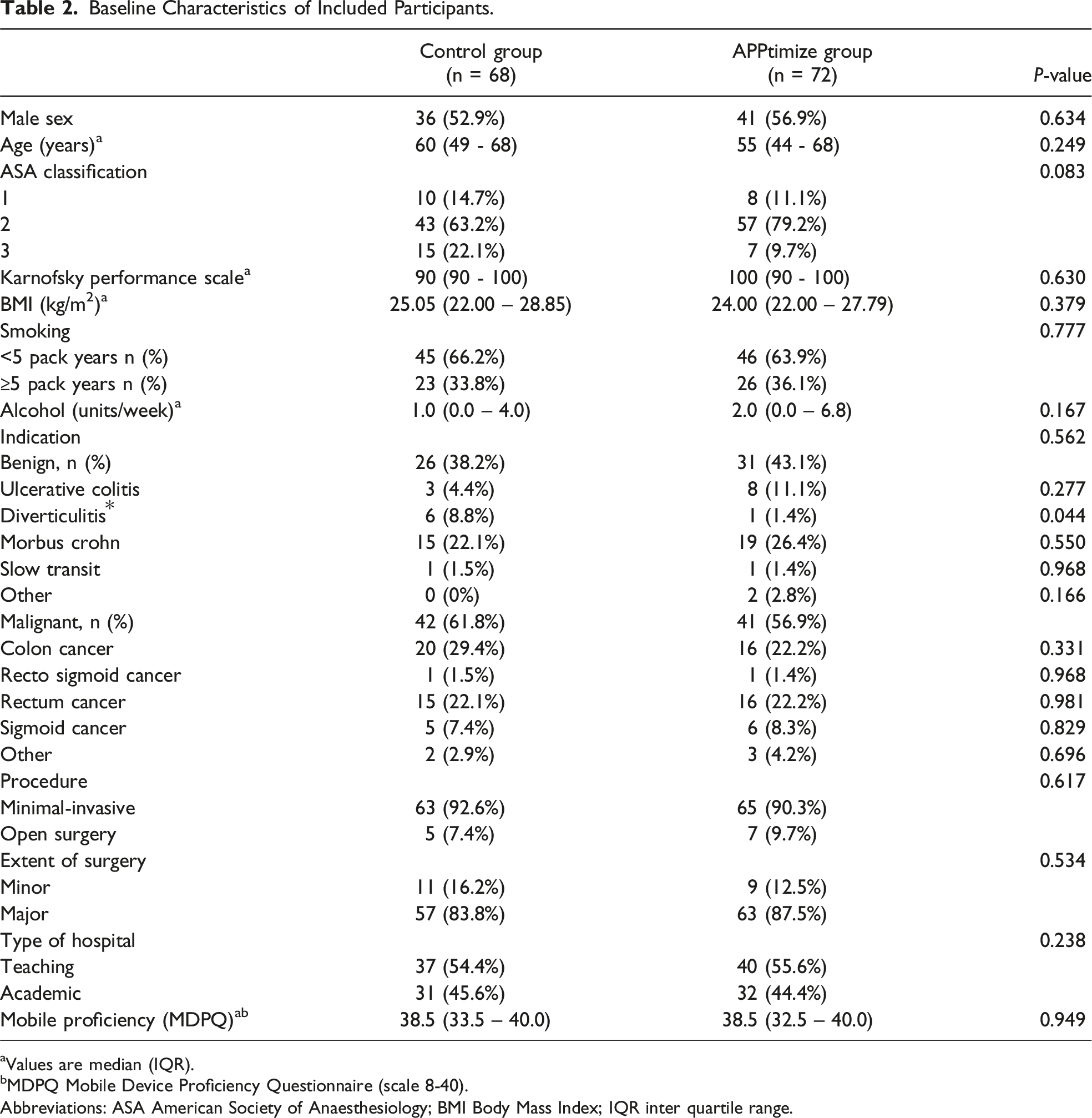
A total of 170 participants provided informed consent and were randomized. Of these participants, respectively 72 and 68 patients were analysed in the intervention and control groups and 30 participants were lost to follow-up (Figure 3). The baseline characteristics of the participants, presented in Table 2, were similar between the 2 groups, with a predominantly male population (n = 77, 54.9%), a median age of 57 years, and a majority of malignant diagnoses (n = 84, 60.1%). Despite randomization, diverticulitis was significantly more prevalent in the control group (P = 0.044). Minimally invasive surgery was the predominant mode of surgery in both groups (n = 128, 91.5%), and both groups had sufficient scores on the mobile proficiency questionnaire. Baseline PROM’s are reported in Figure 4. Treatment assignment and study flow. Baseline Characteristics of Included Participants. aValues are median (IQR). bMDPQ Mobile Device Proficiency Questionnaire (scale 8-40). Abbreviations: ASA American Society of Anaesthesiology; BMI Body Mass Index; IQR inter quartile range. Patient reported outcomes. All reported outcomes are median values. Domains of quality of life are measured in a 0-100 scale, Disabilities is measured in a 12-60 scale, Overall satisfaction is measured in a 7-35 scale, and intervention satisfaction is measured in a 2-10 scale. Abbreviations: QoL = quality of life.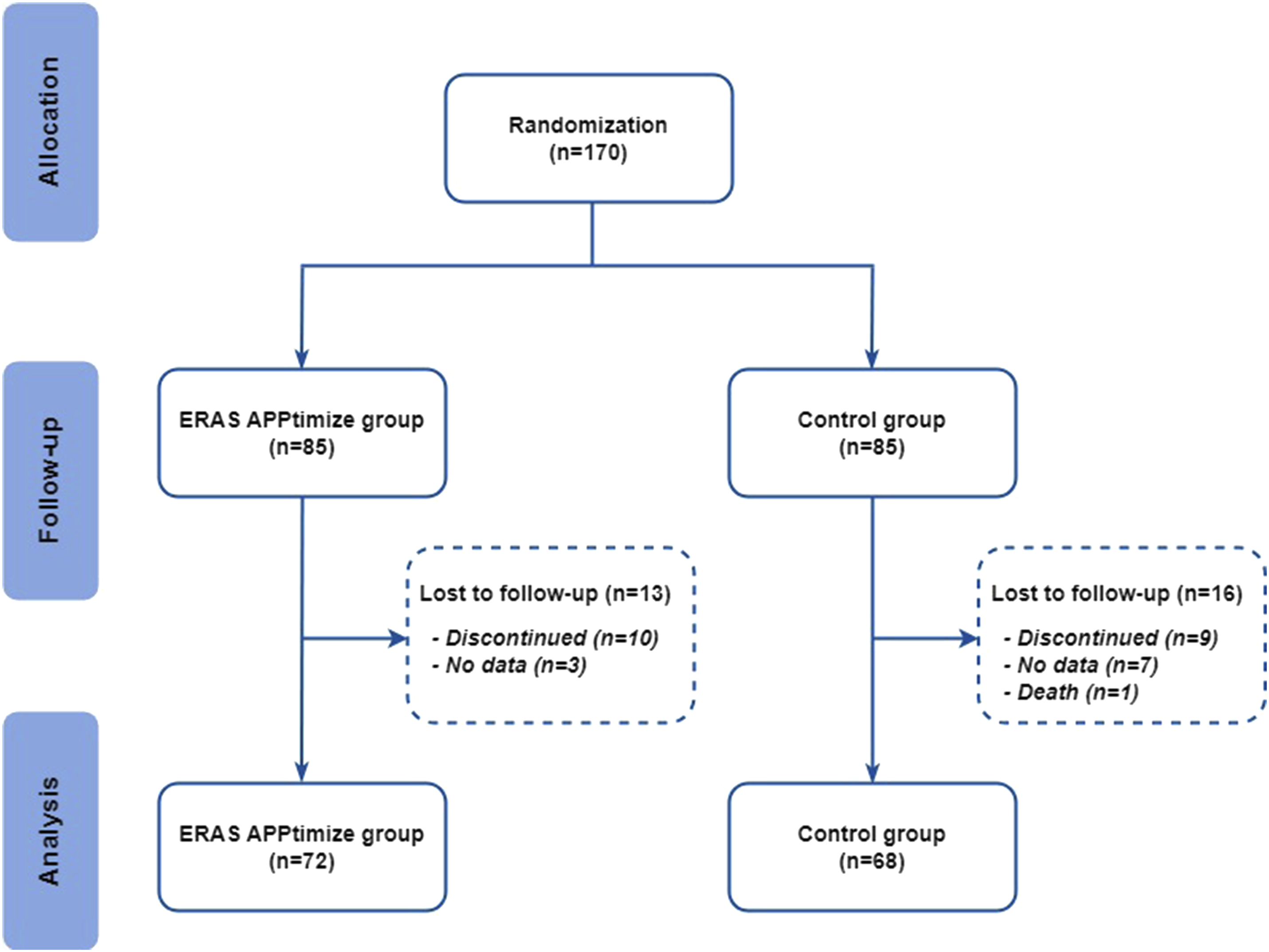
Compliance with the ERAS Protocol
Compliance With the Active ERAS Elements.
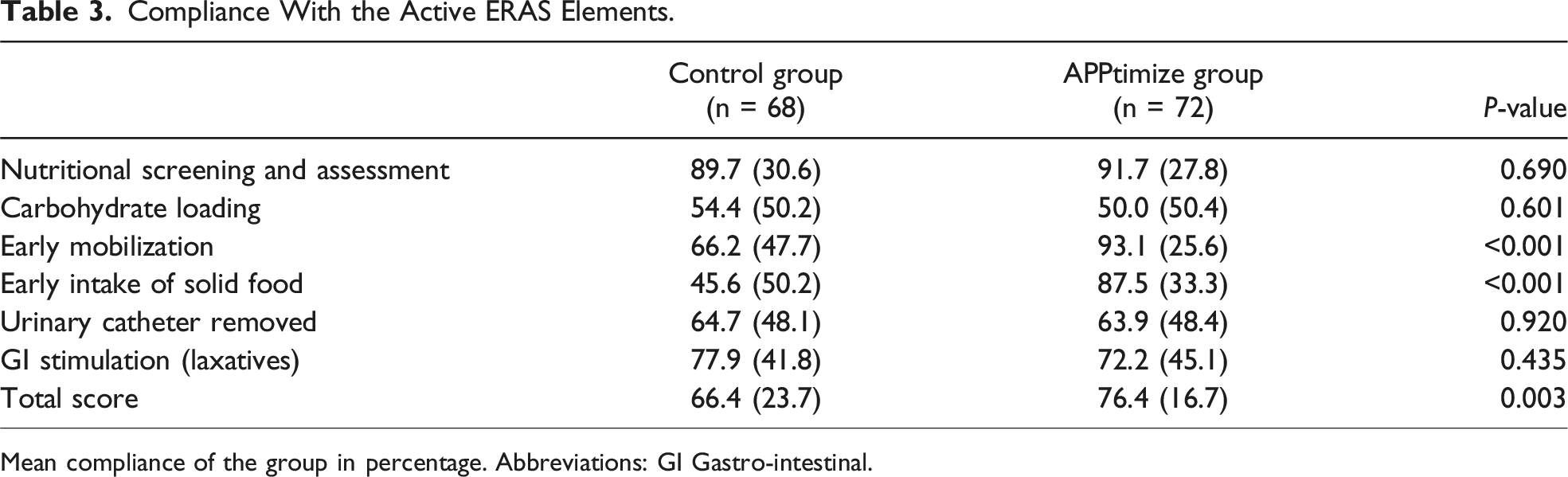
Mean compliance of the group in percentage. Abbreviations: GI Gastro-intestinal.
Multiple Linear Regression Analysis Compliance to ERAS Protocol.
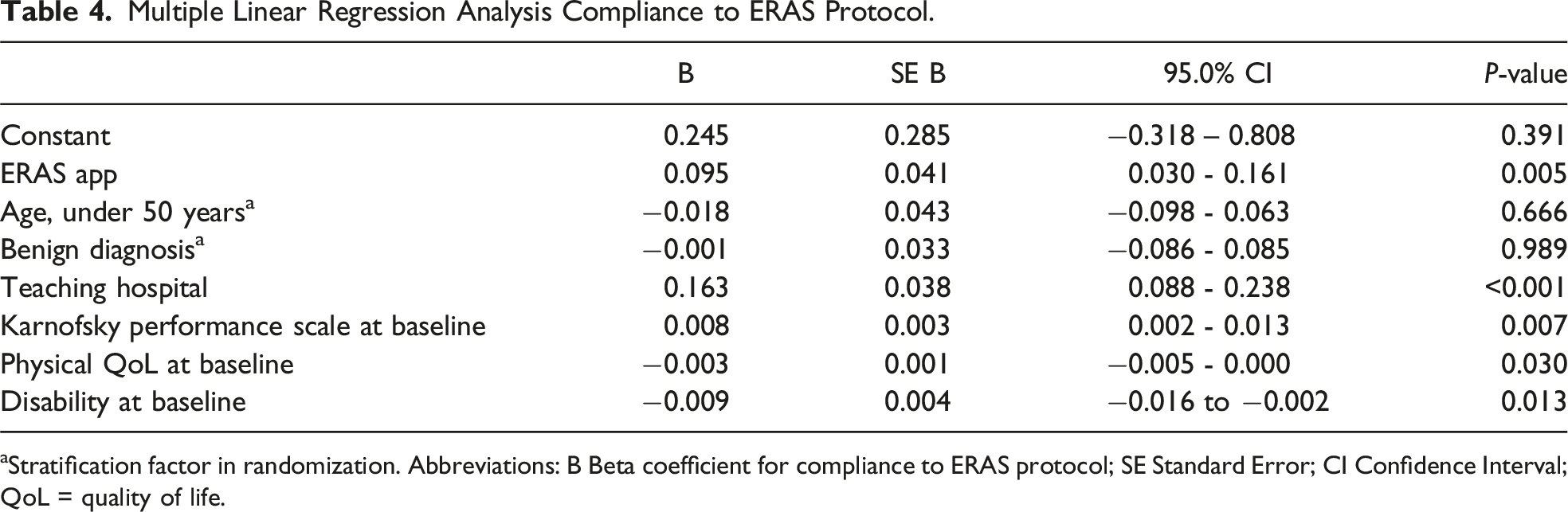
aStratification factor in randomization. Abbreviations: B Beta coefficient for compliance to ERAS protocol; SE Standard Error; CI Confidence Interval; QoL = quality of life.
Secondary Outcomes
Postoperative Outcomes.
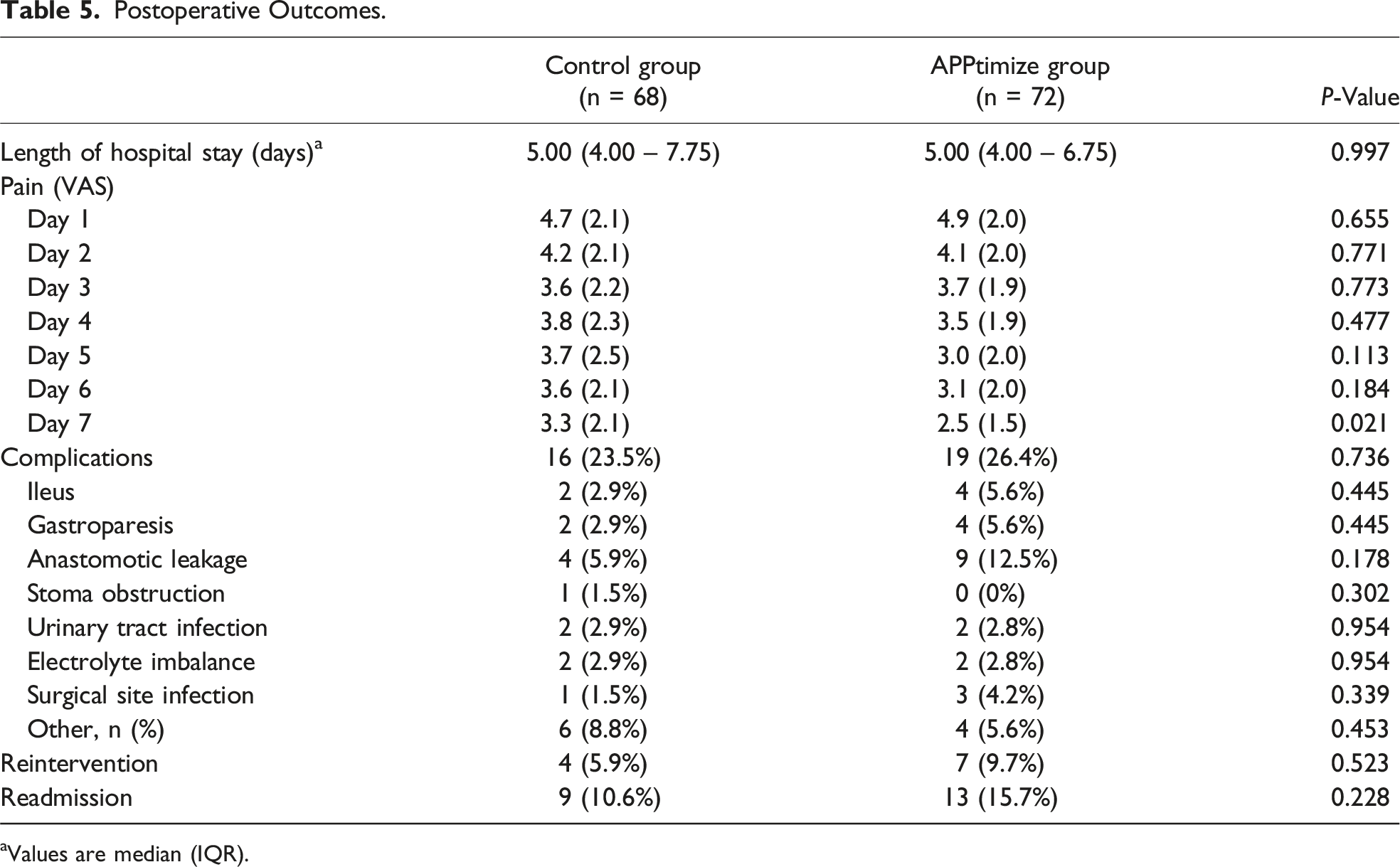
aValues are median (IQR).
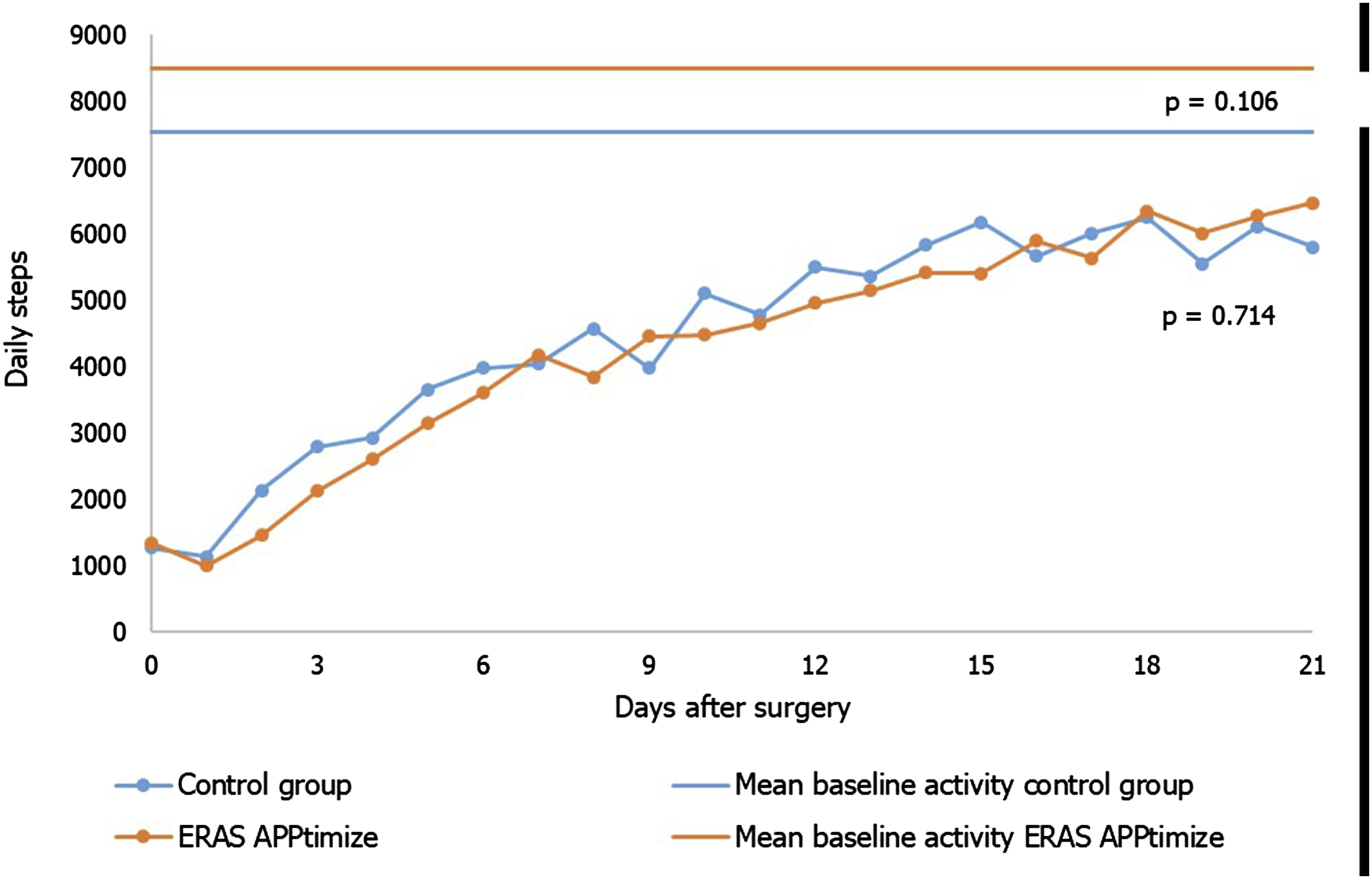
Postoperative activity, measured in steps taken per day.
Discussion
The ERAS protocol has improved perioperative care for patients undergoing colorectal surgery. However, challenges persist in optimizing patient engagement and compliance, prompting exploration into innovative mobile health care solutions. This study investigated the effectiveness of a patient-centred app, the ERAS App, designed to enhance patient education, participation and activation within the ERAS colorectal pathway.
The ERAS App demonstrated a significant improvement in overall compliance with selected active ERAS elements by 10% (P = 0.003), particularly in early solid food intake by 42% (P < 0.001) and early mobilization by 27% (<0.001). Other active ERAS elements remained unchanged, as these elements relies partially on health care providers. Although the postoperative pain was reduced at day 7, the study did not demonstrated improvement in other clinical outcomes. It should be considered that the study was not powered on clinical outcomes as it had already seen significant enhancements since the introduction of the ERAS protocol. Improved adherence to active ERAS elements might translate into clinical benefits in larger study populations. Additionally, the quality of implementation of the ERAS protocol may have varied among health care providers or institutions, leading to inconsistent results across study sites. This highlights the need for standardized implementation and continuous monitoring to ensure protocol effectiveness.
Furthermore, the ERAS App did not improve patient-reported outcomes (PROMs). It is possible that increased adherence to the ERAS protocol may not have a direct impact on PROMs or a potential social desirability bias in self-reported questionnaires could have influenced the observed outcomes. 33 The ERAS App did not lead to improved postoperative activity. Unusually high baseline activity levels (eg, 23 000 steps per day) were reported in both groups, demonstrating the preoperative motivation which have led to an unrepresentative baseline level. Not all participants had optimal postoperative activity goals, as the baseline measurement may have been too short or goals may not have been sufficiently challenging. The intervention group’s increased activity in the final days suggests that the follow-up period might have been too short to capture sustained improvements.
Several limitations to this study need to be addressed. The exclusion of patients undergoing palliative surgery, surgery after neoadjuvant chemotherapy or radiotherapy, or multiple organ resections, may have resulted in a selection bias. These patients may benefit the most from the app, and their exclusion may underestimate the true impact of the app. In addition, the screening process was not thoroughly registered in all study sites, which inhibited presenting complete screening data in Figure 3 as this may lead inaccurate conclusions about the selection bias. Study results should be interpreted with this context in mind. Furthermore, non completing participants had significantly more complications (Table S4), which suggests that the ERAS App may not be optimal for patients with complications. This highlights the need for further research. It is important to note that patients in the control group may have been more actively participating in the ERAS care pathway compared to their peers as. This may have resulted in a decreased compliance difference between the 2 study groups.
Despite the demonstrated effect of the ERAS App, opportunities for further optimization were identified. Dynamic features catering to individual recovery progress and adapting to postoperative complications hold promise. However, it’s important to exercise caution when integrating individual recovery progress because the more personalized the intervention, the less evidence there is to support its overall effectiveness. The integration of prehabilitation with the ERAS App emerges as a potential strategy to improve clinical outcomes. 34 Future research should delve into the feasibility and efficacy of incorporating dynamic features or prehabilitation within the ERAS pathway through the ERAS App. Additionally, exploring barriers and facilitators to the app’s implementation in clinical practice can inform strategies for enhancing its adoption and utilization. Overall, further research and development of the ERAS App can lead to better patient engagement, adherence to the ERAS protocol, and improved clinical outcomes.
Conclusion
The ERAS App successfully increases patient compliance to the ERAS protocol by actively involving patients into their own ERAS care. Although the ERAS App was unable to demonstrate improved patient-related and clinical outcomes, the app is an important step towards optimizing perioperative care for colorectal surgery patients and enabling patients to optimize being in control of their own recovery. Further research and development are necessary to identify ways to improve the app’s efficacy and impact on patient outcomes.
Supplemental Material
Supplemental Material - Improving Enhanced Recovery after Surgery (ERAS): The Effect of a Patient-Centred Mobile Application and an Activity Tracker on Patient Engagement in Colorectal Surgery
Supplemental Material for Improving Enhanced Recovery after Surgery (ERAS): The Effect of a Patient-Centred Mobile Application and an Activity Tracker on Patient Engagement in Colorectal Surgery by Sebastiaan L. van der Storm, Marilou Jansen, Malou D. Mulder, Hendrik A. Marsman, Esther C.J. Consten, Frank C. den Boer, Hans D. de Boer, Willem A. Bemelman, Christianne J. Buskens, Marlies P. Schijven, on behalf of the ERAS APPtimize collaborative study group in Surgical Innovation
Footnotes
Acknowledgements
The ERAS APPtimize collaborative study group consists of local investigators who are responsible for trial execution, patient inclusion and data collection. They have all read and approved the final manuscript. Jurriaan B. Tuynman Amsterdam UMC - location VUmc, Surgery, Amsterdam, the Netherlands. Steven J. Oosterling, Spaarne Gasthuis, Surgery, Hoofddorp, the Netherlands Glaresa P. Molly, OLVG, Amsterdam, Surgery, the Netherlands Belinda M. Vink, Meander Medical Centrum, Surgery Amersfoort, the Netherlands Maaike Corver,, Zaans Medical Center, Surgery, Zaandam, the Netherlands.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sebastiaan van der Storm, Malou Mulder, Hendrik Marsman, Frank den Boer, have no conflicts of interest to disclose. Marilou Jansen is the content developer of the app and Marlies Schijven is the owner of the app. Esther Consten is Proctor for Intuitive Surgical. Willem Bemelman declares that he has received scientific grants from VIFOR and has received speaker fees from Janssen, Medtronic and Galapagos. Christianne Buskens declares that she has received scientific grants from Boehringer Ingelheim and Roche and has received speaker fees from Janssen, Medtronic and Galapagos Hans de Boer is Chairman ERAS Society and Chairman Dutch ERAS Society.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Amsterdam University Medical Centers; Innovation Impuls Grant 2017; Maag Lever Darm Stichting; ZP 18-10; European Crohn’s and Colitis Organisation; ECCO grant 2020.
Ethical Statement
Clinical Trial Registration
International Clinical Trial Registry Platform (ICTRP), NTR7314, prospectively registered on the ninth of November 2017.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
