Abstract
Introduction
Warm ischemia refers to the ischemic injury of cells and tissues under non-hypothermic conditions. During kidney transplantation, warm ischemia time (WIT) may refer to two distinct periods. ‘Donor warm ischemia’ refers to the time during organ procurement from vascular clamping (or asystole in the case of donation after cardio-circulatory death (DCD)) until cold perfusion. ‘Recipient warm ischemia’ or ‘anastomosis time’ refers to the ischemia of rewarming during the vascular anastomosis, occurring from the time of removal from ice until reperfusion. 1 In this paper, any discussion of WIT refers to the latter definition as this tends to be the more lengthy and significant insult to transplanted kidneys, and efforts to minimize it are lacking.
Most peer-reviewed sources report a mean anastomosis time of 30-45 minutes in kidney transplantation with some complex cases taking over an hour.2-8 Renal warm ischemic injury is postulated to occur due to glomerular and tubular metabolic activity occurring under hypoxic conditions. Metabolic activity in cooled organs is minimal at 5°C but approaches normal levels when the organ reaches 15°C.2,8-12 Studies of renal re-warming profiles during transplantation found that as little as 15 minutes was sufficient for kidneys to reach this threshold of metabolic activity, leaving most grafts exposed to 15-45 minutes of harmful warm ischemic activity.2,8,12
In recent years, greater scientific effort has been aimed at determining the true impact of WIT. Prolonged WIT is recognized as an independent risk factor for delayed graft function, impaired long-term graft survival, and decreased patient survival.3-5,13,14 Histologic studies support that WIT of 30 minutes is sufficient to cause loss of 25% of glomeruli in addition to fibrosis and tubular atrophy.4,13,14 Recent studies have shown that each additional 5 minute interval of WIT resulted in an additional day of hospital stay, and prolonged WIT led to worse patient survival and higher incidence of return to dialysis.6,15 Unfortunately, many of the risk factors for prolonged WIT (recipient BMI >25, history of prior renal transplant, receiving a right kidney, and vascular multiplicity) are not modifiable. 16
Vascular anastomosis remains the most critical portion of transplant surgery and should be performed efficiently, yet strategies aimed at maintaining renal hypothermia during anastomosis have not been widely adopted despite considerable evidence that they are effective. The concept of renal allograft cooling has been reported in the literature, as early as 1973, and the benefits have been clearly demonstrated.12,17 Studies have shown that intra-operative cooling leads to lower rates of delayed graft function and acute rejection, in addition to improvements in glomerular filtration rates.18,19
Reported cooling techniques can be divided into three broad categories; cooling irrigation, ice bag immersion, and the use of a cooling jacket. 12 Surface cooling techniques are among the oldest and simplest methods reported; involving intermittent irrigation of the allograft with cold saline, with some including wrapping the kidneys in ice-gauze.2,17,20-23 Although these techniques are simple and inexpensive to adopt, they are ineffective at sufficiently maintaining hypothermia, with median cortical and medullary temperatures of 22.1°C and 22.5°C, respectively, after 30 minutes.2,12,20,22,23
Ice bag immersion techniques generally involve suspending the kidney in a bag containing ice slush, cold saline, and/or other cold preservation solutions.12,18,24-30 Unlike cooling irrigation, sufficient renal cooling has been reported with some of these methods (median cortical and medullary temperatures of 4°C and 5°C after 30 minutes of WIT, respectively).24-26 However, clinical adoption remains very limited, likely because these methods are bulky, clumsy and obscure the surgeon’s view during anastomosis. Moreover, cooling fluid will continually leak into the operative field and, even if well-contained, the fragile and puncturable nature of the bags can necessitate frequent re-filling, adding complexity and additional steps to the vascular anastomosis. 12 These methods also require the surgical assistant to continually hold or position the graft.
Cooling jackets are more complex, and unlike the previous two categories, have not been adopted in clinical transplantation yet. 12 These techniques have great variability, but generally involve a shell or jacket encasing the kidney and incorporating a mechanical cooling system (i.e. polymer gel cylinders, Multitherm® sponges, entrapped air, circulating ethanol/methylene blue). These devices are capable of excellent renal temperature control (median cortical/medullary temperatures of 6.5°C and 6.3°C at 30 minutes of WIT, respectively), and have proven beneficial in all animal studies.12,31-34 Unfortunately, most of these devices are complex and costly to manufacture and may employ non-biocompatible compounds to achieve cooling. This has led to economic and pragmatic barriers to their adoption, and no human studies have been completed. 12
To this end, in this proof-of-concept study, we set out to develop and produce a simple and novel renal cooling device that overcomes the economic and pragmatic barriers of current cooling jacket designs. Our goal was to maintain renal hypothermia at or below 5°C for 60 minutes and to provide a practical device that would optimize rather than impede the vascular anastomosis, allowing for immediate translatability to in-vivo human use.
Methods
This study involved the use of readily available, over the counter animal products generally marketed for human consumption, and so an institutional REB was not required.
Device Construction
A form-fitting cooling jacket was created using malleable food-grade aluminum tubing with 3/16″ outer diameter (ØD) (McMaster-Carr, Cleveland OH) organized in a serpentine pattern. A proprietary method was employed to limit kinking and deformities in the tubing. 3-D printed models of human kidneys were used to design the prototype to fit human kidneys.
Temperature Control
Adult porcine kidneys (n = 4 per arm) were used to test the cooling ability of the device. All kidneys were stored in an ice bath for a minimum of two hours prior to the start of WIT to simulate typical cold storage and allow for temperature equilibration. K-type thermocouples (Omega, Norwalk CT) were implanted in each kidney at a depth of 5 mm and 15 mm, to measure surface (cortical) and core (medullary) temperatures, respectively. Infrared thermal imaging was obtained at various time points using a TG165 thermal camera (FLIR Systems, Wilsonville OR).
At t = 0, kidneys were removed from cold storage and placed at an ambient temperature of 24°C on a mesh cooling rack. The control arm consisted of adult porcine kidneys placed at ambient room temperature (24°C) wrapped in cold-saline soaked gauze. In the interventional group, kidneys were placed in the cooling jacket at t = 0 and then placed on the same mesh cooling rack as control kidneys. Chilled saline solution (4°C) was used as a cooling irrigant, passing through the cooling jacket at a rate of 240 mL/min. Surface and core temperature of the kidneys was recorded at 5-minute intervals until 60 minutes was reached.
Device Usability
Adult porcine kidneys (n = 4 per arm) were used to test feasibility of performing a vascular anastomosis with the device in place. The control arm consisted of adult porcine kidneys suspended with umbilical tapes in the fashion typically used at our centre. Porcine renal artery was anastomosed to GORE-TEX® vascular grafts (W. L. Gore & Associates, Flagstaff AR) in a simulated operative field with the same aperture (15 cm) and depth (9 cm) of a typical iliac fossa renal transplant incision as measured at our institution. Time to complete anastomosis was recorded.
Device Removal
The removal of the cooling device is a critical step that must be managed carefully to avoid complications such as avulsion of the vascular anastomoses. To mitigate these risks, the device is designed to be removed smoothly without applying undue tension to the graft. This is accomplished by the choice of material used in construction, which allows for a form-fitting construction yet great malleability facilitating atraumatic and tension-free removal. The device is removed prior to reperfusion, which allows the surgeon to manipulate the kidney to ensure optimal positioning and address any vascular hemostasis issues. This sequence ensures that the graft remains stable and that the cooling is effective up until the final moments before blood flow is restored.
Statistical Analysis
All calculations were performed using spreadsheeting software (Microsoft Excel). All temperature and anastomosis time data were assumed to be normally distributed. T-tests were conducted to compare surface and core graft temperatures, and time to completion of anastomosis between groups. Statistical significance was accepted at the P < .05 level.
Results
Figure 1(A)–(C) demonstrates renditions of the first cooling jacket prototype. The most recent prototype is shown in Figure 1(D). Design of the renal cooling device. Virtual renderings of the overall view (A), side view (B) and top view (C). (D) Completed construction of device.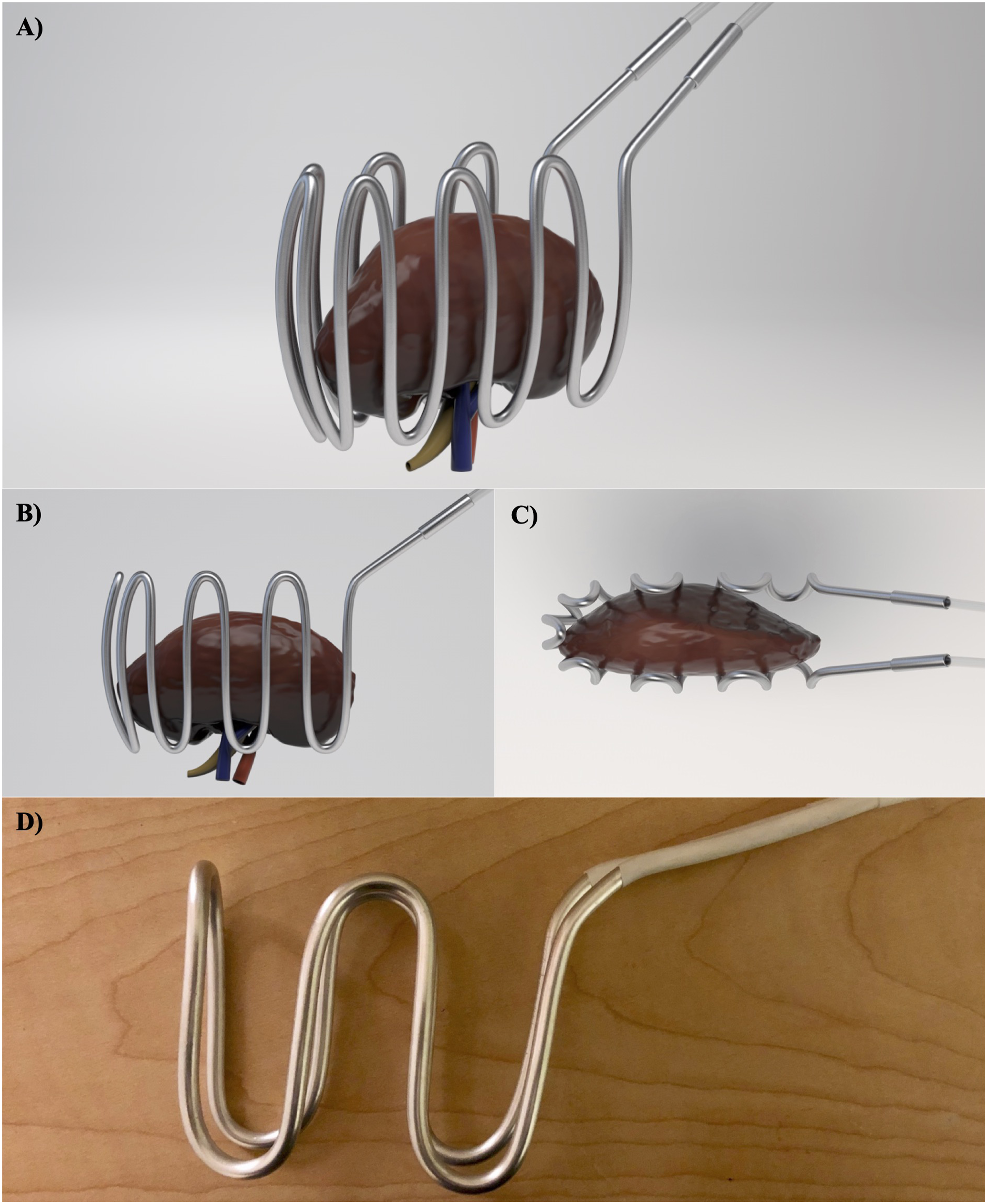
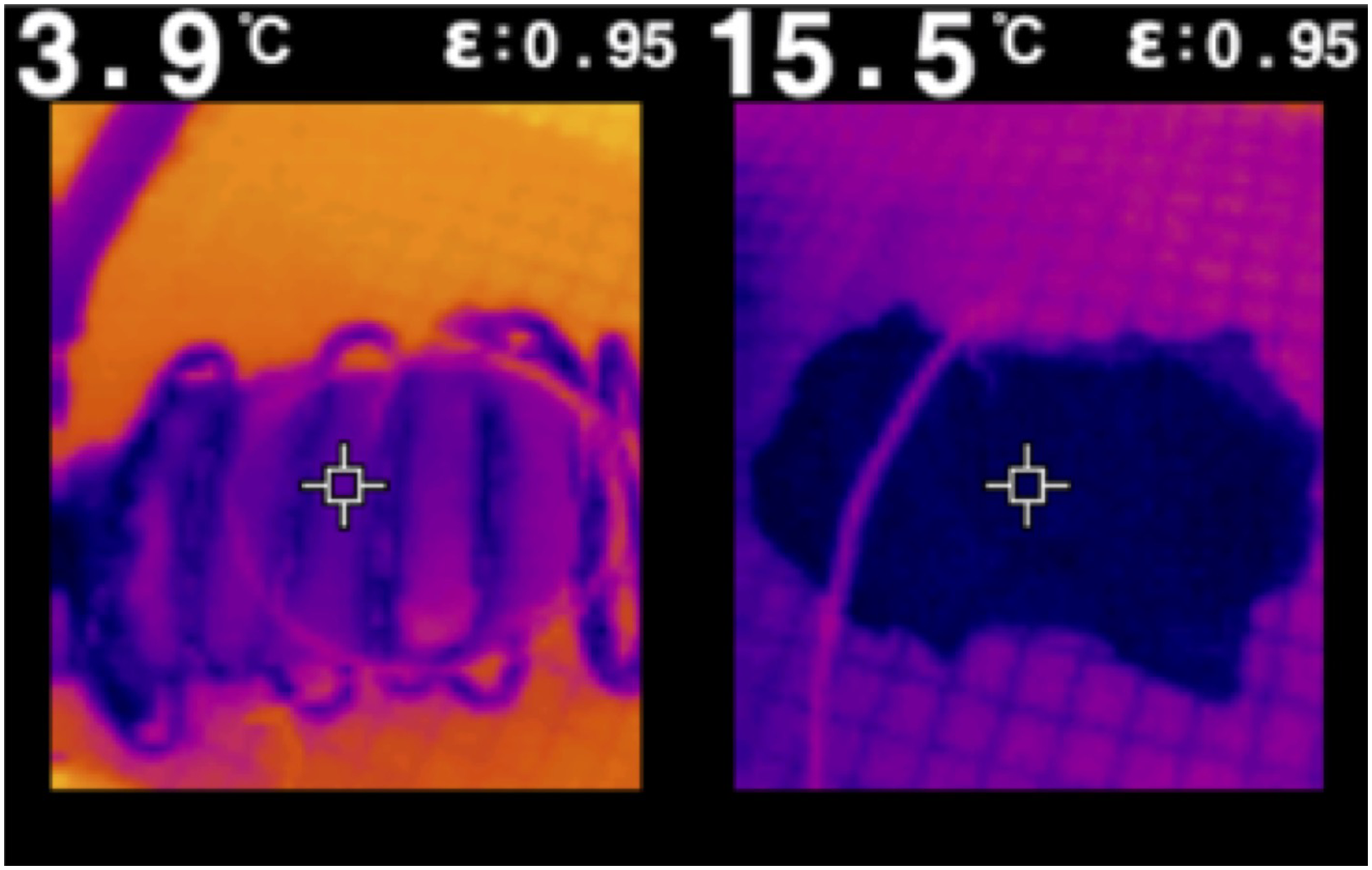
Mean surface and core temperatures after 60 minutes of WIT in the cooling jacket were significantly lower 5.8 ± 0.6°C (mean ± standard deviation) and 5.4 ± 0.5°C respectively vs 16 ± 1.4°C and 16.6 ± 1.2°C (P < .00001) (Figure 2). Moreover, our device mitigated increases in mean surface (2.4 ± 1.3°C vs 12.9 ± 0.9°C) and core temperatures (2.8 ± 1.7°C vs 14.1 ± 1.5°C) after 60 minutes of WIT (P < .00001) (Figure 2). Statistically significant divergence in surface and core (Figure 2) temperatures between cooled and control kidneys was detected at the initial 5-minute time point (P < .05) and sustained throughout the entire duration of the study (difference of 2.1°C and 2.7°C for surface and core temperatures at 5 minutes, 10.8°C and 11.2°C at 60 minutes). Comparison of cortical and medullary temperatures and thermal imaging techniques indicate that the device was able to maintain uniform cooling across both the surface and cross-sectional area (difference of 0.4°C at 60 minutes) (Figures 2 and 3). No statistically significant difference between surface and core temperatures was detected throughout the study. Mean surface and core temperature profiles of cooled and control porcine kidneys. IR thermal images of cooled (left) and control (right) kidneys at 60 minutes.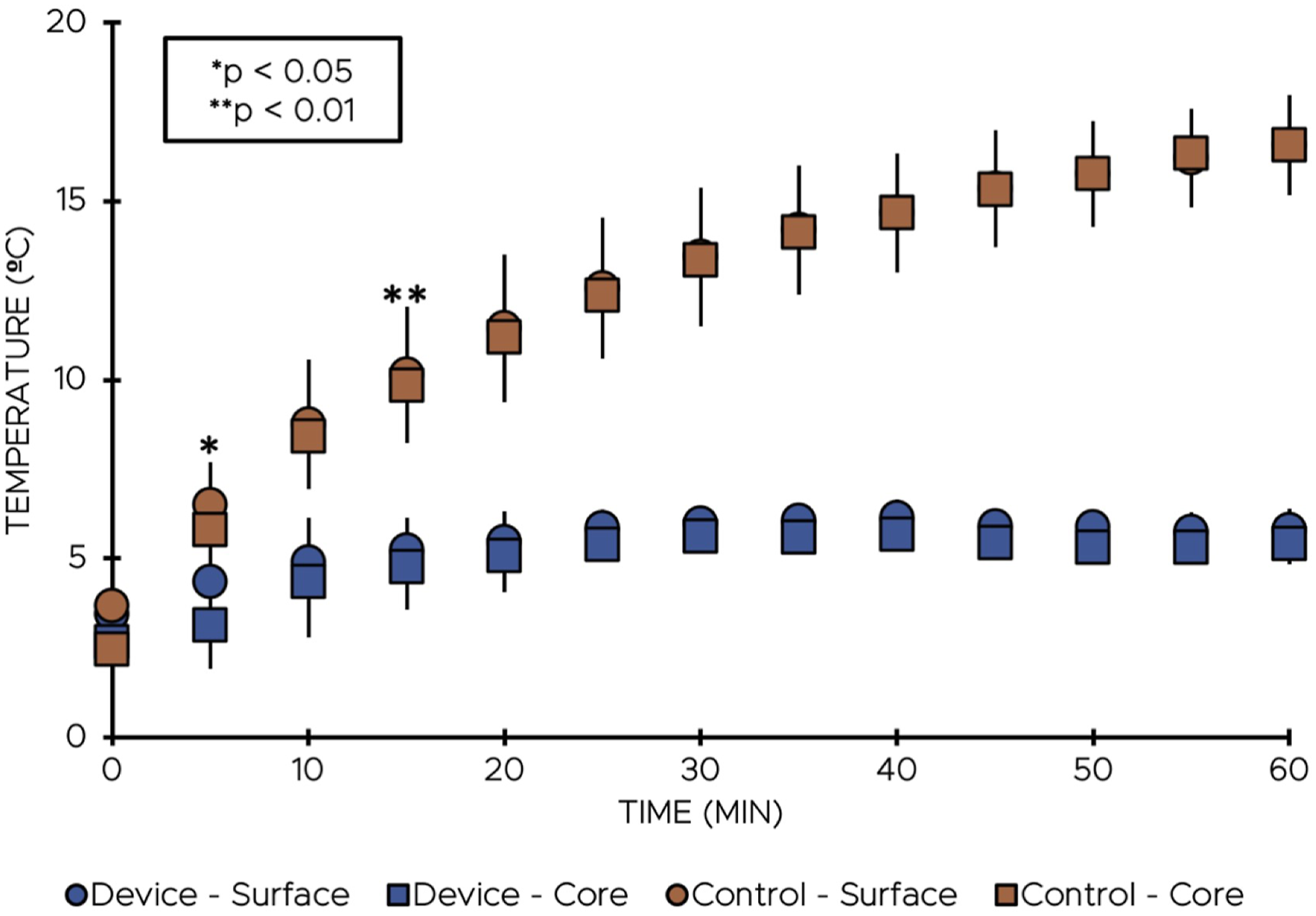
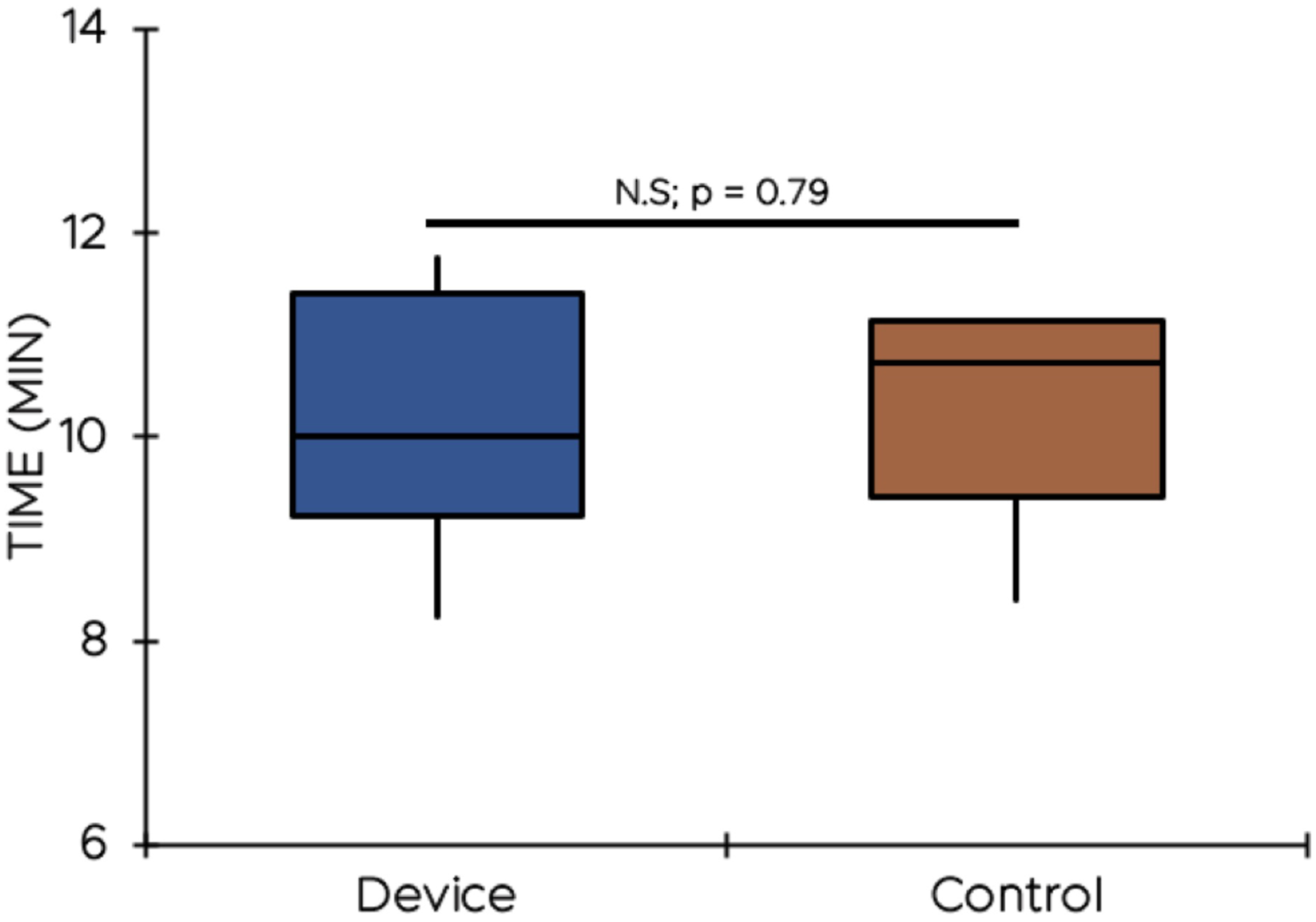
To demonstrate device usability, anastomosis of porcine renal arteries to GORE-TEX® vascular grafts was performed in a simulated operative field (Figure 4). Use of the cooling jacket was compared to the standard allograft support system used at our institution (Figure 4). Mean anastomosis times were compared between the device and control groups. No significant time difference was found (P = .79) between mean anastomosis time of jacket-enclosed kidneys (10.20 minutes) and control kidneys (10.42 minutes), as shown in Figure 5. Surgeons noted that the cooling jacket provided improved support and exposure for the anastomosis and found it easier to control the allograft position. Top: Renal vessel anastomosis to GORE-TEX® graft in simulated operative field. Bottom: suspension of control kidney using umbilical tape. Comparison of anastomosis time of porcine renal vessels to GORE-TEX® graft.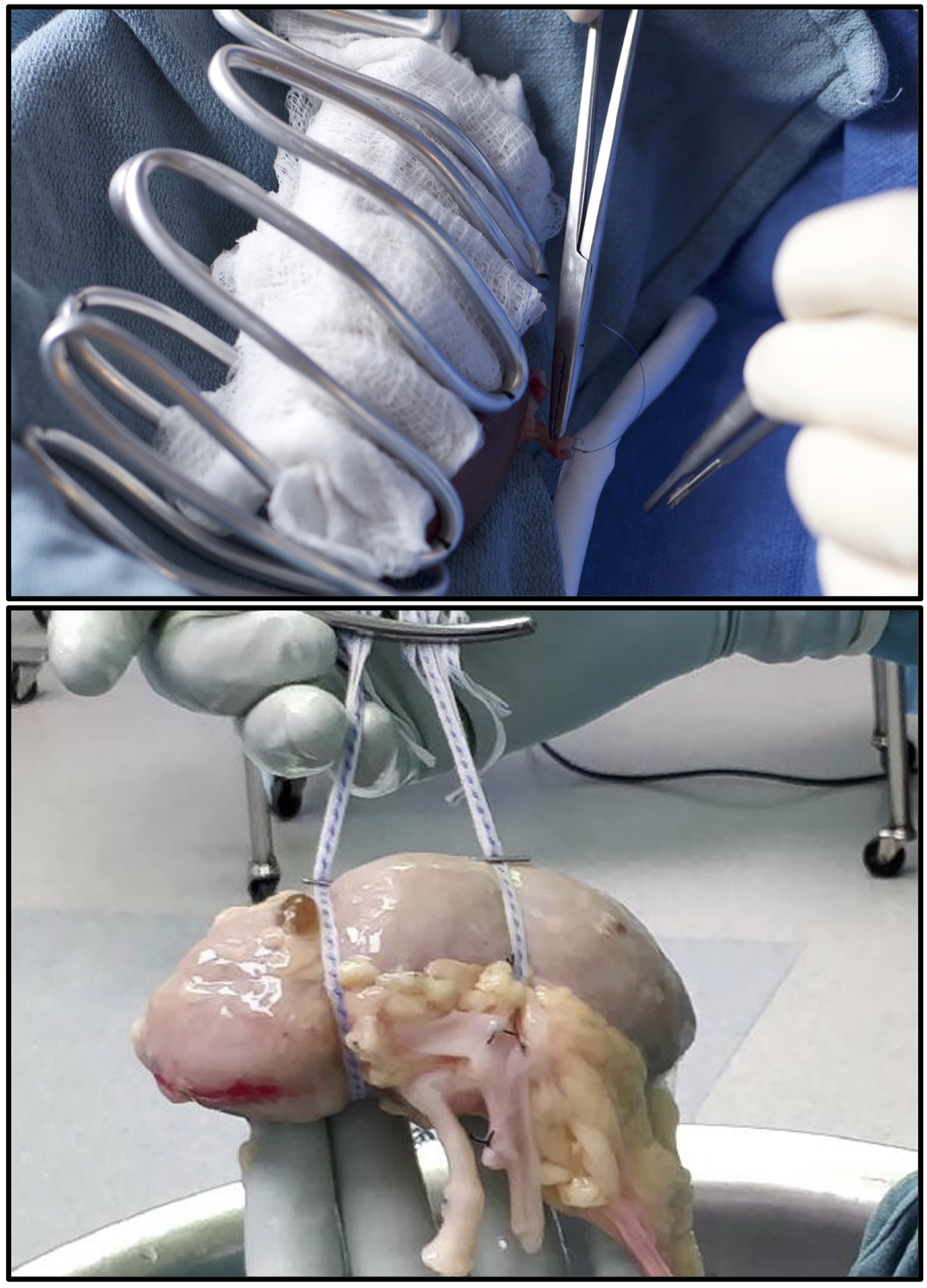
Discussion
Limiting WIT requires efficient vascular anastomoses, while maintaining renal hypothermia during this critical portion of the procedure. To date, three general strategies have been reported in the literature; cooling irrigation, ice bag immersion, and the use of a cooling jacket. Although each strategy comes with inherent limitations (i.e. ineffectiveness, too bulkiness, complexity of use), strategies employing cooling jackets have been shown to be promising. These limitations hampered the widespread adoption of methods to maintain renal hypothermia during kidney transplantation surgery to date, leaving an important warm ischemic threat left unaddressed. We set out to develop a renal cooling jacket that is simple to use and effective in maintaining renal hypothermia, while providing the additional benefit of facilitating vascular anastomosis without obstructing or crowding the operative field. The device we report on here not only provided consistent cooling for 60 minutes, but also provided surgical retraction and support, allowing for hands-free assistance and rapid, unencumbered vascular anastomosis. We are the first to test a cooling jacket for this important practical element required for real world usability.
Malleable aluminum was chosen as the material for construction due to its thermal properties. The single-material construction provides excellent heat transfer capabilities (thermal conductivity of 205
Various tubing diameters were tested, but ØD of 3/16″ was chosen due to the balance of sufficient cooling surface area and internal diameter to allow adequate flow of coolant, and to provide a low profile to promote intra-operative visibility. Larger diameters would add bulkiness limiting the surgical field, while smaller diameters led to limitations in overall cooling ability as a result of flow limitation.
Maintaining renal hypothermia was our primary design criterium. The device was capable of maintaining uniform surface and core temperatures ≤ 5°C for 60 minutes using only gravity-fed chilled saline. These levels of cooling are comparable or better than what has been reported for other cooling jackets and were achieved using a more user-friendly design.12,31-34 Maintaining the allograft at 5°C is sufficient to all but eradicate WIT. Although studies exploring the implications of renal cooling in transplantation are lacking, the available evidence suggests that limiting rewarming during the vascular anastomosis could drastically reduce rates of delayed graft function and graft loss.12,18,23 In addition, our device relieves the time pressure often experienced with complex anatomy, allowing the surgeon to focus on the technical aspects of the anastomosis. It should also be noted that most transplant centres are located at teaching hospitals and by alleviating the element of time pressure, this device could provide an added benefit of improving opportunities for surgical education and instruction.
Our secondary design criterium was to create a device that facilitated rather than impaired the surgical process. By utilizing a semi-rigid material, our device is the first cooling jacket that also acts as a surgical retractor and can allow for hands-free operation. We anticipate attachment to Bookwalter or other similar retractor set-ups could allow this device to easily integrate into the operative environment. In a simulated operative field, we demonstrated that the use of our device allowed for rapid vascular anastomosis times and did not obstruct the surgeon’s view. In fact, the low profile and grasping/ungrasping function allowed for easier handling of allograft when compared to the commonly employed umbilical tape method.
One aspect of renal cooling during anastomosis not addressed by our results is the abrupt temperature shift to normothermia upon reperfusion. Although research into this particular aspect is lacking, there are some reports of abrupt rewarming leading to compromised mitochondrial integrity.35,36 Because we used saline as a coolant, the issue of abrupt rewarming could be mitigated by slowly warming the saline a few moments prior to releasing the vascular clamps. In fact, use of this device would allow the surgeon complete control of the allograft temperature at all times. This represents an area for future development and study.
Ultimately, we designed and produced a cooling jacket that can maintain renal hypothermia for the duration of vascular anastomosis, without adding bulk or obstructing the surgeon’s view in an ex-vivo simulated operative field. The design of this device helps to overcome many practical barriers to cooling jacket use in in-vivo transplantation. Additionally, our device can easily be modified to allow for retroperitoneal cooling of the kidneys during deceased-donor organ procurement, and there is evidence to suggest that kidneys may not be sufficiently cooled during this process with traditional methods.37,38 We hope that the simplicity and functionality of this device will make for diminished barriers to adoption in clinical transplantation and controlled trials. The device is currently protected by US provisional patent (application number 63013691).
Future Directions
Further testing is necessary to evaluate the cooling device’s performance in vivo and across different transplantation scenarios. Future studies should focus on assessing the device’s efficacy in live animal models and human clinical trials, investigating the device’s integration into robotic kidney transplantation, and evaluating the device’s utility in teaching cases. For example, the device’s utility shows promise in teaching environment where it can alleviate time pressure and enhance learning in novice transplant surgeons, however, evaluating it in teaching cases is a necessary subsequent step. Robotic kidney transplantation is another emerging field that presents unique challenges, particularly in maintaining optimal temperatures during the procedure. Our cooling device could be highly beneficial in this context and should be an area of future testing. The current model’s malleable and form-fitting design could allow it to be easily adapted for use in robotic procedures, providing consistent cooling without obstructing the robotic instruments' operational field. Furthermore, its hands-free capability is particularly advantageous in robotic surgery, as it reduces the need for manual adjustments, thereby enhancing the efficiency and safety of the procedure. These studies will help determine whether the device should be universally applied in all transplant surgeries or tailored for specific scenarios such as robotic procedures and teaching cases.
Footnotes
Authorship
AD: Participated in research design, device construction, data collection and writing of paper
LW: Participated in research design, data collection and writing of paper
LP: Participated in writing, and editing of manuscript
JN: Participated in writing, and editing of manuscript
TS: Participated in research design, device construction, data collection and writing of paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
