Abstract
Background
Gastro-esophageal reflux disease (GERD/GORD) is a chronic condition in which gastric acid flows backwards up into the esophagus, causing heart burn and a higher disposition to esophageal cancer. The reflux is caused by impairment of the lower esophageal sphincter (LES). Over the past century gastro-esophageal reflux has become the principal gastrointestinal condition of our time. The proton pump inhibitor class of drugs is effective in ameliorating the symptoms of reflux. The cost of investigation of patients in Europe is €100 billion per annum. The cost in days lost from work is €100 billion per annum in Europe. The global cost is 3 times this amount.
Methodology
The proposed device for treating gastro-esophageal reflux is a biodegradable valve that is placed non surgically in the esophago-gastric junction to prevent reflux from the stomach to the esophagus.
Experiment Results
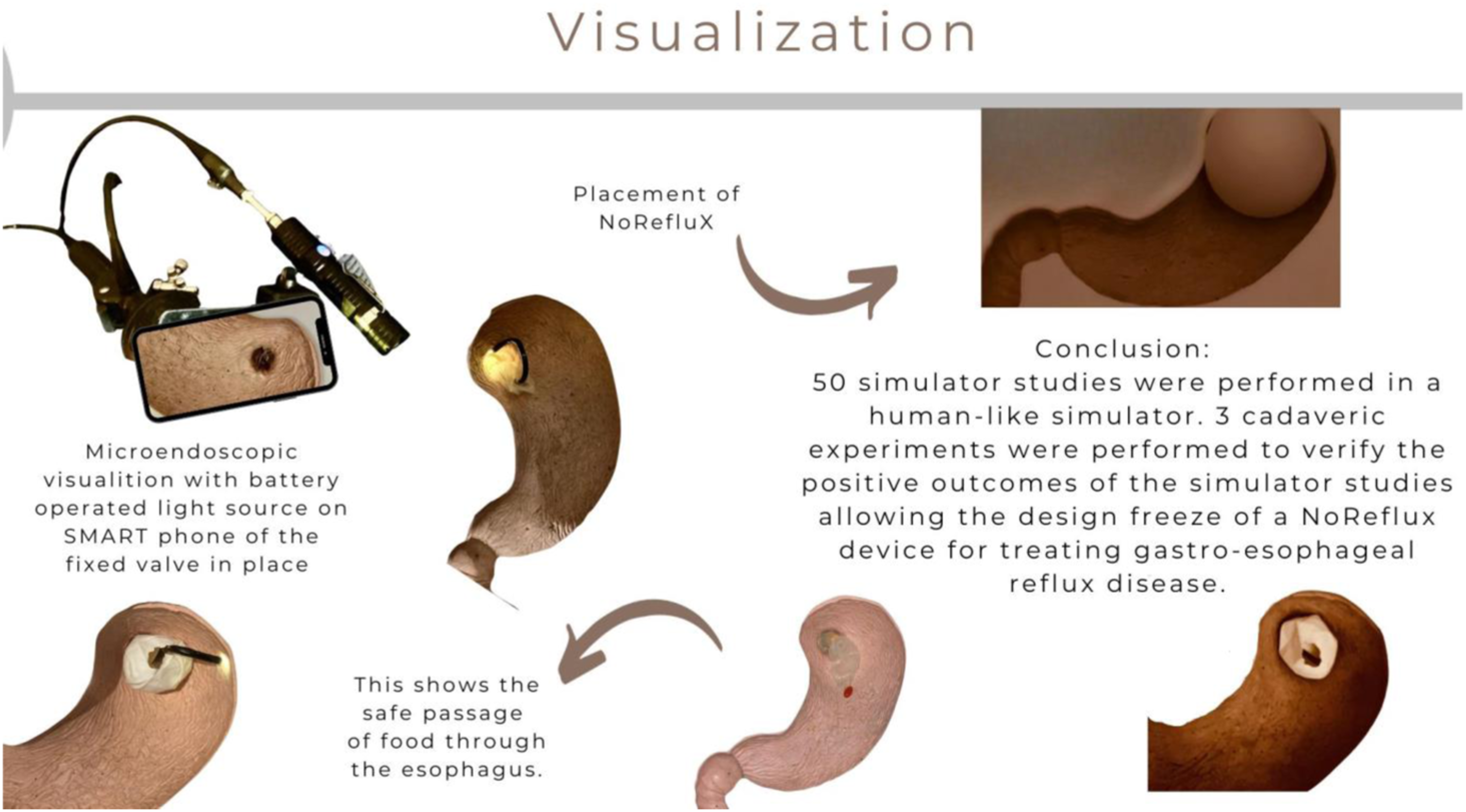
50 simulator studies were performed with the patented device to elucidate the most consistent method of insertion and fixation in a human like simulator. The simulator was designed to replicate the normal human gastro-esophageal anatomy and characteristics. Four animal insertions were performed under ethical regulation at Amsterdam Medical Centre, Netherlands. Three cadaveric experiments were performed at Hackensack University Hospital, New Jersey, USA, to verify the positive outcomes of the simulator studies.
Conclusion
Successful outcomes of simulator studies and cadaveric experiments allowed the design freeze of a NoReflux device for treating gastro-esophageal reflux disease.
Introduction
Gastro-esophageal reflux disease (GERD) is a chronic condition in which gastric acid flows backwards up into the esophagus, causing heart burn, acid regurgitation and a higher disposition to esophageal cancer. The reflux is caused by impairment of the lower esophageal sphincter (LES). Over the past century gastro-esophageal reflux has become the principle gastrointestinal condition of our time. 1 Extra-esophageal symptoms such as laryngitis, asthma and sleep disturbance are related to this condition. The proton pump inhibitor class of drugs is effective in ameliorating the symptoms of reflux.
Gastro-esophageal reflux disease is the most common upper gastrointestinal disorder in the Western world. It is estimated that as much as 20% of the population of Europe experiences symptoms of reflux disease. About 7%–10% of the adult population report daily heartburn, while up to 30%–40% of patients complain of symptoms compatible with GERD monthly. Of this group, 2% shows erosive esophagitis to an extent that warrants pharmacologic therapeutic intervention. 2 Medical management on a lifetime basis costs more than €250,000.
At the more severe end of the spectrum, increasing degrees of esophageal injury can lead to an associated progressive sphincter dysfunction, culminating in the development of Barrett’s metaplasia and even adenocarcinoma. Esophageal adenocarcinoma currently represents one of the fastest-growing categories of cancer in Europe.
Current endoscopic Nissen fundoplication surgical technique suffers 7%-30% failure rate in the most experienced surgeon’s practice and technically difficult for the average surgeon to perform successfully.3,4 Surgical failures result in corrective surgery. Many patients report continuing to take their medications even after fundoplication surgery. Several endoscopic techniques have been launched since 2003, these methods include a variety of endoscopic suturing procedures.
Conclusion
There is a strong unmet need amongst GERD patients and gastroenterologists for an effective treatment option.
System Design
We have developed No-Reflux, a unique biodegradable valve that enables a simple, least-invasive approach to potentially treat GERD. The NoReflux device is a 1-way biodegradable valve that is attached to the gastro-esophageal junction using a CE-and FDA-approved glue. Upon attachment, NoReflux prevents reflux of gastric acid into the esophagus. NoReflux is a simple solution to enable effective treatment of GERD patients.
We have studied the advantage of biomedical implants in cadaver, in vitro and simulator studies. The method of implantation of the valve is perfected to allow a physician to safely place a biomedical synthetic valve in the stomach without recourse to surgery.
The encapsulated valve can be swallowed by a patient thus making this innovative treatment a
The method of insertion is outlined in Insertion of NoReflux. Mode of action of valve. Visualization after placement.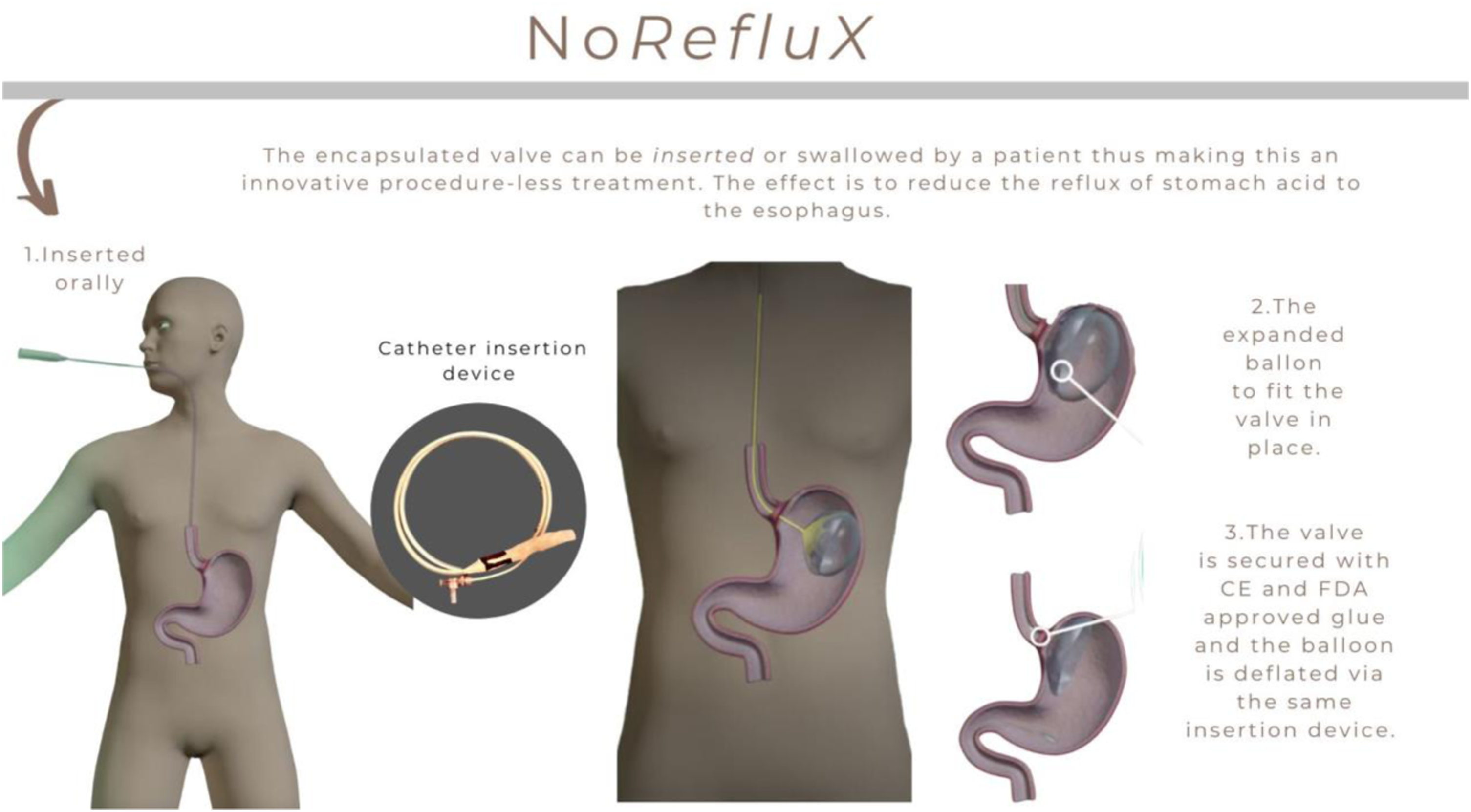

The effect of the valve is to reduce reflux of stomach contents to the esophagus. This allows a healing process to occur. The valve is created from a biodegradable material that is released after approximately 4 months. The valve can be removed endoscopically.
The valve is designed to allow regurgitation in the event of increase in pressure in the stomach.
Supplemental Material
Footnotes
Author Notes
The device is patented in USA and EUROPE.
US Patent Number: 8,915,967
European Patent Number: 1703870
First in human clinical trials will commence in Europe in September 2023.
Author Contributions
Study concept and design:
Acquisition of data:
Analysis and interpretation:
Study supervision:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
